Abstract

Pulmonary embolism (PE) is a major cause of morbidity and mortality, accounting for an estimated 100,000 deaths annually in the United States. 1 Patients often develop PE in the setting of other medical insults such as malignancy, surgery, and prolonged immobility. Few studies have examined the diverse causes of death among patients diagnosed with acute PE, which is critical in determining the potential impact of advanced PE therapies on survival. We conducted this analysis to determine whether deaths in the context of an acute PE are driven primarily by PE directly versus underlying illness. The Beth Israel Deaconess Medical Center institutional review board approved this study.
We included all patients admitted to our institution between August 2012 and July 2018 with a diagnosis of acute PE. Patients were identified using billing codes and the PE response team database. Retrospective chart review was performed to confirm diagnosis of acute PE and to ascertain registry information (as detailed elsewhere 2 ). PE severity was stratified into high-risk, intermediate-risk or low-risk 3 (online supplementary material). The primary outcome was inpatient death, which was independently adjudicated by three physicians (WE, SM, AW) with a fourth physician (BJC) who served as a tie-breaker. Death was determined to be PE-related if the primary mechanism was cardiogenic or obstructive shock, or respiratory failure without infection or alternative underlying pulmonary process. Deaths related to complications from the treatment of PE were counted separately.
Of 2052 patients admitted with acute PE, 53.1% were women, 65.5% were White, and the average age was 63.1 ± 16.3 years. Low-risk PE was most frequent (52.9%, n = 1086/2052), followed by intermediate-risk (42.8%, n = 878/2052), and high-risk PE (4.3%, n = 88/2052). In the overall cohort, 6.4% (n = 131) did not survive to discharge. These patients were older (67.3 ± 12.5 vs 62.1 ± 16.8, p < 0.001), disproportionately non-White (32.4% vs 26.1%, p < 0.001), more likely to have had cancer (48.1% vs 29.7%, p < 0.001), and more likely to have been hospitalized within the past 30 days (27.5% vs 18.0%, p = 0.007) compared to those who survived (online supplementary material Table 1).
Overall mortality and PE-related death stratified by PE severity was 3.6% (n = 39/1086) and 0% for low-risk, 6.7% (n = 59/878) and 0.7% for intermediate-risk, and 37.5% (n = 33/88) and 26% for high-risk PE (p < 0.001), respectively. Overall, the most common cause of death was cancer-related (32%, n = 41), followed by PE (22%, n = 29) and infectious/sepsis (17%, n = 22) (Figure 1). Advanced therapeutics were pursued in 59.1% (n = 52/88) with high-risk PE, and 7.3% (n = 64/878) with intermediate-risk PE, 38.6% and 6.6% of whom died, respectively. The rate of treatment-related death overall was 3.1% (n = 4/131).
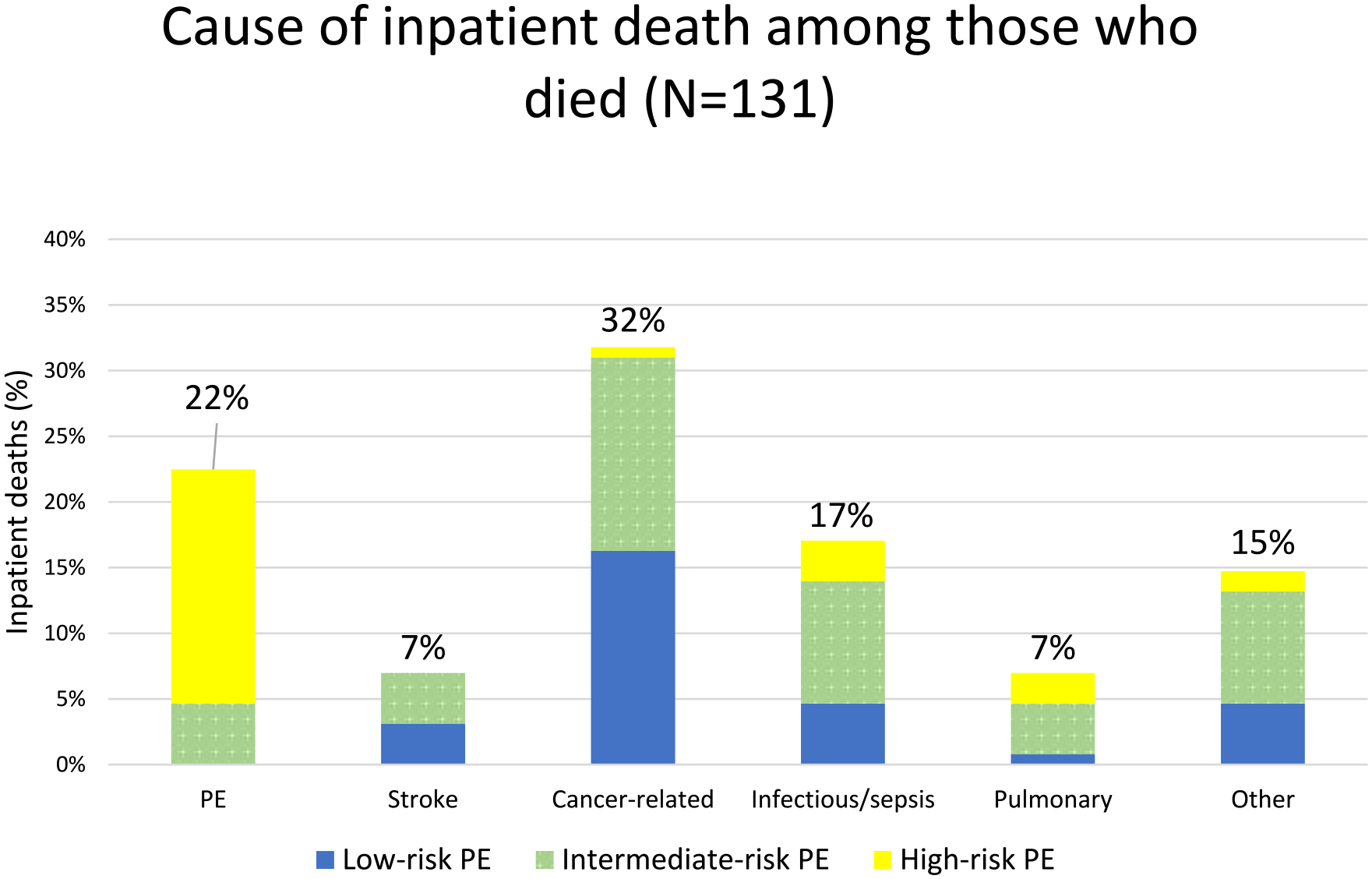
Adjudicated cause of inpatient death stratified by pulmonary embolism (PE) severity.
In this large cohort of patients diagnosed with acute PE, inpatient mortality was 6.4%. PE-related death was the primary cause of death in high-risk PE patients but was rare in intermediate- and low-risk PE.
Risk stratification is essential when determining appropriate therapy for acute PE. Our findings were consistent with prior studies demonstrating substantial PE-related mortality with high-risk PE and low PE-related mortality with low-risk PE. 4 Our study describes the PE-related mortality for intermediate-risk PE, which is poorly defined in the literature. A recent meta-analysis found that only half of PE studies reported a definition for PE-related death and definitions were heterogeneous. 5 Additionally, many studies are limited by reliance on death certificate International Classification of Diseases (ICD) codes, 6 which are susceptible to error and miscoding. These vague definitions make it challenging to interpret studies reporting PE-related mortality. Our focused cause of death adjudication process with access to the medical record allowed for a more rigorous assessment.
Mortality in patients with intermediate-risk PE is estimated at 2.9–14.7%. 7 For further stratification, the 2019 European Society of Cardiology/European Respiratory Society guidelines divided intermediate-risk PE based on the presence of either right ventricular (RV) strain by imaging or biochemical markers (intermediate-low) or RV strain on both imaging and biochemical markers (intermediate-high). 8 A subsequent study found that the mortality rate between intermediate-high and intermediate-low risk PE was similar, 7.7% versus 6%. 9 Of nearly 900 patients with intermediate-risk PE in our study, the rate of PE-related mortality was only 0.7%. Importantly, the practice at our center is to pursue advanced therapy in those deemed highest risk with concerning evidence of RV strain and clinical features. Given the observational nature of our study, it is difficult to quantify how these therapies may have reduced PE-related mortality. However, only 6.9% of intermediate-risk patients received advanced therapies, a rate lower than other contemporary reports. 10
Limitations in our analysis include use of data from a single center, which could limit generalizability. The retrospective nature does not allow us to adequately assess the potential benefit of interventions. Also, key variables were collected with manual chart review, which may have resulted in inadequate reporting as the findings were not externally validated.
In a large, single-center report, we demonstrate that PE-related mortality is rare among intermediate-risk PE with selected use of advanced therapies but remains elevated in high-risk PE patients. These data add to the existing understanding of PE prognosis. Further risk stratification investigation is needed to optimize selection for advanced therapies, particularly in patients with intermediate-risk PE.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231199488 – Supplemental material for Cause of death in patients with acute pulmonary embolism
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231199488 for Cause of death in patients with acute pulmonary embolism by William Earle, Shantum Misra, Alexander Wester, Matthew Herzig, George Abdallah, Charles B Ross, Eric A Secemsky and Brett J Carroll in Vascular Medicine
Footnotes
Acknowledgements
The authors would like to acknowledge Siling Li (Richard A. and Susan F. Smith Center for Outcomes Research in Cardiology; Department of Medicine, Beth Israel Deaconess Medical Center, Boston, MA) for her statistical review of this manuscript. An abstract of this work was presented at the ACC Annual Scientific Session & Expo in New Orleans, LA, March 2023.
Declaration of conflicting interests
Brett J Carroll serves as a consultant to Reliant Medical and Janssen Pharmaceuticals and receives institutional research support from Bristol Myers Squibb. Eric A Secemsky receives research grants to Beth Israel Deaconess Medical Center from NIH/NHLBI K23HL150290, US Food & Drug Administration, Benton Dickinson, Boston Scientific, Cook, CSI, Laminate Medical, Medtronic, and Philips; and consulting/speaking fees from Abbott, Bayer, Benton Dickinson, Boston Scientific, Cook, Cordis, CSI, Inari, InfraRedx, Medtronic, Philips, Shockwave, and VentureMed.
All other authors declare no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
