Abstract
The spectrum of venous thromboembolic (VTE) disease encompasses both acute deep venous thrombosis (DVT) and chronic postthrombotic changes (CPC). A large percentage of acute DVT patients experience recurrent VTE despite adequate anticoagulation, and may progress to CPC. Further, the role of iliocaval venous obstruction (ICVO) in lower-extremity VTE has been increasingly recognized in recent years. Imaging continues to play an important role in both acute and chronic venous disease. Venous duplex ultrasound remains the gold standard for diagnosing acute VTE. However, imaging of CPC is more complex and may involve computed tomography, magnetic resonance, contrast-enhanced ultrasound, or intravascular ultrasound. In this narrative review, we aim to discuss the full spectrum of venous disease imaging for both acute and chronic venous thrombotic disease.
Keywords
Introduction
The spectrum of venous thrombotic disease encompasses both acute deep venous thrombosis (DVT) and its sequelae, including chronic postthrombotic changes (CPC) and postthrombotic syndrome (PTS). In addition, the significance of thrombotic or nonthrombotic iliocaval venous obstruction (ICVO) such as nonthrombotic iliac vein lesions (NIVL) has been increasingly recognized in lower-extremity venous disease. It is estimated that there are as many as 900,000 DVT cases a year in the United States. 1 Recurrent DVT is common despite treatment and the development of PTS can be burdensome for both the patients and the healthcare system.1–5
Imaging plays a crucial role in the diagnosis and management of venous thrombotic disease. Conventional venography was the gold standard in diagnosing venous thrombotic disease, although limited by its invasiveness and contrast load for the patients. Venous duplex ultrasound (VDUS), however, is now the first-line diagnostic modality for both acute DVT and CPC. Despite this, VDUS has known technical limitations, particularly in patients with unfavorable body habitus. Its use is also limited in certain clinical scenarios, particularly for the assessment of CPC or recurrent DVT in the ipsilateral lower extremity. Advanced imaging modalities can help clarify indeterminate diagnosis, and can assess anatomical variants, extent of disease, and thrombus age, including computed tomography venography (CTV), magnetic resonance venography (MRV), MR direct thrombus imaging (MRDTI), contrast-enhanced ultrasound (CEUS), and invasive intravascular ultrasound (IVUS). In this manuscript, we aim to summarize the recent literature on VDUS and advanced imaging modalities for the diagnosis and management of lower-extremity acute and chronic venous thrombotic disease.
Sonographic evaluation of venous thrombotic disease
A variety of sonographic techniques have been described to evaluate both acute and chronic venous thrombotic diseases, including VDUS, US elastography, and CEUS.
Venous duplex ultrasound (VDUS)
VDUS remains the first-line diagnostic modality for suspected acute DVT. There are different technical approaches for performing VDUS, and the three most common methods include: (1) a limited two-point scan of the common femoral vein and the popliteal venous trifurcation; (2) a limited scan of the proximal deep venous system from the common femoral vein to the popliteal venous trifurcation; and (3) a ‘whole-extremity’ extended scan from the common femoral vein to the deep calf veins in 3 cm increments. Per recent recommendations from the Society of Radiologists in Ultrasound Consensus Conference, a whole-extremity ultrasound scan was the preferred diagnostic modality when time and resources permit. 6 Similarly, recent trials have shown that limited or extended VDUS should be utilized in the appropriate clinical setting, depending on the pretest probability and/or D-dimer positivity. 7 Regardless of the applied scanning technique, VDUS is a remarkably reliable diagnostic tool with sensitivities of 100% and 94% and specificities of 98% and 75% for acute proximal and distal DVT, respectively.8,9 With a negative scan, the risk of venous thromboembolism is less than 2% (Table 1).10–12
Summary of multiple modalities, including the advantages and disadvantages of each modality, in the diagnosis of acute and chronic venous thrombotic disease.
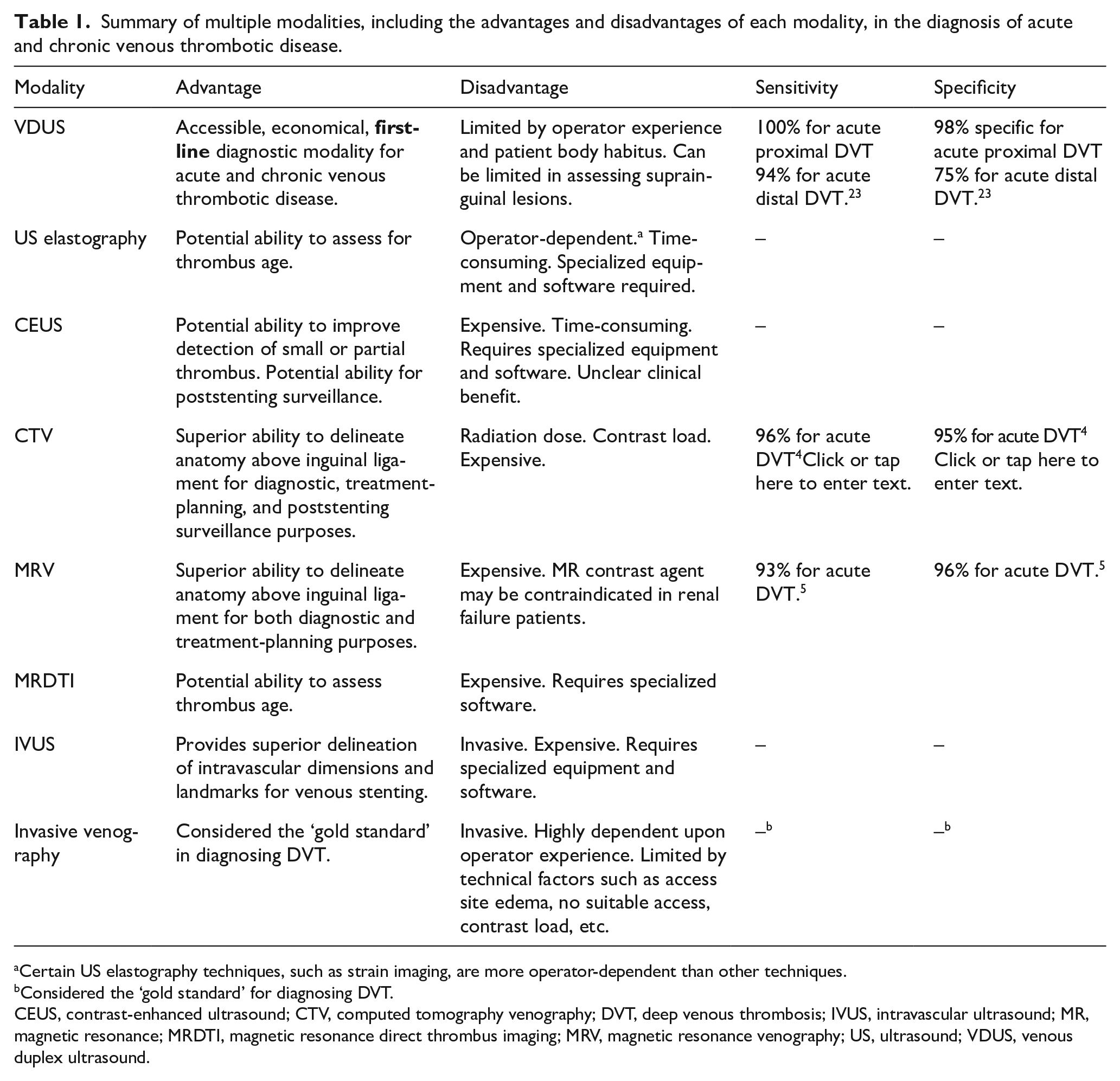
Certain US elastography techniques, such as strain imaging, are more operator-dependent than other techniques.
Considered the ‘gold standard’ for diagnosing DVT.
CEUS, contrast-enhanced ultrasound; CTV, computed tomography venography; DVT, deep venous thrombosis; IVUS, intravascular ultrasound; MR, magnetic resonance; MRDTI, magnetic resonance direct thrombus imaging; MRV, magnetic resonance venography; US, ultrasound; VDUS, venous duplex ultrasound.
However, definitive differentiation between acute recurrent DVT and CPC is more challenging on VDUS. Despite treatment, more than 50% of patients demonstrate chronic postthrombotic changes 1 year after DVT, including the presence of noncompressible venous segments.13,14 Because of the similar imaging appearance of acute recurrent DVT and CPC (Table 2), VDUS is frequently inconclusive in the identification of suspected recurrent ipsilateral DVT. 15 One solution is to obtain a baseline scan after the patient has completed anticoagulation therapy. Future scans are compared to the baseline scan, and changes in sonographic findings can be used to diagnose recurrent DVT.16,17 Another approach is to obtain a short-term follow-up scan after suspected recurrent DVT, 18 as CPC rarely changes over a short timeframe (Figure 1). However, in clinical practice, a baseline scan is frequently unavailable (particularly if patients present to a different institution). Further, obtaining a baseline scan after finishing anticoagulation therapy is against the Society of Vascular Medicine Choosing Wisely recommendation to obtain a repeat VDUS in the absence of clinical changes. Unfortunately, interobserver agreement for acute recurrent DVT is moderate at best without the baseline scan. 19
Features distinguishing acute deep venous thrombosis from chronic postthrombotic changes on various modalities.

Certain US elastography techniques, such as strain imaging, are more operator-dependent than other techniques.
CTV, computed tomography venography; DVT, deep venous thrombosis; MRDTI, magnetic resonance direct thrombus imaging; MRV, magnetic resonance venography; US, ultrasound; VDUS, venous duplex ultrasound.
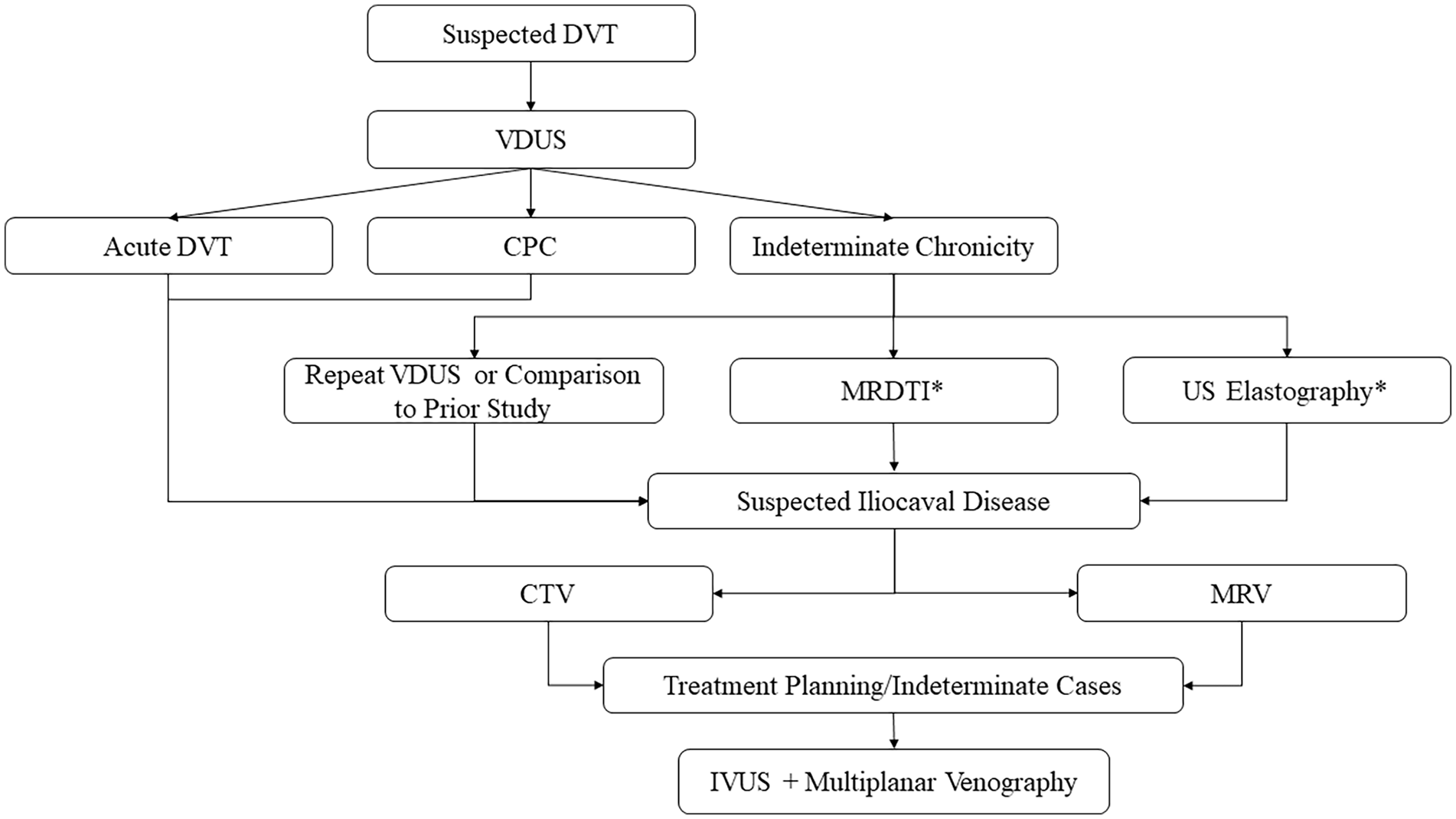
Proposed diagnostic flow chart for acute and chronic venous thrombotic disease, incorporating emerging imaging technologies.
For patients with suspected ICVO, VDUS is readily accessible and therefore considered the preferred first-line diagnostic modality for reflux and infrainguinal venous stenosis. 20 It is important to note that there are no established VDUS diagnostic criteria for ICVO. Direct visualization of the stenosis and flow turbulence may suggest outflow obstruction and correlate well with IVUS findings in patients with favorable body habitus. 21 In patients with unfavorable body habitus (i.e., poor visualization of the common iliac vein), indirect imaging signs on VDUS may include continuous flow, reflux, and poor flow augmentation to Valsalva maneuver.22,23 In general, several small studies have shown that VDUS was not a reliable diagnostic tool for assessment of ICVO.24,25 However, in patients with significant obstructive symptoms, indirect signs of venous obstruction highly correlated with the degree of stenosis on IVUS, which is considered the gold standard. 26 It is also important to note that a large percentage of patients with severe obstructive symptoms may present without evidence of reflux on VDUS yet with obstructive lesions present on IVUS. 27 Therefore, in patients with clinically significant obstructive symptoms, VDUS should be the first-line diagnostic modality and secondary signs suggestive of ICVO should undergo further testing. Imaging modalities such as CTV, MRV, and possibly multiplanar venography with IVUS can be considered in patients with debilitating symptoms, noting there is no consensus on the most optimal modality.
Ultrasound elastography
It is important to differentiate between acute recurrent DVT and CPC, as the management differs. 28 Commonly used in hepatic imaging but not routinely used in vascular imaging, elastography is one emerging sonographic technique that may prove useful in determining thrombus age. As the thrombus matures its composition changes from predominantly ‘elastic’ platelets to ‘stiff’ fibrinous material and this histopathological alteration provides the basis for elastography (Figure 2). A review of various elastography techniques, however, is beyond the scope of this article. In short, elastography techniques can be generally divided into strain imaging (SI) and shear wave imaging (SWI), with the former requiring internal or external compression (which induces shape distortion, hence ‘strain’), and the latter using ultrasound-generated shear wave stimuli (i.e., mechanically generated compressive waves). 29
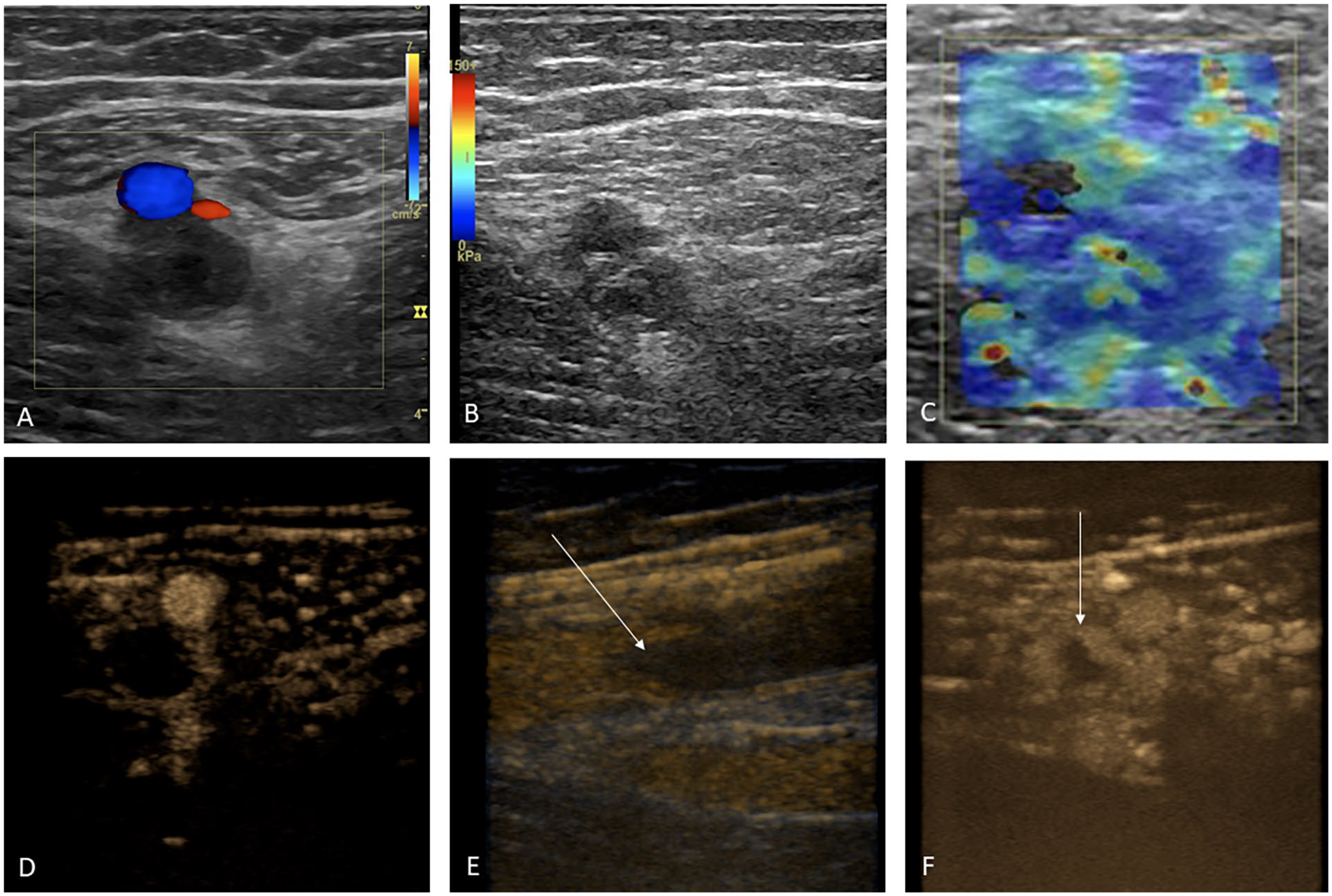
A 42-year-old man with lower-extremity pain and DVT was clinically suspected.
Initial animal studies have shown that chronic thrombi were associated with higher stiffness (measured by the degree of deformation) compared to acute thrombi.30–32 Rubin et al. showed significant differences in stiffness between the acute and chronic thrombi (p < 10–7) in human subjects (thrombus age measured by duration of presence on VDUS). 33 A more recent study of 149 patients has demonstrated significant differences in strain value between acute thrombi and CPC. Further, using a cutoff strain value of 4, the sensitivity and specificity were 99% and 99%, respectively, for acute DVT. 34
All the above studies were performed using the SI technique, which has certain limitations. External compressions are required and these are operator-dependent. In some of the studies, elastography was performed by one or two operators to minimize inherent heterogeneity. Having the same operator may be applicable within a controlled study, but this is not feasible in daily clinical practice. Secondly, a reference point is needed for SI which is arbitrarily chosen at the time of the exam and may not be reproducible in a clinical setting. In contrast, SWI is operator-independent and does not require internal reference. Therefore, SWI is likely to be more applicable in daily clinical practice. A recent study using SWI has revealed significant differences in stiffness between the acute and subacute thrombi (p < 0.001). Further, using a cutoff value of 2.85, the sensitivity and specificity were 96.3% and 91.3% for acute DVT. 35 However, given the small sample size, further studies are warranted to validate these results.
Contrast-enhanced ultrasound (CEUS)
CEUS is an evolving sonographic technique that excels in evaluating vascular pathologies. CEUS contrast microbubbles are strictly intravascular and enable evaluation of solid organ lesions and arterial pathologies. In essence, ultrasound microbubbles are composed of a lipid outer shell containing inert gas. It has an excellent safety profile and is usually renal or pulmonary excreted. 36 It should be noted that CEUS (and contrast microbubbles) has only been US Food and Drug Administration-approved for use in echocardiography and limited hepatic applications. Therefore, CEUS for DVT is currently an ‘off-label’ use in clinical practice.
CEUS should not be the first-line screening test for acute DVT. As previously discussed, VDUS remains a reliable diagnostic modality, particularly with respect to proximal acute DVT. However, VDUS is limited in certain scenarios, such as in obese patients, patients with significant soft tissue swelling, and patients with affected vessels above the inguinal ligament. Previous studies have shown that VDUS sensitivity for calf vein DVT was lower compared to proximal DVT (64% vs 95%). 10
CEUS enables assessment of the entire lower-extremity deep venous system, including the calf veins and internal iliac vein.37,38 Bucek et al. compared CEUS to VDUS in assessing calf vein thrombosis using invasive venography as the reference standard. The authors have found that CEUS reduced the rate of indeterminate scans from 55% to 20% and improved the specificity from 25% to 67%. 39 Similarly, Smith et al. have demonstrated superior visualization of the popliteal and femoral veins with CEUS but not the common femoral vein in patients at high risk for DVT and with a negative initial VDUS scan. 40
It must be emphasized that the added clinical utility of CEUS over VDUS in diagnosing acute DVT is unclear. The current studies have shown that CEUS might improve visualization of distal calf veins but not the proximal venous system. Unlike proximal DVT, there remains an ongoing debate regarding the optimal management strategy of calf vein DVT. 41 Therefore, the added utility of better calf vein DVT visualization may not be necessarily clinically relevant. Similarly, isolated pelvic vein thrombosis is exceedingly rare, such that the added utility of CEUS is unlikely to affect clinical management. 42 Therefore, CEUS (Figure 3) should be reserved when VDUS is inconclusive or when the clinical suspicion is high with a negative initial VDUS scan.
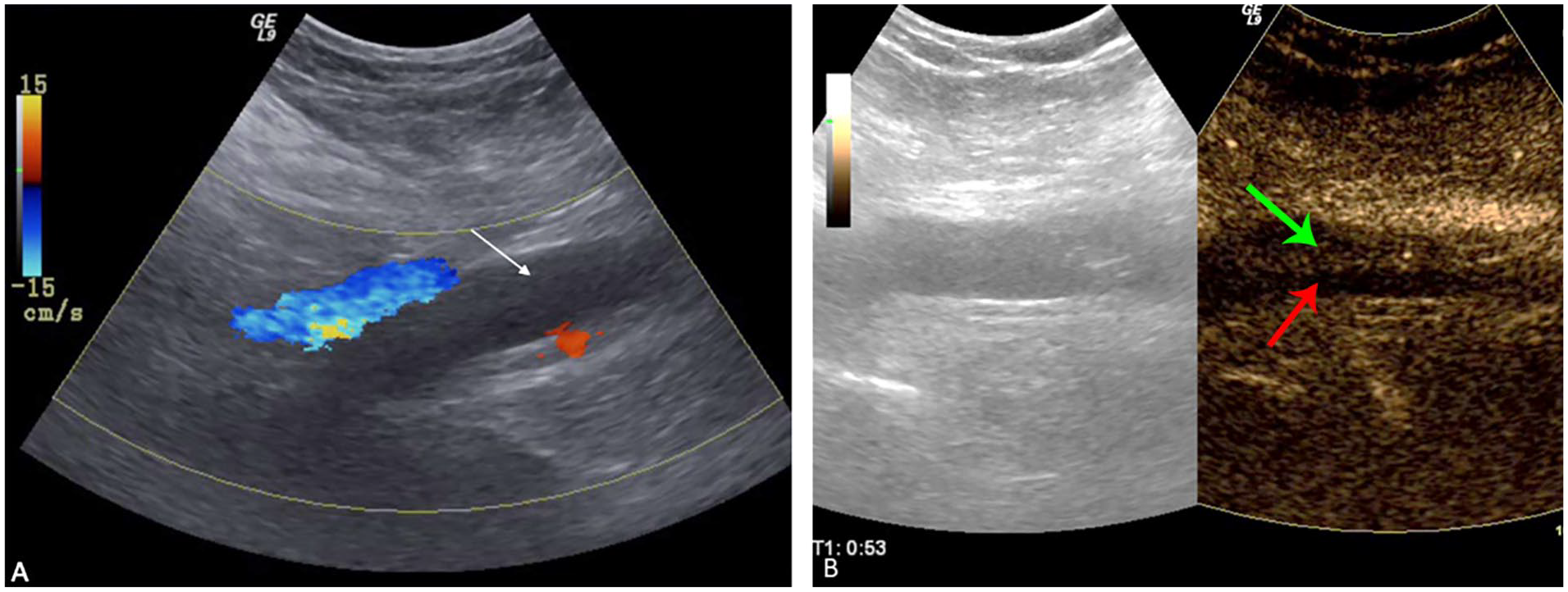
A 45-year-old male presented with lower-extremity swelling.
CEUS may also be helpful for poststenting surveillance in patients with ICVO. Liu et al. compared CEUS and VDUS to CTV in diagnosing in-stent stenosis and the authors found that there was a high degree of concordance between CEUS and CTV (κ = 0.884), resulting in a sensitivity and specificity of 88% and 97%, respectively, for CEUS. 43 It needs to be emphasized that these retrospective CEUS studies included a relatively small number of patients. Prospective trials are warranted to validate these findings.
Computed tomographic evaluation of venous thrombotic disease
Similar to CEUS, CTV should not be the first-line diagnostic modality for acute DVT but may be useful when VDUS is inconclusive and when there is suspected ICVO (Figure 1 and Table 1). Although CTV is associated with high sensitivity and specificity for acute DVT,44,45 the major concerns are the incremental radiation dose (particularly to the pelvis) and contrast load, which may be contraindicated in patients with renal insufficiency. One mitigating strategy to decrease the contrast amount is to reduce the tube voltage while preserving imaging quality. 46 A different strategy is to combine indirect CTV with CT pulmonary angiography (CTPA) in patients with suspected PE, thus negating the need for additional contrast load. However, previous studies have shown that there was little added benefit of indirect CTV while exposing patients to increased radiation.47–49 As bedside VDUS becomes increasingly accessible, CTV should not be the first-line diagnostic modality for acute DVT.
However, in cases of suspected ICVO, CTV is useful in the diagnosis, treatment planning, and posttreatment surveillance. It is important to note that CTV should not be the first-line screening test for ICVO and CTV should be reserved for patients with convincing signs of ICVO. 50 CTV was able to demonstrate anatomic abnormalities in the iliofemoral veins and inferior vena cava, including vascular and nonvascular etiologies, such as iliac vein compression, pelvic malignancy, or benign masses.51,52 Liu et al. have found CTV to be highly sensitive and specific in diagnosing ICVO and in differentiating thrombotic and nonthrombotic iliac compression syndrome. CTV was able to delineate the precise venous anatomy, collateral flow, and extent of iliac thrombosis. 53 In comparison to VDUS, CTV requires decreased exam time and can more readily detect underlying abnormality, particularly within the pelvis. 54 In comparison to IVUS, recent evidence has shown that CTV results were comparable when the degree of venous outflow stenosis exceeded 50%. 55 However, other studies have shown IVUS to be superior to CTV in assessing the degree of stenosis in patients with significant obstructive symptoms. 56 Therefore, in patients with high clinical suspicion and negative CTV results, IVUS may be considered as a reasonable next step.
Poststenting, CTV enabled characterization of morphological changes, including stent occlusion, collapse, and neointimal hyperplasia. 57 Furthermore, as reflux is often uncorrected with iliac vein stenting (thus limiting the utility of VDUS on poststenting surveillance, as the patient will continue to demonstrate reflux), 27 CTV should be used to assess for the presence of new lesions and/or re-stenosis in patients with recurrent or persistent symptoms after initial stenting.
Magnetic resonance evaluation of venous thrombotic disease
MR evaluation of venous thrombotic disease includes MRV and MRDTI. Both techniques can provide valuable information on both the diagnosis and the characterization of DVT.
Magnetic resonance venography (MRV)
MRV with and without contrast is similar to CTV with the obvious benefit of mitigated radiation exposure. MRV can be used to diagnose acute DVT when VDUS is inconclusive. Various MRV techniques have been described, including time-of-flight (TOF), phase contrast (PC) sequence, fresh-blood imaging (FBI), steady-state free precession sequence (SSFP), and contrast-enhanced T1-weighted gradient echo sequence using gadolinium-based contrast agents. A detailed discussion of various MRV techniques is beyond the scope of the article, although MRV has been demonstrated to have adequate sensitivity and specificity (Table 1). 58
Although not definitive, MRV features can be used to differentiate between acute DVT and CPC (Table 2 and Figure 4), which carries important prognostic implications. In the recent post hoc analysis of the Dutch CAVA trial, MRV could differentiate between acute, subacute, and chronic thrombi. In particular, it is noteworthy that 91.7% of self-reported chronic thrombi were re-categorized into acute or subacute thrombi by MRV, though 4% of the self-reported acute thrombi were chronic thrombi by MRV. Further, MRV categories were predictive of procedural duration and success rate: the average durations for catheter-directed thrombolysis (CDT) were 23 hours, 43 hours, and 85 hours for acute, subacute, and chronic thrombi, as defined by MRV (p < 0.0001). CDT was successful in 68.2% of the acute and subacute DVT group versus 16.7% of the chronic thrombus group (OR = 10.71; 95% CI, 2.07 to 55.5; p = 0.006). 59
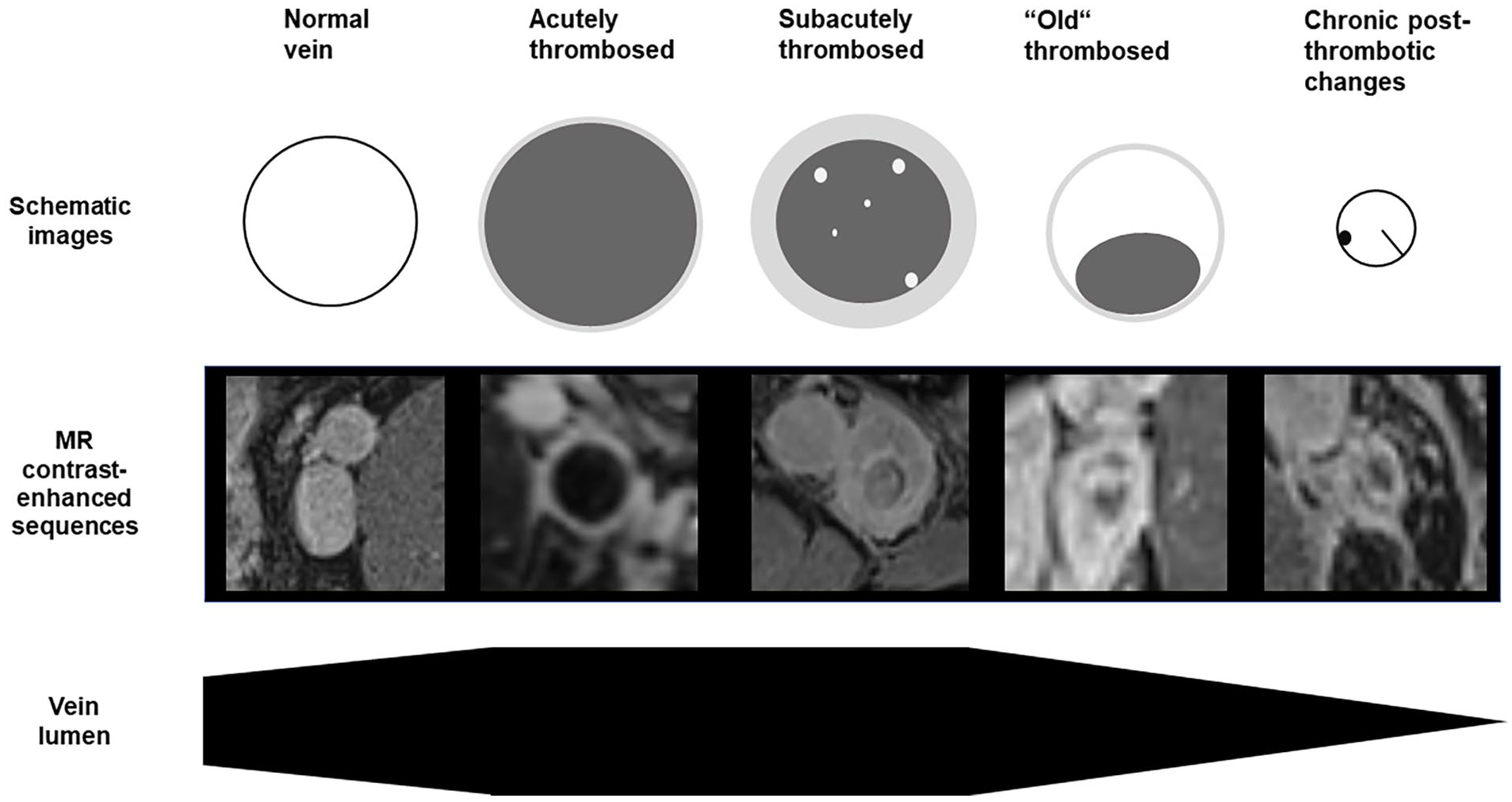
Stages of venous thrombosis by MRI. A normal vein shows a homogeneously opacified vein lumen with normal diameter. An acutely thrombosed vein is dilated with a low signal intensity lumen and a small enhancing rim. A subacutely thrombosed vein is dilated with a low-intensity lumen and a thick enhancing rim of contrast representing the venous wall. There might be small hyperintense areas within the thrombus as a sign of partial recanalization. Over time, the venous luminal size normalizes with a partially patent lumen and peripheral, low signal intensity thrombus (‘old’ thrombosed vein). With chronic postthrombotic changes, the vein lumen is reduced in comparison with a normal vein. There might be intraluminal webs or scars.
Similar to CTV, recent investigative studies have shown that MRV could be used to assess ICVO (Figures 5 and 6). Massenburg et al. retrospectively compared 2D TOF MRV with IVUS and venography findings. The authors have found that MRV had a sensitivity of 100% and specificity of 22.7% in diagnosing the underlying etiology. 60 Similarly, Shi et al. compared multimodal MRV to venography and found that MR imaging yielded an accuracy of 96%, a sensitivity of 97%, and a specificity of 89% in diagnosing ICVO. 61 With regards to posttreatment surveillance, the use of MRV is limited due to venous stent susceptibility artifacts.
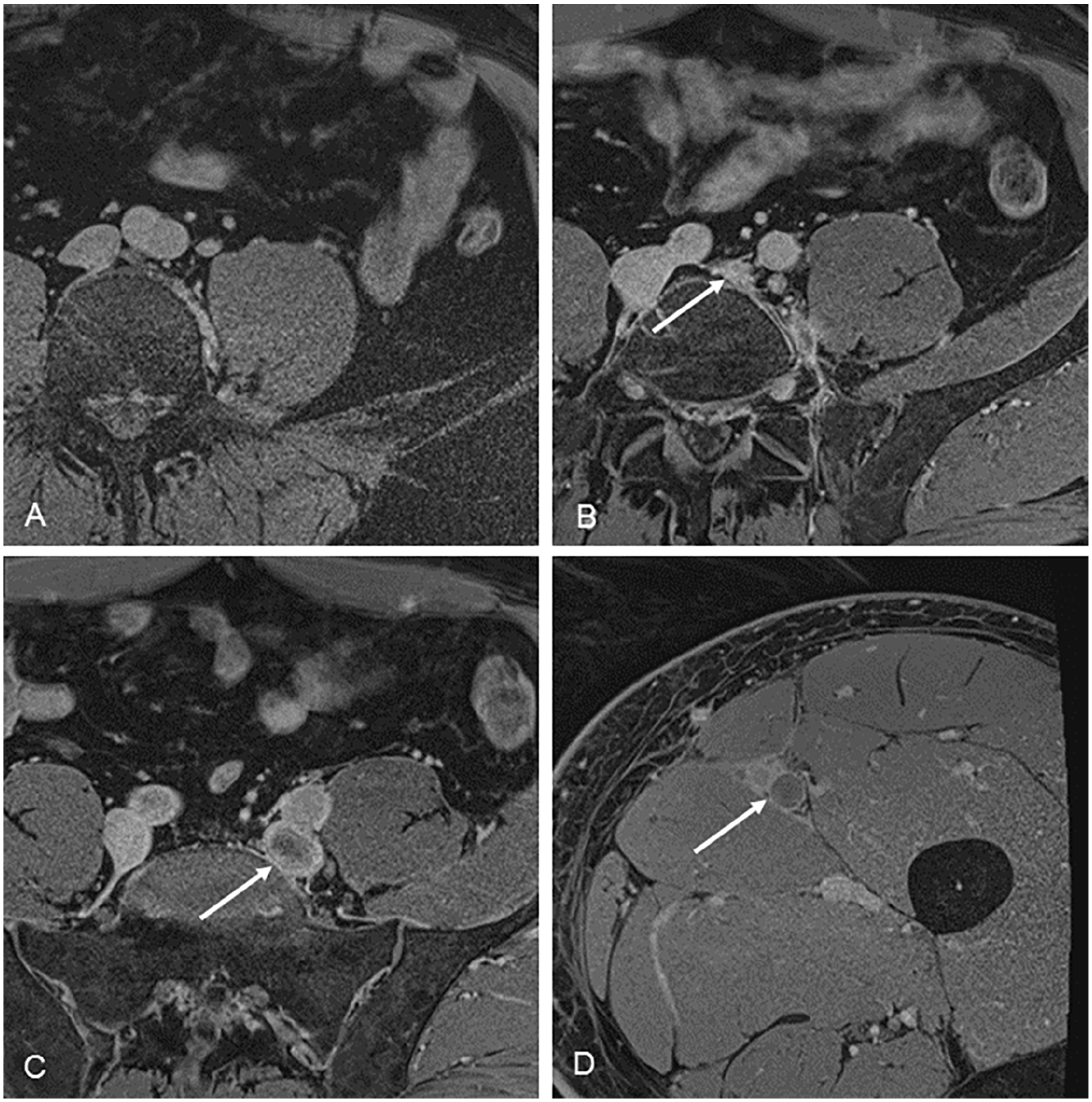
A 58-year-old woman with thrombotic disease of the left common and external iliac vein.
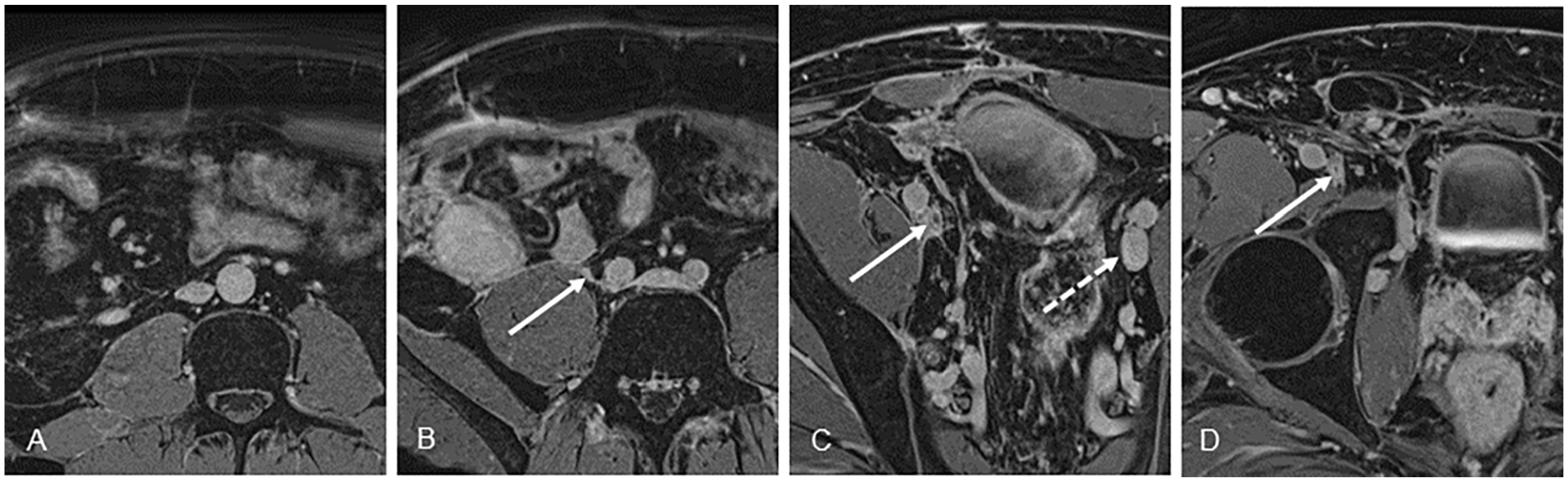
A 44-year-old male with chronic postthrombotic syndrome with occlusive disease of the right common and external iliac veins.
Magnetic resonance direct thrombus imaging (MRDTI)
MRDTI is a novel technique that enables evaluation of the thrombus age, which may have important therapeutic implications. MRDTI is a T1-weighted gradient echo sequence that assesses the changing oxygenation of hemoglobin within the thrombus. The intra-thrombi signal normalized after 6 months, thus providing the basis for differentiating between acute DVT and CPC. 62 Previous studies have shown that MRDTI could accurately diagnose acute DVT above and below the knee with sensitivities and specificities of 87% and 95%, and 97% and 100%, respectively. 63 Similarly, Westerbeek et al. have shown that MRDTI had a sensitivity of 95.3% in identifying acute DVT. 62 In the subsequent study, Tan et al. have demonstrated that MRDTI could accurately differentiate between acute recurrent DVT and CPC with a sensitivity of 95% and a specificity of 100%. The authors have also shown an excellent interobserver agreement (κ = 0.98). 64
Accurate MRDTI assessment of thrombus age carries significant therapeutic implications. In a recent prospective trial, van Dam et al. have evaluated the predictive value of MRDTI in ruling out acute recurrent DVT. A total of 305 patients with suspected recurrent ipsilateral DVT were included in the study. In patients with negative MRDTI findings, no new anticoagulation regimen was initiated, whereas patients with positive MRDTI findings underwent an anticoagulation regimen. Only two patients had inconclusive MRDTI findings secondary to prosthesis and venous stent placement. In patients with negative MRDTI, 1.7% of the patients developed recurrent DVT (95% CI, 0.20% to 5.9%). 65
Intravascular ultrasound evaluation of venous thrombotic disease
IVUS should not be used for acute DVT diagnosis given its invasive nature and high costs. As discussed above, VDUS is likely the most appropriate first-line diagnostic modality for suspected ICVO. However, in patients with significant venous obstructive symptoms and equivalent imaging findings on CTV/MRV/VDUS, or those who have failed conservative management, IVUS was highly sensitive in detecting obstructive lesions (Figure 7). 66 Further, in comparison with other modalities such as CTV and MRV, IVUS has significant advantages given its intravascular nature and ability to assess key parameters and important landmarks. 67 A previous paper has shown that IVUS could assess fine mural details, including venous trabeculations and webs, which may be too small to delineate on CTV or MRV. 68 In comparison to multiplanar venography, a recent study has shown that IVUS was more sensitive in identifying venous stenotic lesions. Further, venography was found to underestimate the degree of stenosis in comparison to IVUS (p < 0.001), which resulted in management changes in 28% of the patients based on IVUS findings. 69 Similarly, Montminy et al. have found that venography failed to identify stenotic lesions in 19% of the assessed limbs in comparison to IVUS. IVUS could better identify key parameters for stenting. For example, the iliocaval junction was on average one vertebral level higher on IVUS compared to venography. Accurate assessment of key landmarks is critical to ensure correct stent sizing and placement. 70
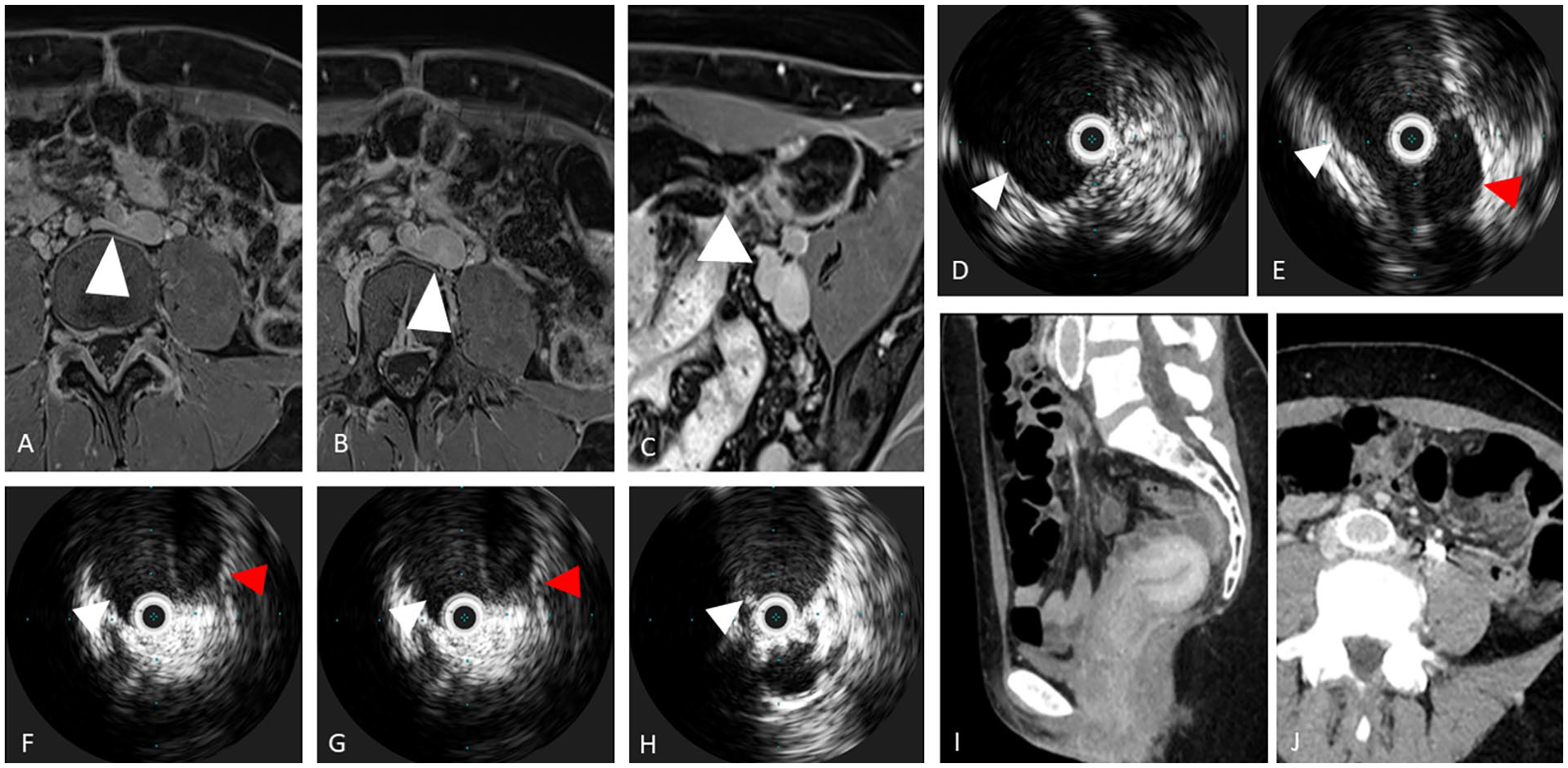
A 46-year-old woman presenting with chronic pelvic pain and left flank pain.
IVUS could be used to assess treatment adequacy and to predict clinical outcome. Previous studies have shown that residual thrombus burden within the iliofemoral segment was associated with worse patient symptomology. 71 At the conclusion of the procedure, IVUS is able to assess for residual thrombus burden and, therefore, subsequent treatment planning. A previous study has shown that IVUS was more sensitive in detecting residual thrombus than venography (p = 0.03). 72 Murphy et al. have found that the residual stenosis was more severe on IVUS than venography (20% vs 10%, p < 0.05). Further, in 36% of the patients, residual thrombotic disease was only visualized on IVUS. 73 With regards to predicting procedure outcome, Gagne et al. examined the relationship between baseline IVUS stenosis and clinical outcomes. The authors have found that greater than 54% stenosis was the optimal threshold for treatment. 74
Most importantly, recent evidence has shown that the use of IVUS is associated with improved patient outcomes. Tran et al. compared IVUS and multiplanar venography to venography alone in the management of thrombotic and nonthrombotic iliofemoral venous disease. The authors have found the additional use of IVUS was associated with a longer total length of the stent and a higher rate of infrainguinal stent extension. Further, the mean diameter of the stent was larger when IVUS was used. Both 30-day and 2-year primary patency rates were significantly higher in the IVUS with venography group (p = 0.02 and 0.03, respectively). 75
Invasive venography of venous thrombotic disease
Invasive venography has been considered the ‘gold standard’ for diagnosing acute DVT. The four cardinal signs of DVT include constant filling defects, abrupt termination of the contrast column, nonfilling of the venous system, and diversion of flow, as described in the original paper by Rabinov and Paulin. 76 However, venography remains technically challenging and in up to 30% of the cases, venography failed to visualize some of the venous segments. 77 In addition, venography is limited secondary to its invasiveness and association with large contrast load, which can be contraindicated in certain patient populations.
With regards to iliac vein stenting for ICVO, venography, and in particular multiplanar venography, remains an important modality. As discussed above, IVUS has been shown to be superior in its ability to delineate landmarks and key parameters for iliac venous stenting. However, venography can offer a more ‘global’ view of the diseased segment, including visualization of collateral flow and its ‘road-mapping’ ability in recanalizing chronically occluded venous segment. Venous stenting is routinely performed with both venography and IVUS with clinical benefits for the patients.
Conclusion
Lower-extremity venous thrombotic disease encompasses both acute and chronic thrombotic disease and can lead to significant morbidity and mortality. In particular, PTS has been increasingly recognized as a major quality of life issue in the recent decade. As endovascular therapy has evolved, there is a need for superior pre- and postoperative assessment of venous thrombotic disease. VDUS remains the first-line diagnostic tool given its ease of access and cost effectiveness. Further, it is a remarkably reliable modality for acute DVT diagnosis. However, its role in assessing chronic or recurrent DVT, and ICVO, is less clear. More advanced imaging modalities, including various sonographic techniques, CTV, MRV, MRDTI, and IVUS, have been shown to provide added utility in certain clinical scenarios. In particular, CEUS, US elastography, and MRDTI may prove useful in cases of diagnostic uncertainties and to provide additional information regarding the thrombus composition/age. CTV and MRV are particularly useful in delineating underlying etiologies in venous outflow obstruction. Lastly, IVUS is critically important in assessing venous stenosis and endovenous pathology, which is important for iliac stenting. Although promising, current studies are including a relatively small and heterogeneous patient population. Future clinical trials are required to further validate the role of various imaging techniques in the management of acute and chronic lower-extremity venous thrombotic disease.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
