Abstract
Background:
The natural history of patients with a pacemaker-related upper-extremity deep vein thrombosis (UEDVT) has not been consistently studied.
Methods:
We used the RIETE registry data to compare the outcomes during anticoagulation and after its discontinuation in noncancer patients with symptomatic UEDVT associated with a pacemaker, other catheters, or no catheter. The major outcome was the composite of symptomatic pulmonary embolism or recurrent DVT.
Results:
As of February 2022, 2578 patients with UEDVT were included: 156 had a pacemaker-related UEDVT, 557 had other catheters, and 1865 had no catheter. During anticoagulation, 61 patients (2.3%) developed recurrent VTE, 38 had major bleeding (1.4%), and 90 died (3.4%). After its discontinuation, 52 patients (4.4%) had recurrent acute venous thromboembolism (VTE) and six had major bleeding (0.5%). On multivariable analysis, there were no differences among subgroups in the rates of VTE recurrences or major bleeding during anticoagulation. After its discontinuation, patients with a pacemaker-related UEDVT had a higher risk for VTE recurrences than those with no catheter (adjusted OR: 4.59; 95% CI: 1.98–10.6).
Conclusions:
Patients with pacemaker-related UEDVT are at increased risk for VTE recurrences after discontinuing anticoagulation. If our findings are validated in adequately designed trials, this may justify changes in the current recommendations on the duration of anticoagulation.
Keywords
Background
Based on extrapolation of randomized trials from lower-extremity deep vein thrombosis (DVT) and small cohort studies, current guidelines from the European Society of Vascular Surgery recommend that in patients with catheter-associated upper-extremity DVT (UEDVT), 1 anticoagulant therapy can be discontinued after 3 months if the catheter is removed. If the catheter is not removed, the guidelines suggest that anticoagulation should be considered for a minimum of 3 months, or continued as long as the catheter remains.
Since the placement of the first permanent transvenous pacemaker in 1965, asymptomatic UEDVT has been frequently associated with this procedure.2–5 The symptomatic form in which swelling and pain prevail in the arm is much less frequent;6,7 when it develops, UEDVT may lead to severe local morbidity and can be a source of pulmonary embolism (PE).8–10
In patients with a pacemaker-related UEDVT, there is scarce information to provide insight into the association between transvenous leads and UEDVT over time to elucidate whether they should be regarded a transient or persistent risk factor. Although anticoagulant treatment is the most widely used, the adequate duration is not known, nor are the complications associated with the treatment, or its suspension. An answer to this question would provide clarity as to whether anticoagulation may be discontinued after the initial 3 months of treatment for a first DVT or extended anticoagulant therapy should be considered.
RIETE (
Methods
We enrolled in RIETE consecutive patients with symptomatic, acute DVT, or PE. DVT were confirmed by compression ultrasonography or contrast venography. PE were confirmed by ventilation perfusion lung scintigraphy, helical computed tomography (CT) scan or angiography. If patients currently participated in a therapeutic clinical trial with a blinded therapy they were excluded. All patients (or their relatives) provided written or oral consent for their participation in the registry, in accordance with local ethics committee requirements.
Study design
Given the high association between cancer and VTE recurrences, we excluded patients with active cancer, or with history of cancer prior to the diagnosis of DVT. The major outcome was the incidence rate of VTE recurrences, defined as the composite of symptomatic (objectively confirmed) PE and/or DVT recurrences (in the ipsilateral arm, contralateral, or in the lower limb). When an episode of clinically suspected recurrent VTE was suspected, the investigation consisted of repeat compression ultrasonography, lung scintigraphy, helical-CT scan or pulmonary angiography, as appropriate. Secondary outcomes were major bleeding and death. Major bleeding was defined as a bleed that required two or more units of blood transfusion, was retroperitoneal, spinal or intracranial, or was fatal. Fatal bleeding was defined as any death occurring within 10 days of a major bleeding episode, in the absence of an alternative cause of death. Fatal PE, in the absence of an autopsy, was defined as any death appearing within 10 days of a PE event, in the absence of any alternative cause of death.
Baseline variables
The following parameters were recorded in RIETE: patient’s characteristics; VTE signs and symptoms at baseline; clinical status including any coexisting or underlying conditions such as hypertension, diabetes, atrial fibrillation, peripheral artery disease, ischemic stroke, chronic heart failure or chronic lung disease, recent (< 30 days before) major bleeding, anemia or renal insufficiency; concomitant disorders; additional risk factors for VTE; blood tests at baseline (including hemoglobin, leukocyte, and platelet count; creatinine clearance levels); the treatment received upon VTE diagnosis (drugs, doses, and duration); concomitant drugs (including corticosteroids, antiplatelets, or statins); and the outcomes. Anemia was defined as hemoglobin levels < 13 g/dL for men and < 12 g/dL for women. The RIETE registry restricted all values of these variables to the nearest recorded to the time of VTE diagnosis. We imputed missing values where necessary.
Treatment and follow up
Patients were managed according to the clinical practice of each participating hospital (i.e., there was no standardization of treatment). The type, dose, and duration of anticoagulant therapy were recorded. The decision to treat patients in hospital or at home was left to the attending physicians. After hospital discharge, patients were followed up in the outpatient clinic (or telephone interviews with patients who could not show up for a clinic visit). During each visit, any signs or symptoms suggesting VTE recurrences or major bleeding were noted.
Statistical analysis
Continuous variables were reported with the mean, SD, median, and IQR, and categorical variables with absolute frequencies and percentages. Differences between groups were assessed by the chi-squared test for categorical variables and a t-test for continuous data. The risk for VTE recurrences appearing during the course of anticoagulant therapy, and after its discontinuation, was separately assessed using logistic regression models. Covariates entering into the model were selected by a significance level of p < 0.10 on univariable analysis or by a well-known association reported in the literature. The doses of anticoagulant drugs were not included because its choice might have been influenced by the physician’s assessment of a patient’s risk of bleeding or recurrent VTE. Adjusted odds ratio (OR) with 95% CIs were estimated. IBM SPSS Statistics, Version 25 (IBM Corp., Armonk, NY, USA) was used for the statistical management of the data, and a two-sided p < 0.05 was considered to be statistically significant.
Results
From March 2001 to March 2022, there were 4505 patients with UEDVT in RIETE. Of these, 1924 (43%) had cancer (active cancer, 1312; history of cancer, 612) and were excluded from this study. Among the remaining 2578 patients, 156 (5.9%) had a pacemaker-related UEDVT, 557 (22%) had UEDVT related to other catheters (central venous catheters, 404; peripheral, 153), and 1865 (72%) had UEDVT with no catheter. Overall, 256 patients presented with concomitant UEDVT and PE (5.1%, 8.1%, and 11%, respectively).
Patients with a pacemaker-related UEDVT were more likely to be men, much older, and more likely to have hypertension, atrial fibrillation, chronic heart failure, prior myocardial infarction, or to be using antiplatelet drugs or statins at baseline than those with UEDVT secondary to other catheters, or those with no catheter. Women accounted for 1193 (46.2%) patients. The proportion of women in each subgroup was: 30% of patients with VTE associated with a pacemaker, 43% of those with VTE associated to other catheters, and 49% with spontaneous (noncatheter-related) UEDVT (Table 1).
Baseline characteristics of patients with upper-extremity deep vein thrombosis, according to the study subgroups.
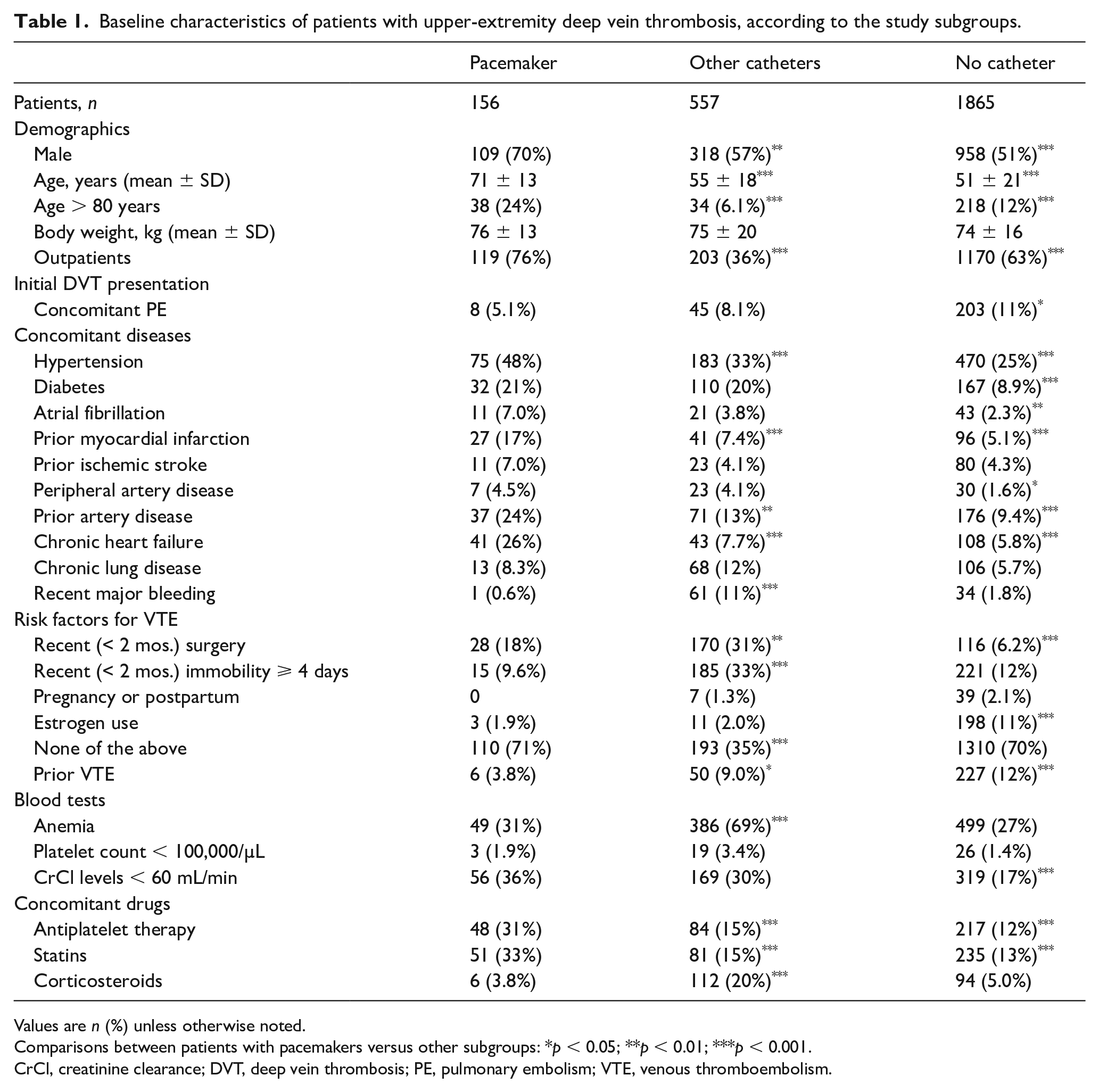
Values are n (%) unless otherwise noted.
Comparisons between patients with pacemakers versus other subgroups: *p < 0.05; **p < 0.01; ***p < 0.001.
CrCl, creatinine clearance; DVT, deep vein thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism.
Among 1492 patients diagnosed with UEDVT as outpatients, 839 (56%) required hospitalization to treat the DVT (50%, 64%, and 56%, respectively), as shown in Table 2. Most patients in all three subgroups (90%, 83%, and 82%, respectively) received initial therapy with low-molecular-weight heparin (LMWH). Then, 54% of patients with a pacemaker-related UEDVT switched to vitamin K antagonists, 32% kept receiving LMWH, and 12% switched to direct oral anticoagulants. The duration of anticoagulation was longer in patients with a pacemaker-related UEDVT (median, 148 days) than in those in the other two subgroups (median, 102 and 130 days, respectively). The duration of follow up after discontinuing anticoagulation was also longer in patients with a pacemaker-related UEDVT than in those in the other two subgroups (median, 445 vs 295 and 178 days, respectively).
Treatment strategies for upper extremity deep vein thrombosis.
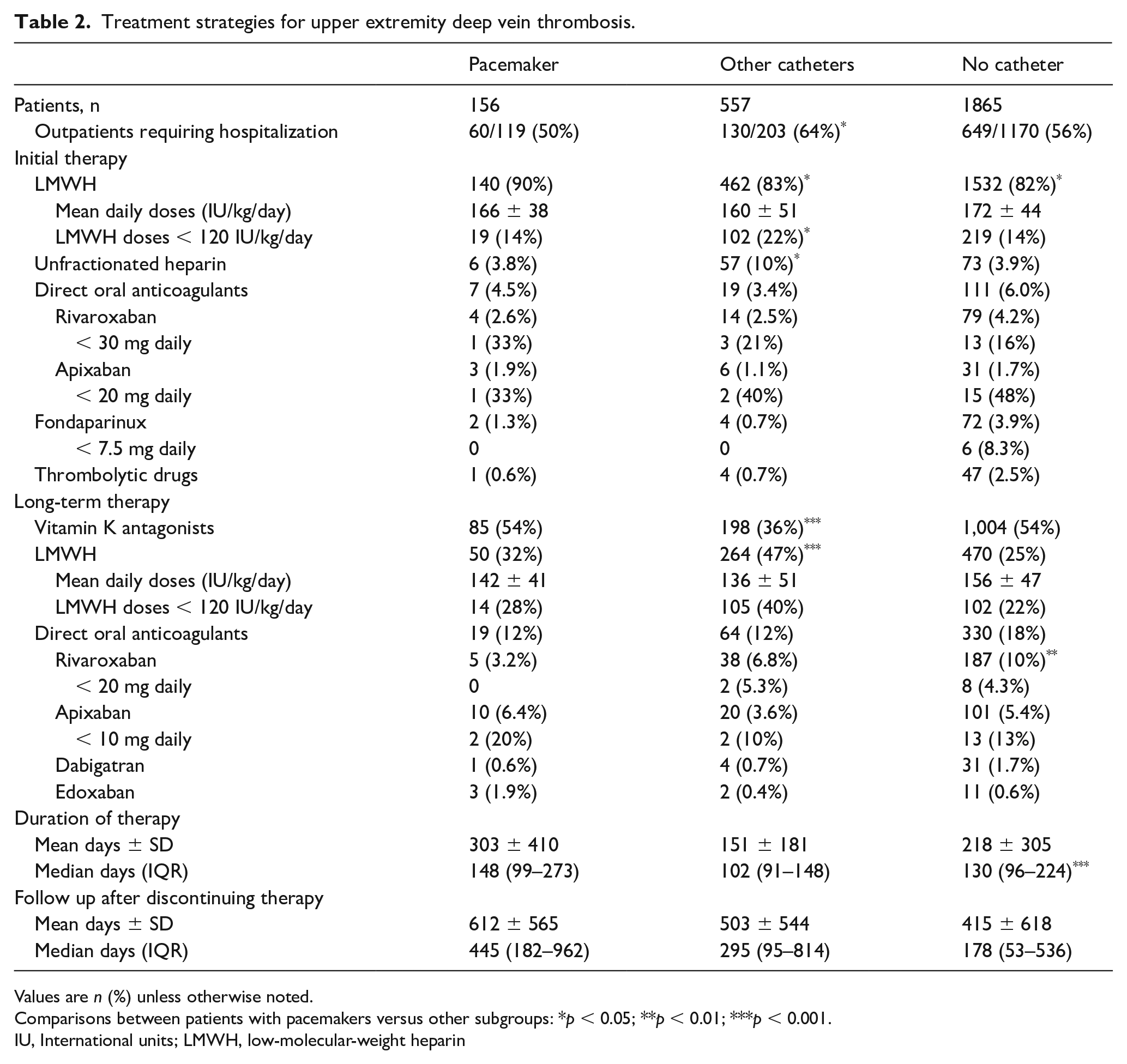
Values are n (%) unless otherwise noted.
Comparisons between patients with pacemakers versus other subgroups: *p < 0.05; **p < 0.01; ***p < 0.001.
IU, International units; LMWH, low-molecular-weight heparin
During the course of anticoagulation, 18 patients (0.7%) developed symptomatic PE, 43 (1.6%) had DVT recurrences (ipsilateral UEDVT, 27; contralateral, 11; lower-limb DVT, 5), 38 (1.4%) suffered major bleeding (gastrointestinal, 13; intracranial, 4), and 90 (3.4%) died (fatal PE, 6; fatal bleeding, 4). Among the six patients who died of PE, two died of the index PE and four died of recurrent PE. During anticoagulation, no patient with a pacemaker-related UEDVT developed symptomatic PE, and the rate of DVT recurrences in patients with a pacemaker-related DVT was close to the rate of DVT recurrences in the other two subgroups (Table 3). All VTE recurrences in patients with a pacemaker-related UEDVT appeared beyond the third month of anticoagulant therapy. At variance with this, all VTE recurrences in patients with UEDVT related to other catheters appeared within the first 3 months (online Supplementary Figure 1). The rate of major bleeding in patients with a pacemaker-related UEDVT was similar to the rate of major bleeding in patients with no catheter, and much lower than in those with UEDVT related to other catheters.
Clinical outcomes during anticoagulation and after its discontinuation.
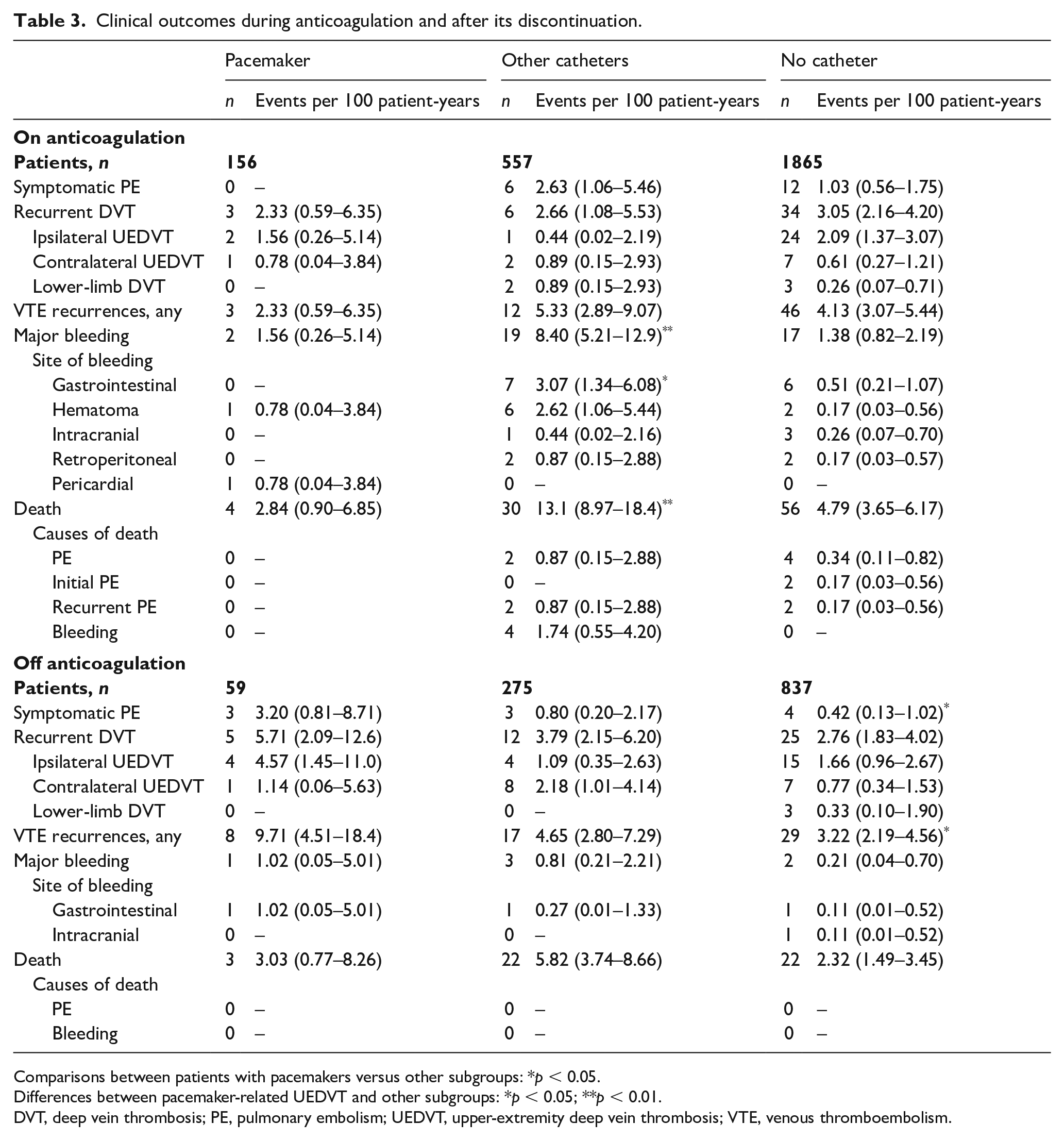
Comparisons between patients with pacemakers versus other subgroups: *p < 0.05.
Differences between pacemaker-related UEDVT and other subgroups: *p < 0.05; **p < 0.01.
DVT, deep vein thrombosis; PE, pulmonary embolism; UEDVT, upper-extremity deep vein thrombosis; VTE, venous thromboembolism.
After discontinuing anticoagulant therapy, 10 patients (0.8%) developed symptomatic PE, 42 (3.6%) had DVT recurrences (ipsilateral UEDVT, 23; contralateral, 16; lower-limb DVT, 3), six (0.5%) suffered major bleeding (gastrointestinal, 3; intracranial, 1), and 47 (4.0%) died (no fatal PE nor fatal bleeding). Among patients with a pacemaker-related UEDVT, all VTE recurrences (either symptomatic PE or DVT recurrences) appeared within the first 90 days after discontinuing anticoagulation, half of them (three of six) within the first 30 days (online Supplementary Figure 2). On the other side, less than half of the VTE recurrences in patients with UEDVT related to other catheters (four of 10) or no catheter (nine of 19) appeared within the first 3 months. The rate of symptomatic PE was significantly higher in patients with a pacemaker-related DVT than in those with no catheters, and nonsignificantly higher than in those with other catheters (Table 3).
Variables entering in the multivariable analyses were: age, sex, body weight, inpatient condition, chronic lung disease or heart failure, hypertension, diabetes, recent major bleeding, additional risk factors for VTE, initial VTE presentation (isolated UEDVT or with concomitant PE), prior VTE, anemia, platelet count, creatinine clearance levels at baseline, and type of catheter (pacemaker, other catheters, or no catheters). On multivariable analysis, there were no differences among subgroups in the risk for VTE recurrences (considering together symptomatic PE and DVT recurrences) during the course of anticoagulation, but patients with a pacemaker-related UEDVT (adjusted OR: 4.59; 95% CI: 1.98–10.6) or UEDVT secondary to other catheters (adjusted OR: 2.60; 95% CI: 1.35–4.98) were at increased risk for VTE recurrences after its discontinuation (Table 4).
Uni- and multivariable analyses for VTE recurrences during and after discontinuing anticoagulant therapy.
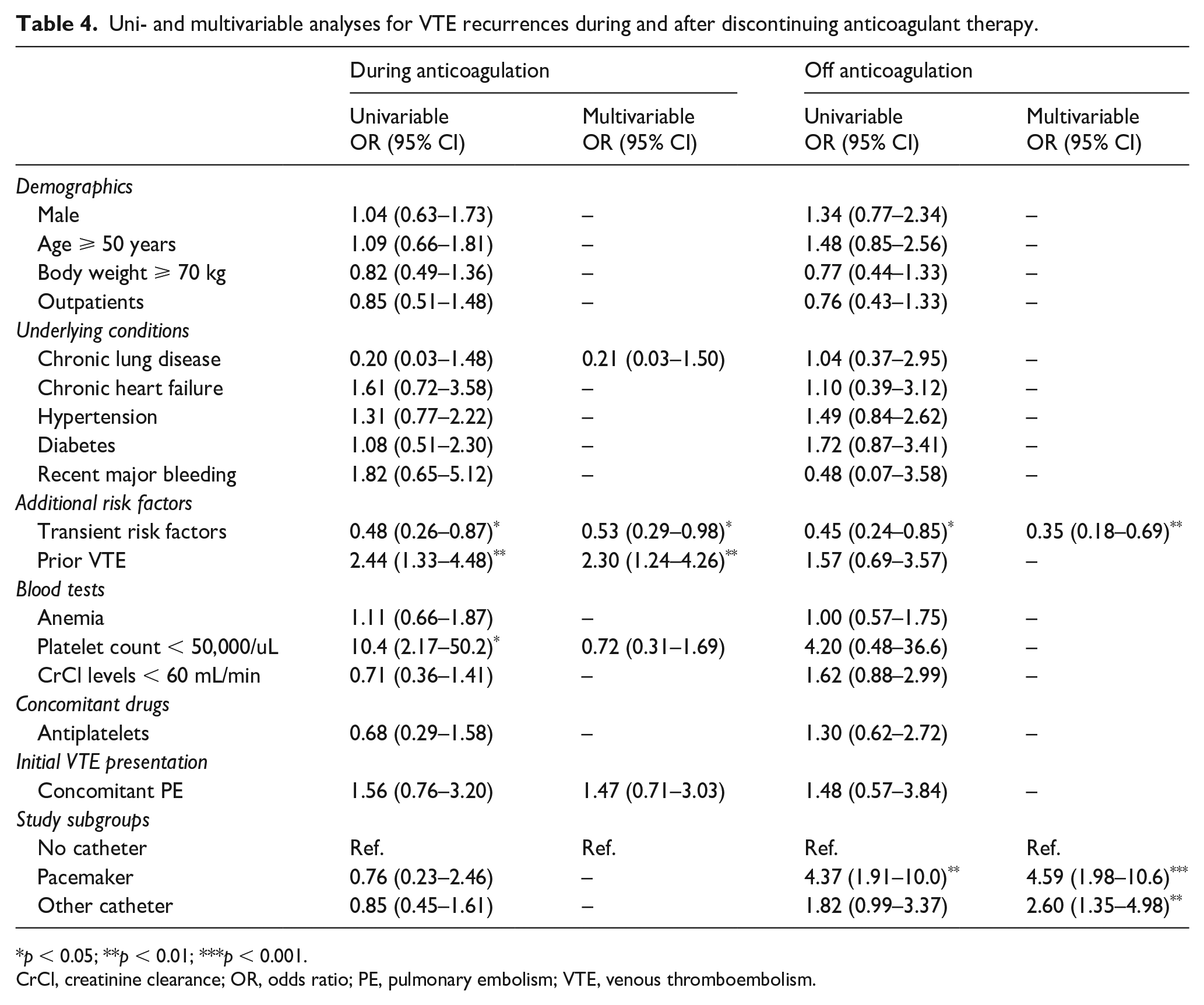
p < 0.05; **p < 0.01; ***p < 0.001.
CrCl, creatinine clearance; OR, odds ratio; PE, pulmonary embolism; VTE, venous thromboembolism.
Discussion
Previous studies reported that symptomatic UEDVT occurred in 0.3–0.5% of the patients, mainly within the first month after pacemaker implantation,6,7 though may occur up to multiple years after. Recently, Duijzer et al.’s review and meta-analysis collected 72 patients with symptomatic UEDVT related to a pacemaker over 34 years (from 1985 to 2019). The authors requested ‘further research to elucidate whether trans-venous leads should be considered a transient or persistent risk factor’. 10
In our cohort of 156 patients with a symptomatic pacemaker-related UEDVT, over 21 years, there was a twofold higher incidence of VTE recurrences (the composite of symptomatic PE or DVT recurrences) after discontinuing anticoagulation that persisted after adjusting for potentially confounding factors. Compared to the other two subgroups of patients, the incidence rate of VTE recurrences in patients with a pacemaker-related UEDVT was higher and appeared early after discontinuing anticoagulant therapy, thus suggesting they could likely benefit from prolonging the duration of anticoagulation. Certainly, patients with a pacemaker-related UEDVT were older, more likely to be using antiplatelets concomitantly or to have renal insufficiency, and thus might have been considered to also be at high risk for bleeding during anticoagulation. However, their incidence rate of major bleeding was similar to that in the other two subgroups.
During the course of anticoagulation, no patient with a pacemaker-related UEDVT in our cohort developed symptomatic PE, as compared to six patients with UEDVT secondary to other catheters and 12 with noncatheter-related UEDVT. Of note, six of these 18 patients (33%) died of the PE. Thus, the clinical relevance of PE in patients with UEDVT should not be underestimated. Interestingly, no patient with a pacemaker-related UEDVT developed DVT recurrences during the first 3 months of therapy, the time when most recurrences had appeared in the other two subgroups of patients. However, the incidence rate of recurrent DVT in patients with a pacemaker-related UEDVT increased quickly beyond the third month of therapy. The explanation for the previously mentioned data is that pacemakers, with few exceptions, will never be removed, and therefore their responsibility as a pathogenic factor in venous thrombosis will persist over time. It would be reasonable for these patients with symptomatic pacemaker-related UEDVT to be included in the group with venous thromboembolic disease related to a permanent risk factor. Therefore, the extension of anticoagulant treatment could be considered if there is no high risk of bleeding. The results of our study show that prolonging anticoagulant treatment in this group of patients is not associated with a higher risk of bleeding.
During anticoagulation, overall rates of recurrence (2.4%), major bleeding (1.4%) and mortality (3.4%) are lower than those previously documented in case series and reviews.13–21 The referenced publications include patients with cancer, a condition that justifies higher percentages of recurrence (between 5.1% and 7.5%), major bleeding (between 3.1% and 6.7%), and mortality in the first months (between 11% and 27%).
Study limitations
Our study has a number of limitations that should be emphasized. First, the sample size of patients with a pacemaker-related UEDVT in our cohort is rather small. And the amount of those who were followed-up after discontinuing anticoagulant therapy was even smaller. It is therefore dangerous to draw conclusions from such a sample size. However, ours is the largest cohort of patients with a pacemaker-related UEDVT published thus far. Second, unfortunately, in RIETE, we gathered no information on the number of leads (i.e., a dual or single-lead pacemaker) to assess its influence on the development of symptomatic pacemaker-related UEDVT. Unlike catheters, which offer the option of removal, device therapies are often permanent and removal introduces additional risk for vascular injury, and the need for device therapy either subcutaneously or intracardiac if possible. Third, ours is an observational study where the therapeutic decisions (and the decision to discontinue or not) were left to the criteria of the attending physicians. Thus, our findings may be only considered as hypothesis-generating. However, to our knowledge, there are no randomized controlled clinical trials comparing different therapeutic options in patients with a pacemaker-related UEDVT.
Conclusion
In summary, patients with a pacemaker-related UEDVT are at increased risk for VTE recurrences after discontinuing anticoagulation. If our findings were validated in adequately designed trials, this may justify changes in the current recommendations on the duration of anticoagulant therapy.
Supplemental Material
sj-docx-2-vmj-10.1177_1358863X231175185 – Supplemental material for Risk for recurrence of symptomatic upper-extremity deep vein thrombosis in patients without cancer: Analysis of three RIETE cohorts
Supplemental material, sj-docx-2-vmj-10.1177_1358863X231175185 for Risk for recurrence of symptomatic upper-extremity deep vein thrombosis in patients without cancer: Analysis of three RIETE cohorts by Manuel J Núñez Fernández, Emilio M Padín Paz, Nuria Vázquez Temprano, José A Nieto Rodríguez, Pablo J Marchena Yglesias, Egidio Imbalzano, Ana Cristina Montenegro, Begoña Fernández Jiménez, Alberto Rivera, Olivier Espitia and Manuel Monreal in Vascular Medicine
Supplemental Material
sj-docx-3-vmj-10.1177_1358863X231175185 – Supplemental material for Risk for recurrence of symptomatic upper-extremity deep vein thrombosis in patients without cancer: Analysis of three RIETE cohorts
Supplemental material, sj-docx-3-vmj-10.1177_1358863X231175185 for Risk for recurrence of symptomatic upper-extremity deep vein thrombosis in patients without cancer: Analysis of three RIETE cohorts by Manuel J Núñez Fernández, Emilio M Padín Paz, Nuria Vázquez Temprano, José A Nieto Rodríguez, Pablo J Marchena Yglesias, Egidio Imbalzano, Ana Cristina Montenegro, Begoña Fernández Jiménez, Alberto Rivera, Olivier Espitia and Manuel Monreal in Vascular Medicine
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231175185 – Supplemental material for Risk for recurrence of symptomatic upper-extremity deep vein thrombosis in patients without cancer: Analysis of three RIETE cohorts
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231175185 for Risk for recurrence of symptomatic upper-extremity deep vein thrombosis in patients without cancer: Analysis of three RIETE cohorts by Manuel J Núñez Fernández, Emilio M Padín Paz, Nuria Vázquez Temprano, José A Nieto Rodríguez, Pablo J Marchena Yglesias, Egidio Imbalzano, Ana Cristina Montenegro, Begoña Fernández Jiménez, Alberto Rivera, Olivier Espitia and Manuel Monreal in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
