Abstract
Gastric cancer is the fifth most common malignancy worldwide. Venous thromboembolism is an independent predictor of death among patients with gastric cancer. We aimed to describe the factors associated with mortality, thrombosis recurrence, and bleeding complications in patients with gastric cancer who develop venous thromboembolism. We included 612 patients with gastric cancer and venous thromboembolism in the Registro Informatizado de la Enfermedad TromboEmbólica (RIETE) registry from 2001 to 2018. We used Cox proportional hazard ratios and a Fine–Gray model to define factors associated with outcomes. The overall mortality at 6 months was 44.4%. Factors associated with increased 6-month mortality included immobility (HR 1.8, 95% CI 1.3–2.4;
Introduction
The relationship between cancer and venous thromboembolism (VTE) was well described by Armand Trousseau, who paradoxically died with his eponymous syndrome when he developed gastric cancer. 1 In fact, 20% of all VTEs are related to a malignancy. 2 Patients with cancer who develop VTE have decreased survival compared to those without VTE. 3 Gastric cancer has been identified as one of the most prothrombotic tumor types and after accounting for prevalence, it may be responsible for a significant burden of cancer-associated thrombosis (CAT). 4 VTE occurs in approximately 13% of patients with gastric cancer and is an independent predictor of death among those with gastric cancer.5–7 Patients with gastric cancer face the highest, disability-adjusted life years lost and the occurrence of VTE in these patients carries serious prognostic implications.8–10 Given that gastric cancer is the fifth most common type of cancer worldwide, VTE in gastric cancer represents a serious epidemiological problem. 11
The management of cancer-associated VTE is challenging because the risks of recurrence and bleeding are significantly higher in patients with cancer compared to patients without cancer.12,13 These competing risks are particularly high in patients with gastric cancer when compared to other solid tumors. 6 Gastric cancer confers an increased risk of gastrointestinal bleeding, and when bleeding occurs, 30-day mortality is greater than 40%.14,15 Unfortunately, VTE occurrence, recurrence or bleeding cannot be accurately assessed with current assessment models. Hence, these patients have been largely underrepresented and excluded from major primary thromboprophylaxis and treatment trials.16,17
To better assess the outcomes of VTE in patients with gastric cancer, we utilized the Registro Informatizado de la Enfermedad TromboEmbólica (RIETE) database. With over 80,000 patients with VTE, this prospective database is the largest thrombosis database worldwide, including over 12,000 patients with CAT.18,19 Comparatively, RIETE has captured more than three times the sum of patients with cancer-associated thrombosis than all therapy randomized controlled trials on cancer thrombosis to date.17,20
The purpose of this study is to determine the risk of mortality in patients with gastric cancer who develop VTE using the RIETE database, as well as identify risk factors contributing to VTE recurrence and bleeding events in gastric cancer thrombosis.
Methods
Patient population
This study is an analysis of prospectively collected data in the RIETE database from 209 hospitals in Europe, America, and Asia (see Appendix in supplemental material). RIETE includes consecutive patients with deep vein thrombosis (DVT) or pulmonary embolism (PE) confirmed by objective tests (compression ultrasonography or contrast venography for suspected DVT; pulmonary angiography, ventilation-perfusion lung scan or helical computed tomography scan for suspected PE) since March 2001. Its design, rationale, and methodology have been reported elsewhere. 21 Informed consent was obtained from all participants in accordance with local ethics committee requirements, such as Institutional Review Boards (IRBs). Patients were managed according to the clinical practice of each participating hospital (i.e. there was no standardization of treatment). After VTE diagnosis, all patient outcomes were followed up for at least 3 months.
Patients with biopsy-proven gastric cancer who were receiving treatment or had metastatic disease at VTE diagnosis were included in the study. We excluded patients who received the diagnosis of gastric cancer after the acute VTE episode because they were at risk of not receiving the standard CAT therapy and potentially biased the outcomes of this study.22–24 Exclusion criteria also included current enrollment in a therapeutic clinical trial with a blinded therapy.
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions. Informed consent to be included in the study, or the equivalent, was obtained from all patients.
Measured variables
Characteristics of the index VTE episode and of patients (demographics, comorbidities, additional risk factors for VTE, concomitant medications, initial and long-term therapy, and outcomes during anticoagulation) were recorded. We abstracted other provoking factors such as surgery, prolonged immobilization, trauma, and pregnancy. VTE was considered to be secondary to surgery if occurring within 2 months of the procedure and secondary to immobilization if within 2 months of confinement to bed with bathroom privileges for ⩾ 4 days. An increased shock index (SI) (heart rate/systolic blood pressure) is a predictor of in-hospital mortality in patients with acute PE. 25 We defined anemia as hemoglobin < 10 g/dL and leukocytosis as a white blood cell count > 11 × 109/L. These cut-offs were adopted given prior literature specific to cancer-associated thrombosis.26,27 Taller height has been associated with CAT outcomes and was defined as the upper quartile of height per sex. 28 Other quantitative variables such as platelet count, time from cancer diagnosis to VTE, and glomerular filtration rate were categorized according to quartiles or by pre-existing criteria as available.
Outcomes
Outcomes studied were all-cause mortality, VTE recurrence, and bleeding. Bleeding complications were classified as ‘major’ if they: (1) were overt and required a transfusion of two units of blood or more; (2) were retroperitoneal, spinal or intracranial; and (3) were fatal. We also tested the performance of VTE-BLEED, a six-variable bleeding prediction score for anticoagulated patients with VTE.29,30 This score has been externally validated in a post-hoc analysis of the randomized, double-blinded, double-dummy Hokusai-VTE study comparing edoxaban versus warfarin for the treatment of VTE. 30 The variables include active cancer, uncontrolled hypertension, anemia, history of any major or non-major bleeding, age ⩾ 60 years, and renal dysfunction.
Time zero was the date of diagnosis of VTE, and patients were censored at 6 months, at the time of death or at the last date for which outcome data were available.
Statistical analysis
Mortality analysis was done using Cox proportional hazard ratios (HR) and corresponding 95% CIs. Quantitative variables with multiple categories were collapsed to dichotomous according to their strength of association with the outcome in the univariate analysis. We used backward modeling in the multivariate analysis for factors associated with death. To account for the competing risk of very high mortality in gastric cancer thrombosis, we applied the Fine–Gray model for VTE recurrence and bleeding outcomes.31,32 Covariates were included in the multivariate modeling if the univariate
Statistical analyses using Cox proportional HRs were conducted with SPSS for Windows, version 20 (IBM Corp., Armonk, NY, USA). A
Results
We included 612 patients with gastric cancer and acute VTE (Table 1). These patients were mostly male (
Patients with gastric cancer and VTE.
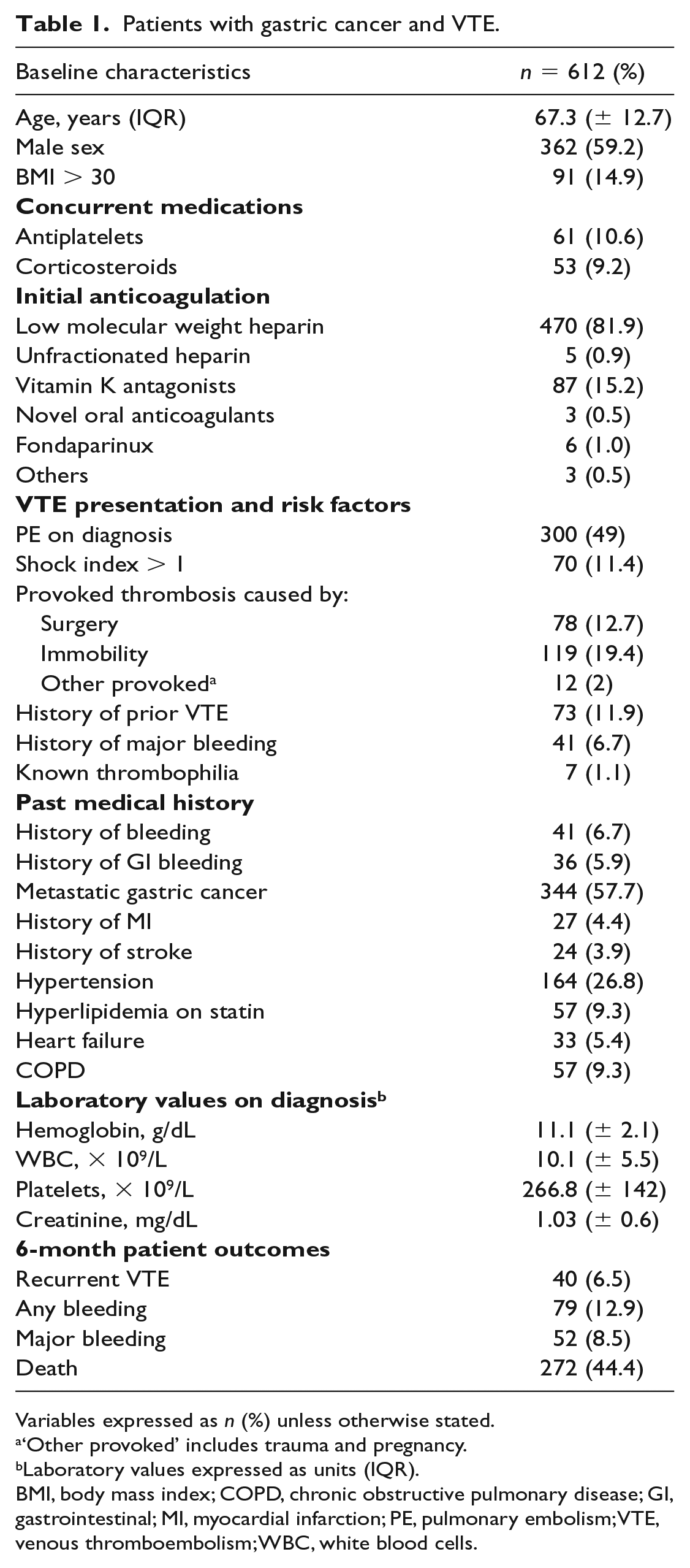
Variables expressed as
‘Other provoked’ includes trauma and pregnancy.
Laboratory values expressed as units (IQR).
BMI, body mass index; COPD, chronic obstructive pulmonary disease; GI, gastrointestinal; MI, myocardial infarction; PE, pulmonary embolism; VTE, venous thromboembolism; WBC, white blood cells.
Mortality
The overall mortality at 6 months was 44.4% (
In the univariate analysis (Table 2), we measured an increased likelihood of mortality in patients with SI > 1 (HR 1.7, 95% CI 1.2–2.3), metastatic disease (HR 4.2, 95% CI 3.2–5.5), and prior VTE (HR 1.3, 95% CI 0.9–1.8). While PE on presentation was not categorically associated with mortality, statin treatment was modestly related to survival (OR 0.6, 95% CI 0.3–1.02;
Univariate analysis for risk of mortality.
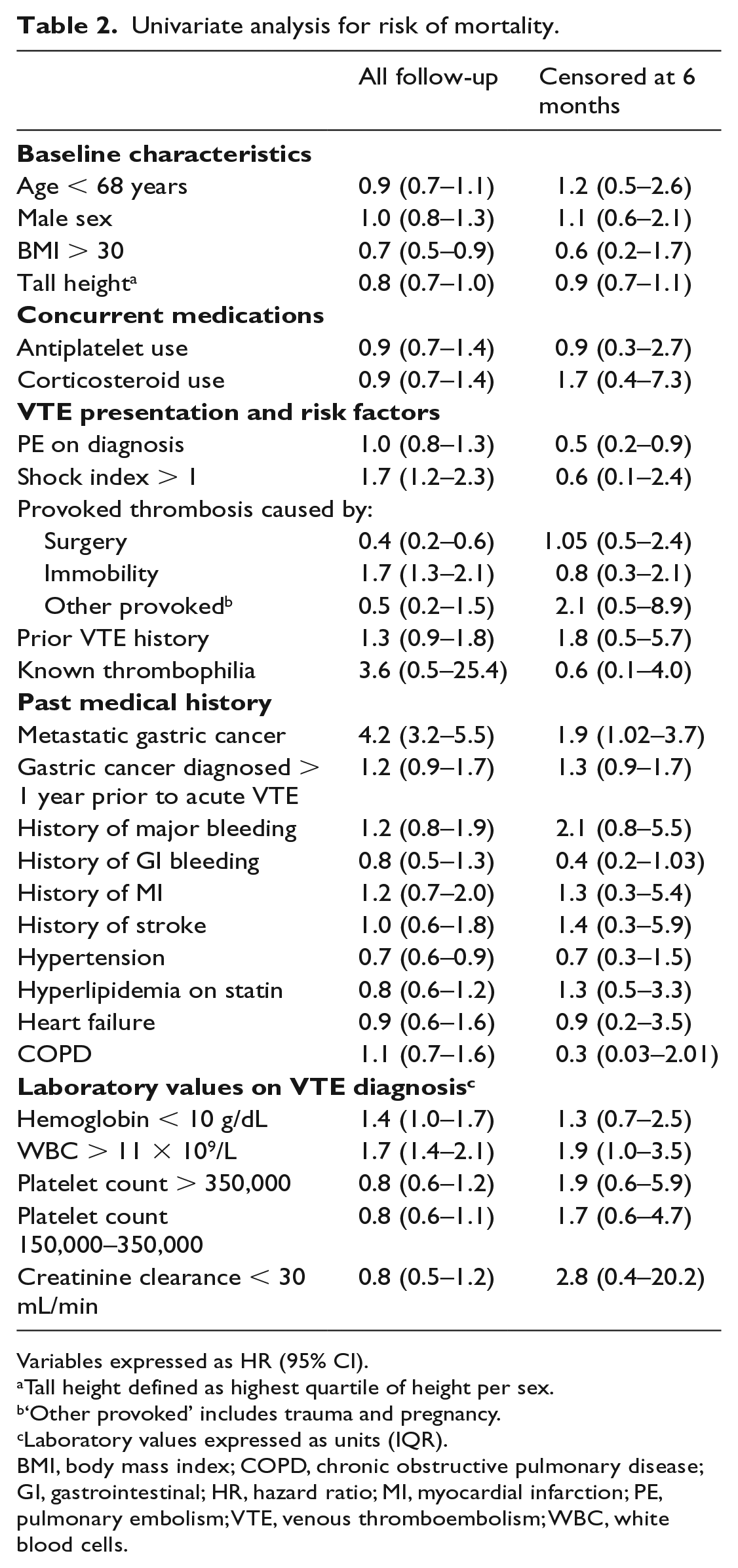
Variables expressed as HR (95% CI).
Tall height defined as highest quartile of height per sex.
‘Other provoked’ includes trauma and pregnancy.
Laboratory values expressed as units (IQR).
BMI, body mass index; COPD, chronic obstructive pulmonary disease; GI, gastrointestinal; HR, hazard ratio; MI, myocardial infarction; PE, pulmonary embolism; VTE, venous thromboembolism; WBC, white blood cells.
Multivariate analyses for VTE recurrence, bleeding, and mortality.
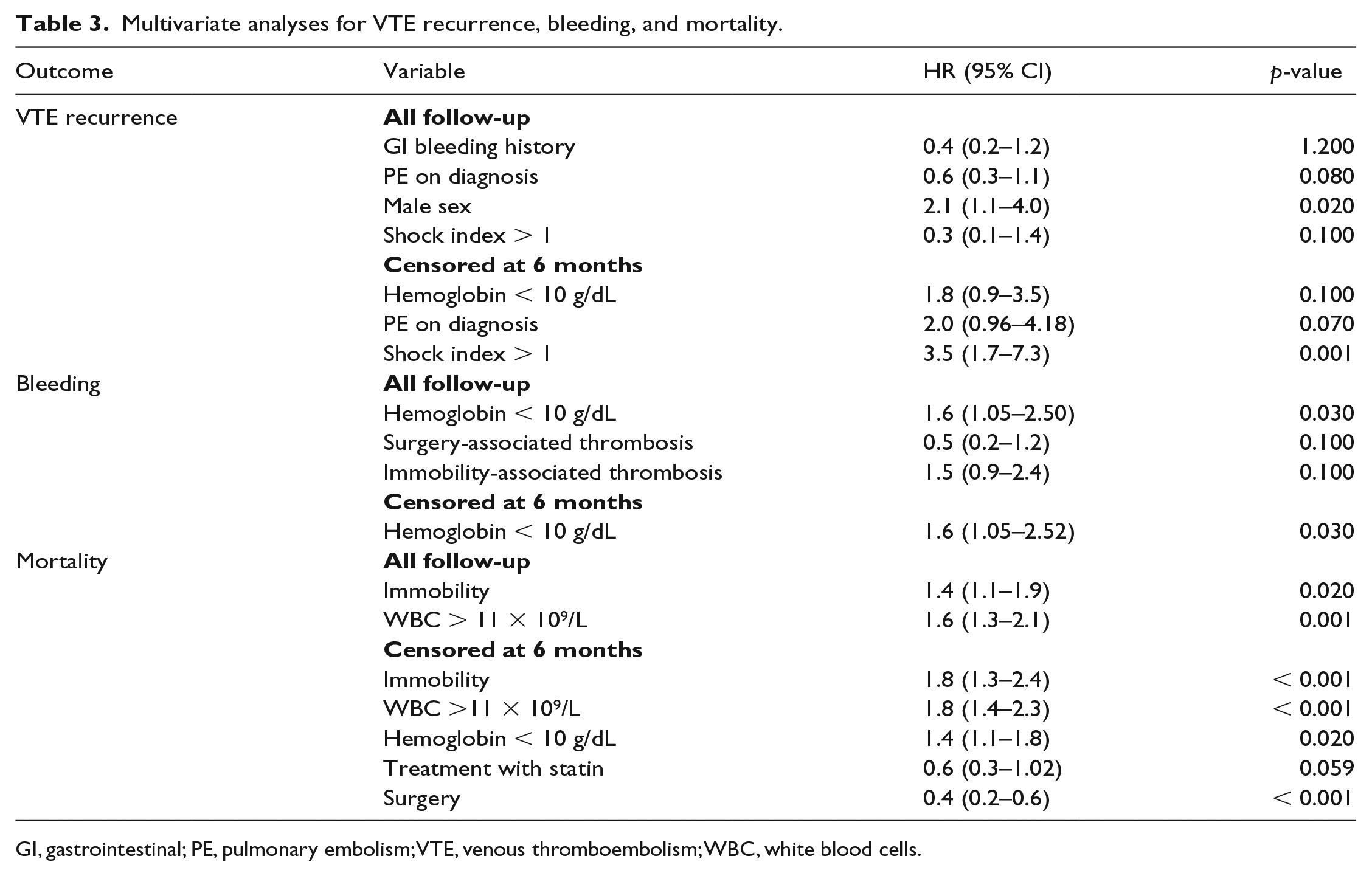
GI, gastrointestinal; PE, pulmonary embolism; VTE, venous thromboembolism; WBC, white blood cells.
VTE recurrence
Recurrent thrombosis occurred in 6.5% (
Recurrent VTE with death as competing risk using Fine–Gray modeling: Univariate analysis censored at 6 months.
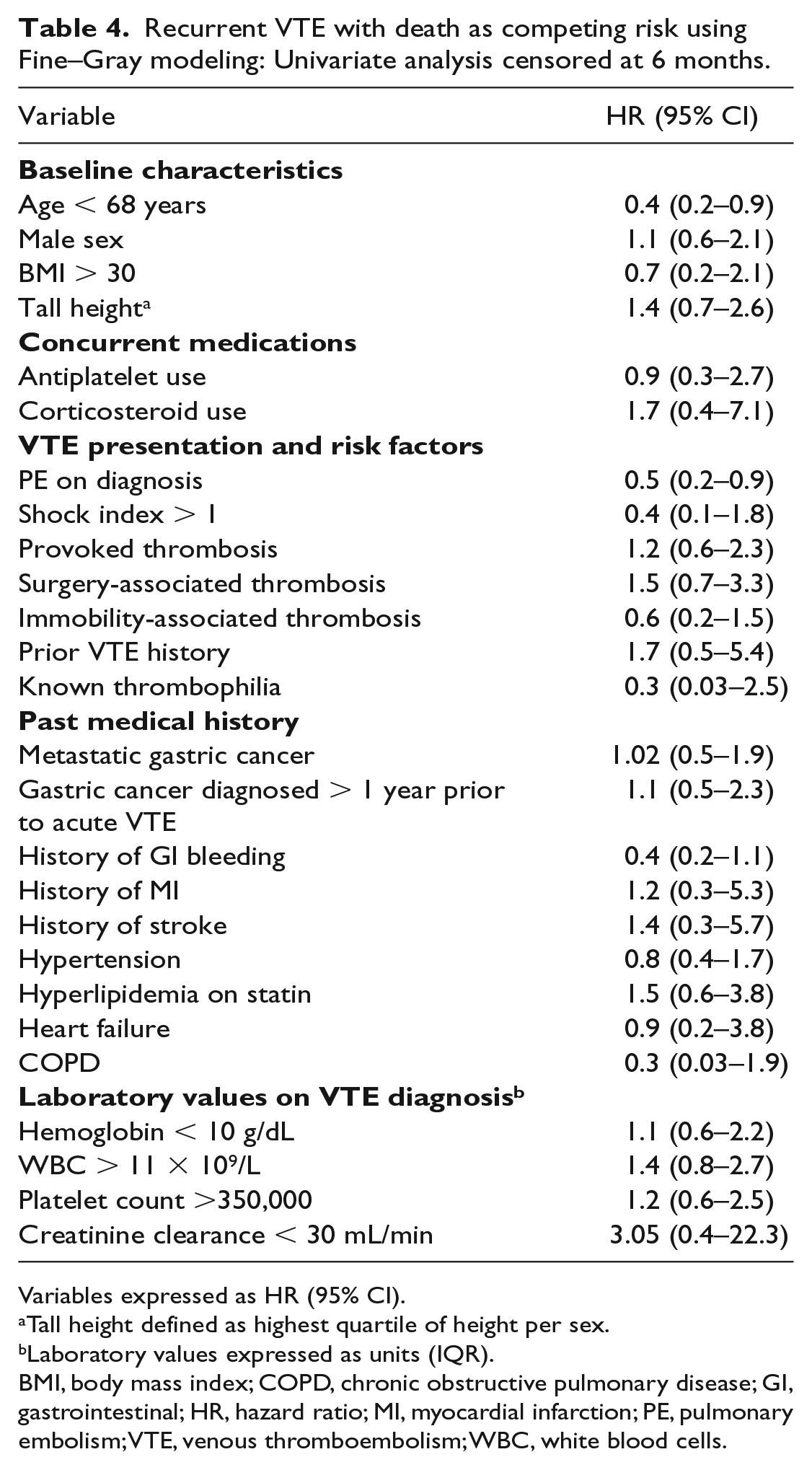
Variables expressed as HR (95% CI).
Tall height defined as highest quartile of height per sex.
Laboratory values expressed as units (IQR).
BMI, body mass index; COPD, chronic obstructive pulmonary disease; GI, gastrointestinal; HR, hazard ratio; MI, myocardial infarction; PE, pulmonary embolism; VTE, venous thromboembolism; WBC, white blood cells.
Bleeding
Bleeding occurred in 12.9% (
Bleeding with death as competing risk using Fine–Gray modeling: Univariate analysis censored at 6 months.
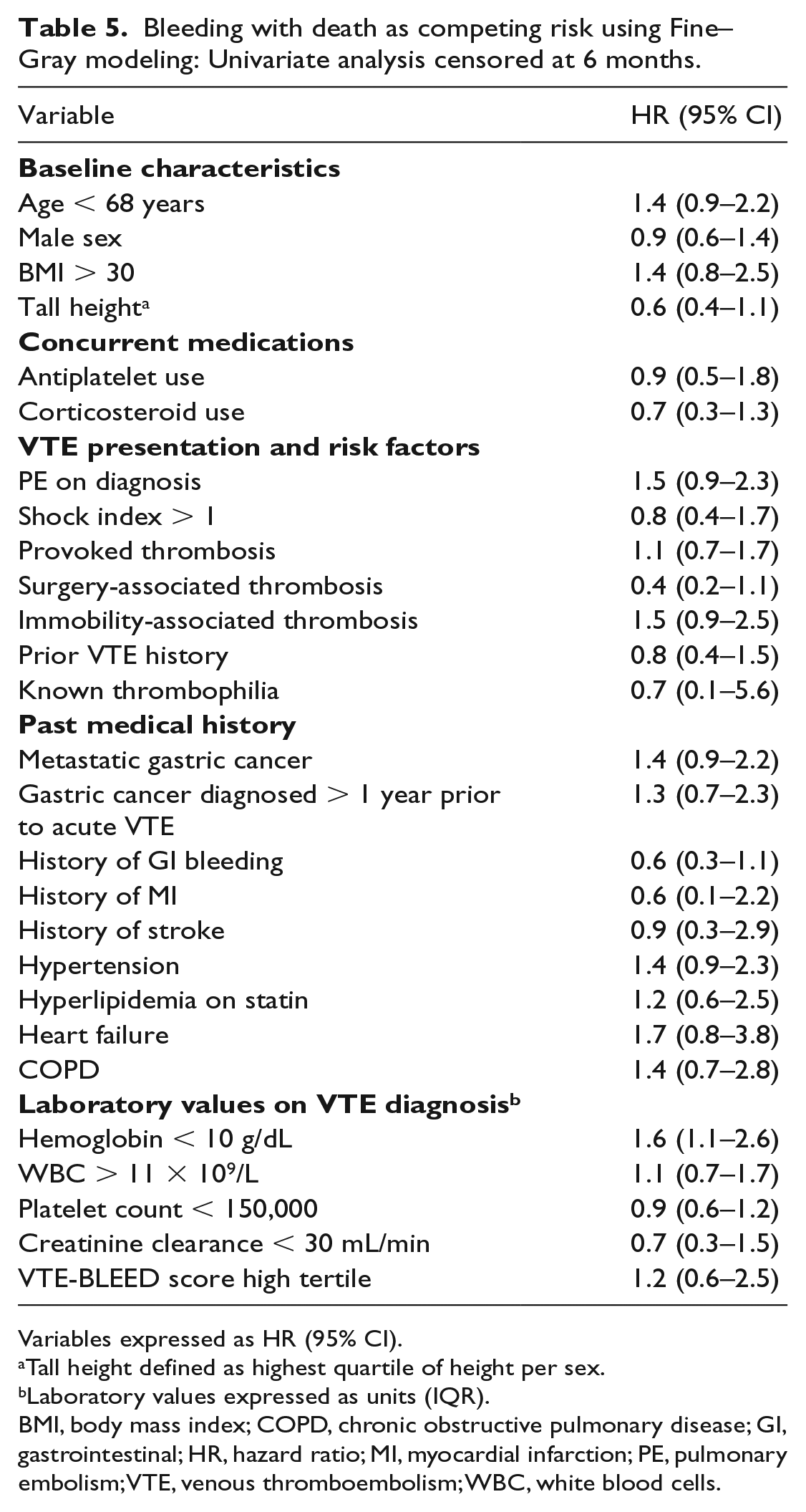
Variables expressed as HR (95% CI).
Tall height defined as highest quartile of height per sex.
Laboratory values expressed as units (IQR).
BMI, body mass index; COPD, chronic obstructive pulmonary disease; GI, gastrointestinal; HR, hazard ratio; MI, myocardial infarction; PE, pulmonary embolism; VTE, venous thromboembolism; WBC, white blood cells.
Discussion
Our results demonstrated a very high rate of mortality in patients with gastric cancer and VTE. In our cohort, 44.4% died within 6 months of thrombosis. This sobering mortality rate is not unexpected: in 13,840 patients with gastric cancer in the Surveillance, Epidemiology, and End Results (SEER) registry, the median overall survival was only 4 months for those with metastatic disease. 39 Indeed, the majority of patients in our cohort had metastatic disease at VTE diagnosis. In addition, our findings are in line with the evidence of cancer-associated thrombosis as an independent factor associated with increased risk of death in gastric cancer. Patients with gastric cancer who develop VTE are nearly three times more likely to die than those who do not develop cancer-associated thrombosis. 6 Notably, most of the patients who died had metastatic disease when the VTE occurred; thus, cancer-associated thrombosis seems to be a late event in gastric cancer. In contrast, a third of the patients had either surgery or immobility preceding the VTE. This begets improved targeted primary thromboprophylaxis; in the randomized, controlled, PROTECTOR trial, the use of prophylactic LMWH postoperatively in 682 patients with gastric cancer undergoing gastrectomy reduced the rates of postoperative VTE from 3.6% to 0.6%. 40 Indeed, the current guidelines are in favor of extended thromboprophylaxis for cancer-associated abdominal surgery.7,41 However, only 19.4% of the patients were postsurgical thrombosis in our cohort. Thus, post-hospitalization prophylaxis and outpatient thromboprophylaxis during chemotherapy still need to be better explored. The status of cancer-associated thrombosis prediction is encouraging, but not yet successful for gastric cancer.42–44
Immobilization, including nonsurgical and hospitalization-associated, was found to be a factor associated with mortality. Indeed, immobilization due to the malignancy or its treatment has been identified as a factor associated with mortality in other cancers.27,45,46 With the advent of the results of the APEX trial, in which betrixaban was effective in reducing the VTE likelihood after hospitalization, it is plausible that patients with gastric cancer also would benefit from extended post-hospitalization thromboprophylaxis. 47 Notably, immobility is not accounted for in the cancer-specific VTE prediction scores and few patients with cancer were recruited in the APEX trial.
Given the underscored limitations in VTE prediction among patients with gastric cancer, predicting the probability of bleeding becomes an important point of clinical relevance. In our cohort, death was more likely after bleeding than after VTE recurrence. This emphasizes the need for a bleeding prediction tool in gastric cancer-related thrombosis. We looked into VTE-BLEED as a tool to stratify bleeding risk in patients with gastric cancer-related thrombosis. The score was not developed specifically for a cancer demographic; thus it did not perform well in our study. The single variable that clearly predicted bleeding events in patients with gastric cancer thrombosis was anemia. A recent analysis of the RIETE database demonstrated that anemia is a risk factor for bleeding in cancer-associated thrombosis. 19 In this recent analysis of over 12,000 patients with cancer-associated thrombosis, those with anemia had significantly higher rates of major and fatal bleeding compared to those without. The risk of bleeding in patients with gastric cancer and thrombosis is higher than for other malignancies. This underscores the need for awareness of anemia as a morbidity predictor when considering therapeutic and prophylactic options in patients with gastric cancer.
The prediction of VTE recurrence in patients with cancer remains elusive. For instance, the Ottawa score is a clinical prediction tool recently developed to predict recurrent VTE in patients with cancer. Multiple attempts by different groups to externally validate this score were unsuccessful.48,49 Indeed, our findings should be hypothesis-generating only, but an indicator of clinical severity in the presentation was promising. A SI ⩾ 1 has been well validated in PE as an independent predictor of 30-day mortality in acute PE. 50 A high SI was associated with VTE recurrence; this finding may represent the deconditioned status and poor compensatory mechanisms of the patient with gastric cancer in response to thrombosis. Sex was a factor associated with thrombosis recurrence in our cohort; indeed, male sex has been found to be predictive of recurrence in the general population as well as in cancer-associated thrombosis.51,52 However, continued research is necessary in this area of cancer-associated thrombosis to help identify who may benefit the most from extended anticoagulation despite the bleeding risk. Finally, our results are concordant with the recent validation of immobility and leukocytosis as major predictors of 30-day mortality in cancer-associated thrombosis. 53 Moreover, our data suggest that leukocytosis may be a factor associated with mortality even longer than 30 days in gastric cancer-associated thrombosis. There was an association between statins and lower 6-month mortality. In non-small cell lung cancer, the use of statins is independently associated with survival. 54 Given the strong association of lung and gastric cancer with tobacco and the recognized association between cancer-associated thrombosis and arterial events, the plausible biological mechanism of statins in cancer survival deserves to be further explored. 55
A limitation of our study is that while the database is prospective, RIETE is an observational registry that does not standardize treatments. Varied treatments by local institutions and practitioners could lead to differing treatment choices and in turn affect events such as bleeding. Similarly, we were unable to control for the indication of anticoagulation interruption that may have mediated both bleeding and thrombosis likelihood. While the VTE-BLEED score takes into account any history of bleeding, the RIETE database only accounts for history of major bleeding within 1 month prior to acute thrombosis. We were unable to find a strong association between the platelet count and bleeding events. This has been well described as a U-shaped association in the RIETE database. 56 Although those findings were not specific to the gastric cancer population, it remains plausible that the limited number of bleeds precluded definitive observations to many of the variables. Additionally, the database did not include histologic classification of gastric cancer, location of bleeding or if any therapies were attempted to manage bleeding events. Known prognostic factors such as TNM (tumor, node, metastasis) stage, surgical curativity, history of chemotherapy, histological type, and depth of invasion were not collected in this database and thus we could not incorporate them as confounders into our survival analysis. Finally, patients were not mandated to undergo autopsy; in patients in the final stages of malignancy, any efforts to perform diagnostic procedures to be sure about the cause of death are often dismissed. Given the considerable number of deaths in our cohort, it remains plausible that many were secondary to a massive thrombosis and not analyzed as such. Our analysis, nonetheless, stems from the largest cohort of patients with cancer-associated thrombosis and constitutes the most robust description of gastric cancer thrombosis to date.
Conclusion
In conclusion, we set out to investigate the mortality impact of venous thromboembolism in gastric cancer using the RIETE database. Our results demonstrated a significantly high 6-month mortality rate but call for caution with the bleed risk associated with anemia.
Supplemental Material
APPENDIX – Supplemental material for Outcomes after venous thromboembolism in patients with gastric cancer: Analysis of the RIETE Registry
Supplemental material, APPENDIX for Outcomes after venous thromboembolism in patients with gastric cancer: Analysis of the RIETE Registry by Kaushal Majmudar, Iva Golemi, Alfonso J Tafur, Jorge Del Toro, Adriana Visonà, Conxita Falgá, Joan Carles Sahuquillo, Manuel Alejandro Lorente, Antonella Tufano, Ido Weinberg and Manuel Monreal in Vascular Medicine
Footnotes
Acknowledgements
We express our gratitude to Sanofi Spain for supporting this Registry with an unrestricted educational grant. We also express our gratitude to Bayer Pharma AG for supporting this Registry. Bayer Pharma AG’s support was limited to the part of RIETE outside Spain, which accounts for 25.28% of the total patients included in the RIETE Registry. We also thank the RIETE Registry Coordinating Center, S&H Medical Science Service, for their quality control data, and logistic and administrative support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sanofi Spain supports this Registry with an unrestricted educational grant. Bayer Pharma AG supports this Registry limited to the part of RIETE outside Spain. Neither sponsors have a role in the study design, analysis, manuscript writing, or decision about where to publish the paper.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
