Abstract
Background:
Persons living with human immunodeficiency virus (HIV, PLWH) have an increased risk of peripheral artery disease (PAD) in comparison to the general population. However, a gap remains in understanding optimal management for this condition. This study assesses longitudinal outcomes associated with peripheral endovascular intervention (PVI) for PAD among PLWH.
Methods:
All Medicare fee-for-service patients undergoing femoropopliteal artery PVI between April 1, 2015 and December 31, 2018 were identified and stratified by HIV serostatus. The primary outcome was major adverse limb events (MALE), defined as major amputation or arterial embolism/thrombosis following an index procedure. The subdistribution hazard was used to evaluate the association between HIV serostatus and MALE, accounting for the competing risk of death. Results were adjusted for sociodemographics and major comorbidities.
Results:
Of 168,553 patients who underwent PVI, 357 (0.21%) were PLWH. The average age was 77.0 ± 7.6 years; 80.3% had hypertension, 70.3% had hyperlipidemia, and 24.6% had tobacco use disorder. Compared to those without HIV, PLWH were younger and had a higher burden of cardiovascular risk factors. MALE were substantially more frequent among PLWH, with a cumulative incidence of 24.6%, compared to 14.5% among those without HIV. The adjusted subdistribution hazard ratio was 1.26 (95% CI 1.00–1.58, p = 0.05). The use of guideline-directed statin therapy was low in both groups in the 90 days following revascularization (57.9% in PLWH vs 58.1% in those without HIV, p = 0.95).
Conclusion:
Among US Medicare beneficiaries, PLWH had poorer long-term outcomes following PVI. Greater attention to the management of symptomatic PAD is warranted for the HIV population, particularly following revascularization.
Background
Human immunodeficiency virus (HIV) infection is associated with an increased risk of atherosclerotic cardiovascular disease, including stroke, myocardial infarction,1,2 and peripheral vascular disorders.3,4 Appropriate management of these conditions is essential to minimize excess morbidity and mortality as the population of persons living with HIV (PLWH) ages in the United States. 5 Peripheral artery disease (PAD) is of particular concern, due to its strong association with impaired physical function, loss of independence, and poor overall prognosis in older adults.6–9 PAD is a common condition in the general US population and continues to increase in prevalence worldwide. 10 The pathophysiology overlaps that for HIV-associated atherosclerosis and includes lifestyle exposures such as smoking, chronic arterial inflammation and, in PLWH, metabolic dysregulation associated with antiretroviral therapy.11–13 Consequently, PLWH have an increased risk of both asymptomatic 14 and symptomatic 15 PAD in comparison to the general population.
There is a growing interest in understanding both the epidemiology of PAD among PLWH and the optimal approach to treatment in this unique patient population. However, there is currently limited evidence on response to PAD therapy specifically among PLWH. In particular, outcomes following the invasive management of symptomatic PAD among PLWH remain poorly described. The present study seeks to address this evidence gap by utilizing longitudinal Centers for Medicaid and Medicare Services (CMS) data to better understand whether HIV serostatus is associated with adverse long-term outcomes among older adults with femoropopliteal artery disease referred for peripheral vascular intervention (PVI).
Methods
Data source and study population
This was a retrospective cohort study using all available samples of inpatient and outpatient CMS data to identify all femoropopliteal PVIs performed between April 1, 2015 and December 31, 2018 among Medicare fee-for-service (FFS) beneficiaries in the US. Individuals were eligible for inclusion if they were aged 66 or older, were enrolled in Medicare FFS for at least 1 year prior to their index procedure (to allow for ascertainment of comorbidities), and underwent a femoropopliteal PVI either during a hospital admission or as an outpatient. Only the first procedure for each participant was included.
PVIs were identified using the International Classification of Diseases, 10th Revision (ICD-10) Procedural Coding System (PCS) and Current Procedural Terminology (CPT) codes (online supplemental Table 1). PVI could include balloon angioplasty alone or with stent implantation. The study was conducted in compliance with the data use agreement in place between CMS and Beth Israel Deaconess Medical Center. The study was approved by the institutional review board of the Beth Israel Deaconess Medical Center, with a waiver of informed consent for retrospective data analysis.
Exposure classification and covariates
The primary exposure of interest was HIV serostatus as defined by the CMS Chronic Conditions Warehouse (CCW) Other Conditions algorithm for HIV. 16 Demographic data were obtained from Medicare enrollment records to determine participant age, self-reported sex, and race/ethnicity. Other baseline comorbidities were assessed using the CCW Chronic Conditions coding algorithm. Critical limb ischemia (CLI) and prior amputation were defined by ICD 9th and 10th Revisions Clinical Modification claims codes ascertained via a 1-year lookback period (online supplemental Table 2). Hospital characteristics were retrieved from the 2016 Hospital Association Annual Survey File, including teaching status, region, and bed capacity. The use of cardiovascular medications was assessed by querying prescription claims in the 90-day period postprocedure. Prescription analysis was restricted to participants enrolled in Medicare Part D at the time of PVI.
Outcomes
The primary outcome for this study was the cumulative incidence of major adverse limb events (MALE) through December 31, 2019. As in prior studies, MALE were defined as arterial thrombosis/embolism or major lower-extremity amputation. 17 The ICD 9th and 10th Revisions Clinical Modification claims codes were used to identify MALE following an index procedure (online supplemental Table 2). Secondary endpoints included all-cause mortality and any lower-extremity amputation through study follow up.
Statistical analysis
Sociodemographics and comorbidities were compared between groups with t-tests for continuous variables and chi-squared tests for categorical variables. To assess the risk of MALE and major amputation alone following index PVI, survival methods accounting for the competing risk of death were used. Cumulative incidence functions were estimated using Fine–Gray methods and differences were compared using the Fine–Gray test. The subdistribution hazard ratio (HR) was also estimated with and without adjustment for relevant covariates. 18 Covariates included age, sex, race, diabetes mellitus, chronic kidney disease, ischemic heart disease, heart failure, and CLI.
The cumulative incidence of death and the combination endpoint of death plus MALE were estimated using Kaplan–Meier methods. Cox proportional hazards regression was used to calculate the HR for each outcome, adjusted for the covariates listed above. However, due to violation of the proportional hazards assumption for death plus MALE over the total follow-up period, the results for the combined endpoint are not reported.
Statistical significance was defined as p < 0.05 for all analyses and no adjustments were made for multiple comparisons. Data linkage and analyses were performed using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Study cohort
Of 287,866 PVIs performed during the study period, 168,553 met criteria and were included in the analysis. A total of 70,677 procedures were excluded due to age < 66 years, or < 1 year of Medicare FFS coverage; 48,265 procedures were excluded as they were repeat interventions in the same individual; and 371 procedures were excluded due to missing age or insurance coverage (Figure 1). Of the 168,553 participants included in the final analysis, 357 (0.21%) were PLWH.
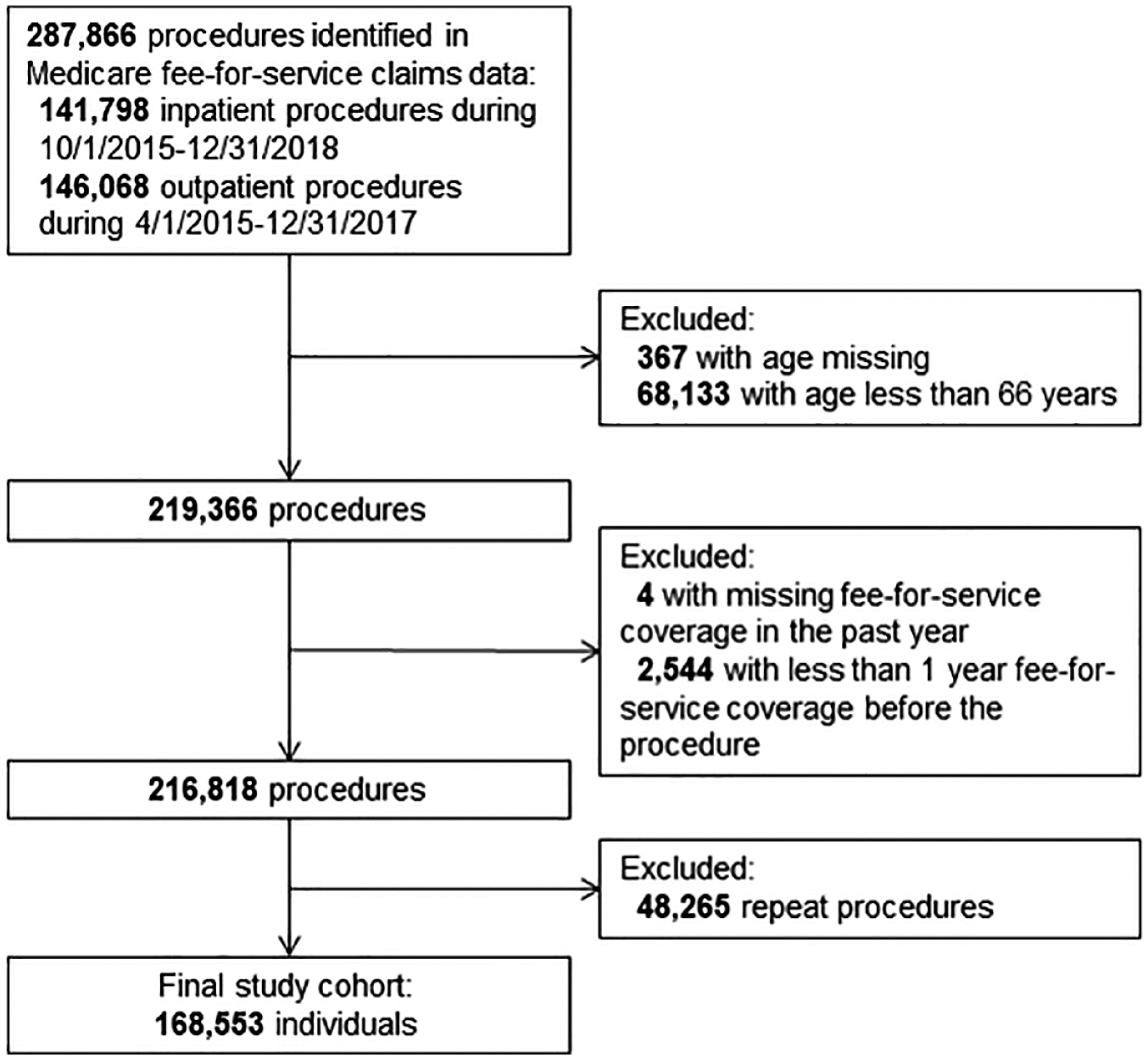
Patient flow diagram for establishment of the final cohort. Procedures were initially identified based on claims codes. After exclusion criteria were applied, repeat procedures were then removed to allow for analysis of individual beneficiaries.
Baseline demographic and clinical characteristics of the study population are presented in Table 1. The average age of the total population was 77.0 ± 7.6 years; 80.3% had hypertension, 70.3% had hyperlipidemia, and 24.6% had tobacco use disorder. A large proportion (46.6%) had CLI and 7.9% had undergone a prior amputation. Among the study cohort, 130,919 participants (77.7%) had pharmacy coverage through Medicare.
Baseline characteristics of study participants.
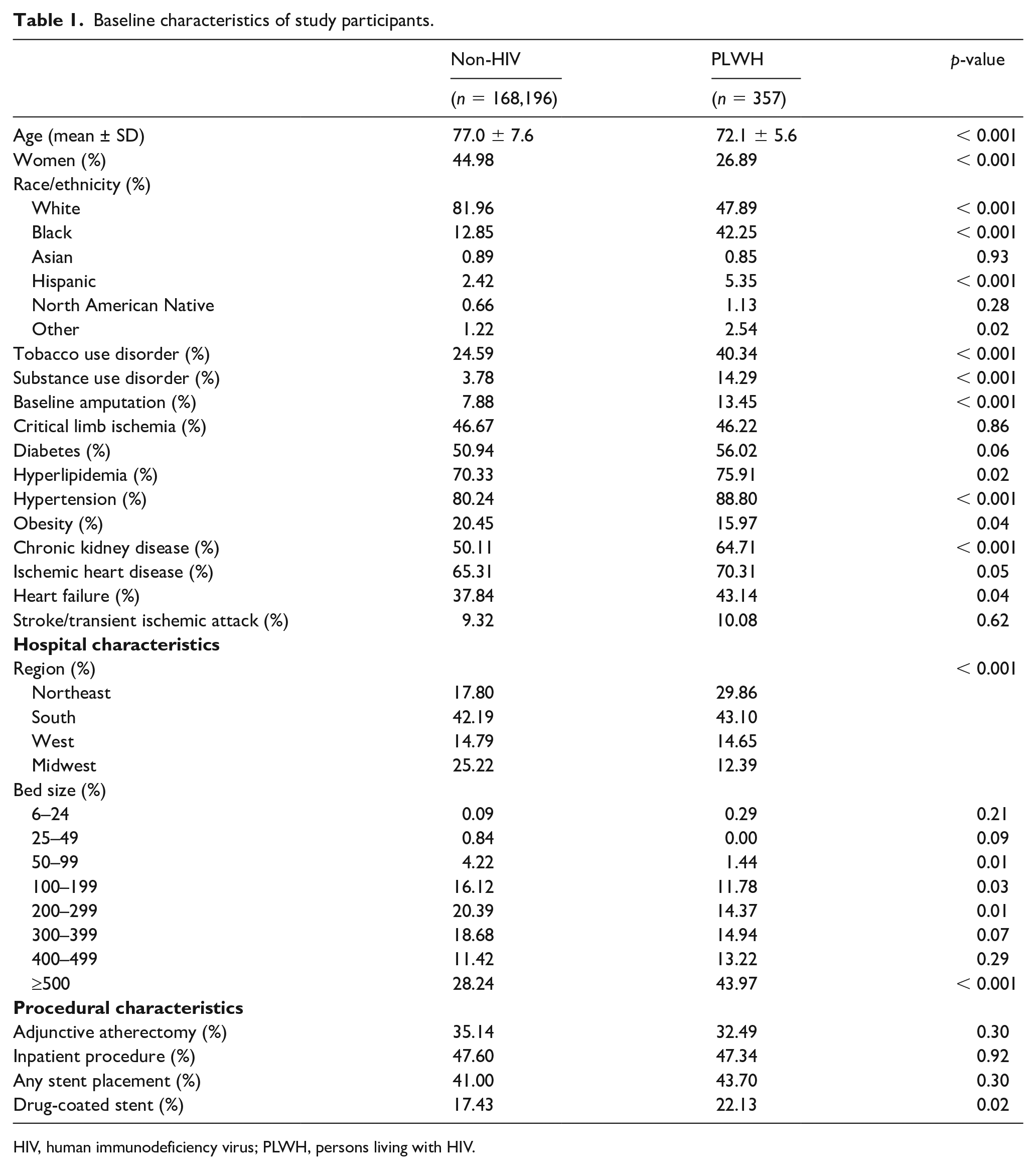
HIV, human immunodeficiency virus; PLWH, persons living with HIV.
Baseline characteristics between groups
Compared to those without HIV, PLWH were younger, less likely to identify as White or women, and more likely to smoke or have a history of substance use disorder. PLWH also exhibited an increased risk of multiple baseline cardiovascular comorbidities, including hypertension, hyperlipidemia, heart failure, ischemic heart disease, and chronic kidney disease. There was no difference in baseline rates of CLI between the two groups, but PLWH had nearly twice the prevalence of prior amputation (13.5% vs 7.9%, p < 0.001). PLWH were also more likely to undergo PVI at a teaching hospital and at hospitals with greater than 500 beds compared to persons without HIV (Table 1).
Procedural characteristics, including the use of atherectomy, stent placement, and patient location at the time of procedure (inpatient vs outpatient) did not differ by HIV serostatus. Notably, there was no difference in the rate of any stent placement between the two groups, but PLWH were more likely to receive a drug-eluting stent compared to those without HIV (22.1% vs 17.4%, p = 0.02).
The use of cardiovascular medications was similar by HIV serostatus (Table 2), including use of statins and antiplatelet agents. Among PLWH, 57.9% received a prescription for a statin, 42.4% received a prescription for non-aspirin antiplatelet agent, and 73.4% received a prescription for any antiretroviral therapy within 90 days following PVI.
Medication prescription fills to 90 days postprocedure among Medicare Part D beneficiaries.
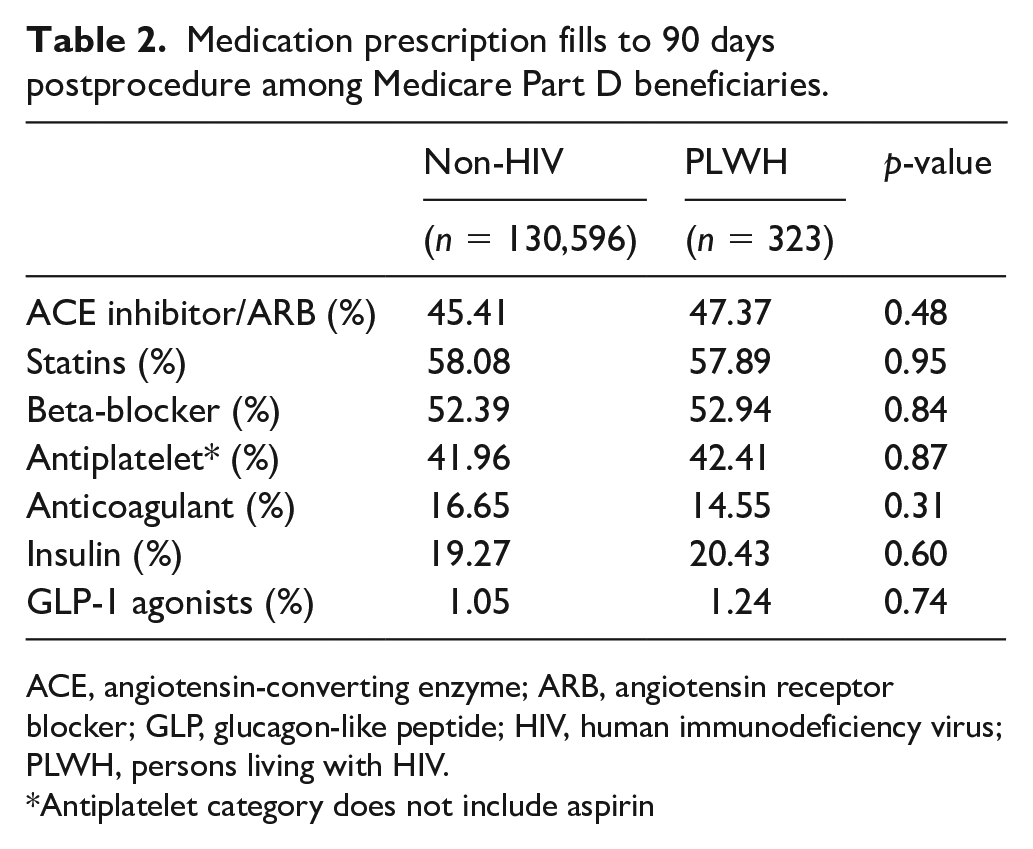
ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; GLP, glucagon-like peptide; HIV, human immunodeficiency virus; PLWH, persons living with HIV.
Antiplatelet category does not include aspirin
Primary outcome
The median study follow-up time was 2.4 years in both groups, with a maximum follow up of 4.8 years. PLWH had a higher risk of MALE following PVI over the study follow up. The rate of MALE was 9.4 events per 100 person-years among PLWH and 5.8 events per 100 person-years in the non-HIV group (Table 3). The cumulative incidence of MALE at the end of follow up was 24.6% for PLWH and 14.5% among those without HIV (Figure 2). The unadjusted HR for MALE was 1.56 (95% CI 1.23–1.97, p < 0.01). This association persisted after multivariable adjustment (HR 1.26, 95% CI 1.00–1.58, p = 0.05).
Risk of adverse events following peripheral vascular intervention by HIV serostatus.
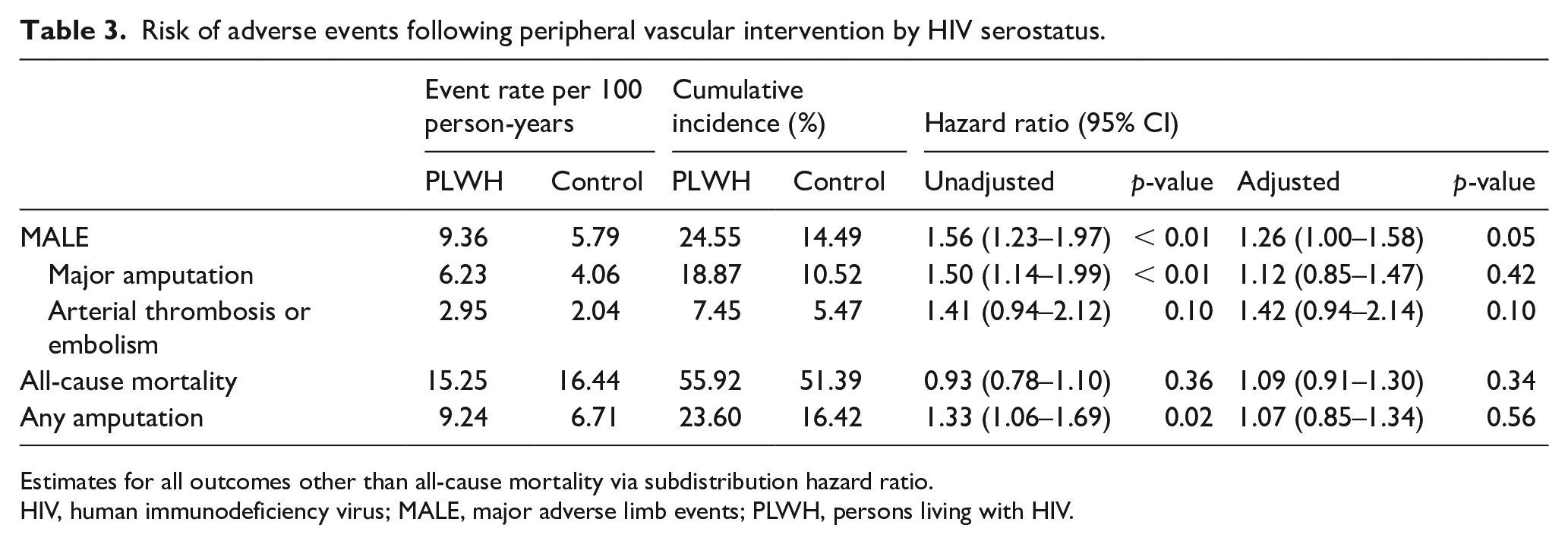
Estimates for all outcomes other than all-cause mortality via subdistribution hazard ratio.
HIV, human immunodeficiency virus; MALE, major adverse limb events; PLWH, persons living with HIV.
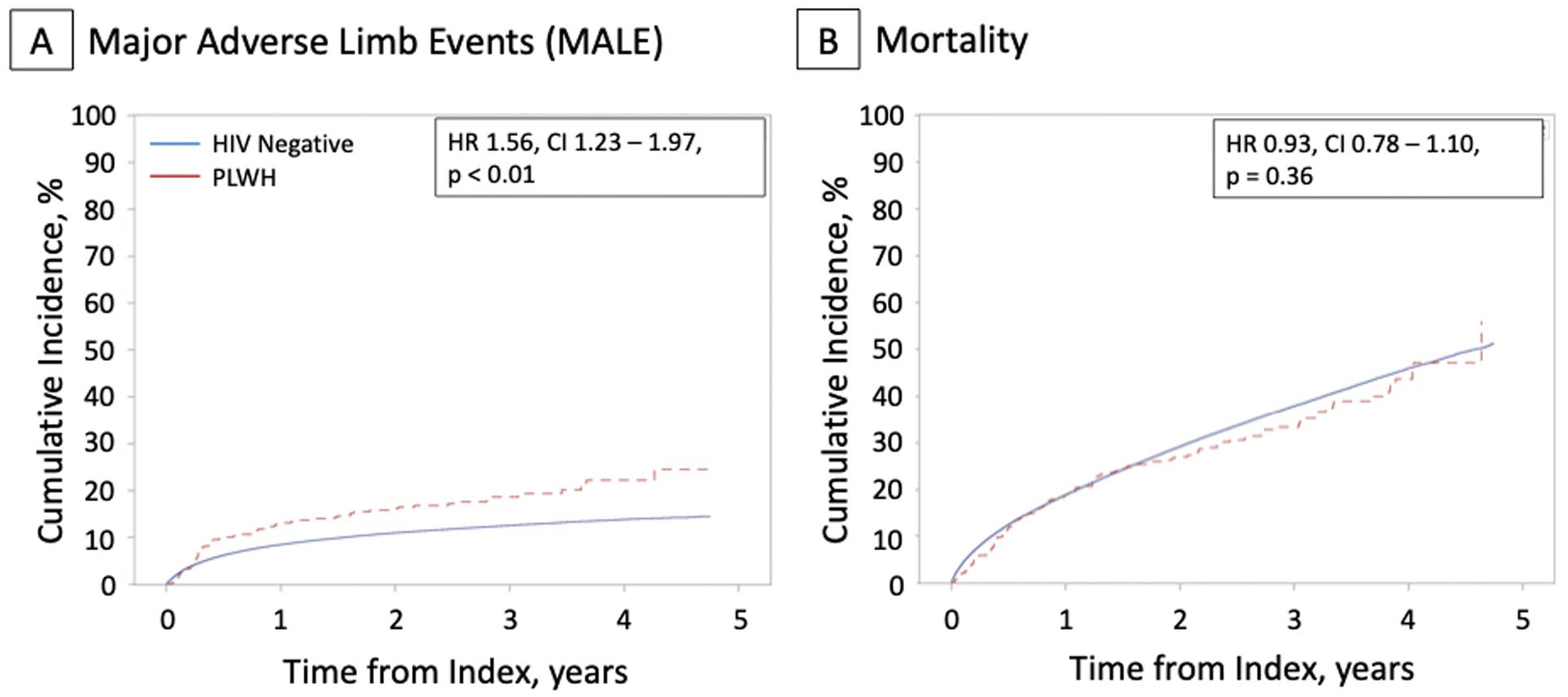
Cumulative incidence of MALE and mortality, stratified by HIV serostatus. Displayed are the cumulative incidence functions for study endpoints among persons living with HIV (PLWH, red) compared to HIV-negative controls (blue). (
Secondary outcomes
Among the individual endpoints, the unadjusted HR for major amputation was 1.50 (95% CI 1.14–1.99, p < 0.01) and for arterial thrombosis or embolism was 1.41 (95% CI 0.94–2.12, p = 0.10, Table 3). After multivariable adjustment, the HR for major amputation was attenuated (1.12, 95% CI 0.85–1.47, p = 0.42).
The all-cause mortality rate was 15.3 deaths per 100 person-years in the HIV group and 16.4 deaths per 100 person-years in the non-HIV group. There was no statistically significant difference in the risk of death over the study follow up (adjusted HR 1.09, 95% CI 0.91–1.30, p = 0.34).
Discussion
In this study of older adults undergoing PVI for PAD, HIV was associated with a nearly twofold increase in risk of MALE over 4.8 years of follow up. PLWH had an increased burden of cardiovascular comorbidities at baseline and were more likely to have had a prior amputation. Notably, pharmacotherapy for atherosclerotic disease was underutilized for both study cohorts. Fifty-eight percent of analyzed subjects were provided with a statin prescription in the 90-day post-PVI, despite prior evidence demonstrating improved outcomes for PAD with statin therapy. 19
The current study is novel as it investigates long-term outcomes following endovascular intervention for PAD among PLWH, and includes procedures performed both in inpatient and outpatient settings. Previous work has demonstrated that PLWH with PAD have an increased risk of perioperative mortality following open surgical revascularization, as well as a higher risk of subsequent amputation, 20 but have not investigated longitudinal outcomes following a percutaneous approach. Data on in-hospital outcomes immediately following PVI among PLWH in the National Inpatient Sample did not demonstrate increased rates of in-hospital amputation or mortality, but did not explore events after discharge.21,22
Multiple factors likely contribute to the increased risk of MALE observed among PLWH in our population. Our data are consistent with prior studies which have reported a high prevalence of traditional cardiac risk factors among PLWH with suboptimal control of modifiable contributors to disease, such as smoking. 23 Similar to previous analyses using CMS claims data, our study demonstrated high rates of tobacco and substance use among PLWH undergoing treatment for PAD in comparison to those without HIV in the general Medicare population. 24 Our study also demonstrated notable differences in the baseline health status of Medicare beneficiaries living with HIV as a cohort, including higher rates of hypertension, hyperlipidemia, kidney disease, and heart failure. Prior work has demonstrated that these comorbidities contribute to an increased risk of adverse events following PVI among older adults in the general population. 25 The increased risk of MALE among PLWH was attenuated in the current study after adjustment for medical comorbidities, supporting the hypothesis that poor overall health status in these individuals is a major contributor to subsequent risk of adverse events.
Our findings are also consistent with recent work by Parikh et al. demonstrating an increased risk in 5-year mortality following percutaneous intervention for coronary artery disease among US veterans aging with HIV. This increased risk was observed despite well-controlled HIV infection and a similar procedural approach to HIV-negative controls. 26 As in our study, the authors report significantly higher rates of tobacco and substance use disorder among PLWH, suggesting the critical importance of addressing these modifiable risk factors among PLWH with advanced atherosclerosis requiring intervention.
Whether HIV itself is a direct causative factor in adverse arterial events is a subject of ongoing investigation. The overall physiologic links between HIV and atherosclerotic cardiovascular disease are complex, with immune dysregulation and chronic inflammation contributing to accelerated arterial dysfunction over time, even in individuals with well-controlled infection.27 –29 Various studies have assessed the association between HIV and subclinical atherosclerosis and have demonstrated independent increases in coronary artery calcium, 30 noncalcified coronary plaque, 31 and carotid intima–media thickness. 32 These changes contribute to the increased burden of overt cardiovascular disease among PLWH, and may extend to the increased risk of PAD that has been observed in multiple HIV populations.3,14,15 It remains plausible that these same changes may contribute to adverse events following PVI, though a complete causal analysis of the direct effects of the virus on MALE risk is outside the scope of this study.
One additional major contributing factor may be the underutilization of guideline-based medical therapy. Several large studies have reported that individuals with HIV are less likely be prescribed statins despite meeting criteria for lipid-lowering therapy.33,34 Statins also continue to be woefully under-prescribed for PAD in the general population.35–37 This includes cohorts undergoing peripheral vascular intervention, despite data demonstrating an increased risk of MALE in individuals who have not been prescribed statin and antiplatelet therapy following revascularization. 25 Although no differences in cardiovascular medication use were observed between groups in our current study, the overall low frequency of key medications including statin and antiplatelet agents is concerning and may disproportionately affect PLWH due to their higher burden of cardiovascular comorbidities. Further attention is needed to maximize optimal medical therapy, treat uncontrolled comorbidities, and intervene on tobacco utilization if long-term PVI outcomes are going to be comparable to the general population. A team-based model, including representation from vascular medicine, vascular surgery, and providers experienced in management of HIV, may offer opportunities for improved quality for this patient group.
Study limitations
The current study has several limitations. First, because these data are derived from Medicare FFS records, younger patients and those with alternative insurance (e.g., Medicare Advantage) are not represented. The prescription medication utilization results were further restricted to patients who enrolled in both Medicare FFS and Medicare Part D in order to obtain complete prescription claims, and do not include aspirin for this reason. Second, residual confounding cannot be excluded in this nonrandomized sample – we conclude that PLWH as a group have an overall increased risk of MALE, but we are not able to define the HIV-specific attributable risk as compared to the risk accrued from underlying differences in baseline health and socioeconomic status. Third, claims data do not allow for evaluation of detailed clinical, anatomical, and procedural information, including the presence of multilevel arterial disease, laterality of procedures, and duration/control of HIV infection. Finally, although this was one of the largest samples of PAD patients above the age of 65 with long-term limb data, the proportion of PLWH overall was small and no data were included on surgical revascularization for either group.
Conclusions
PLWH enrolled in Medicare FFS who are referred for endovascular treatment of symptomatic PAD are younger overall but have a high rate of comorbidities in comparison to HIV-negative controls. Following PVI, these patients are at an increased risk of long-term MALE and are under prescribed goal-directed medical therapy. Additional studies investigating the etiology behind adverse outcomes and optimization of high-quality, guideline-based medical treatment for PAD are warranted.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231191822 – Supplemental material for Longitudinal outcomes following peripheral vascular intervention among older persons living with HIV
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231191822 for Longitudinal outcomes following peripheral vascular intervention among older persons living with HIV by Katherine Kentoffio, Tianyu Sun, Jiaman Xu, Rushi V Parikh, Priscilla Y Hsue and Eric A Secemsky in Vascular Medicine
Footnotes
Data availability
The data underlying this article were provided by the Centers for Medicaid and Medicare Services under a license. Data will be shared on request to the corresponding author with permission of the Centers for Medicaid and Medicare Services.
Declaration of conflicting interests
Rushi V Parikh receives research support from the American Heart Association, Janssen, Infraredx, Abbott Vascular, and Bayer, and consulting fees from Abbott Vascular. Priscilla Y Hsue reports honoraria from Gilead and Merck. Eric A Secemsky receives research support from the US Food & Drug Administration, Harvard Medical School’s Shore Faculty Development Award, Becton Dickinson, Boston Scientific, Cook, Cardiovascular Systems, Inc., Laminate Medical, Medtronic, and Philips, and reports consulting and speaking fees from Abbott, Bayer, Becton Dickinson, Boston Scientific, Cook, Cardiovascular Systems Inc., Janssen, Medtronic, Philips, and VentureMed. The remaining authors report no other relevant disclosures.
Funding
Priscilla Y Hsue is supported by NIH grant K24AI112393; Eric A Secemsky is supported by NIH/NHLBI grant K23HL150290.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
