Abstract
The development of highly active antiretroviral therapy (HAART) has significantly improved the life expectancy of patients with human immunodeficiency virus (HIV), but has led to the rise of chronic conditions including peripheral artery disease (PAD). However, trends and outcomes among patients with HIV undergoing lower extremity revascularization are poorly characterized. The aim of this study was to investigate the trends and perioperative outcomes of lower extremity revascularization among patients with HIV and PAD in a national database. The National Inpatient Sample (NIS) was reviewed between 2003 and 2014. All hospital admissions with a diagnosis of PAD undergoing lower extremity revascularization were stratified based on HIV status. Outcomes were assessed using propensity score matching and multivariable regression. Among all patients undergoing lower extremity revascularization for PAD, there was a significant increase in the proportion of patients with HIV from 0.21% in 2003 to 0.52% in 2014 (
Keywords
Introduction
The development of highly active antiretroviral therapy (HAART) has significantly improved the life expectancy of patients with human immunodeficiency virus (HIV).1,2 As a result of this increased longevity, the prevalence of chronic conditions, including cardiovascular diseases, in this population has continued to rise. 3 HIV and HAART medications have been shown to be risk factors for accelerated atherosclerosis and cardiovascular events.4–7 Recent studies also demonstrate increasing rates of peripheral artery disease (PAD) in patients with HIV.8–10 The underlying mechanisms for this risk remain unclear, but are likely due to a combination of factors including increased systemic inflammation and immune activation, metabolic changes, antiretroviral medications, and high rates of concomitant risk factors for atherosclerosis.11–14
However, HIV is a documented risk factor for perioperative morbidity and mortality with interventions. An analysis of 1641 patients with HIV matched with patients without HIV across a variety of surgical specialties in the United States Veterans Health Administration Database found that patients with HIV were at increased risk for 30-day mortality, with low CD4 cell counts associated with even higher rates of mortality. 15 Similarly, perioperative outcomes among patients with HIV and PAD undergoing revascularization are poor.16,17 Botes et al. described their series of revascularization for chronic limb-threatening ischemia (CLTI) in 66 patients with HIV. 16 Procedures consisted of primarily open revascularization, with high rates of early bypass failure, amputation, and mortality. However, contemporary outcomes among patients with HIV undergoing endovascular interventions are not well described, and large-scale studies investigating the association between HIV and outcomes following revascularization for PAD are lacking. As the burden of patients with HIV continues to rise, it is important to understand the trends in lower extremity revascularization and outcomes in this population. The aim of this study was to examine perioperative outcomes of lower extremity revascularization for PAD in patients with HIV in a large, nationwide database.
Methods
Database
The National Inpatient Sample (NIS) database was retrospectively reviewed between the years of 2003 and 2014. Given the transition to the International Classification of Diseases, Tenth Revision (ICD-10) coding system in 2015, which had major changes in the coding of diseases and procedures with an unclear impact on the prevalence of HIV, PAD, and other diseases, the analysis was restricted to the years 2003 through 2014. The NIS is the largest inpatient database in the United States, and is sponsored by the Agency for Healthcare Research and Quality (AHRQ) Healthcare Cost and Utilization Project (HCUP), and provides a sampling of abstracted discharge data representing a 20% stratified sample of all discharges from a national survey of all nonfederal acute care hospitals in the United States. Discharge data are deidentified patient-level data, including up to 25 primary and secondary diagnoses and up to 15 primary and secondary procedures, using the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) coding system. Patients, providers, and hospitals are deidentified, and consent was not necessary. This study was approved by Yale University’s institutional review board.
Variables
All hospital admissions with a principal diagnosis of PAD were identified by ICD-9 diagnostic codes (online supplemental Table 1). Intermittent claudication was defined with ICD-9 code 440.21. CLTI included those with rest pain or tissue loss. Rest pain was defined with ICD-9 code 440.22. Tissue loss was defined as those listed under procedure codes in the table for tissue loss, or a primary diagnosis code of atherosclerosis of the extremities (ICD-9 codes 440.20, 440.22–443) with a secondary code including one of those listed under tissue loss. Patients with PAD were stratified for analysis based on the presence or absence of a history of HIV. Patients with a history of HIV infection were identified using Clinical Classification Software (CCS) diagnosis code 5.18,19 Demographic information included age, sex, race, primary payer, hospital region, hospital teaching status, median household income, and hospital bed size. Age is reported as both a continuous variable and stratified by decade to demonstrate differences between groups. Patients younger than 18 years of age were excluded. Race was categorized as white, African American, Hispanic, or other. Other race included Asian or Pacific Islander, Native American, and other. Primary payer was classified by Medicare, Medicaid, private insurance, uninsured, or other. Hospital regions were classified as Northeast, Midwest, South, and West. Hospital teaching status was classified as either teaching or nonteaching. Hospital bed size was classified as small, medium, or large. Hospital bed size is dependent on hospital region and teaching status. Further information regarding hospital bed size can be found at: https://www.hcup-us.ahrq.gov/db/vars/hosp_bedsize/nisnote.jsp. Median household income was based on patient’s zip code, and classified in quartiles as $1–43,999, $44,000–55,999, $56,000–73,999, and $74,000 or more. A complete description of all variables can be found at: hcup-us.ahrq.gov.
Comorbid conditions included hypertension, hyperlipidemia, coronary artery disease, congestive heart failure, diabetes, diabetes with complications, chronic kidney disease, chronic obstructive pulmonary disorder (COPD), and Charlson Comorbidity Index (CCI). Comorbidities were identified with the CCS diagnosis codes based on ICD-9 diagnosis codes. Open and endovascular surgical procedures were identified by ICD-9 procedure codes (online supplemental Table 1).
Outcomes included in-hospital mortality, major amputation, length of stay, and total cost. Total in-hospital cost was estimated based on a cost-to-charge ratio created by HCUP that is based on data from the Centers for Medicare and Medicaid Services. In-hospital amputation rates were obtained from secondary procedure codes for above-knee and below-knee amputations, which included ICD-9 procedure codes 84.15 and 84.17.
Statistical analysis
Differences between patients with and without HIV undergoing lower extremity revascularization were assessed using chi-squared and
Two separate methods were used to adjust for confounders in the analysis including propensity score matching and multivariable regression. Two forms of analysis were chosen to address limitations with each method and support the associations found.
20
Propensity score matching was used to match patients by HIV status who underwent lower extremity revascularization for PAD. A non-parsimonious multivariable logistic regression model was used to estimate the propensity score using age, sex, race, income, primary payer, hospital location, hospital teaching status, hospital bed size, hospital region, hypertension, hyperlipidemia, coronary artery disease, congestive heart failure, diabetes, chronic kidney disease, COPD, indication, and procedure type. The double robust method was then used to generate treatment weights, and the inverse probability of treatment weighting was used to match cases with controls using generalized linear models as validated previously for survey data.
21
In the second method, a multivariable regression model was used to identify predictors of in-hospital mortality and amputation with a forward selection model. The inclusion threshold was set at
Results
Trends in revascularization among patients with HIV
Among patients undergoing lower extremity revascularization for PAD, there was a significant increase in the proportion of patients with HIV from 0.21% in 2003 to 0.52% in 2014 (
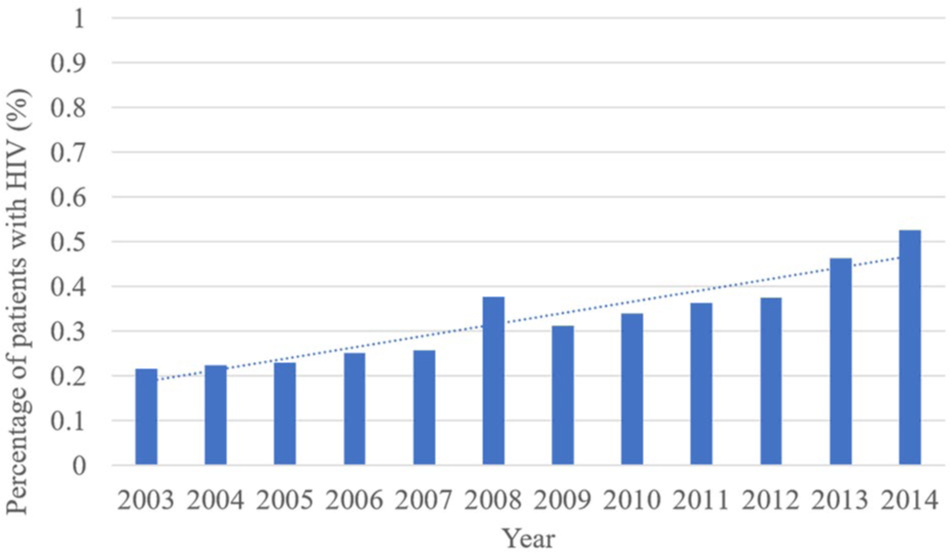
Trends in the proportion of patients with HIV undergoing lower extremity revascularization over time (2003-2014).
Demographics and comorbidities
Among patients with PAD undergoing lower extremity revascularization, there were 1,667,466 (99.69%) patients without HIV and 5154 (0.31%) patients with HIV (Table 1). Patients with HIV were significantly younger than those without HIV at the time of surgery at 55.74 and 68.72 years old, respectively (
Demographics, comorbidities, and characteristics of patients undergoing lower extremity revascularization by HIV status.
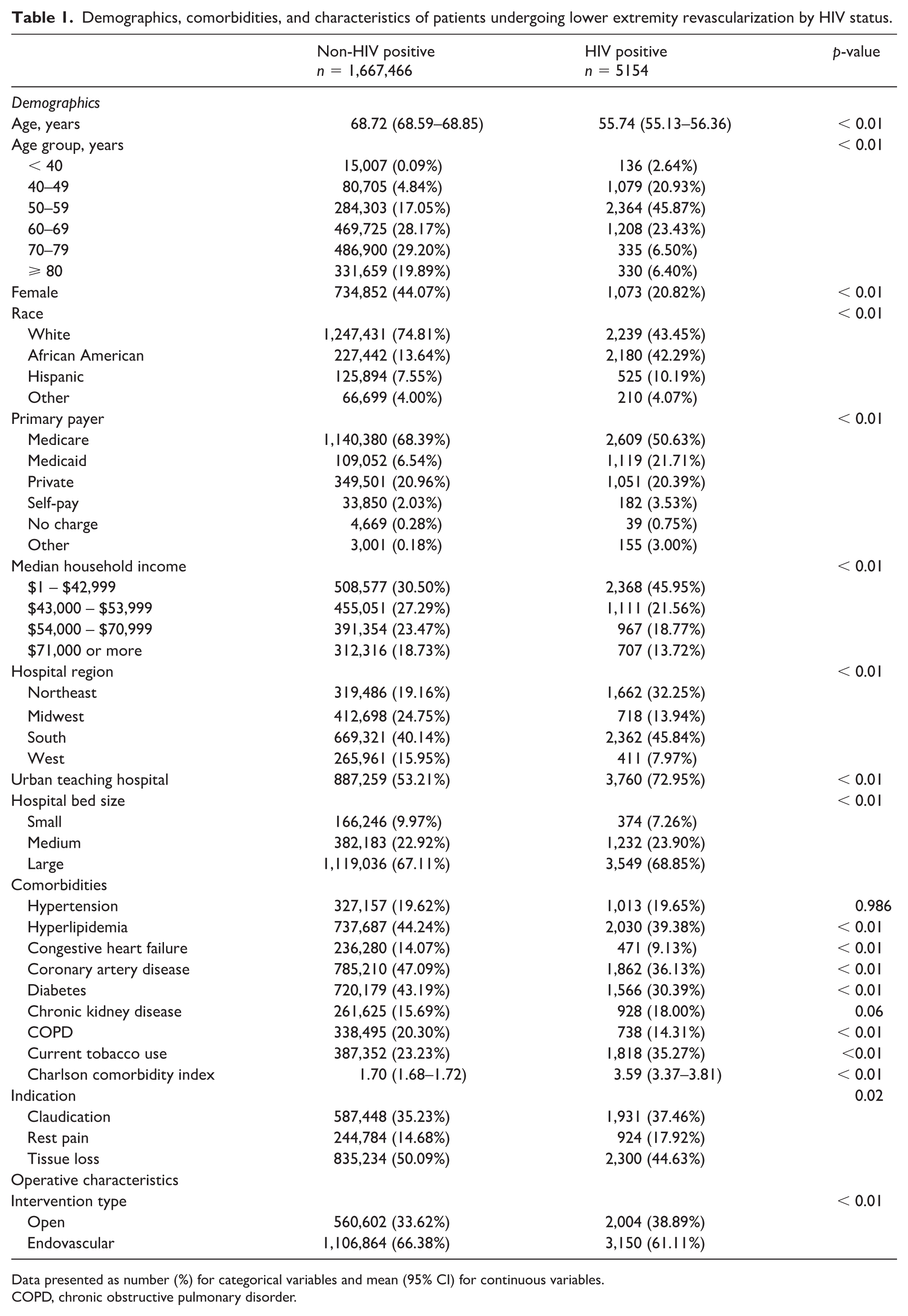
Data presented as number (%) for categorical variables and mean (95% CI) for continuous variables.
COPD, chronic obstructive pulmonary disorder.
Outcomes among patients with HIV
Among patients with PAD undergoing revascularization, patients with HIV had higher rates of in-hospital major amputation compared to patients without HIV, although not significant at 1.30% and 0.84%, respectively (
Outcomes of patients undergoing lower extremity revascularization by HIV status.
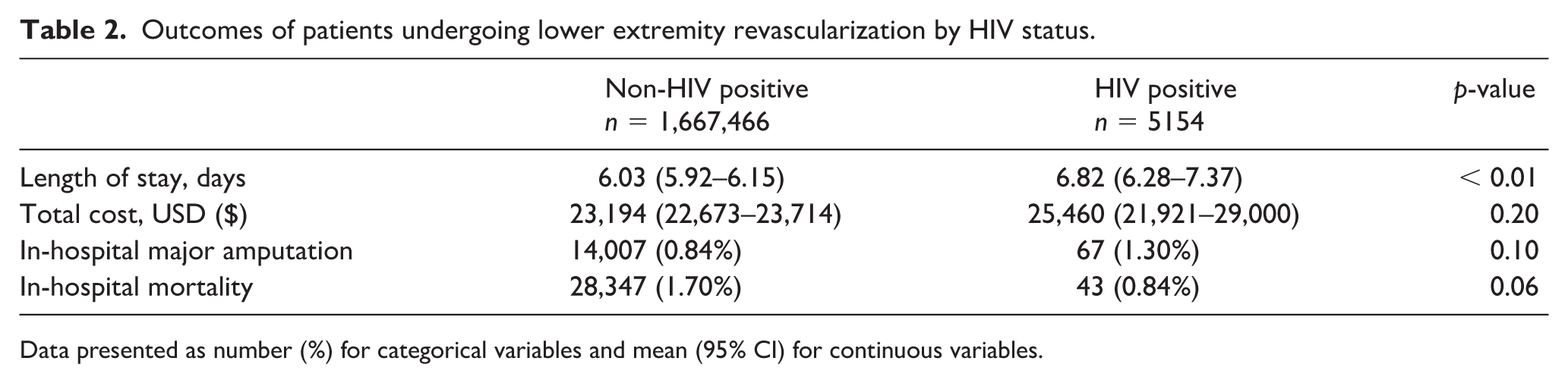
Data presented as number (%) for categorical variables and mean (95% CI) for continuous variables.
Adjusted outcomes among HIV-positive patients
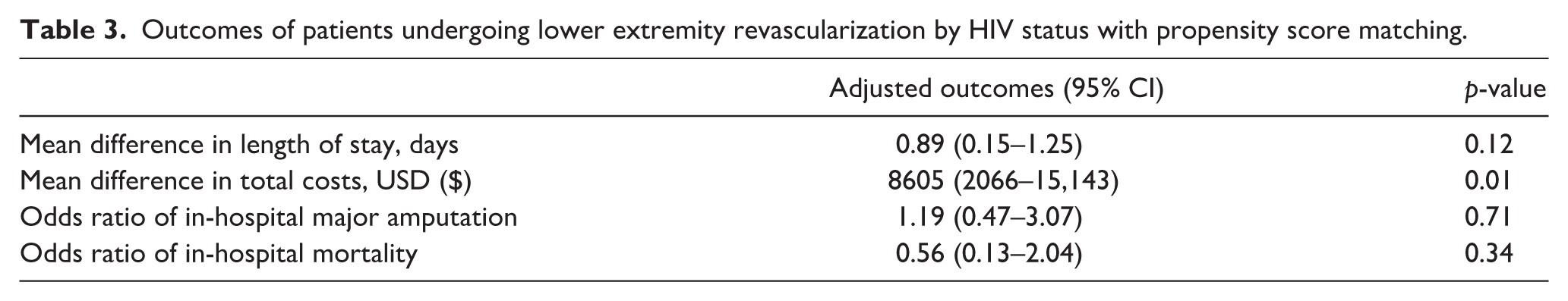
With propensity score matching, HIV status was not associated with increased major amputation (OR = 1.19, 95% CI 0.47–3.07,
Outcomes of patients undergoing lower extremity revascularization by HIV status with propensity score matching.
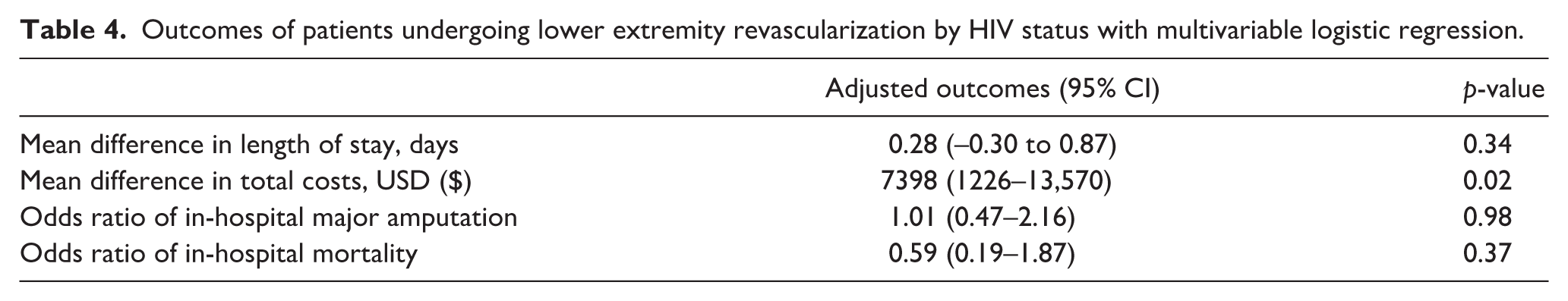
With multivariable logistic regression, HIV status was not associated with in-hospital major amputation (OR = 1.01, 95% CI 0.47–2.16,
Outcomes of patients undergoing lower extremity revascularization by HIV status with multivariable logistic regression.
Discussion
In this study of patients with PAD undergoing lower extremity revascularization, the proportion of patients with HIV steadily increased over an 11-year period. Although patients with HIV presented at a younger age and had a more costly hospital stay, there was no significant difference in in-hospital amputation or mortality between those with and without HIV.
Patients with HIV and PAD undergoing lower extremity revascularization were significantly younger than patients without HIV. However, it remains unclear if the younger age observed among PAD patients with HIV undergoing revascularization is due to the younger age of this population as a whole, or is secondary to an accelerated atherosclerotic process. 22 HIV causes chronic inflammation and infection of vascular smooth muscle cells that has been associated with an accelerated atherosclerotic process and has also been linked to early coronary artery disease.11–13 Additionally, patients with HIV have high rates of dyslipidemia and insulin resistance. 14 Early development of atherosclerosis and PAD in HIV-positive patients has also been demonstrated in several prior studies. Periard et al. found that 9.8% of 92 patients with HIV had evidence of PAD by ankle–brachial indices at a mean age of 49.5 years old. 23 Similarly, PAD was found in 12% of patients with HIV under 40 years of age in the COCOMO study, which is much higher than historical controls without HIV. 24 In the Veterans Aging Cohort Study, HIV was associated with a 19% increased risk of PAD, and occurred primarily in patients with sustained CD4 counts less than 200 cells/mm3. 8 Protease inhibitors, a common component of HAART, have also been linked to premature atherosclerosis and coronary artery disease, although their link to PAD is not well understood.25,26 This class of medications cause dyslipidemia and insulin resistance by dysregulating lipoprotein and glucose metabolism.27,28 A prospective study of patients with HIV demonstrated that those exposed to protease inhibitors were at increased risk of myocardial infarction with prolonged use. 29
Patients with HIV and PAD were not at increased risk of in-hospital amputation or mortality in this study compared to those without HIV. Perioperative outcomes among patients with HIV have been mixed. Several studies investigating perioperative outcomes in patients undergoing colorectal, cardiac, and thoracic surgery in the United States demonstrated no differences in mortality.30–32 However, other studies demonstrated an increased risk of perioperative and 1-year mortality among patients with HIV, with CD4 count, HIV viral loads, and hypoalbuminemia as risk factors for complications.15,33 Lin et al. investigated the outcomes of open abdominal aortic surgery in 48 patients with HIV with aneurysms or aortoiliac occlusive disease. 34 Postoperative complications were common, and perioperative mortality occurred in 15% of patients. Low CD4 counts and hypoalbuminemia were risk factors for worse outcomes. Van Marle et al. investigated the outcomes of 154 patients with HIV in South Africa with PAD, with 44 patients who underwent open revascularization for CLTI. 35 Limb salvage rates were poor, with a high rate of early graft thrombosis and a perioperative mortality rate of 7%. However, only a minority of patients had been initiated on HAART prior to presentation in the study. In contrast, the population investigated in the current study included a high proportion of patients with claudication. Additionally, this study represents more contemporary outcomes, with revascularization including high rates of endovascular-based interventions. Few studies have investigated the outcomes of endovascular interventions for PAD, although we found no increased risk for perioperative amputation or mortality. 16
As the treatment and care of patients with HIV improves their life expectancy, the prevalence of chronic diseases such as PAD will continue to increase. Similarly, a nationwide study of carotid interventions found that the proportion of patients with HIV undergoing carotid endarterectomy or stenting significantly increased over the study period from 2004 through 2014. 36 Lower extremity revascularization in this population appears to be relatively safe with no increased risk of in-hospital amputation or mortality. Despite this, long-term outcomes following revascularization are unknown. It is unclear if the accelerated atherosclerotic process associated with HIV leads to reduced long-term patency and increased rates of reintervention and limb loss.
Increased hospitalization costs were also observed after adjustment in patients with HIV. Similar findings of increased costs have also been observed in several other studies in patients with HIV undergoing various procedures.37,38 This may be due to additional medications being needed and the increased risk of infection in this population. 39 This population of patients is also younger without many of the same risk factors found typically in patients with PAD, which may delay recognition and workup prior to revascularization.
Study limitations
Limitations include those inherent to the NIS database. The NIS is based on billing codes abstracted from hospital discharges, causing some ICD-9 codes to be underreported. Additional characteristics including CD4 count, serum albumin levels, and HAART medications are not available. The true proportion of patients with HIV undergoing lower extremity revascularization is likely underreported in this database, as two states prohibit the release of information about inpatient records of patients with HIV, and one state masks information if a patient has HIV. Patients with HIV and PAD undergoing lower extremity revascularization in outpatient centers and those who underwent same-day procedures without a hospital admission are also not captured within the NIS. However, the NIS represents a large, nationwide database that allows for the analysis of HIV, which is otherwise not possible in many other PAD-related databases. The change to ICD-10 coding in 2015 also limited the years available for analysis, given the unclear changes in prevalence of diseases due to coding differences. An additional limitation includes patient selection, as it is possible that patients with HIV and more severe comorbidities may have had surgery deferred and been treated conservatively, or are more likely to undergo primary amputation and thus have been excluded from the current study.
Conclusion
Lower extremity revascularization for PAD can be safely performed in patients with HIV without risk of increased perioperative major amputation or mortality, although there are increased hospital costs. Further studies are needed to clarify the long-term outcomes of patients with HIV following revascularization.
Supplemental Material
hiv_pad_supplemental_tables – Supplemental material for Trends and perioperative outcomes of patients with human immunodeficiency virus (HIV) undergoing lower extremity revascularization
Supplemental material, hiv_pad_supplemental_tables for Trends and perioperative outcomes of patients with human immunodeficiency virus (HIV) undergoing lower extremity revascularization by Tanner I Kim, Anand Brahmandam, Timur P Sarac and Kristine C Orion in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
