Abstract
Endovascular therapy is a viable treatment option for symptomatic peripheral artery disease (PAD). Diabetes mellitus is a major risk factor for PAD. We evaluated the long-term impact of diabetes mellitus in patients with PAD who underwent endovascular therapy. In this prospective registry of 765 patients who underwent endovascular therapy for symptomatic PAD, 560 had diabetes and 205 did not have diabetes. Propensity score matching (PSM) analysis was performed to adjust for any potential confounders. The primary endpoints were 5-year major adverse cardiac and cerebrovascular events (MACE) and major adverse limb events (MALE). After PSM analysis, baseline clinical and limb characteristics were similar in patients with diabetes and in patients without diabetes. Patients with diabetes had a higher rate of major hematoma (> 4 cm) (9.9% vs 3.3%, p = 0.038). At 5-year clinical follow-up, patients with diabetes and patients without diabetes had similar rates of MACE (20.7% vs 20.7%, log rank p = 0.989) and MALE (19.8% vs 24.5%, log rank p = 0.312), as well as the individual endpoints. In subgroup analysis adjusted by PSM, female patients with diabetes had a higher risk of MALE (hazard ratio 0.31, 95% CI 0.13–0.69, p = 0.004). Although there was a higher rate of major hematoma, patients with diabetes who underwent endovascular therapy had similar 5-year cardiac and vascular outcomes compared with patients without diabetes. Bleeding avoidance strategies, including optimal antiplatelet and antithrombotic therapy, and meticulous vascular access techniques, are needed in patients with diabetes to reduce the risk of bleeding and its deleterious impact on morbidity and mortality.
Keywords
Introduction
The World Health Organization estimates that 420 million adults worldwide have diabetes mellitus. 1 In 2012, in addition to 1.5 million deaths, complications of diabetes included myocardial infarction, stroke, kidney disease, and lower limb amputation. Peripheral artery disease (PAD) affects over 202 million people worldwide. 2 An estimated one out of every three people with diabetes over the age of 50 have PAD. 3 In fact, diabetes mellitus is one of the most common causes of nontraumatic lower limb amputations. 4 Patients with diabetes have a 100-fold increase in the incidence of gangrene of the lower extremities. 5
In patients with diabetes, hyperglycemia and insulin resistance alter endothelial hemostasis, accelerating atherosclerosis. 6 High levels of glucose and free fatty acids may lead to the accumulation of reactive oxygen species. This can lead to the alternation of pro-oxidant and inflammatory pathways including factor kappa-B, which promotes transcriptional programs leading to the overexpression of adhesion molecules, monocyte adhesion, and subsequent formation of foam cells.
The atherosclerotic plaque observed in patients with diabetes with coronary artery disease (CAD) and PAD is commonly heavily calcified, which increases the complexity and peri-procedural complications of percutaneous coronary intervention (PCI) and endovascular therapy. 7 In addition, they tend to have plaque that is commonly long and diffuse in nature, with involvement of the distal vascular bed. 8 In the OLIVE registry, diabetes mellitus was not a predictor of 3-year major adverse limb events (MALE). 9 Similarly, analysis from the 2008–2014 Vascular Quality Initiative demonstrated no significant differences in 1-year primary patency (81% vs 79%, p = 0.36), major amputation (14% vs 11%, p = 0.09) or mortality (6% vs 7%, p = 0.30) among patients with diabetes and patients without diabetes undergoing endovascular therapy. 10 Despite the absence of significant differences in clinical outcomes after endovascular therapy between patients with diabetes and patients without diabetes at short and intermediate-term follow-up, data on long-term follow-up are limited. We report the 5-year clinical results of patients with diabetes who underwent percutaneous transluminal angioplasty (PTA) for symptomatic PAD compared with patients without diabetes.
Methods
Study population
This prospective study included 765 patients (560 with diabetes and 205 without diabetes) who underwent endovascular therapy for symptomatic PAD from 2006 at Korea University Guro Hospital (KUGH), Seoul, Korea. A standardized case report form was filled out by a study coordinator and outcomes data were entered into the PTA registry of KUGH. Baseline clinical, procedural, and outcomes data were obtained from face-to-face interviews, medical chart reviews, and telephone contact. Informed consent was obtained in all patients. This study protocol was approved by the Institutional Review Board.
Study endpoints and definitions
The primary endpoints were the 5-year rates of major adverse cardiac and cerebrovascular events (MACE), defined as the composite of total death, myocardial infarction, repeat coronary revascularization (percutaneous or surgical), and stroke, as well as MALE, defined as the composite of target extremity revascularization (TER) or target extremity surgery including amputation and lower extremity bypass surgery.11,12 Myocardial infarction was defined as symptoms of unstable angina with new ST-segment elevation or elevation of cardiac biomarkers to at least twice the upper limit of normal. Target lesion revascularization (TLR) was defined as revascularization of the target lesion due to restenosis or re-occlusion within the balloon angioplasty site, stent or in an adjacent 5 mm of the distal or proximal segment. Target extremity revascularization was defined as clinically driven revascularization of the target lesion or any segment of the same limb containing the target lesion. Diabetes mellitus was defined as fasting glucose levels ⩾ 126 mg/dL or the use of glucose-lowering medication. Critical limb ischemia was defined as ischemic pain at rest, ulcer, or gangrene in one or both legs attributed to objectively proven arterial disease with transcutaneous oxygen pressure.
Percutaneous transluminal angioplasty
Standard techniques for PTA were used. For infrapopliteal lesions, a 5 Fr HeartrailTM guiding catheter (Terumo, Tokyo, Japan) and a 0.014” guide wire were used to cross the lesions. If intraluminal wiring was unsuccessful, a subintimal angioplasty or retrograde approach was attempted. Upon successful wire passage, balloon inflations of at least 120–180 seconds with sizes ranging from 1.5 to 3.0 mm were used. Self-expanding nitinol stents (Xpert™ (Abbott Vascular, Santa Clara, CA, USA) or Maris Deep™ (Medtronic-Invatec, Santa Rosa, CA, USA)) were used if balloon angioplasty results were suboptimal.
For chronic total occlusion (CTO) of the superficial femoral artery (SFA) and iliac artery, dedicated 0.018” CTO wires were used to remain inside the true lumen. If unsuccessful, subintimal angioplasty was performed using a 0.035 soft Terumo wire (1.5 J curve) and a 5 Fr angiocatheter support for longer CTO lesions. Provisional spot stenting using self-expanding nitinol stents was performed if the true lumen was wired for shorter CTO lesions. Re-entry with CTO wires, or a re-entry device (OUTBACK catheter; Cordis, Miami Lakes, FL, USA), was achieved if the subintimal wiring failed to re-enter the distal true lumen. If re-entry into the true lumen was unsuccessful, retrograde access was obtained from the distal SFA, or popliteal or infrapopliteal arteries.
Statistical analysis
Continuous data were presented as mean ± SD and compared with the unpaired t-test or Mann–Whitney rank test. Categorical data were presented as number and percentage and compared with χ2 or Fisher’s exact tests. A two-tailed p-value of < 0.05 was considered statistically significant. Statistical analyses were performed using SPSS 20.0 (IBM Corp., Armonk, NY, USA). To adjust for any potential confounders, propensity score matching (PSM) analysis was performed using the logistic regression model. Matching was performed via a 1:1 matching protocol using the nearest neighbor matching algorithm, with a caliper width equal to 0.01 of the SD of the propensity score, yielding 121 well-matched pairs. The variables included in the PSM model were: sex, age, body mass index, left ventricular ejection fraction, wound, claudication, resting pain, Buerger’s disease, hypertension, dyslipidemia, stroke, chronic kidney disease, congestive heart failure, atrial fibrillation, history of smoking, alcohol drinking habit, presence of coronary stenosis, hemoglobin, high-sensitivity C-reactive protein (hs-CRP), creatinine, aspirin, clopidogrel, cilostazol, ticlopidine, angiotensin II receptor blockers, angiotensin-converting enzyme inhibitors, calcium channel blockers, β-blockers, diuretics, statins, ankle–brachial pressure index, Rutherford classification, and PTA lesion site. Clinical outcomes up to 5 years were estimated by the Kaplan–Meier analysis, and differences between the groups were compared with the log-rank test before and after PSM. Cox regression models were constructed to identify whether diabetes mellitus was a predictor for clinical outcomes. For all analyses, a two-sided p < 0.05 was considered statistically significant.
Results
There was considerable imbalance between patients with diabetes and patients without diabetes in baseline clinical characteristics, such as initial diagnosis of PAD, hypertension hemorrhagic stroke, chronic kidney disease, smoking, alcohol use, coronary stenosis, and laboratory findings (Table 1). After PSM analysis, baseline clinical and limb characteristics were similar in both groups, except for fasting glucose (154 ± 81 mg/dL vs 112 ± 30 mg/dL, p < 0.01) and hemoglobin A1c (7.48 ± 1.83% vs 5.95 ± 0.73%, p < 0.01).
Baseline characteristics and post-procedural medications in the entire cohort and propensity-matched groups.
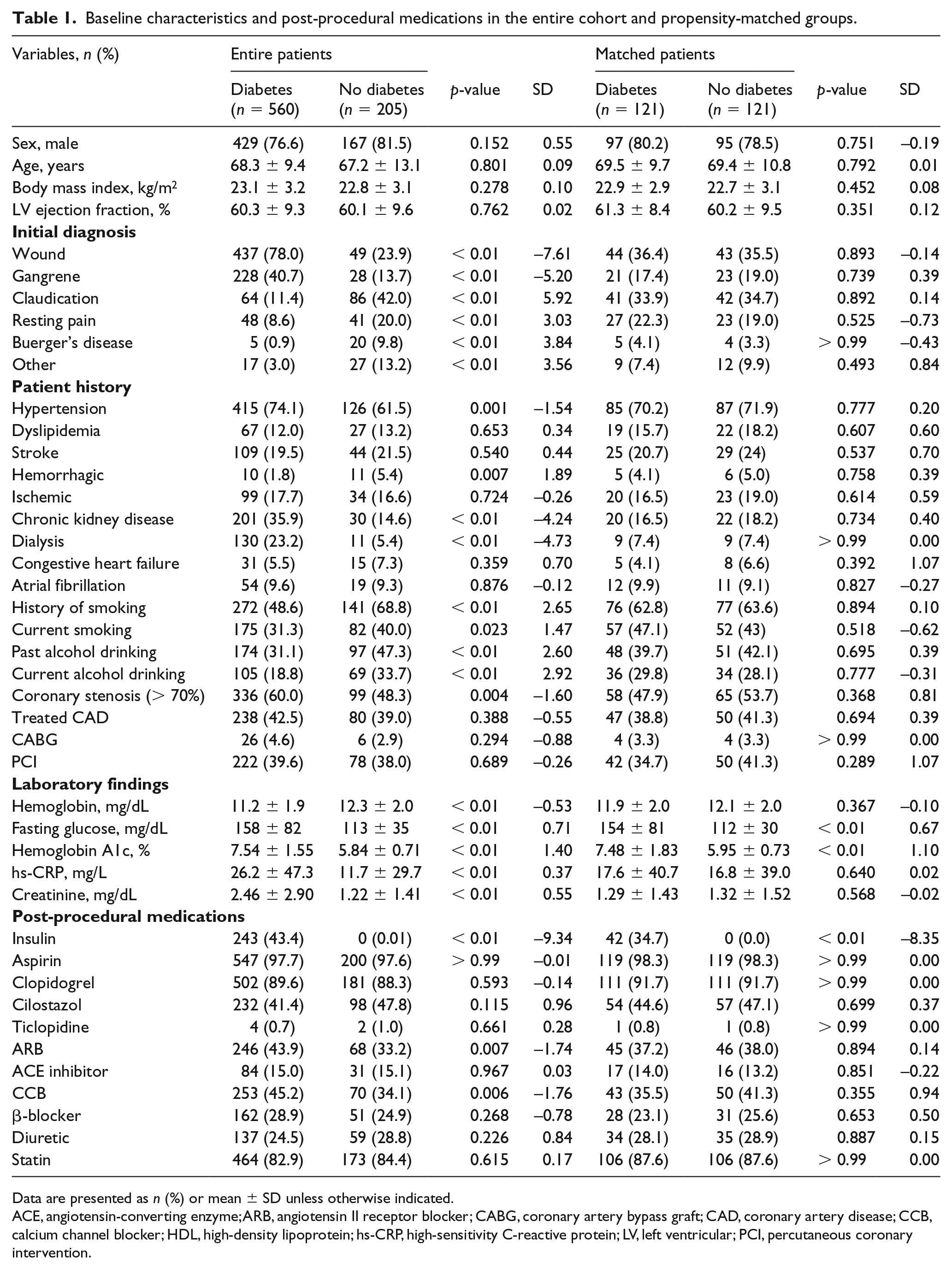
Data are presented as n (%) or mean ± SD unless otherwise indicated.
ACE, angiotensin-converting enzyme; ARB, angiotensin II receptor blocker; CABG, coronary artery bypass graft; CAD, coronary artery disease; CCB, calcium channel blocker; HDL, high-density lipoprotein; hs-CRP, high-sensitivity C-reactive protein; LV, left ventricular; PCI, percutaneous coronary intervention.
Patients with diabetes were more likely to have disease involving the anterior tibial artery (57.4% vs 26.1%, p < 0.01), posterior tibial artery (45.1% vs 17.5%, p < 0.01), and peroneal artery (22.6% vs 10.1%, p < 0.01) (Table 2). After PSM analysis, the clinical limb characteristics were well matched.
Clinical limb characteristics in the entire cohort and propensity-matched groups.
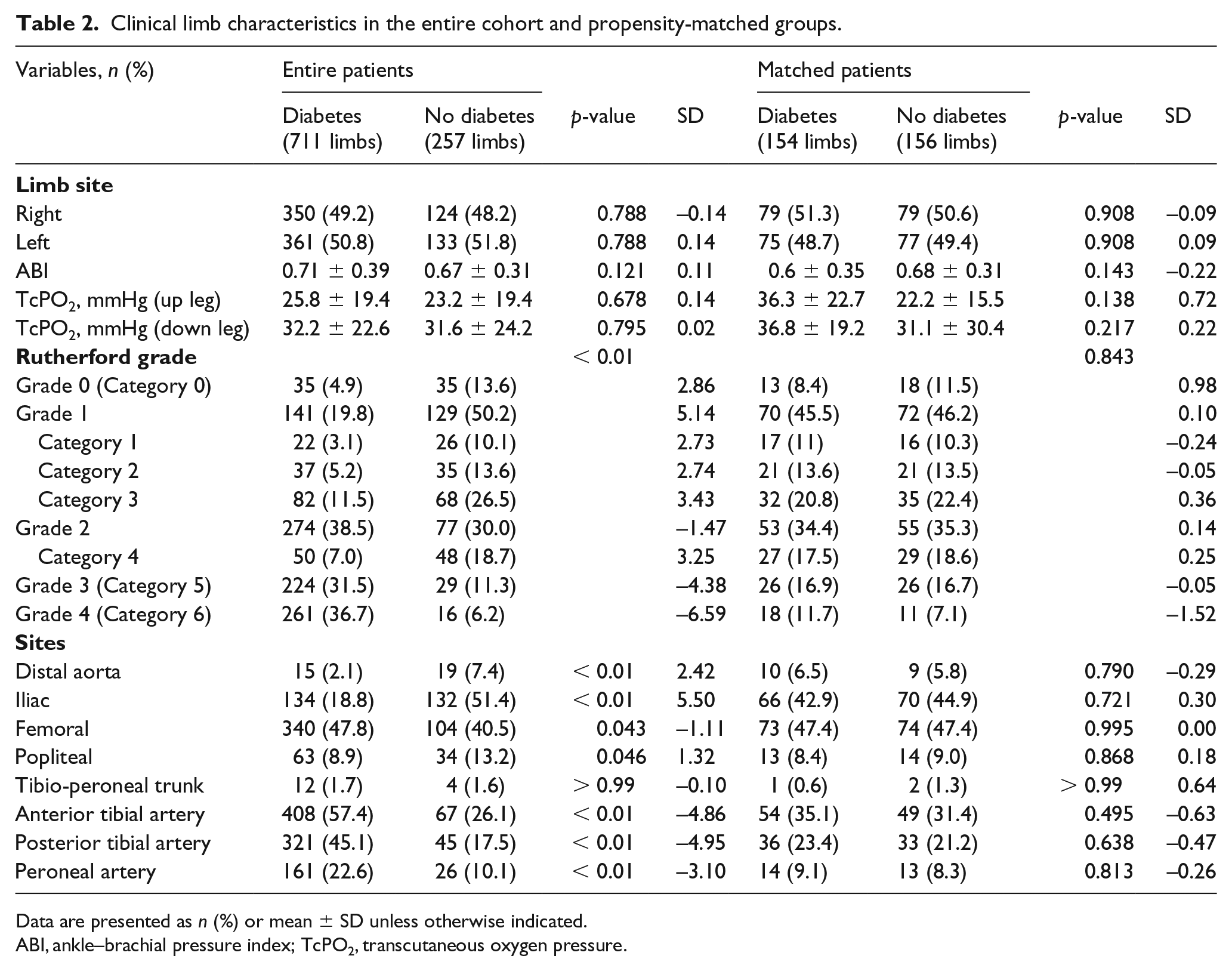
Data are presented as n (%) or mean ± SD unless otherwise indicated.
ABI, ankle–brachial pressure index; TcPO2, transcutaneous oxygen pressure.
Diabetic patients had a higher rate of bleeding complications (23.4% vs 13.7%, p = 0.003) and need for blood transfusion (39.8% vs 25.4%, p < 0.01) (Table 3). After PSM analysis, the incidence of major hematoma (> 4 cm) was higher in patients with diabetes (9.9% vs 3.3%, p = 0.038).
In-hospital complications and variable clinical outcomes up to 5 years by Kaplan–Meier curved analysis with log-rank test.
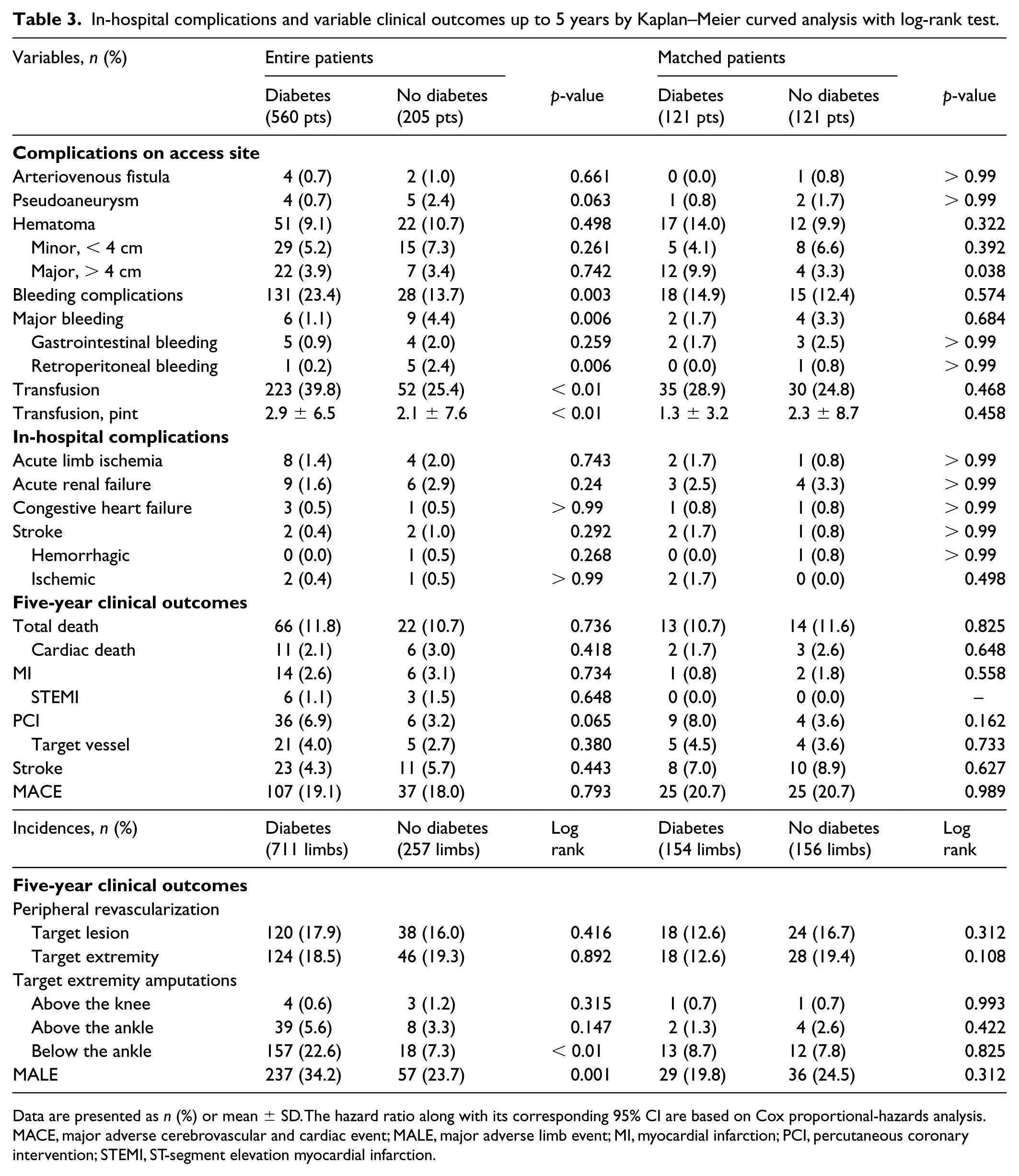
Data are presented as n (%) or mean ± SD. The hazard ratio along with its corresponding 95% CI are based on Cox proportional-hazards analysis.
MACE, major adverse cerebrovascular and cardiac event; MALE, major adverse limb event; MI, myocardial infarction; PCI, percutaneous coronary intervention; STEMI, ST-segment elevation myocardial infarction.
The mean follow-up period was 4.3 ± 2.8 years (median: 3.8 years, IQR: 1.9–6.2 years). Clinical outcomes up to 5 years were estimated by Kaplan–Meier analysis, and differences between the groups were compared with the log-rank test before and after PSM. During the 5-year clinical follow-up, patients with diabetes and patients without diabetes had similar rates of MACE (19.1% vs 18.0%, p = 0.793). After PSM, patients with diabetes also had similar rates of MACE (20.7% vs 20.7%, log rank p > 0.9), as well as the individual endpoints of death (10.7% vs 11.6%, p = 0.825), myocardial infarction (0.8% vs 1.8%, p = 0.558), PCI (8.0% vs 3.6%, p = 0.162), and stroke (7.0% vs 8.9%, p = 0.627). Patients with diabetes had a higher rate of MALE (34.2% vs 23.7%, p = 0.001). After PSM analysis, patients with diabetes and patients without diabetes had similar rates of MALE (19.8% vs 24.5%, log rank p = 0.312). Patients with diabetes and patients without diabetes had similar rates of target extremity revascularization (12.6% vs 19.4%, p = 0.108), above-the-ankle amputation (1.3% vs 2.6%, p = 0.422), and below-the-ankle amputation (8.7% vs 7.8%, p = 0.825).
At 5-year follow-up, MACE was similar in patients with diabetes that required insulin and diabetes that did not require insulin (20.6% vs 18.0%, p = 0.467) (Figure 1). At 5-year follow-up, MALE was higher in patients with diabetes that required insulin compared with patients with diabetes that did not require insulin (40.9% vs 29.3%, p = 0.002).
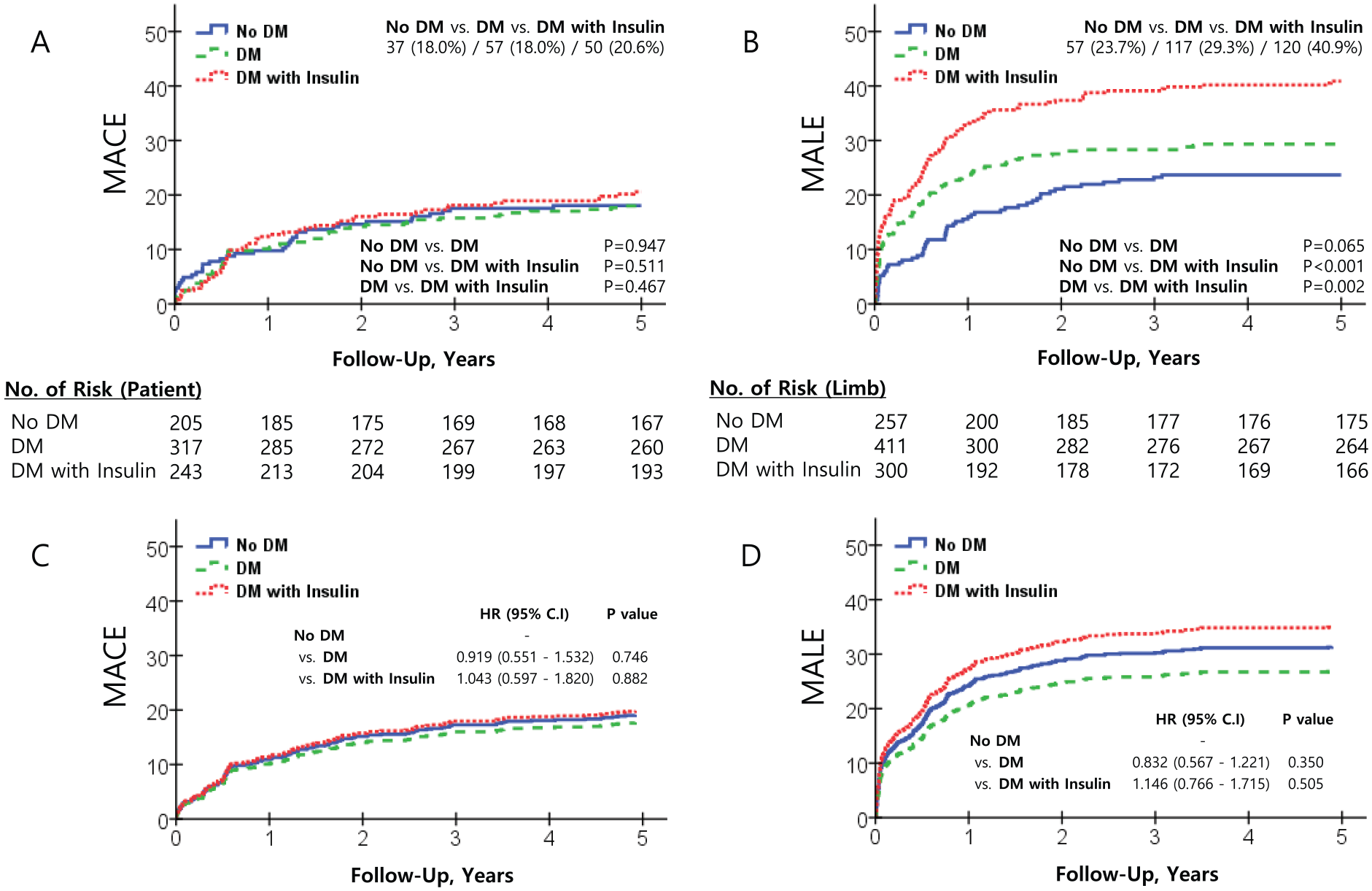
Five-year follow-up results of MACE and MALE by Kaplan–Meier curved analysis and Cox proportional-hazards ratio analysis adjusted by propensity score.
In subgroup analysis adjusted by PSM, patients with diabetes who were female had a higher risk of MALE (hazard ratio (HR) 0.31, 95% CI 0.13–0.69, p = 0.004) (Figure 2; online Supplementary Table 1).
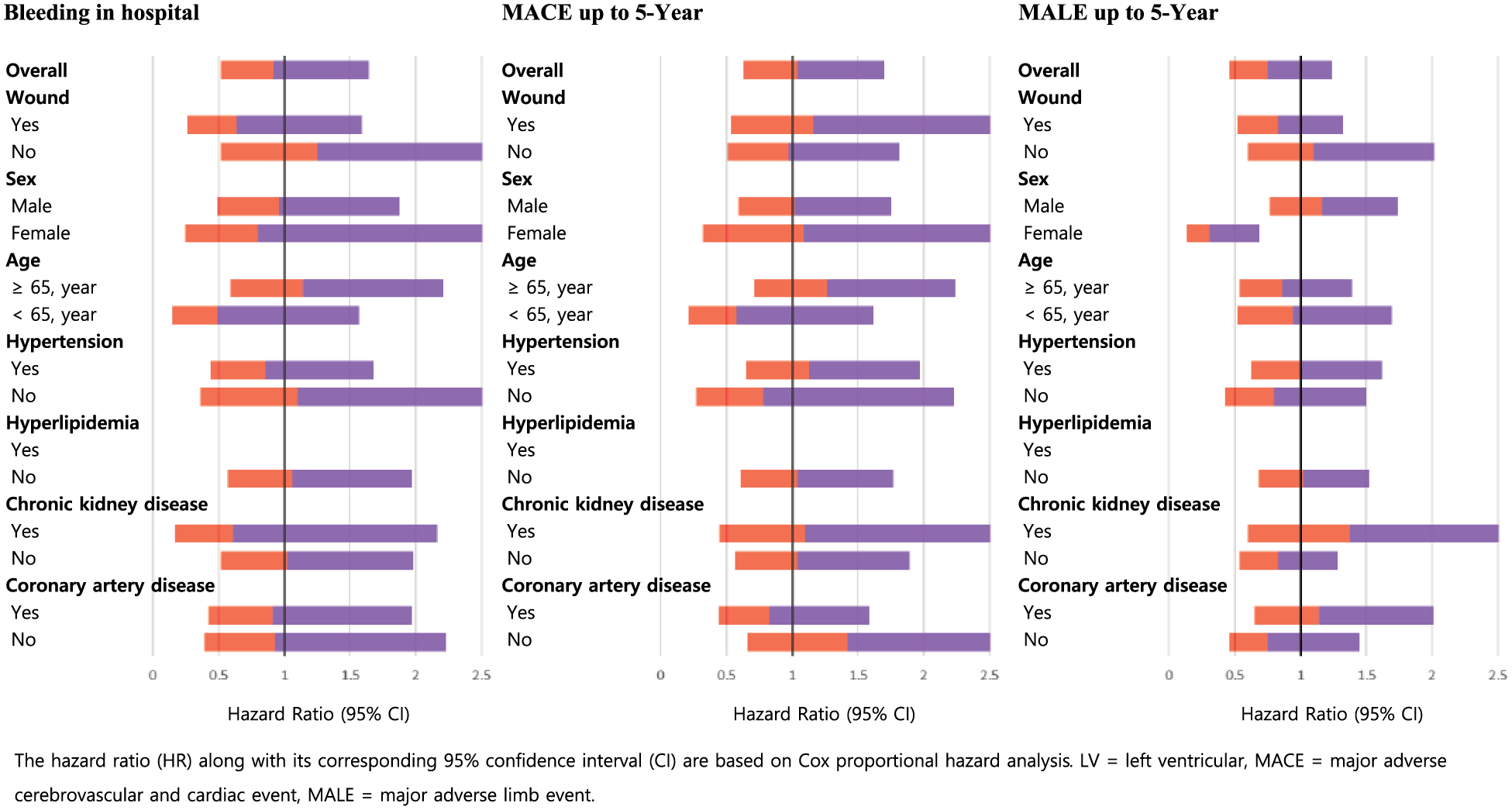
Impact of diabetes mellitus on variable clinical outcomes up to 5 years in variable subgroups analysis with Cox proportional-hazards ratio analysis adjusted by propensity score. Orange (light gray) and purple (dark gray) represent the range of the lower and upper limits of 95% confidence intervals. Note: This figure is in color online.
Predictors of in-hospital bleeding were chronic renal insufficiency (HR 1.77, 95% CI 1.18–2.64, p = 0.005) and the presence of a wound (HR 2.22, 95% CI 1.29–3.82, p = 0.004) (Figure 3; online Supplementary Table 2). Predictors of MACE up to 5 years were chronic renal disease (HR 1.64, 95% CI 1.14–2.37, p = 0.008) and significant CAD (HR 1.46, 95% CI 1.02–2.08, p = 0.035). Predictors of MALE up to 5 years were chronic renal insufficiency (HR 1.35, 95% CI 1.04–1.73, p = 0.019), stroke (HR 1.53, 95% CI 1.17–1.99, p = 0.002), smoking (HR 1.38, 95% CI 1.06–1.81, p = 0.015), involvement of the femoral artery (HR 1.63, 95% CI 1.28–2.07, p < 0.001) and arteries below the knee (HR 1.72, 95% CI 1.24–2.40, p = 0.001), and the presence of a wound (HR 2.31, 95% CI 1.69–3.17, p < 0.001).
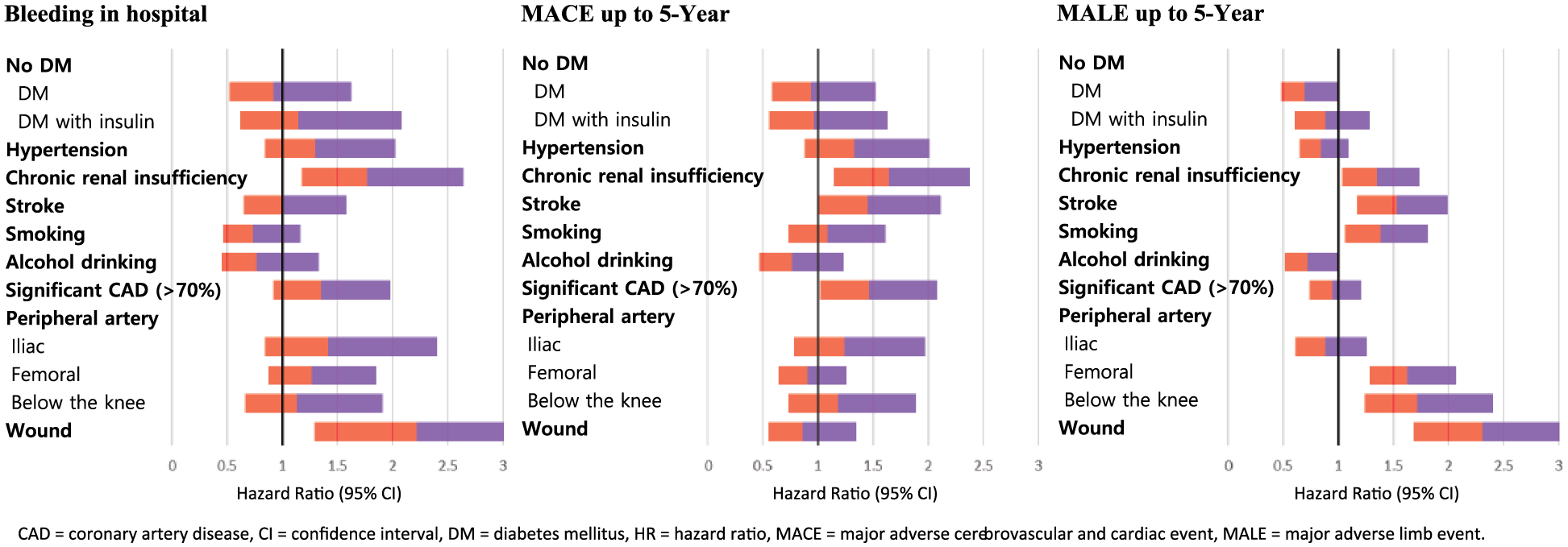
Predictors of variable clinical outcomes up to 5 years in entire population by multivariable Cox proportional-hazards ratio analysis. Orange (light gray) and purple (dark gray) represent the range of the lower and upper limits of 95% confidence intervals. Note: This figure is in color online.
Discussion
The results of our study demonstrate that patients with diabetes had similar rates of MACE and MALE at long-term follow-up. Patients with diabetes were at increased risk for major hematoma. Patients with diabetes who were female were at increased risk of MALE.
The leading causes of morbidity and mortality in patients with diabetes are PAD and CAD. Despite advances in treatment strategies, patients with diabetes are also at higher risk for complications and poorer outcomes following acute coronary events and revascularization procedures, translating into significant morbidity and mortality for this growing patient population.13–15 Almost 1.5 million revascularization procedures – coronary artery bypass grafting and PCI – are performed each year in the United States and approximately 25% of these procedures are for patients with diabetes. 16 The RESEARCH registry highlighted diabetes as an independent multivariable predictor of restenosis (odds ratio 2.63; 95% CI 1.14–6.31, p = 0.02); an increase seen in both patients with diabetes on insulin and not on insulin. 17 Inflammation and a prothrombotic state are key factors for diabetes-associated CAD and poor outcomes following revascularization procedures, in addition to influences from endothelial dysfunction, negative vascular remodeling, increased protein glycosylation, and increased vascular matrix deposition.18,19 In our analysis, after PSM, patients with diabetes and patients without diabetes had similar MACE at long-term follow-up. One potential explanation of similar MACE rates in the patients with diabetes and patients without diabetes may be due to aggressive treatment of CAD. Rather than noninvasive evaluation, patients with severe PAD underwent routine coronary angiography and subsequent revascularization if severe CAD was present. We previously reported our experience with this strategy, which yielded low rates of ischemic complications. 20
Patients with diabetes have a prothrombotic state, characterized by elevated levels of fibrinogen and plasminogen activator inhibitor-1, as well as platelet hyper-reactivity.21,22 Despite this, major hematoma occurred more frequently in patients with diabetes. Possible reasons that may explain this include a smaller diameter of the common femoral artery, which may increase the level of difficulty in obtaining vascular access. Patients with metabolic syndrome have a higher risk of developing diabetes. These patients often have increased waist and abdomen measurements, making it more difficult to obtain optimal vascular access. Common femoral arteries are more heavily calcified in patients with diabetes, which may have led to suboptimal hemostasis when vascular closure devices were used.
Females with diabetes had a higher risk of MALE. Our results are similar to the outcomes observed in previous studies examining gender-related differences in the outcomes of patients with PAD undergoing endovascular treatment. In a study of 372,692 PAD-related surgical hospital admissions, women had a higher risk of postoperative complications, major amputation, and death, even after adjusting for baseline covariates. 23
Limitations
This was a retrospective analysis at a single center with a relatively small number of patients. This study included patients with either lifestyle-limiting claudication as well as critical limb ischemia. Coronary angiographic follow-up was not available for all patients. Furthermore, peripheral vascular imaging with invasive angiography, computed tomography angiography, or arterial duplex to determine the restenosis rates were not available for all patients. A complete-case analysis was performed as cases with missing variables were excluded from PSM. 24 However, this method introduces bias. 25
Conclusion
Among patients undergoing endovascular revascularization for symptomatic PAD, those with diabetes did not have increased rates of late adverse cardiovascular outcomes compared to patients without diabetes. Bleeding avoidance strategies including optimal pharmacotherapy and meticulous vascular access techniques are needed in patients with diabetes to reduce the risk of bleeding and its deleterious impact on morbidity and mortality. In addition to disease management and associated risk factor reduction, it is important to continue assessing current management strategies and investigating new approaches to improve long-term outcomes in this patient population.
Supplemental Material
10.1177_1358863X19879751_Supplementary_tables – Supplemental material for Impact of diabetes mellitus on 5-year clinical outcomes following successful endovascular revascularization for peripheral artery disease
Supplemental material, 10.1177_1358863X19879751_Supplementary_tables for Impact of diabetes mellitus on 5-year clinical outcomes following successful endovascular revascularization for peripheral artery disease by Michael S Lee, Byoung Geol Choi and Seung-Woon Rha in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
