Abstract

Patients with peripheral artery disease (PAD) are at increased risk for major adverse cardiovascular events (MACE) and major cardiovascular limb events (MALE), with those after prior lower extremity revascularization (LER) at particularly high risk of the latter. 1 Guideline-directed medical therapies (GDMT) reduce the risk of MACE and MALE in PAD patients, including lipid-lowering therapy, antithrombotic therapy, and angiotensin-converting enzyme inhibitor (ACEi)/angiotensin receptor blockers (ARB).2,3 The 2021 American Heart Association (AHA) Scientific Statement on PAD highlighted underuse of GDMT despite a guideline update 5 years previously. 4 Prior analyses have observed differences in outcomes on the basis of sex, and differences in diagnosis and treatment may contribute to these observations.5,6
We examined sex differences in the implementation of GDMT to understand whether subpopulations with PAD should be targeted to improve care. Using de-identified data from TriNetX (Cambridge, MA, USA), which collects real-world data from electronic medical records, 7 we identified adults with healthcare encounters with a diagnosis of PAD from May 2021 to April 2022 in the University of Colorado health system using International Classification of Diseases, 10th Revision (ICD-10) codes, as previously described. 8 Those with symptomatic PAD were identified by ICD codes for intermittent claudication, ischemic rest pain, ischemic ulceration, and gangrene. 8 Categories were compared by chi-squared analysis. The Colorado Multiple Institutional Review Board provided a waiver of informed consent.
Among 9810 patients with PAD identified in the dataset, 50% (n = 4910) were women. Men had a higher prevalence of hypertension, hyperlipidemia, diabetes mellitus (DM), current or prior smoking, coronary artery disease (CAD), and chronic kidney disease (CKD). Moreover, previous peripheral revascularization, amputation, and symptomatic PAD were more frequent in men (all p < 0.0001, online Supplementary Table 1). Overall, patients with PAD had low rates of GDMT regardless of sex and symptoms. Women were less likely than men to receive GDMT. In addition, in the overall PAD cohort, medication use was lower in patients without comorbid CAD, with the lowest rates in women without a history of CAD including statin use of (38%), antiplatelet therapy (53%), and ACEi/ARB (43%) (all p < 0.0001) (Figure 1: upper panel). Among patients with symptomatic PAD, GDMT was less often prescribed in women than men irrespective of concomitant CAD (Figure 1: lower panel). Even among high-risk patients with concurrent DM or CKD, women were less likely to receive ACEi/ARB than men (all p < 0.0001). Among patients with diabetes, women had poorer glycemic control than men, with hemoglobin A1c < 7.0% in 25% versus 28% (p < 0.01). Among patients with LER, GDMT was numerically less often prescribed in women than men: statin (84% vs 90%, p = NS), ACEi/ARB (74% vs 79%, p = NS), and dual antiplatelet therapy (84% vs 89%, p = NS). Finally, GDMT was less often prescribed among Black women compared with White women: statin (35% vs 43%, p < 0.01), antiplatelet therapy (50% vs 63%, p < 0.01), and ACEi/ARB (42% vs 50%, p = 0.01).
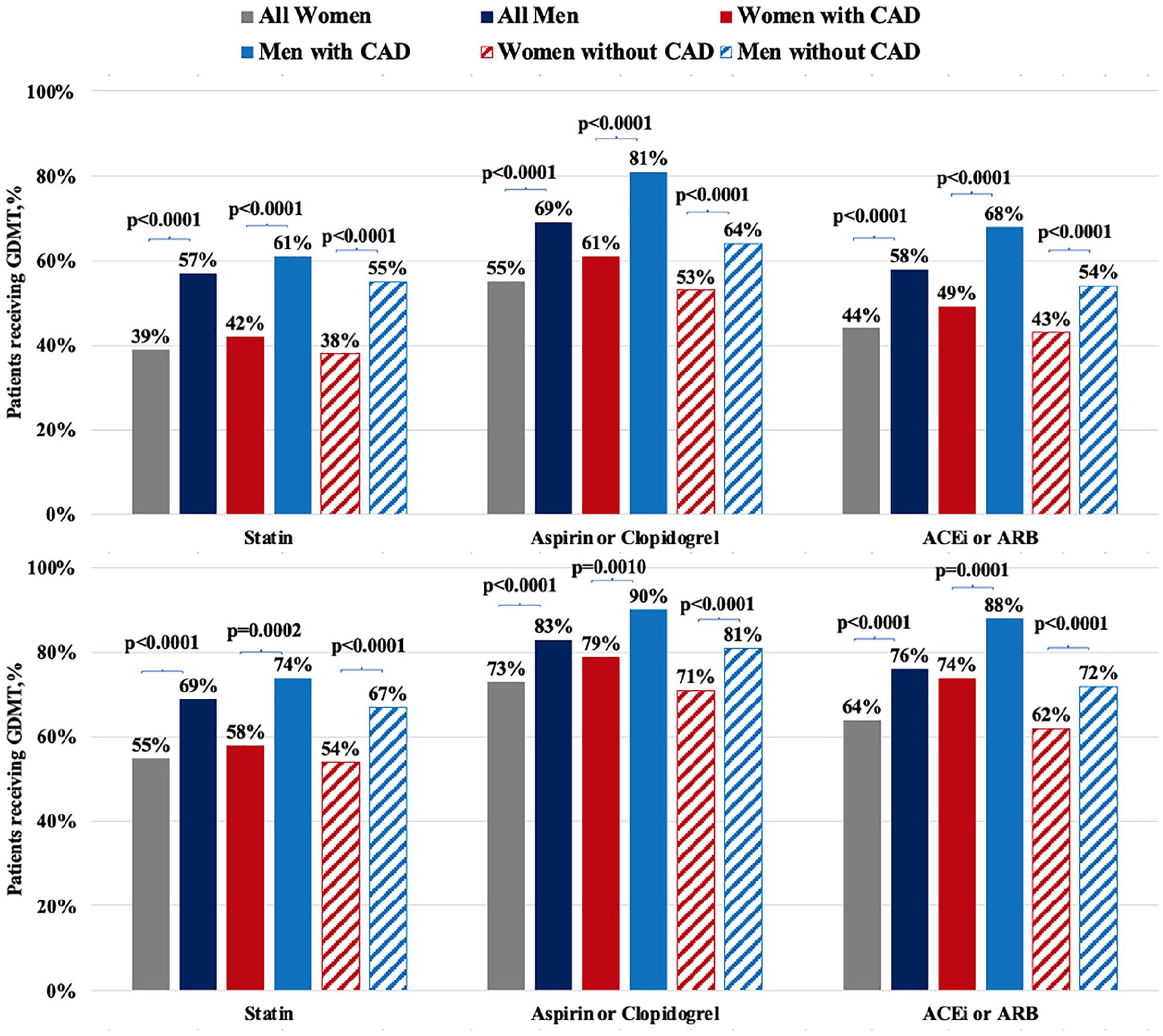
GDMT use in women and men with PAD overall and with or without concomitant CAD. The upper panel describes the overall cohort of patients with PAD and the lower panel includes only patients with symptomatic PAD.
This contemporary real-world analysis represents a snapshot of the 2021–22 GDMT prescription in a diverse population of patients with PAD receiving care across the spectrum from large academic centers to small rural practices. We observed sex-based differences in use of GDMT in patients with PAD, with overall low use and even lower GDMT prescription among women, irrespective of concurrent CAD, CKD, DM, symptomatic PAD or after LER. Moreover, Black women were less likely to receive GDMT than White women. Even with updated guidelines and a recent AHA call to action, less than half of women with PAD were receiving statins or ACEi/ARB regardless of known concomitant CAD, despite having the same risk of adverse limb events as men in the EUCLID and VOYAGER PAD trials.5,6 Although this analysis focused on pharmacologic therapy, GDMT includes smoking cessation and supervised exercise therapy 4 ; 14% of women and 21% of men in the cohort reported current smoking and we have previously reported a very low rate (3%) of referral within this health system for supervised exercise following LER. 9 Despite some discrepancies among specialty-specific guideline recommendations (e.g., ACEi were not included in Society for Vascular Surgery guidelines – 2015), each component (i.e., lifestyle, medications, and exercise therapy) of GDMT has been shown to improve outcomes, function, and/or quality of life, particularly in high-risk patients such as those with previous LER. 4
Limitations of this analysis include its use of retrospective, albeit recent, pooled observational data which lacked patient-level information. In addition, these findings reflect a single, large health system. Finally, owing to the nature of the data, we were unable to assess lifestyle modification such as smoking cessation and physical activity.
Understanding the key drivers, including provider behaviors, patient preferences, and costs, other issues should be the focus of future studies. Targeted efforts to understand reasons for low utilization can be conducted in parallel with implementation science studies to identify practical and effective approaches to improving the health care of patients with PAD, particularly focused in high-risk subpopulations patients. 10
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231155308 – Supplemental material for Sex differences in guideline-directed medical therapy in 2021–22 among patients with peripheral artery disease
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231155308 for Sex differences in guideline-directed medical therapy in 2021–22 among patients with peripheral artery disease by Mario Enrico Canonico, Judith Hsia, Connie N Hess and Marc P Bonaca in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs Hsia, Hess, and Bonaca receive salary support through their universities from CPC Clinical Research, a nonprofit academic research organization affiliated with the University of Colorado, that receives research grant/consulting funding from: Abbott, Agios, Alexion Pharma, Alnylam, Amgen, Angionetics, ARCA Biopharma, Array, AstraZeneca, Atentiv, Audentes, Bayer, Better Therapeutics, Brigham and Women’s Hospital, Bristol Myers Squibb, Cardiol Therapeutics, CellResearch, Cook Medical, Cook, CSL Behring, Eidos Therapeutics, EP Trading Co, Esperion Therapeutics, EverlyWell, Faraday, Fortress Biotech, HDL Therapeutics, HeartFlow, Hummingbird Bioscience, Insmed, Janssen, Kowa Research, Lexicon, Merck, Medtronic, Moderna, Novate Medical, Novo Nordisk, Pfizer, PhaseBio, PPD Development, Prairie Education and Research, Prothena Biosciences, Regeneron, Regio Biosciences, Sanifit Therapeutics, Sanofi, Smith and Nephew, Stealth BioTherapeutics, University of Colorado, University of Pittsburgh, Worldwide Clinical Trials, Wraser, and Yale Cardiovascular Research Group. Dr Hsia also reports owning AstraZeneca stock. Dr Bonaca reports salary support from an AHA SFRN under award numbers 18SFRN3390085 (BWH-DH SFRN Center) and 18SFRN33960262 (BWH-DH Clinical Project), stock in Medtronic and Pfizer, and consulting fees from Audentes. Dr Canonico has no disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
