Abstract
Patients undergoing major vascular surgery have an increased risk of perioperative major adverse cardiovascular events (MACE). Accordingly, in this population, it is of particular importance to appropriately risk stratify patients’ risk for these complications and optimize risk factors prior to surgical intervention. Comorbidities that portend a higher risk of perioperative MACE include coronary artery disease, heart failure, left-sided valvular heart disease, and significant arrhythmic burden. In this review, we provide a current approach to risk stratification prior to major vascular surgery and describe the strengths and weaknesses of different cardiac risk indices; discuss the role of noninvasive and invasive cardiac testing; and review perioperative pharmacotherapies.
Keywords
Introduction
Between 2000 and 2010, the prevalence of patients worldwide with peripheral artery disease increased by 23.5%, with a corresponding increase in the rate of annual vascular interventions.1,2 As compared to most other noncardiac operative interventions, major vascular procedures (e.g., open abdominal aortic aneurysm (AAA) repair, infrainguinal bypass surgery, open reconstruction of the renal or mesenteric arteries) are associated with a significantly elevated risk of major adverse cardiovascular events (MACE). 3 Among a Medicare population undergoing elective open or endovascular surgery, the prevalence of adverse events was ⩾ 20% – lowest in those undergoing endovascular aortic procedures and highest in those undergoing open aortic procedures. 4 The level of procedural risk attributable to different vascular interventions is shown in Table 1.
Level of procedural risk of different vascular interventions.

MACE, major adverse cardiovascular events.
There are several features inherent to vascular procedures that increase the risk of MACE. This may be due to large intravascular and extravascular fluid shifts, prolonged duration of aortic cross-clamping with aortic procedures, large volume blood and insensible fluid losses, and hypothermia induction. This is associated with a heightened stress response, characterized by elevated circulating levels of cortisol and catecholamines in the operative and perioperative period, promoting a hypercoagulable state and increased myocardial demand. 5
Postoperative myocardial infarction occurs frequently following major vascular surgery, which may reflect the high prevalence of coronary artery disease in this population. 6 Postoperative myocardial infarction has been independently associated with poor prognosis due to an increased risk of future cardiac events, including 30-day cardiac arrest, all-cause mortality, and 6-month MACE.7,8 Open aortic procedures are especially associated with a higher risk of postoperative myocardial infarction, which is in turn associated with up to 650% increased risk of 1-year mortality.9,10 Although postoperative stroke in noncarotid vascular surgery is rare (< 1%), it is associated with a significantly increased risk of 30-day perioperative mortality (approximately three to sixfold increase).11,12
Postoperative arrhythmias affect a large percentage of patients undergoing noncardiac surgery, with the incidence of 7–8% – most frequently atrial fibrillation (~4.4%). 13 The risk is even higher in those undergoing vascular surgery (10–11%).14,15 In a retrospective cohort study of > 120,000 patients undergoing major vascular surgery, the incidence of clinically significant postoperative arrhythmias (requiring medication or cardioversion) was ~5%, with the highest risk in those undergoing open AAA repair (14.4%). 16
In addition to risks inherent to surgery, patients are at a high risk of MACE due to underlying medical comorbidities that are prevalent in this patient population. Risk factors associated with peripheral vascular diseases often mirror those of coronary artery disease. As such, a significant number of patients undergoing major vascular surgery have underlying coronary artery disease.3,17 Physical limitations in this population may be concealed by their underlying vascular disease, leading to both an inaccurate representation of their symptoms and preoperative assessment of their surgical readiness. In order to minimize the risk of cardiac events in the peri-procedural period, a comprehensive preoperative cardiovascular evaluation is of upmost importance.
Here, we highlight important recommendations made by the American College of Cardiology/American Heart Association (ACC/AHA), European Society of Cardiology/European Society of Anaesthesiology (ESC/ESA), and Canadian Cardiovascular Society (CCS) on perioperative cardiovascular evaluation and management of patients undergoing noncardiac surgery,18 –20 and the evidence for these recommendations as it pertains to patients undergoing vascular surgery. These recommendations are summarized in Table 2. The suggested approach to preoperative evaluation is shown in Figure 1.
ACC/AHA, American College of Cardiology / American Heart Association; CCS, Canadian Cardiovascular Society; ESC/ESA, European Society of Cardiology / European Society of Anaesthesiology; MACE, major adverse cardiovascular events; METs, metabolic equivalents.
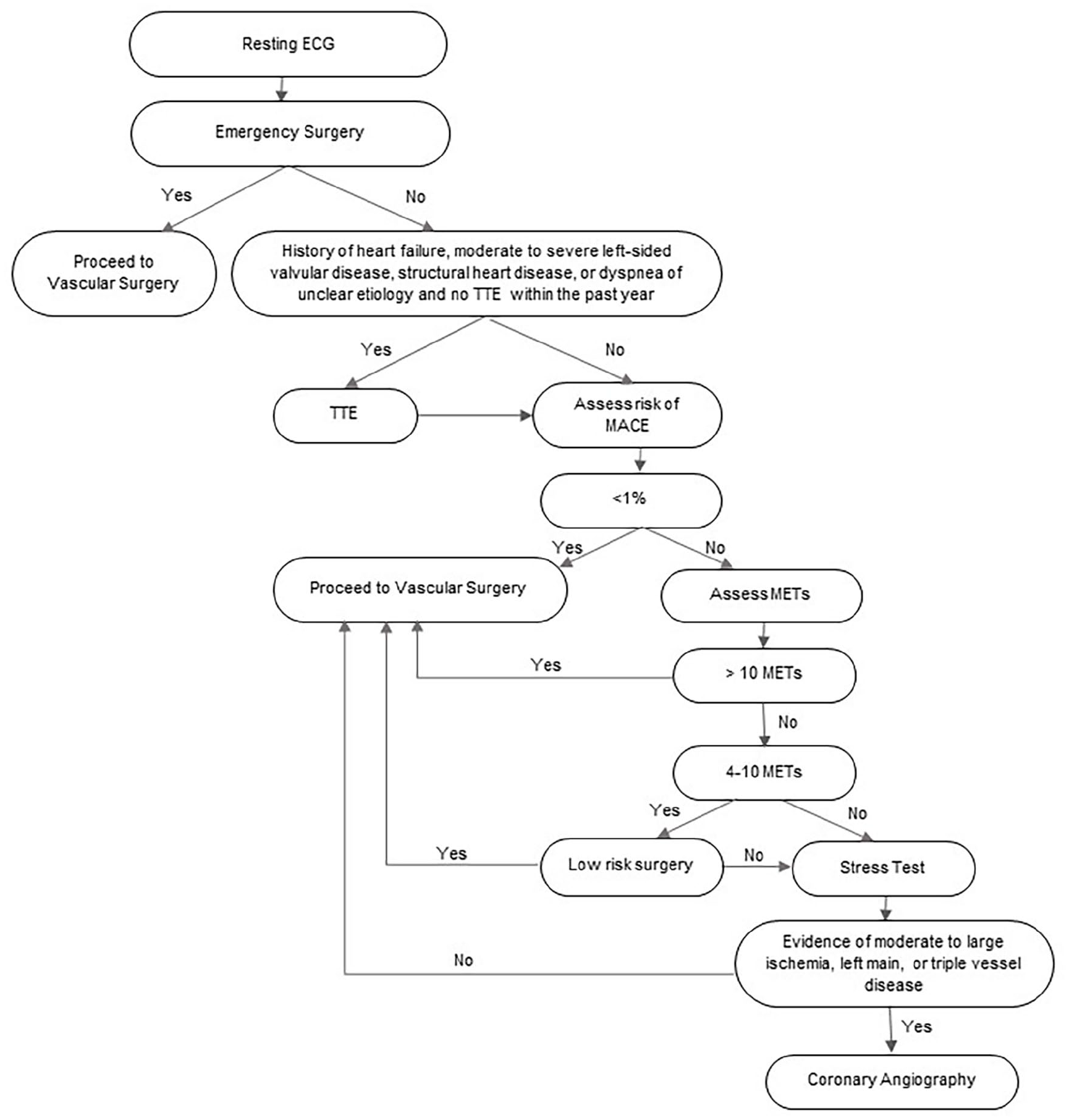
Preoperative evaluation prior to major vascular surgery.
History and physical exam
A preoperative cardiovascular evaluation should start with a detailed history and physical examination. The history should include a thorough investigation of cardiac symptoms (e.g., angina, dyspnea) along with the patient’s functional status and physical limitations. The presence of acute coronary syndrome should delay elective vascular surgery, as the risk of 30-day and 1-year mortality is markedly increased if surgical intervention is pursued. 21 Findings of decompensated congestive heart failure should be investigated before proceeding with surgery.
Cardiac risk indices
Revised Cardiac Risk Index (RCRI)
There are various risk prediction models that have been derived to aid clinicians in the preoperative evaluation of patients undergoing surgical procedures. The original preoperative risk prediction index was first published by Goldman et al. in 1977 and revised by Lee et al. in 1999.22,23 The simplicity and general accuracy of this surgical risk prediction index has made it an oft-cited and utilized prediction tool. The components of this risk model include high-risk surgery (intraperitoneal, intrathoracic, or supra-inguinal vascular surgery); history of ischemic heart disease, heart failure, or cerebrovascular disease; preoperative treatment with insulin; or preoperative creatinine greater than 2 mg/dL. The RCRI has been found to discriminate moderately well between low and high-risk patients for all types of noncardiac surgery. However, a meta-analysis of 24 studies with more than 792,000 patients found that the ability of the index to predict cardiac events after vascular surgery was not as accurate as compared to other noncardiac surgeries. 24
Vascular Surgery Group of New England Cardiac Risk Index (VSG-CRI)
In 2010, Bertges et al. derived a surgical risk prediction index, the VSG-CRI, with the goal of creating a preoperative risk stratification tool specifically for patients undergoing vascular surgery, as RCRI significantly underestimated the risk of in-hospital MACE in these patients (6.5 to 7.4-fold for low-risk patients and 1.7-fold for high-risk patients). 25 The components of the VSG-CRI include: age; history of coronary artery disease, congestive heart failure, or chronic obstructive pulmonary disease (COPD); creatinine greater than 1.8 mg/dL; smoking status; insulin-dependent diabetes; and long-term beta-blockade. Procedure-specific cardiac risk models were developed for carotid endarterectomy, open AAA repair, endovascular AAA repair, and lower-extremity bypass. The models best predict adverse events in those undergoing carotid endarterectomy (receiver operating characteristic [ROC] 0.75) and perform the worst in those undergoing open AAA repair (ROC 0.69).
Gupta / MICA score
A risk calculator based on the National Surgical Quality Improvement Program (NSQIP) was developed in 2011 by Gupta et al. 26 The components of the model include age, functional status, American Society of Anesthesiologists (ASA) classification, creatinine greater than 1.5 mg/dL, and type of surgery to predict the risk of intraoperative or postoperative myocardial infarction or cardiac arrest. When the RCRI and Gupta/MICA scores were both applied to the 2008 NSQIP registry, the Gupta/MICA score had a better ability to predict myocardial infarction or cardiac arrest in patients undergoing aortic or noncardiac vascular surgery (C-statistics 0.75 vs 0.71).
American University of Beirut-Pre-Operative Cardiovascular Evaluation Study (AUB-POCES) / AUB-HAS2
The AUB-HAS2 cardiovascular risk index was developed in 2019, validated using the NSQIP database. 27 The components include age ⩾ 75 years, history of heart disease, symptoms of angina or dyspnea, hemoglobin < 12 mg/dL, any vascular surgery, and emergency surgery. Only 4.5% of the original derivation cohort was comprised of patients undergoing vascular surgery, yet the risk of death, myocardial infarction, or stroke was highest in this population (6.7% prevalence of death, myocardial infarction, or stroke at 30 days). The AUB-HAS2 risk index had better discriminatory power than the RCRI, including in patients undergoing vascular surgery.28,29
Noninvasive testing
Resting electrocardiogram (ECG)
Given the large proportion of patients undergoing major vascular surgery who have underlying coronary artery disease, an ECG is of particular importance.3,17,30 The ECG should be evaluated for the presence of active myocardial ischemia, prior myocardial infarction, atrial or ventricular arrhythmias, conduction abnormalities, and QT prolongation. A preoperative ECG also provides a useful baseline for future comparison. ACC/AHA and ESC/ESA guidelines recommend a preoperative ECG if patients are undergoing intermediate to high-risk surgical intervention, and have known cardiovascular disease, arrhythmias, or structural heart disease.19,20 The CCS does not give a formal recommendation on the acquisition of a preoperative ECG. 18
Resting echocardiogram
In patients undergoing intermediate and high-risk vascular surgery, ACC/AHA guidelines recommend that a resting transthoracic echocardiogram (TTE) should be obtained in the preoperative period if there is any clinical suspicion for decompensated heart failure, valvular disease, structural heart disease, or dyspnea of unclear etiology. 19 In contrast, ESC/ESA guidelines only recommend a resting TTE for patients undergoing high-risk surgery and the CCS recommends against routinely obtaining a TTE unless physical examination suggests severe valvular disease, pulmonary hypertension, or an undiagnosed cardiomyopathy.18,20
Stress testing
ACC/AHA guidelines recommend assessment with stress testing based on operative risk, which is determined with the use of a preoperative risk calculator, such as the RCRI. 19 However, stress testing should only be considered in patients who are undergoing nonemergent surgical intervention. Patients with vascular emergencies should not undergo additional testing prior to operative intervention as surgical intervention may be unnecessarily delayed. In patients who may be appropriate candidates for preoperative stress testing, patients at low risk of postoperative MACE (< 1%) do not require stress testing prior to major vascular surgery. However, patients who are at higher risk of postoperative MACE (⩾ 1%), should be further risk-stratified based on functional status. In fact, evaluation of a patient’s functional status is particularly important. Patients who regularly achieve greater than 10 metabolic equivalents (METs) without any limiting symptoms have excellent functional capacity and can proceed without further ischemic evaluation. In patients who can achieve 4–10 METs of activity, noninvasive stress testing may be considered if there is a high operative risk. If a patient’s functional status is unknown or less than 4 METs, an ischemic workup is justified, if the results will change preoperative management (e.g., pharmacotherapy, performance of invasive angiography, or choice of surgical procedure). 19 Poor preoperative functional status (< 4 METs) and/or preoperative frailty are associated with higher risk of perioperative MACE, including cardiac arrest, perioperative myocardial infarct, or 30-day readmission.31 –34 If possible, an exercise-based stress test is preferred over pharmacologic modalities to allow for quantification of functional capacity. Figure 2 shows the ACC/AHA guidelines on stress testing in the preoperative setting in patients undergoing noncardiac surgery.
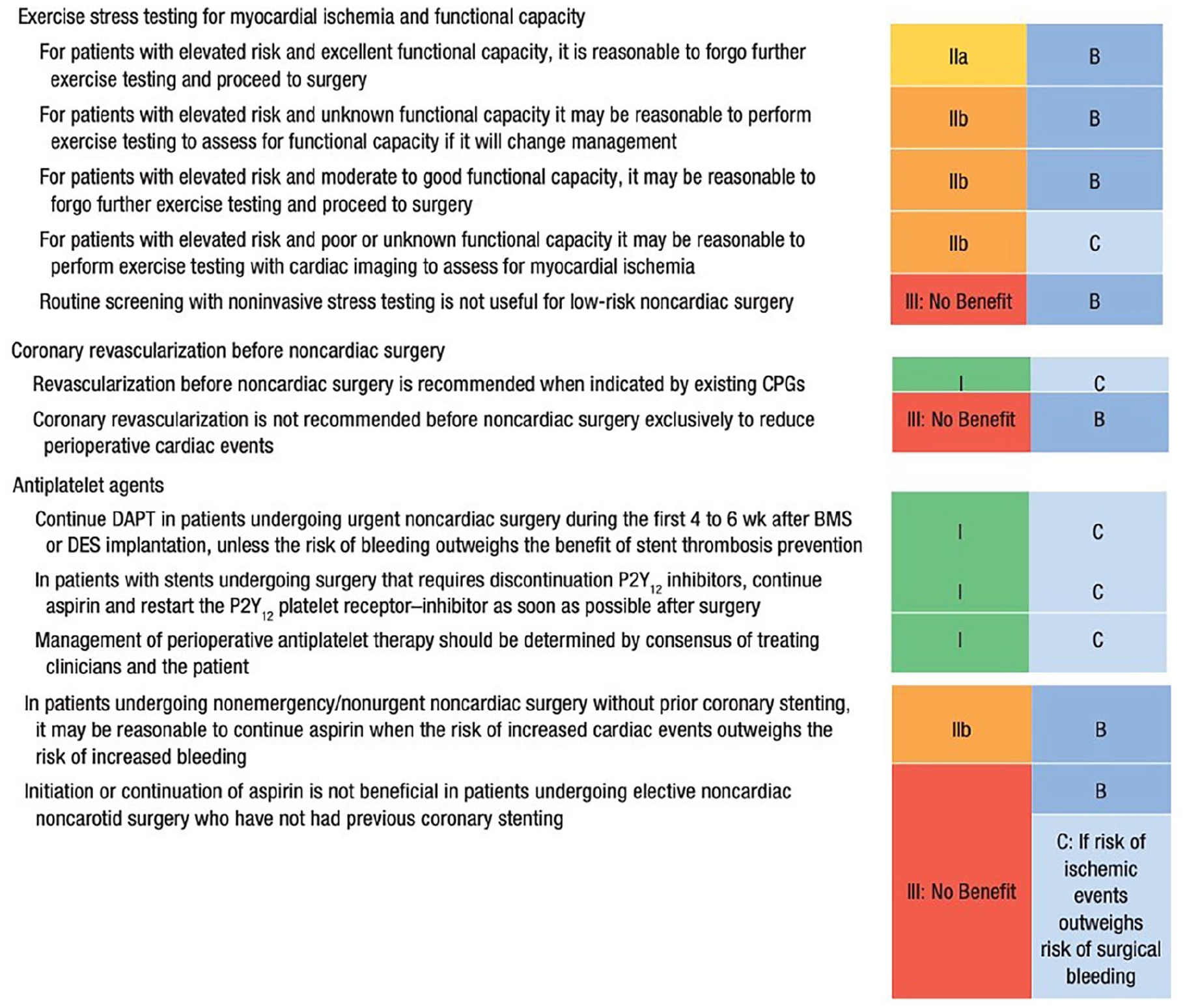
ACC/AHA guidelines on exercise stress testing for myocardial ischemia and functional capacity, coronary revascularization, and antiplatelet recommendations prior to noncardiac surgery. First column is class of recommendation. Second column is level of evidence. ‘A’ is data derived from multiple randomized clinical trials or meta-analyses of these trials; ‘B’ is data derived from one or more randomized trials or meta-analyses of these trials; ‘C’ is data derived from non-randomized trials.
The ESC/ESA only recommends a preoperative stress test in patients undergoing high-risk surgery who achieve < 4 METs and have more than two clinical risk factors (ischemic heart disease, heart failure, stroke/transient ischemic attack, renal dysfunction, or insulin-dependent diabetes mellitus) (class I recommendation); or in patients undergoing intermediate or high-risk surgery who can achieve < 4 METs and have one or two clinical risk factors (class IIb recommendation). 20 The CCS recommends against performing preoperative stress testing regardless of patient functional status due to costs and delays associated with testing without significant improvement in operative risk. 18
Some centers routinely perform noninvasive stress testing prior to major vascular surgical intervention, irrespective of the specific guideline recommendations noted above.35,36 Higher frequency of stress testing was not associated with lower risk of MACE, 36 and patients with a negative stress test did not have a lower risk of postoperative MACE compared with patients who did not have a stress test. 35 Thus, there appears to be no benefit to routine stress testing prior to major vascular intervention.
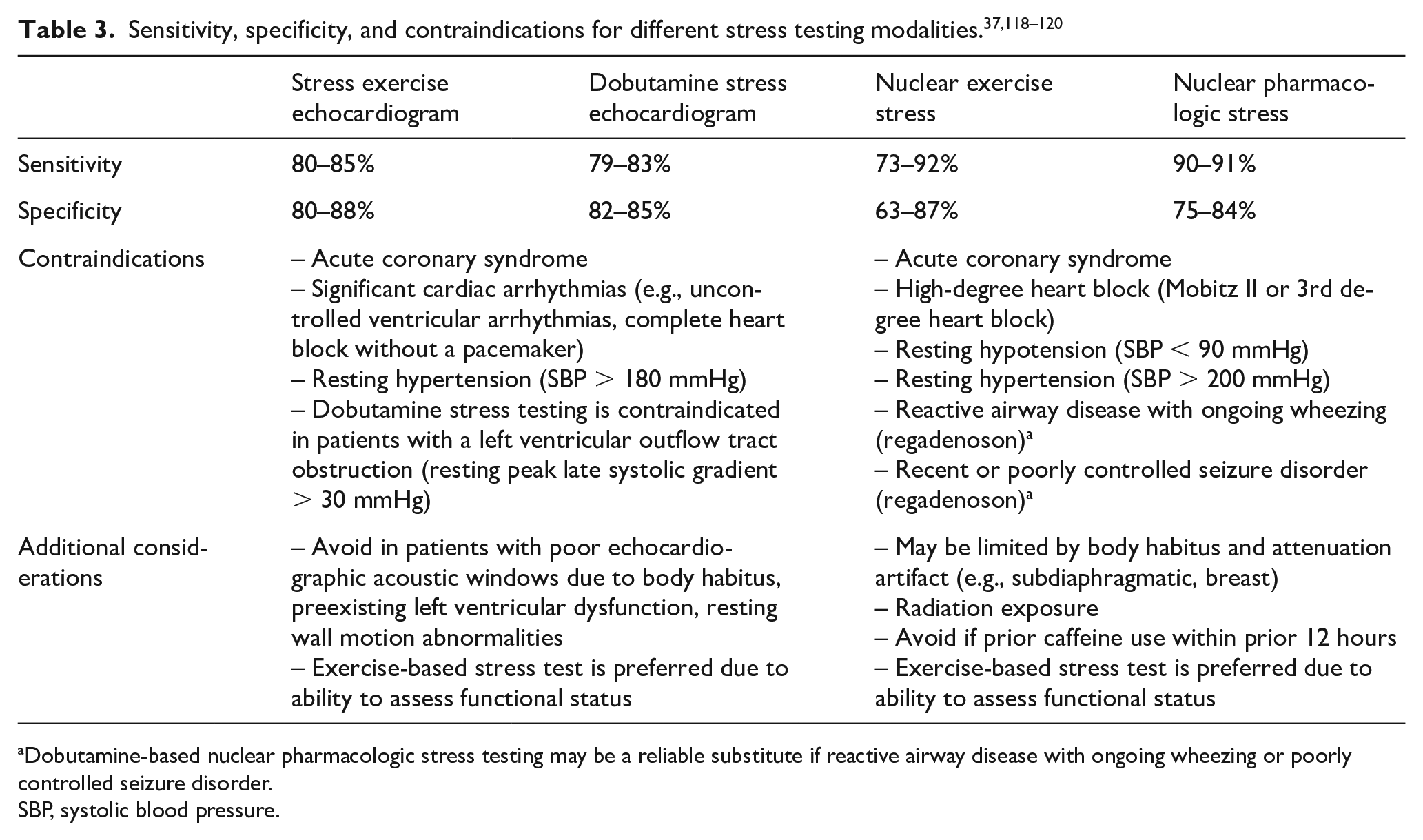
There are several modes of stress testing that can be used to assess cardiac risks in patients undergoing major vascular surgery. The sensitivities, specificities, and contraindications to echocardiographic and nuclear-based stress testing modalities are shown in Table 3 and a suggested algorithm to guide modality of stress testing is shown in Figure 3. Availability and local expertise in performing each of the different modalities of stress testing should also help to guide the optimal choice of ischemic evaluation.
Dobutamine-based nuclear pharmacologic stress testing may be a reliable substitute if reactive airway disease with ongoing wheezing or poorly controlled seizure disorder.
SBP, systolic blood pressure.
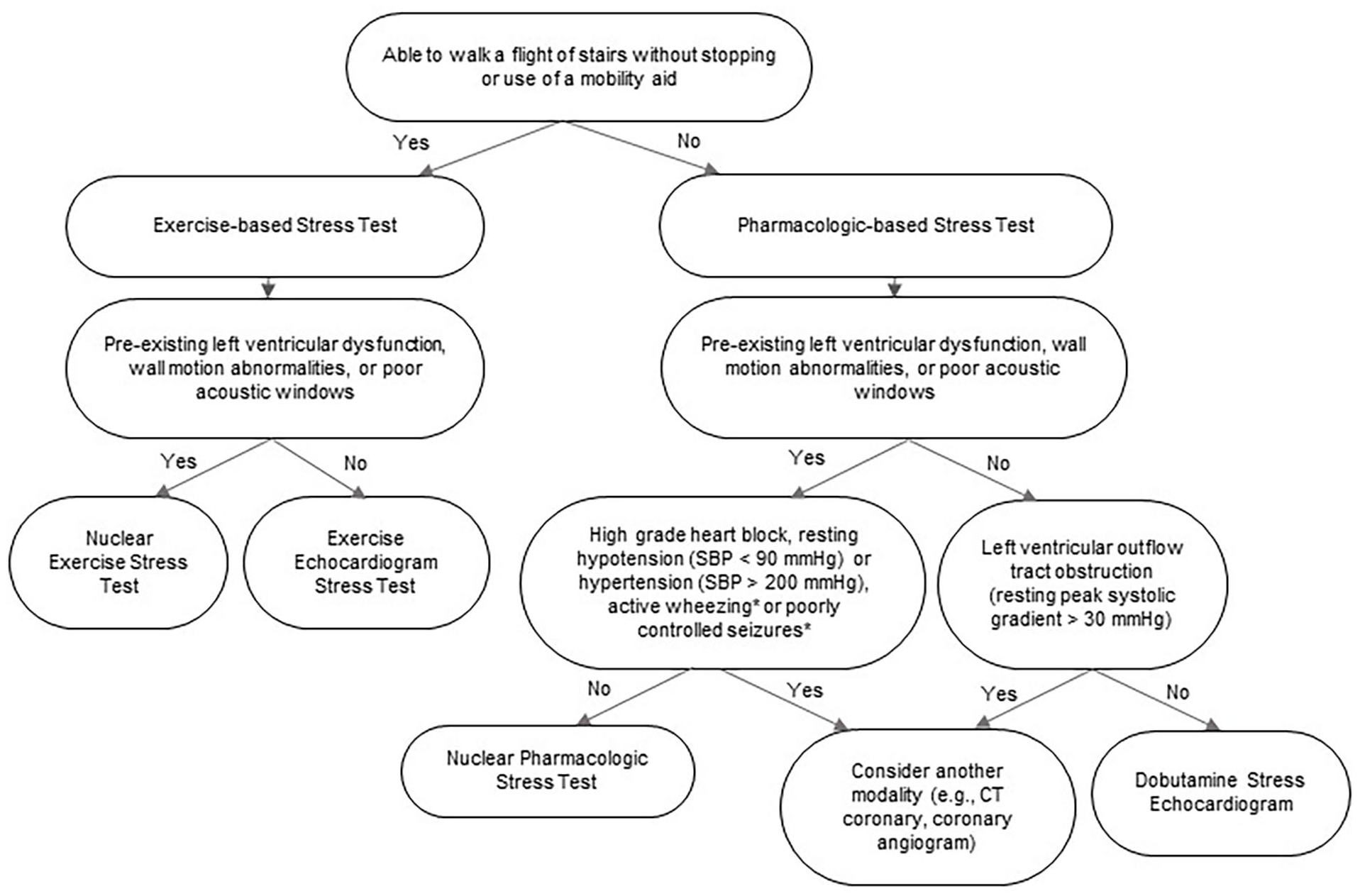
Algorithm to guide stress testing modality prior to major vascular surgery.
Stress echocardiography is based on the premise that obstructive coronary artery disease will manifest as a regional wall motion abnormality in the vascular territory on echocardiogram at peak stress. Other abnormal stress echocardiographic findings include transient ischemic left ventricular dilation (TID), a decrease in left ventricular function with stress, or a myocardial biphasic response with dobutamine, during which left ventricular function augments with low-dose dobutamine but decreases with higher doses of dobutamine. 37 The stress echocardiogram can be performed via exercise (treadmill or bicycle) or pharmacologically. Pharmacologic-based stress echocardiograms are usually performed with an inotrope; dobutamine (DSE) is the most commonly used agent in the United States. 38 DSE has not been extensively studied in patients with stable AAA but appears to be safe and well-tolerated, with few case reports of AAA expansion or rupture.39,40 However, two single-center studies (total of 347 patients, majority with AAA diameter > 5 cm) did not show an increased incidence of aneurysmal rupture or hemodynamic instability with DSE.41,42
Radionuclide myocardial perfusion imaging (rMPI) stress testing utilizes a radioactive tracer that is administered intravenously at peak stress. Following injection, gamma photons are captured either via single photon emission computer tomography (SPECT) or positron emission tomography (PET). Images are obtained at both rest and following stress to detect changes in regional tracer uptake and myocardial perfusion. Other parameters, such as TID, reduced post-stress left ventricular ejection fraction, and stress-induced wall motion abnormalities, can also be detected to indicate underlying coronary artery disease. Myocardial blood flow (MBF) can routinely be quantified with PET stress, aiding in the detection of coronary disease. 43 Several radioactive tracers can be utilized for SPECT rMPI, the most common of which are technetium-99m or thallium-201. Thallium is less commonly used because it is associated with higher radiation as compared to technetium. For PET rMPI, the most commonly utilized radioactive tracers are rubidium-82 or ammonia N-13. With SPECT, similar to echocardiography-based stress testing, rMPI can be accomplished via an exercise or pharmacologic modality with the use of vasodilators (e.g., adenosine, dipyridamole, regadenoson) or dobutamine. With PET, only pharmacologic modalities are used, given the short half-life of the radiotracers used with PET.
Invasive testing
Coronary angiography and routine revascularization of coronary artery disease
The ACC/AHA guidelines suggest that invasive coronary angiography prior to major vascular surgery should only be pursued in patients with active cardiac ischemia who would otherwise require angiography per standard indications (Figure 2). 19 The guidelines from the ESC/ESA and CCS provide similar recommendations.18,20
The largest randomized trial evaluating routine preoperative coronary artery revascularization prior to major vascular surgery was the Coronary Artery Revascularization Prophylaxis (CARP) trial, which randomized patients with stable ischemic heart disease undergoing elective vascular surgery to preoperative coronary revascularization or no revascularization. 44 There was no significant difference in 30-day risk of MACE or long-term mortality between the two groups. However, a criticism of the CARP trial was its exclusion of higher-risk patients, such as those with significant left main disease, a left ventricular ejection fraction of less than or equal to 20%, or severe aortic stenosis, leaving a trial population of patients with lower-risk disease (mostly single or two-vessel coronary artery disease). A post hoc analysis of patients screened for the CARP trial found that preoperative coronary artery revascularization was associated with mortality benefit in patients with significant left main disease. 45 In a smaller single-center prospective study randomizing all-comers to selective (coronary angiography based on the results of noninvasive stress testing) or a systematic (routine coronary angiography) strategy prior to elective vascular surgery, there was no difference in in-hospital MACE. 46 Long-term follow-up demonstrated a lower rate of MACE in those undergoing a systematic strategy, likely due to the increased detection of multivessel and left-main coronary artery disease. Coronary angiography and subsequent revascularization prior to major vascular surgery should be focused on the urgency of surgical intervention, individual symptoms, risk factors, and coronary anatomy (if known).19,47 If surgical intervention can be delayed, a coronary angiogram should be considered for patients with moderate to large areas of reversible ischemia, or concern for high-risk multivessel or left main disease (e.g., transient ischemic dilation).
Perioperative management of comorbidities
Heart failure
In the preoperative evaluation, the presence of decompensated heart failure should prompt further assessment prior to major vascular surgery. Patients with either acute or chronic heart failure have worse outcomes as compared to patients without a history of heart failure. In a study utilizing Medicare claims data, individuals undergoing above and below-knee amputations, lower-extremity bypass, or open AAA repair, the 30-day risk of mortality was up to twice as high for patients with a history of heart failure compared to those without heart failure. 48
The risk of mortality in patients with heart failure undergoing major vascular surgery may even outweigh that posed by coronary artery disease. A Canadian population-level study of more than 38,000 patients with heart failure, atrial fibrillation, or coronary artery disease assessed the risk posed by each of these conditions in patients undergoing noncardiac surgery. 49 In adjusted analyses, patients with ischemic and nonischemic cardiomyopathy were at an approximately two and an approximately three times increased risk of 30-day postoperative mortality, respectively, compared to individuals with coronary artery disease without cardiomyopathy. In a Veterans Affairs retrospective cohort study of patients undergoing noncardiac surgery, patients with symptomatic heart failure (either preserved or reduced ejection fraction) had a 10.1% risk of mortality within 90 days, as compared to 4.9% in patients with asymptomatic heart failure, and 1.2% if no history of heart failure. 50 Patients with an ejection fraction < 30% had a risk of mortality that was 50% to 100% higher as compared to patients with an ejection fraction of ⩾ 40%.
Valvular heart disease
Patients with moderate to severe stenotic and regurgitant disease of the mitral and aortic valves should have a transthoracic echocardiogram in the preoperative setting if there has not been an echocardiogram completed within the prior year. 19 Based on original RCRI data, severe aortic stenosis was associated with significantly higher risk of perioperative mortality (13.6% vs 1.6% in those without severe aortic stenosis). 22 However, a meta-analysis of nine studies with over 29,000 patients with severe aortic stenosis undergoing noncardiac surgery found no significant difference in mortality risk between patients with and without severe aortic stenosis, unless symptomatic, in which case they had a significantly higher risk of perioperative myocardial infarction and mortality. 51 The ACC/AHA and ESC valvular heart disease guideline recommendations are reflective of these findings.52,53 In symptomatic patients who meet standard indications for aortic valve replacement (AVR), AVR should be pursued prior to noncardiac surgery. In asymptomatic patients with severe aortic stenosis without a decreased left ventricular ejection fraction or symptomatic obstructive coronary artery disease, it is reasonable to proceed with low to intermediate-risk operative intervention with close hemodynamic monitoring. However, for high-risk operative interventions, such as major vascular surgery, aortic valve intervention with an AVR or balloon valvuloplasty should be considered.52,53
As compared to aortic stenosis, less is known about the impact of mitral stenosis in patients undergoing noncardiac surgery. Similar to aortic stenosis, the ACC/AHA and ESC valvular heart disease guidelines discuss the importance of symptoms when making preoperative decisions.52,53 In asymptomatic patients with severe mitral stenosis and a pulmonary artery systolic pressure of less than 50 mmHg, it is reasonable to perform elective noncardiac surgery without any valvular intervention. The presence of pulmonary hypertension due to mitral stenosis should lead to consideration of valvular intervention prior to major vascular surgery.54,55 The ACC/AHA and ESC valvular heart disease guidelines suggest that in patients with symptomatic severe mitral stenosis, mitral valve intervention should be attempted prior to major vascular surgery. In patients with rheumatic mitral stenosis and favorable anatomy, percutaneous mitral commissurotomy can be considered.52,53
Limited studies on patients with moderate to severe mitral regurgitation undergoing noncardiac surgery found higher risks of adverse perioperative outcomes, such as myocardial infarction, heart failure, stroke, and short-term mortality.56,57 Similarly, moderate to severe aortic regurgitation is associated with higher risks of intraoperative hemodynamic instability, circulatory collapse, and in-hospital mortality. 58 Still, left-sided regurgitant lesions appear to be better tolerated in the perioperative setting than stenotic lesions. ACC/AHA and ESC/ESA guidelines recommend that patients with mitral or aortic regurgitation undergo noncardiac surgery without preoperative valvular intervention if left ventricular ejection fraction is preserved and the patient is asymptomatic.19,20 The CCS does not give a formal recommendation on the preoperative management of valvular disease prior to surgical intervention. 18
Arrhythmias
Patients with a history of significant arrhythmias, such as rapid supraventricular arrhythmias (e.g., atrial fibrillation or flutter), sustained ventricular arrhythmias, or significant conduction system disease (e.g., complete heart block, Mobitz type II heart block), should undergo evaluation and correction prior to major vascular surgery. Patients with significant ventricular and supraventricular arrhythmic burden may have underlying structural heart disease or inherited electrical disorders and should have an electrocardiogram and echocardiogram as part of their preoperative evaluation, along with an evaluation by a cardiovascular specialist. Frequent ventricular premature contractions and ventricular arrhythmias may portend underlying coronary artery disease.
Data are limited on perioperative outcomes following major noncardiac surgery in patients with cardiac arrhythmias. The original cardiac risk index associated any preoperative rhythm other than sinus rhythm or premature atrial contractions with a higher risk of postoperative adverse events. 22 However, subsequent small studies have not demonstrated an increase in mortality or MACE following major noncardiac surgery in patients with preoperative ventricular arrhythmias, though the presence of these arrhythmias in the preoperative setting is associated with an increased risk of perioperative arrhythmias.59,60
Perioperative management of medications
Beta-blockers
The use of beta-blockers in the perioperative setting has been theorized to reduce the risk of myocardial infarction by decreasing heart rate and reducing myocardial contractility, and thereby lowering myocardial oxygen demand. In the PeriOperative ISchemic Evaluation (POISE) trial, > 8000 patients undergoing noncardiac surgery (42% undergoing vascular surgery) were randomized to extended-release metoprolol 2–4 hours prior to surgery versus placebo. 61 There was an overall decrease in the primary endpoint of cardiovascular death, nonfatal myocardial infarction, and nonfatal cardiac arrest in the beta-blocker group (5.8% vs 6.9%) – almost completely driven by the reduction in myocardial infarction. However, there was a significant increase in the risk of mortality (3.1% vs 2.3%) and stroke (1% vs 0.5%) in the beta-blocker group. Post hoc analysis suggested that the increased risk of death in the treatment group was explained by clinically significant hypotension, bradycardia, and ischemic stroke, likely due to large doses of extended-release metoprolol prior to procedure. Multiple meta-analyses have reached similar conclusions.62 –64 Thus, beta-blockers should not be started in the immediate preoperative period in low-risk patients. However, for patients on chronic beta-blocker therapy, ACC/AHA, ESC/ESA, and CCS guidelines recommend that patients be continued on these medications,18 –20 as preoperative withdrawal of beta-blockers has been associated with up to 350% increased risk of mortality. 65
Patients with intermediate to high-risk myocardial ischemia on noninvasive stress testing, known ischemic disease, heart failure with reduced ejection fraction, or with multiple RCRI risk factors, may be considered for a preoperative beta-blocker (ACC/AHA and ESC/ESA class IIb recommendation).19,20 In contrast, the CCS recommends against the initiation of beta-blockers, noting that there is no reliable data to support this practice. 18 The preoperative benefits of beta-blocker therapy must be carefully weighed against the risks. If initiated, beta-blocker therapy should be started at least 7 days prior to surgical intervention and should avoid large fluctuations in blood pressure and heart rate.19,66 –69
Statins
Several trials studying the effects of preoperative statin therapy in patients undergoing vascular surgery showed fewer postoperative cardiovascular events and improved long-term overall mortality in patients who had received statin therapy in the perioperative setting.70,71 Acute discontinuation of statin therapy in the perioperative period is associated with a significantly increased risk of myocardial infarction and cardiovascular death. 72 A meta-analysis of 23,536 patients found that preoperative statin therapy was associated with a decreased risk of all-cause mortality and myocardial infarction. 73 The beneficial effects of perioperative statin therapy appear to be due to both the plaque-stabilizing effect and antiinflammatory properties of statins. Thus, the ACC/AHA and ESC/ESA guidelines recommend that patients who are undergoing major vascular surgery should either be continued on their home high-intensity statin or started on a high-intensity statin in the preoperative period.19,20 The CCS also recommends that patients should be continued on chronic statins in the preoperative setting; however, they note that the evidence is too weak to recommend that patients be newly started on a statin in the preoperative setting. 18
Angiotensin-converting enzyme inhibitors (ACEI) / angiotensin II receptor blockers (ARB)
Studies on the use of ACEIs and ARBs in the perioperative setting of major vascular surgery are limited. ACEIs and ARBs are commonly used in the setting of heart failure as cardio-protective agents with reverse remodeling properties. However, the perioperative use of ACEIs and ARBs carries the simultaneous risk of hypotension.
Several studies have evaluated the role of ACEIs and ARBs prior to noncardiac surgery;74 –80 however, there have not been any large, randomized trials on the use of these medications prior to major vascular surgery. Preoperative ACEI/ARB may be associated with perioperative hypotension whereas the transient interruption of chronic therapy is not associated with inferior outcomes in most patients.74,78,80 In the Vascular Events in Noncardiac Surgery Patients Cohort Evaluation (VISION) study of 14,687 patients (3.3% undergoing major vascular surgery), preoperative withholding of ACEI or ARB decreased intraoperative hypotension by 20%, with 18% relative risk reduction in composite outcome of postoperative all-cause death, stroke, or myocardial injury. 79
On the contrary, there may be a benefit of continuing ACEI or ARB therapy in patients with a reduced left ventricular ejection fraction. A study of 511 patients with a left ventricular ejection fraction < 30% undergoing major vascular surgery found that the perioperative use of ACEI was associated with a 67% reduction in mortality. 75 If chronic ACEI or ARB therapy is discontinued preoperatively, these medications should be restarted as soon as feasible postoperatively as restarting ACEI or ARB therapy has been associated with a decrease in 30-day mortality. 77 Based on these findings, the ACC/AHA guidelines recommend continuing ACEIs or ARBs perioperatively (class IIa recommendation). ESC/ESA guidelines recommend that if ACEIs or ARBs are to be started in patients with heart failure with reduced ejection fraction, they should be started at least 1 week prior to operative intervention (class IIa recommendation).19,20 In contrast, the CCS recommends withholding ACEIs or ARBs starting 24 hours prior to noncardiac surgery due to the increased risk of intraoperative hypotension. 18 As with any perioperative pharmacologic therapy, the benefits of ACEI and ARB therapy should be weighed closely against the risks.
Antiplatelet therapy
Antiplatelet therapy is often used in patients undergoing major vascular surgery. The ACC/AHA guideline recommendations are mostly based on the PeriOperative ISchemic Evaluation-2 (POISE-2) trial.19,81 In this randomized trial of > 10,000 patients with history of coronary or peripheral artery disease, or stroke who underwent noncardiac surgery, aspirin (compared to placebo) was not associated with a reduction in the primary composite outcomes of death or myocardial infarction after noncardiac surgery. Conversely, there was a ~ 20% increase in bleeding in the aspirin group. Subgroup analyses of 6% of enrolled patients undergoing major vascular surgery did not show any difference between the groups. Patients undergoing carotid endarterectomy were excluded. 82 A meta-analysis of > 30,000 patients showed that perioperative aspirin therapy (81–500 mg daily) was associated with a 14% higher risk for blood transfusion without increased risk for surgical reintervention. 83 The POISE-2 trial excluded patients who had received a bare metal stent (BMS) 6 weeks prior or drug-eluting stent (DES) 1 year prior to operative intervention. Subgroup analyses of patients with a history of percutaneous coronary intervention (PCI) outside of the defined exclusion timeframe showed that perioperative aspirin reduced the incidence of the composite outcome of death or myocardial infarction by 50%, with a ~ 20% increase in major bleeding. 84 An observational study also corroborated the risk of increased MACE and stroke in patients with prior PCI when oral antiplatelet therapy was interrupted for more than 5 days prior to surgical intervention. 85 The ACC/AHA and CCS guidelines recommend against the initiation or continuation of aspirin prior to noncardiac, noncarotid surgery if there have been no coronary stents placed in the past, but recommend continuation of aspirin, if possible, in patients with a prior history of PCI.18,19 The ESC/ESA guidelines recommend an individualized approach based on the risk of perioperative bleeding as compared to thrombotic complications. 20
There is a benefit to continuing antiplatelet therapy in patients undergoing carotid endarterectomy (CEA). A randomized trial of 232 patients undergoing carotid endarterectomy found that preoperative aspirin 75 mg daily was associated with a significantly lower risk of stroke at 6 months, without a difference in major bleeding. 86 A subsequent larger, randomized trial of 2849 patients undergoing CEA randomized patients to aspirin 81 mg, 325 mg, 650 mg, or 1300 mg daily and determined that the risk of stroke, myocardial infarction, and death up to 3 months following CEA was lower in patients on low-dose aspirin (81 mg or 325 mg) as compared to patients taking higher doses of aspirin (650 mg or 1300 mg). 87 Guidelines from the American Academy of Neurology on the use of aspirin prior to CEA recommend the use of low-dose aspirin (81 mg or 325 mg) before and after surgical intervention to reduce the rate of stroke and MACE. 88
Perioperative treatment with dual antiplatelet therapy (DAPT) with aspirin and a thienopyridine is often encountered in patients with vascular disease, due to the frequency of coronary artery disease and prior PCI. In this population, DAPT is of particular importance due to the risk of in-stent thrombosis if DAPT is discontinued too early after PCI. Current ACC/AHA guidelines recommend that for elective noncardiac surgery, surgery should ideally be delayed 30 days after BMS implantation and 6 months after DES implantation. 89 For patients undergoing major vascular surgery, surgical timing should be an individualized decision, weighing the urgency of surgical intervention with the need to complete a minimum amount of dual antiplatelet therapy prior to discontinuation of thienopyridine therapy. Figure 2 shows the ACC/AHA guidelines on the use of antiplatelet agents in the perioperative setting in patients undergoing noncardiac surgery. Table 4 summarizes the use of pharmacotherapy prior to major vascular surgery.
Pharmacotherapy prior to major vascular surgery.
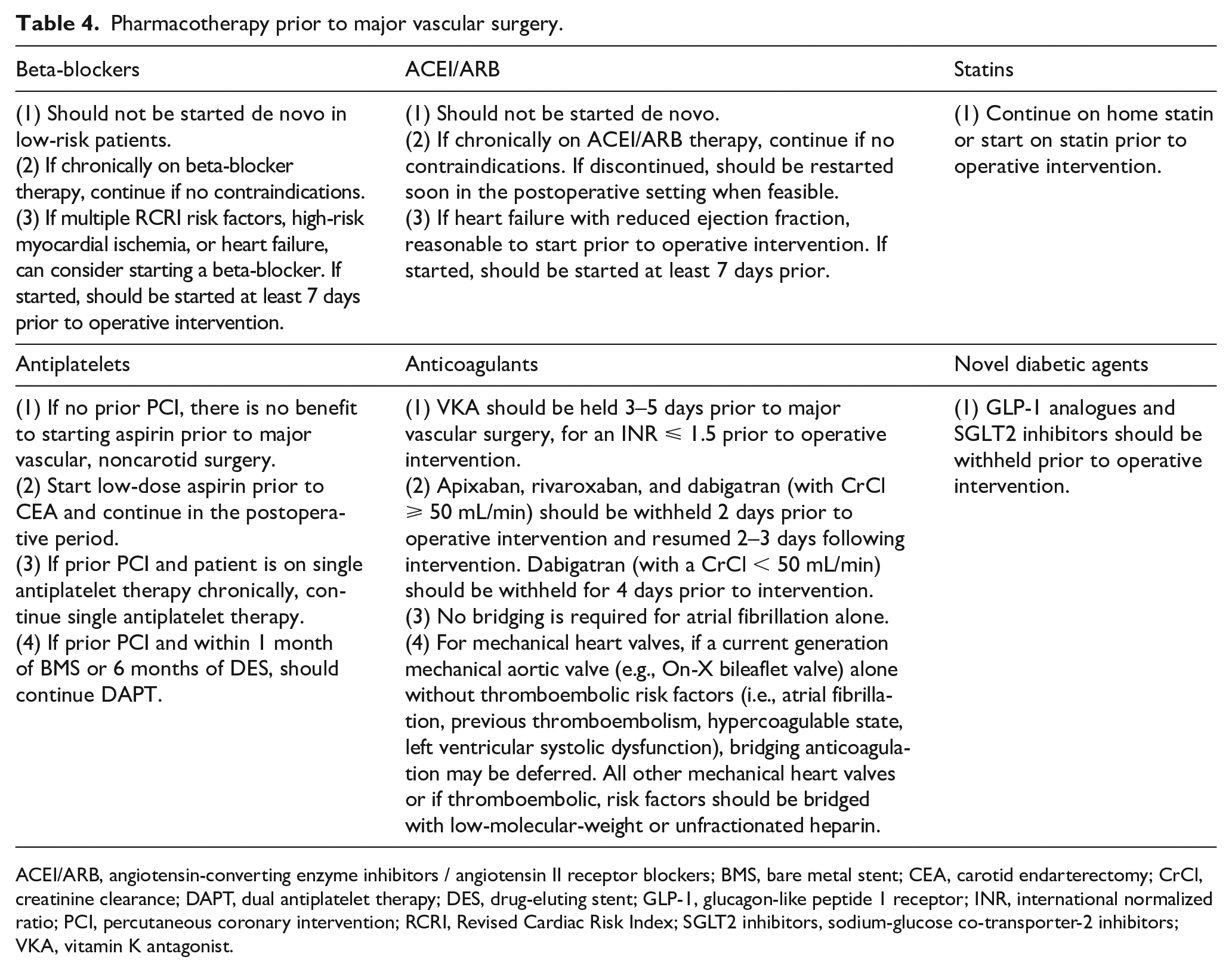
ACEI/ARB, angiotensin-converting enzyme inhibitors / angiotensin II receptor blockers; BMS, bare metal stent; CEA, carotid endarterectomy; CrCl, creatinine clearance; DAPT, dual antiplatelet therapy; DES, drug-eluting stent; GLP-1, glucagon-like peptide 1 receptor; INR, international normalized ratio; PCI, percutaneous coronary intervention; RCRI, Revised Cardiac Risk Index; SGLT2 inhibitors, sodium-glucose co-transporter-2 inhibitors; VKA, vitamin K antagonist.
Anticoagulants
The use of oral anticoagulation is frequently seen in patients for a multitude of reasons, ranging from atrial fibrillation to mechanical heart valves. This often presents a challenging situation for patients undergoing major vascular surgery, as the continuation of oral anticoagulation through the operative period can increase the risk of major bleeding, though interruption of anticoagulation can increase the risk of thromboembolism. Patients undergoing surgical intervention that places patients at unacceptably elevated risk of major bleeding should be taken off their oral anticoagulation in the preoperative setting. For patients whose vitamin K antagonist (VKA; e.g., warfarin) is interrupted for operative intervention, the question of whether patients require bridging anticoagulation in the pre- and perioperative period is frequently encountered. In the Bridging Anticoagulation in Patients who Require Temporary Interruption of Warfarin Therapy for an Elective Invasive Procedure or Surgery (BRIDGE) trial, bridging with low-molecular-weight heparin while withholding VKA prior to surgery was not associated with decreased rates of arterial thromboembolism at 30 days (0.3% vs 0.4%). 90 As such, patients with atrial fibrillation who need interruption of their anticoagulation for operative intervention should not routinely be bridged in the pre- and perioperative period.
In contrast, patients with mechanical heart valves are at an elevated risk for thromboembolism and valve thrombosis if VKA is held. There is a lack of large studies assessing the risk of thromboembolism in this population who undergo operative intervention. The Mayo Clinic Thrombophilia Center assessed the risk of thromboembolism and bleeding in patients with mechanical heart valves, comparing outcomes of patients who were bridged with heparin versus not bridged, according to an individualized protocol. 91 Patients undergoing (1) minor procedures (e.g., dental procedures) were anticoagulated at the lower limit of therapeutic anticoagulation (INR 2–2.5); (2) major surgery with an aortic bileaflet mechanical prosthesis had their warfarin discontinued 3–5 days prior to operative intervention without bridging; (3) major surgery with a caged ball-valve, lifting disc, or tilting disc aortic valve mechanical prosthesis, a mechanical mitral valve, multiple valve prostheses, or a prosthetic valve with risk factors for thromboembolism had their warfarin discontinued 3–5 days prior to operative intervention and were bridged with low-molecular-weight or unfractionated heparin. There was a low overall risk of thromboembolism, with a cumulative incidence of 0.9%. The risk was lowest in the no-bridge group (0.5%), and highest in the group receiving unfractionated heparin for bridging (0.8%), which likely reflects underlying risk of thromboembolism. The ACC/AHA guidelines recommend that in patients with a current-generation mechanical aortic valve (e.g., On-X bileaflet valve) who are undergoing noncardiac procedures, anticoagulation may be held without bridging and restarted when feasible in the postoperative setting if there are no thromboembolic risk factors (e.g., atrial fibrillation, previous thromboembolism, hypercoagulable state, left ventricular systolic dysfunction). However, patients with any thromboembolic risk factors, an older generation mechanical aortic valve, or a mechanical valve in a nonaortic position should be bridged in the preoperative and perioperative setting. 52 The ESC/ESA guidelines have similar recommendations, though do not differentiate between generations of mechanical valves, and recommend withholding VKA for 3–5 days prior to surgery until the INR is ⩽ 1.5, with bridging in the preoperative and perioperative setting.20,53 The CCS does not give a formal recommendation on anticoagulation management in the perioperative period. 18
Guidelines on the use of direct oral anticoagulants (DOACs) in the peri-procedural period are lacking. The Perioperative Anticoagulation Use for Surgery Evaluation (PAUSE) trial was a multicenter, prospective cohort trial that assessed the frequency of major bleeding and arterial thromboembolism following a structured holding pattern of DOACs among patients with atrial fibrillation undergoing a planned operative intervention. 92 A total of 3007 patients were enrolled and the schedule of withholding anticoagulation was based on surgical bleeding risk. For patients taking apixaban, rivaroxaban, or dabigatran with a creatinine clearance ⩾ 50 mL/min and undergoing a low-bleeding-risk procedure (e.g., colonoscopy/gastroscopy, dental procedures), the DOAC was withheld 1 day prior to the procedure and resumed 1 day afterwards. For patients taking dabigatran with a creatinine clearance < 50 mL/min and undergoing a low-bleeding-risk procedure, patients withheld their dabigatran for 2 days prior to the procedure and resumed it 1 day afterwards in the postoperative period. If undergoing a high-bleeding-risk procedure (e.g., major vascular surgery – aortic aneurysm repair, aortobifemoral bypass, popliteal bypass, major intracranial or neuraxial surgery), apixaban, rivaroxaban, and dabigatran with a creatinine clearance ⩾ 50 mL/min were withheld 2 days prior and resumed 2–3 days afterwards in the postoperative period (for dabigatran with a creatinine clearance < 50 mL/min, dabigatran was held for 4 days prior and resumed 2–3 days afterwards in the postoperative period). Total 30-day postoperative rates of major bleeding were low. Patients who underwent a low-bleeding-risk procedure and were taking apixaban, rivaroxaban, and dabigatran had a 1.35%, 1.85%, and 0.90% rate of major bleeding, respectively. For patients undergoing a high-bleeding-risk procedure, there was a 2.96%, 2.95%, and 0.88% risk of major bleeding, respectively. The overall risk of arterial thromboembolism was 0.16% in the apixaban group, 0.37% in the rivaroxaban group, and 0.60% in the dabigatran group. Given these findings and the lack of guidelines on the peri-procedural holding pattern of DOACs, it is reasonable that anticoagulant management in the pre- and postoperative period in patients treated with DOACs should follow the PAUSE trial algorithmic approach.
In addition to traditional uses for oral anticoagulation, patients with polyvascular disease also may benefit from combination therapy with low-dose rivaroxaban (2.5 mg twice daily) in addition to aspirin. In the Cardiovascular Outcomes for People Using Anticoagulation Strategies (COMPASS) peripheral artery disease trial, 7470 patients with stable peripheral or carotid artery disease were randomized to rivaroxaban alone (5 mg twice daily); or rivaroxaban (2.5 mg twice daily) and low-dose aspirin; or low-dose aspirin alone. 93 The combination of low-dose rivaroxaban and low-dose aspirin significantly decreased the incidence of major adverse limb events and amputations (1%) as compared to aspirin alone (2%). Though there was a higher incidence of major bleeding in the combination group as compared to aspirin alone (hazard ratio [HR] 1.61), most of the bleeding episodes were due to nonfatal gastrointestinal bleeds, with the net clinical benefit significantly favoring a combination of rivaroxaban and aspirin. 94 In addition, a combination of low-dose rivaroxaban (2.5 mg twice daily) and aspirin also benefits patients following lower-extremity revascularization. In the Vascular Outcomes Study of Acetylsalicylic Acid Along with Rivaroxaban in Endovascular or Surgical Limb Revascularization for Peripheral Artery Disease (VOYAGER-PAD) trial, patients undergoing peripheral revascularization were randomized to a combination of low-dose rivaroxaban (2.5 mg twice daily) and low-dose aspirin versus low-dose aspirin alone. 95 After 3 years of follow up, the primary composite outcome (combination of acute limb ischemia, major amputation, myocardial infarction, ischemic stroke, or cardiovascular death) occurred less frequently in the rivaroxaban and aspirin combination group as compared to the aspirin alone group (17.3% vs 19.9%, respectively). Although the risk of bleeding was higher in the combination group (HR 1.43), there was no difference in the number of patients with intracranial hemorrhage or fatal bleeding. Given these findings, after major vascular surgery, appropriate patients who are at a lower risk of bleeding should be considered for low-dose rivaroxaban in addition to low-dose aspirin.
Novel diabetic agents
Novel diabetic agents, such as glucagon-like peptide 1 receptor (GLP-1) analogues and sodium-glucose co-transporter-2 (SGLT2) inhibitors, are encountered with increasing frequency in patients undergoing major vascular surgery. GLP-1 analogues (semaglutide and liraglutide) have shown benefit in reducing the composite of death from cardiovascular causes, nonfatal myocardial infarction, and nonfatal stroke.96,97 The potential benefits of GLP-1 analogues on patients with peripheral vascular disease are still being actively investigated. A post hoc analysis of the Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results (LEADER) trial found decreased diabetic foot ulcer-related amputations in the liraglutide group as compared to placebo. 98 Clinical trials, such as the STARDUST trial, which is an open-label, randomized trial investigating the effect of liraglutide on lower-limb perfusion in diabetic patients with peripheral artery disease, are ongoing (NCT04881110).
SGLT2 inhibitors (empagliflozin, canagliflozin, and dapagliflozin) have been shown to be beneficial in reducing death due to cardiovascular causes, heart failure hospitalizations, and death from any cause.99 –101 Though canagliflozin was initially associated with an approximately twofold increased risk of below the ankle amputations (6.3 vs 3.4 per 1000 patient-years) in the CANVAS trial, there was no significantly increased risk of amputations in the subsequent CREDENCE trial.99,102 The United States Food and Drug Administration subsequently removed the boxed warning on amputation risk with canagliflozin that had previously been placed after the CANVAS trial. 103 Repeat analyses have demonstrated that there does not appear to be any incremental risk of amputations in patients treated with SGLT2 inhibitors.104,105 As with any perioperative pharmacologic therapy, the benefits should be weighed closely against the risks. However, in patients undergoing major vascular surgery, it is reasonable to hold outpatient, noninsulin diabetes medications in the perioperative setting to facilitate inpatient glucose control and avoid hypoglycemic episodes.
Postoperative management
Postoperative arrhythmias
Atrial fibrillation is frequently encountered in the postoperative setting following major vascular surgery. In this situation, reversible factors that may be contributing to the dysrhythmia (e.g., sepsis, volume overload, pain, hemorrhage) should be identified and treated. In the perioperative period, management of atrial fibrillation with rapid ventricular response (> 110 beats per minute) is usually through rate or rhythm control, depending upon the hemodynamic status of the patient. In the hemodynamically stable patient, typical rate control medications that can be utilized include beta-blockers and nondihydropyridine calcium channel blockers, although calcium channel blockers should be avoided in individuals with heart failure with reduced ejection fraction. In the hemodynamically unstable patient, direct current cardioversion and intravenous antiarrhythmic agents, such as amiodarone, can be utilized to attempt to restore sinus rhythm. Patients with new-onset atrial fibrillation following noncardiac surgery have an elevated risk of thromboembolism that is four- to fivefold higher than patients without atrial fibrillation and have a risk of stroke that is similar to patients with nonsurgical, nonvalvular atrial fibrillation.106,107 Thus, ESC guidelines recommend long-term oral anticoagulation after noncardiac surgery in patients who develop postoperative atrial fibrillation and are suitable for anticoagulation (class IIa). 108
Postoperative heart failure
Patients with evidence of an acutely decompensated heart failure exacerbation in the perioperative period should initially be assessed with hemodynamic monitoring and diagnostic tests such as an ECG, chest X-ray, and measurement of laboratory values including a complete blood count, basic metabolic panel, lactic acid, brain natriuretic peptide, and troponin. Patients with evidence of acute coronary syndrome (e.g., chest pain, ECG changes, troponin elevation) can be considered for invasive coronary angiography, if appropriate. Physical examination should include an accurate assessment of the patient’s volume status. A transthoracic echocardiogram can be performed to assess biventricular function and the presence of valvular abnormalities. Patients with hemodynamically stable heart failure exacerbations can be managed with intravenous diuretic therapy and can be treated with their home doses of beta-blocker and ACEI/ARB (in the absence of acute renal injury). Patients with evidence of shock (e.g., hemodynamic compromise and end-organ hypoperfusion) should be managed in the critical care setting. Pharma-cologic therapy should include intravenous diuresis and discontinuation of medications that have negative inotropic effects (e.g., beta-blockers, nondihydropyridine calcium channel blockers). Inotropes (e.g., dobutamine, milrinone) and/or vasopressors can be utilized for blood pressure support or to augment cardiac output.
Postoperative troponin measurement
In patients undergoing major vascular surgery, perioperative screening for myocardial infarction or myocardial injury after noncardiac surgery (MINS) is frequently undertaken via perioperative troponin measurements. MINS, defined as myocardial injury occurring within 30 days of operative intervention, is associated with a three- to fourfold increase in 30-day mortality. 109 In the VISION study, 21,842 patients undergoing noncardiac surgery had high-sensitivity troponins measured serially (hsTnT; 6 hours, 12 hours, daily) for 3 days. 110 Patients with peak hsTnT of 20 to < 65 ng/L, 65 to < 1000 ng/L, and 1000 ng/L or higher had 30-day mortality rates of 3%, 9.1%, and 29.6%, respectively. Patients with MINS and ischemic features (i.e., symptoms, ECG changes, echocardiographic wall motion abnormalities, or defects on radionuclide imaging) had an 8.5% risk of 30-day mortality as compared to 2.9% for patients with MINS without ischemic features. Given that most postoperative myocardial infarctions occur within 48 hours of operative intervention and are associated with an elevated risk of 30-day postoperative mortality, there appears to be a role in trending troponins in the immediate postoperative period to aid in the detection of postoperative myocardial infarction. 111 As such, the CCS guidelines recommend the measurement of daily troponins in patients for 2–3 days in the postoperative period for patients who are at high risk (> 5%) for cardiovascular death or myocardial infarction. 18 Both the ACC/AHA and ESC/ESA guidelines, however, note that screening troponins in the perioperative period may be considered in high-risk patients (class IIb recommendation), but these guidelines were published prior to the publication of the results of the VISION study.19,20
Blood pressure management postcarotid endarterectomy
Hemodynamic changes following carotid intervention can occur due to changes in carotid baroreceptor function. 112 A retrospective cohort study found that in patients undergoing carotid endarterectomy, the incidence of postoperative hypertension was 9%, and hypotension was up to 12%. 113 Hemodynamic changes in this setting that require intravenous medications are associated with worse 30-day mortality, stroke, myocardial infarction, and length of stay.114 –116 As such, the goal in the postprocedural period is to maintain normotensive blood pressures (systolic blood pressure ⩽ 160 mmHg) while minimizing hypotension to maintain appropriate cerebral perfusion. 117
Conclusion
Patients undergoing major vascular surgery have an elevated risk of perioperative MACE. They commonly have multiple medical comorbidities that predispose them to adverse events, while the superimposed stress response to the vascular intervention leads to an increased risk for an acute myocardial infarction. A thorough evaluation prior to planned operative intervention can minimize postprocedural risk. A preoperative ECG should be obtained in all patients undergoing major vascular surgery, but a preoperative echocardiogram, stress testing, or coronary angiography should only be obtained if the results will change perioperative management. Significant arrhythmias (e.g., rapid supraventricular arrhythmias, sustained ventricular arrhythmias, or high-degree conduction system disease) should be corrected prior to surgery. Patients who are on chronic beta-blocker or ACEI/ARB therapy should continue to be treated with these medications; initiating these medications de novo less than 1 week prior to surgical intervention should be avoided due to higher risk of perioperative hypotension. It is reasonable to start high-intensity statins prior to major vascular surgery. Patients who are on single agent antiplatelet therapy (e.g., low-dose aspirin, thienopyridine) due to a prior PCI should be continued on single agent antiplatelet therapy through the operative period but should not be newly started on an antiplatelet agent if no stents have previously been placed (unless undergoing a carotid endarterectomy). Patients on DAPT due to prior PCI should be continued on DAPT for at least 1 month for a BMS and at least 6 months for a DES. Similarly, anticoagulation should be held prior to major vascular intervention. Patients with mechanical valves should be considered for pre- and perioperative bridging, although patients with current-generation mechanical aortic valves without other thromboembolic risk factors may have anticoagulation held without bridging. Patients who are on traditional and novel antidiabetic agents (e.g., GLP-1 analogues and SGLT2 inhibitors), should have their medications withheld in the perioperative period to avoid hypoglycemia.
The risk of postoperative complications, including myocardial infarction and arrhythmia, mirror the level of invasiveness of the surgical intervention. Ideally, a multidisciplinary team approach prior to and following major vascular surgery should be considered to optimize patient outcomes. Key points to consider are summarized in Table 5.
Key points to remember for patients undergoing vascular surgery.
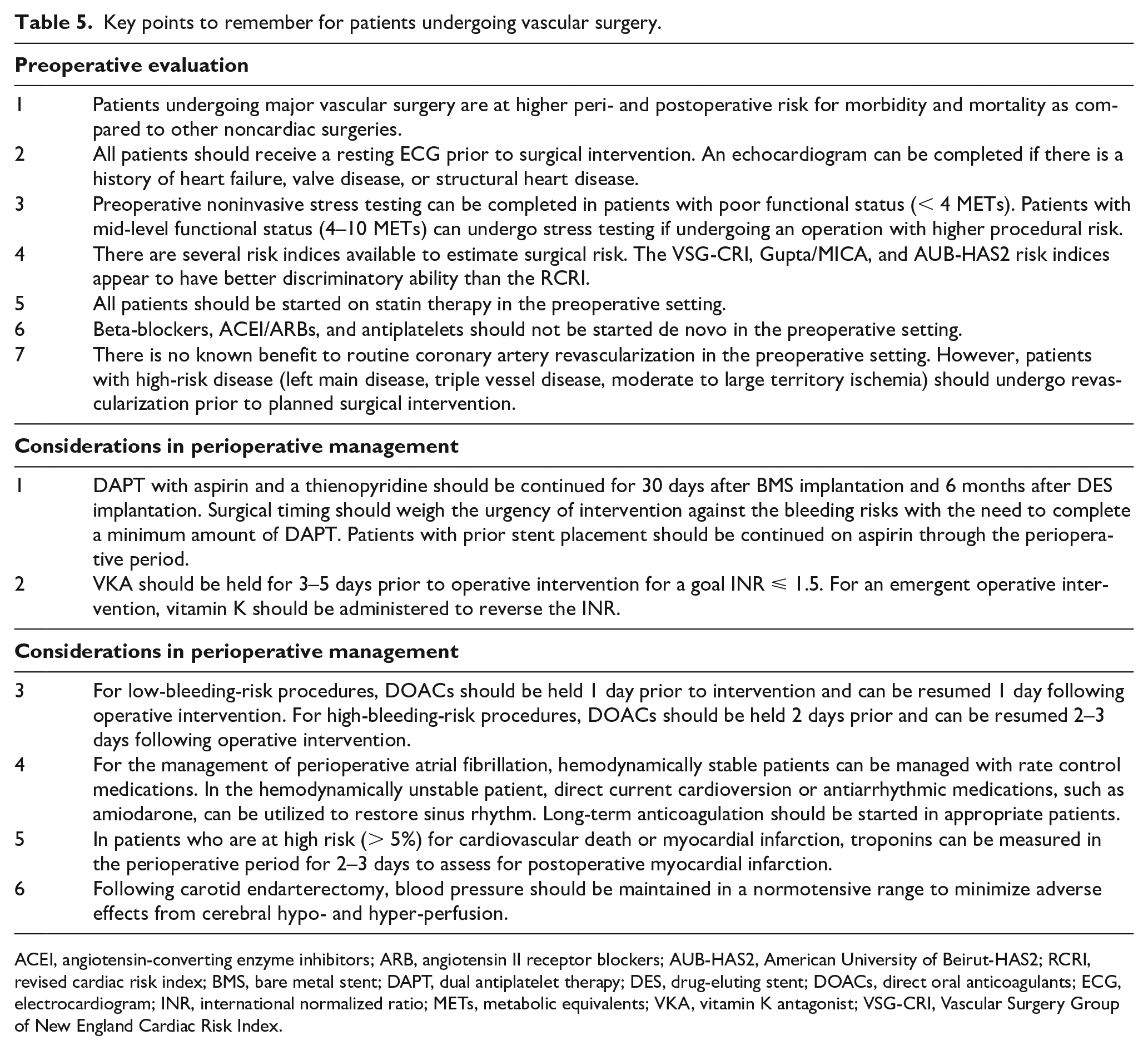
ACEI, angiotensin-converting enzyme inhibitors; ARB, angiotensin II receptor blockers; AUB-HAS2, American University of Beirut-HAS2; RCRI, revised cardiac risk index; BMS, bare metal stent; DAPT, dual antiplatelet therapy; DES, drug-eluting stent; DOACs, direct oral anticoagulants; ECG, electrocardiogram; INR, international normalized ratio; METs, metabolic equivalents; VKA, vitamin K antagonist; VSG-CRI, Vascular Surgery Group of New England Cardiac Risk Index.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.