Abstract
Background:
Despite optimal treatment, type II diabetes mellitus remains associated with an increased risk for future cardiovascular events. We sought to determine the association between baseline fasting plasma insulin levels and major adverse cardiovascular outcomes in patients with type II diabetes mellitus and high-risk vascular disease enrolled in the ACCELERATE (Assessment of Clinical Effects of Cholesteryl Ester Transfer Protein Inhibition with Evacetrapib in Patients at a High Risk for Vascular Outcomes) trial.
Methods:
We included all patients with type II diabetes mellitus who had a central laboratory measured fasting plasma insulin level drawn at baseline as part of the study protocol. Hazard ratios were generated for the risk of major adverse cardiovascular outcomes (composite of cardiovascular death, non-fatal myocardial infarction, stroke, hospitalization for unstable angina and coronary revascularization) with increasing quartile of baseline fasting plasma insulin level. We then performed a multivariable regression adjusting for significant baseline characteristics.
Results:
Among 12,092 patients in ACCELERATE, 2042 patients with type II diabetes mellitus had a baseline fasting plasma insulin level drawn. Median follow-up was 28 months. The study population had a mean age of 66.6 years, 79.2% male and 96.2% had established coronary artery disease. During follow-up, major adverse cardiovascular outcomes occurred in 238 patients (11.6%); of these events, 177 were coronary revascularization (8.7%). We observed a statistically significant relationship between rates of revascularization and rising quartile of baseline fasting plasma insulin level which was not noted for the other individual components of major adverse cardiovascular outcomes. Patients with type II diabetes mellitus who underwent revascularization were noted to have significantly higher baseline fasting plasma insulin levels (27.7 vs 21.4 mU/L, p-value = 0.009) although baseline haemoglobin A1c (6.63% vs 6.55%), body mass index (31.5 vs 31.1 kg/m2) and medical therapy were otherwise similar to the group not undergoing revascularization. Following multivariable regression adjusting for significant characteristics including exposure to evacetrapib, the log of baseline fasting plasma insulin level was found to be an independent predictor for major adverse cardiovascular outcomes (hazard ratio = 1.36, 95% confidence interval = 1.09–1.69, p-value = 0.007); this was driven by need for future revascularization (hazard ratio = 1.56, 95% confidence interval = 1.21–2.00, p-value = 0.001).
Conclusion:
In a contemporary population of patients with type II diabetes mellitus and high-risk vascular disease on optimum medical therapy, baseline hyperinsulinaemia was an independent predictor for major adverse cardiovascular outcomes and need of future coronary revascularization. These results suggest a pathophysiological link between hyperinsulinaemia and progression of atherosclerotic vascular disease among diabetics.
Introduction
Type II diabetes mellitus (T2DM) is highly prevalent and poses a significant and rising burden on the health care system. 1 Numerous studies have described an association between T2DM and coronary artery disease (CAD). 2 Although several new agents have recently been proven to reduce cardiovascular risk in T2DM,3–5 the residual risk of developing atherosclerotic cardiovascular events despite optimal treatment remains significantly higher in this population when compared to non-diabetics.6,7 While a large proportion of the accentuated risk in patients with T2DM is attributable to the presence of traditional risk factors, further identification of modulating novel risk factors is crucial to developing novel therapeutic interventions.
Previous studies have examined the association between hyperinsulinaemia and incidence of CAD in a variety of clinical settings, although primarily in healthy individuals without T2DM or prior history of established CAD.8–13 A majority of these studies are now of historic importance as subjects were not optimally medically managed with current guideline-recommended therapies. Consequently, the association between baseline fasting insulin levels and residual risk in high-risk patients with T2DM has not been adequately investigated.
The Assessment of Clinical Effects of Cholesteryl Ester Transfer Protein Inhibition with Evacetrapib in Patients at a High Risk for Vascular Outcomes (ACCELERATE) trial was a randomized, double-blinded placebo-controlled trial investigating the use of evacetrapib, a cholesteryl ester transfer protein inhibitor, on patients with high-risk vascular disease. 14 We examined the association between baseline fasting plasma insulin levels and major adverse cardiovascular outcomes (MACE) in patients with T2DM and high-risk vascular disease enrolled in the ACCELERATE trial.
Methods
The trial design of ACCELERATE has previously been described. 15 Briefly, approximately 12,000 patients with high-risk vascular disease, including those with recent acute coronary syndrome, peripheral arterial disease, cerebrovascular disease and DM with established history of CAD, were randomized in a 1:1 fashion to evacetrapib 130 mg versus placebo. The trial was event-driven with a primary endpoint of MACE which included cardiovascular death, myocardial infarction, cerebrovascular accident, coronary revascularization or hospitalization for unstable angina all of which were adjudicated by a blinded Clinical Endpoints Committee. Due to clinical futility, the trial was terminated prematurely after accrual of 1363 of the planned 1670 primary endpoint events and a median of 26 months study drug exposure. Follow-up was comprehensive and the end of study visit was completed by 98.8% of patients.
Baseline fasting plasma insulin level was collected as part of the study protocol at randomly selected study sites identified at study initiation. We performed a subgroup analysis among those who had a central laboratory measured fasting plasma insulin level collected at baseline and were known to be diabetic. Patients with type 1 diabetes mellitus and insulin-dependent diabetes mellitus were excluded from this analysis. Baseline patient characteristics, medications and laboratory parameters were compiled. Percentages and means ± standard deviations were computed for categorical and continuous variables, respectively. Categorical variables were compared using the chi-square test or Fisher exact tests, when appropriate, while continuous variables were analysed using the two-tailed Student’s t test or the Mann–Whitney U test, when appropriate. Kaplan–Meier methods generated survival curves to graphically demonstrate the risk with increasing quartile in baseline fasting plasma insulin. Multivariable Cox’s proportional hazard models with stepwise selection identified significant factors associated with each endpoint. Hazard ratios (HRs) with 95% confidence intervals (CI) are reported for the log of fasting plasma insulin after adjustment for other clinical covariates associated with the endpoint. A p-value ⩽ 0.05 was considered statistically significant.
Results
A total of 12,092 patients were enrolled in ACCELERATE. As described in Figure 1, 8236 patients had diabetes mellitus with 166 patients excluded due to insulin dependence; of the remaining patients, 2042 patients with T2DM had a baseline fasting plasma insulin level drawn. The overall median follow-up was 28 months. The average age was 66.6 years, 79.2% were male and 96.2% had established CAD. At baseline, 85.2% of patients were taking an aspirin, 95.4% a statin, 79.1% an angiotensin-converting enzyme inhibitor or angiotensin-receptor II antagonist, and 75.3% a β-blocker. Overall, 83.6% of patients were taking an oral hypoglycaemic agent. At study initiation, baseline low-density lipoprotein cholesterol (LDL-C) was 80.6 mg/dL, high-density lipoprotein cholesterol (HDL-C) was 44.6 mg/dL, triglycerides were 150.5 mg/dL and haemoglobin A1c was 6.6%.
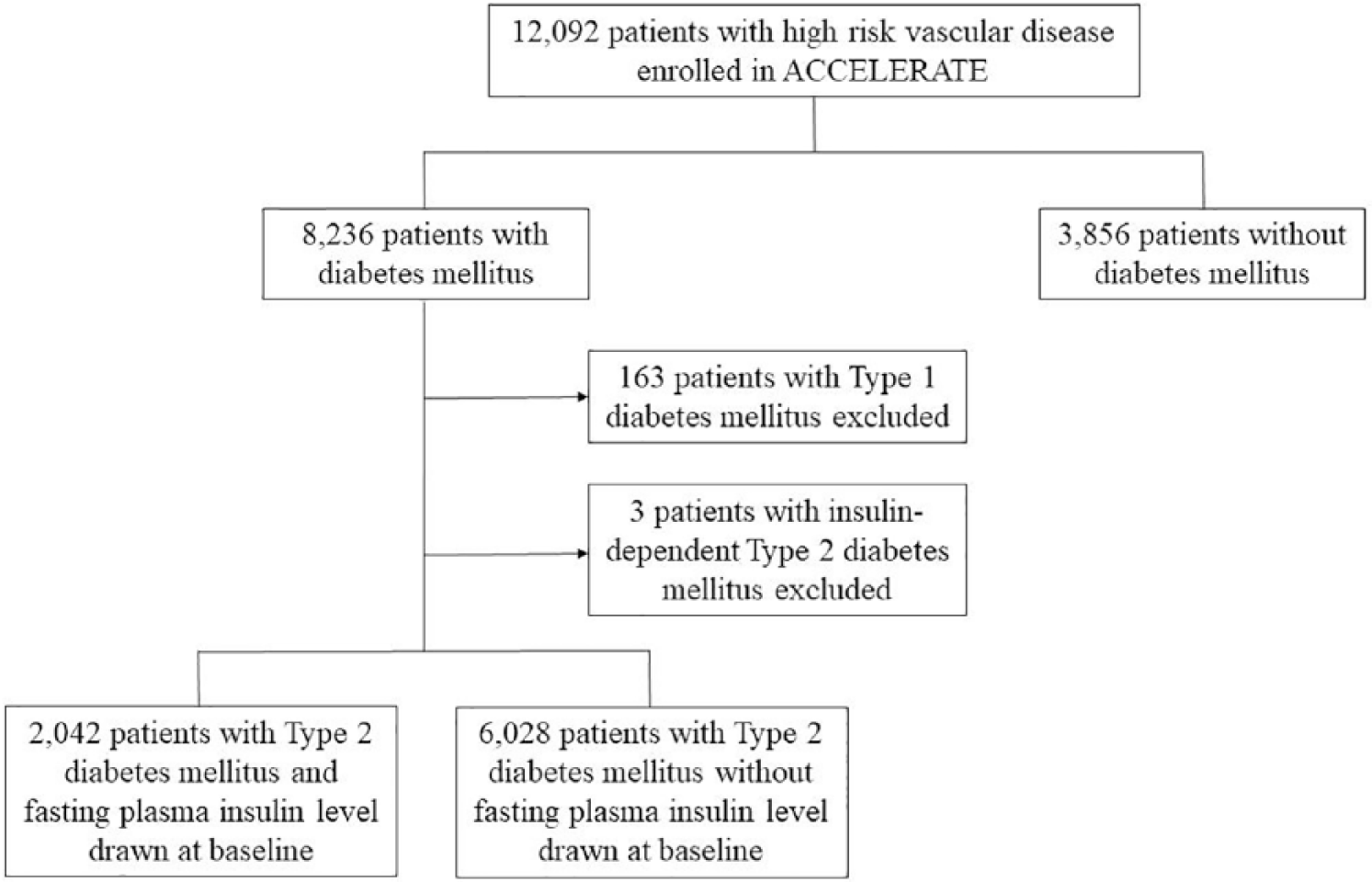
Summary of included patients.
During follow-up, MACE occurred in 238 patients (11.6%); among these events, 177 (8.7%) were coronary revascularization. As seen in Figure 2, Kaplan–Meier event curves demonstrated an increase in MACE by quartiles of baseline fasting plasma insulin level which appeared to be predominantly driven by need for revascularization. Baseline characteristics of patients with and without revascularization following randomization are shown in Table 1. Patients with T2DM who underwent revascularization had a 29.4% higher baseline fasting plasma insulin level (27.7 vs 21.4 mU/L, p-value = 0.009) and were younger (65.2 vs 66.7 years, p-value = 0.020), more likely to be current smokers (16.9% vs 12.0%, p-value = 0.06), have undergone prior percutaneous coronary intervention (83.6% vs 73.5%, p-value = 0.004) and have a higher baseline LDL-C (83.9 vs 80.3 mg/dL, p-value = 0.03). The baseline haemoglobin A1c (6.63% vs 6.55%), body mass index (31.5 vs 31.1 kg/m2), baseline medical therapy and remainder of characteristics were similar regardless of need for revascularization during follow-up. Table 2 demonstrates that while there was no difference in use of any oral anti-glycaemic medication (81.9% vs 83.8%, p = 0.529), those who underwent revascularization were significantly less likely to be prescribed a thiazolidinedione (2.3% vs 6.2%, p-value = 0.032).
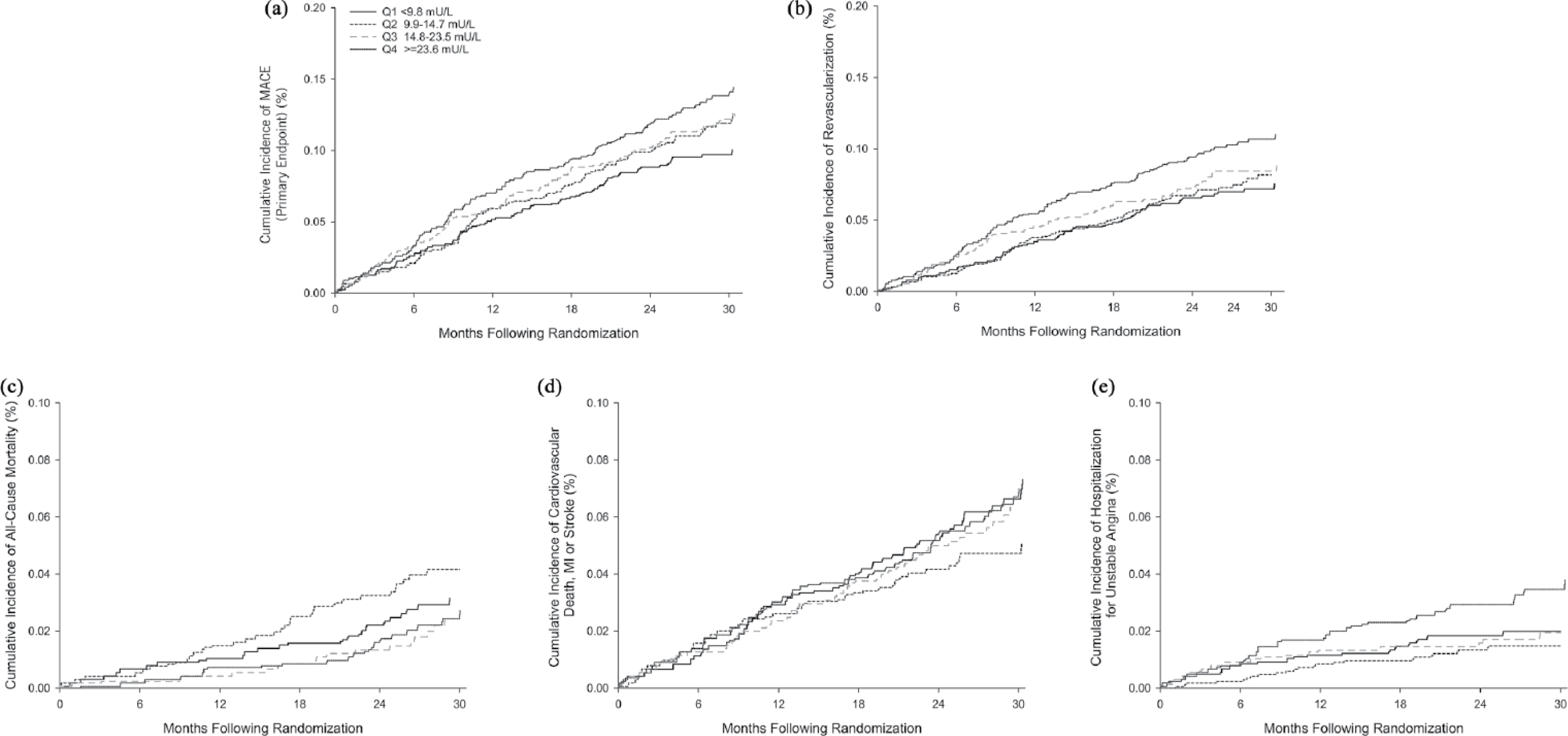
Event curves for each endpoint by quartile of baseline fasting plasma insulin level: (a) primary composite of major adverse cardiac events; (b) revascularization; (c) all-cause mortality; (d) composite of cardiovascular death, myocardial infarction and cerebrovascular accident; and (e) hospitalization for unstable angina.
Baseline characteristics of patients who underwent revascularization in comparison to those who did not.
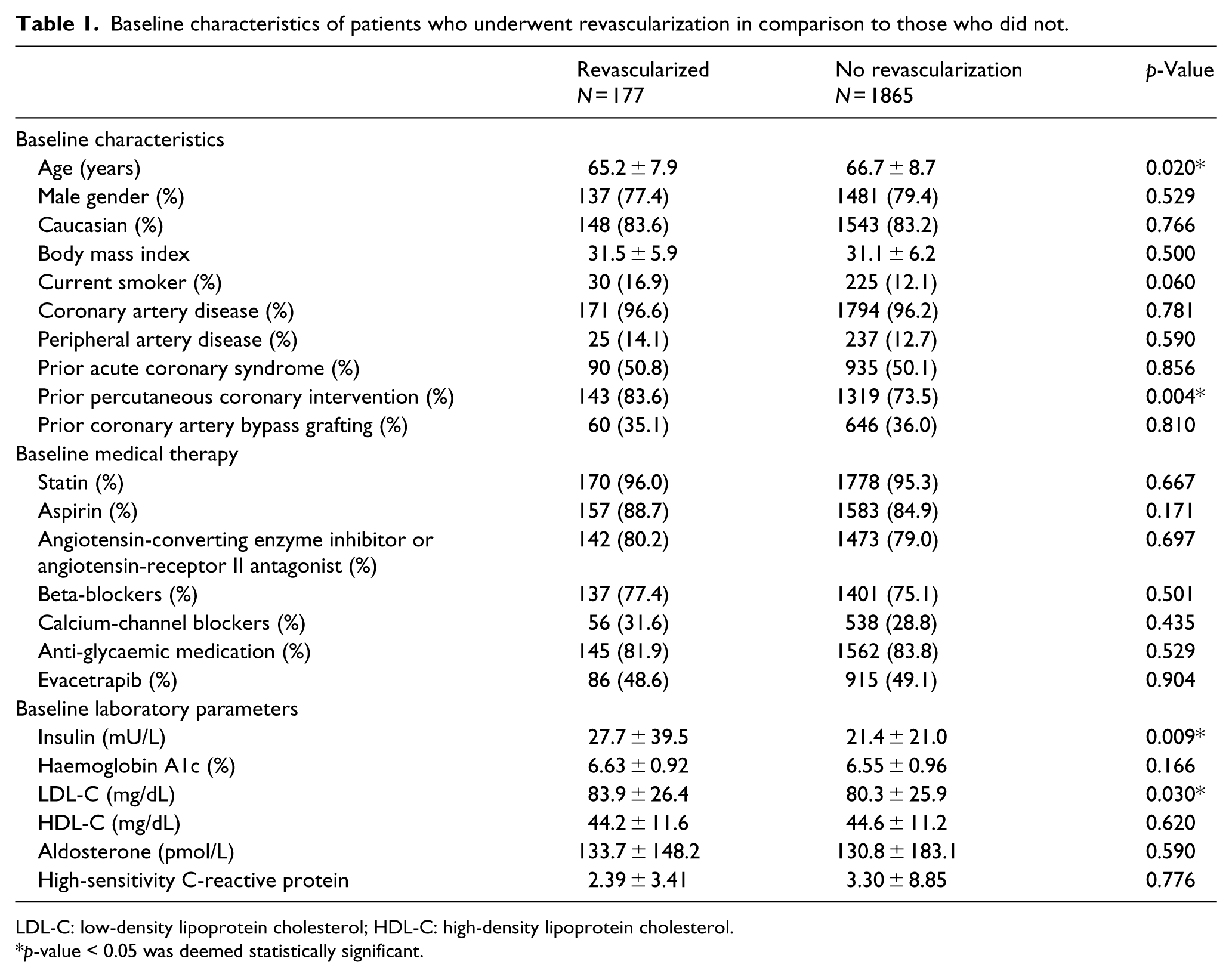
LDL-C: low-density lipoprotein cholesterol; HDL-C: high-density lipoprotein cholesterol.
p-value < 0.05 was deemed statistically significant.
Use of oral anti-glycaemic medications among patients who underwent revascularization in comparison to those who did not.
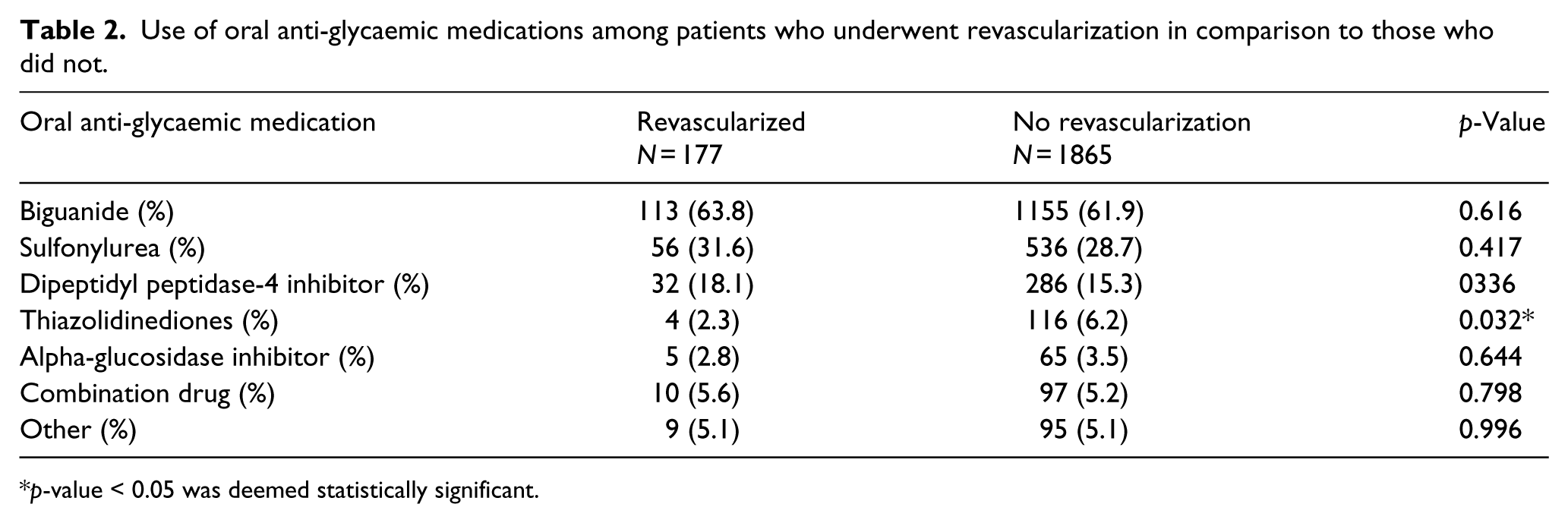
p-value < 0.05 was deemed statistically significant.
Multivariable regression adjusting for baseline haemoglobin A1c, fasting glucose, log of triglycerides, LDL-C, body mass index, age, race, current smoking, prior percutaneous coronary intervention and receipt of evacetrapib noted log of baseline fasting plasma insulin level to be an independent predictor for overall MACE (HR = 1.36, 95% CI = 1.09–1.69, p-value = 0.007) and coronary revascularization (HR = 1.56, 95% CI = 1.21–2.00, p-value = 0.001).
Discussion
The burden of cardiovascular disease among patients with T2DM remains substantial. 16 Although treatment with multiple new agents has proven to improve cardiovascular outcomes, further mechanistic insight into the modulators of risk in this high-risk population remains a priority. This study demonstrates that among a contemporary patient population with T2DM and known high-risk vascular disease on appropriate guideline-directed medical therapy, baseline fasting insulin level was an independent predictor for MACE mainly mediated through the clinical outcome of need of revascularization. Our results suggest a pathophysiological link between baseline insulin levels and future risk for atherosclerotic vascular disease progression among patients with T2DM.
Although the association between hyperinsulinaemia and cardiovascular disease has been previously investigated in several patient populations with varied results, minimal contemporary data, if any, exist regarding the association between fasting hyperinsulinaemia and progression of atherosclerotic vascular disease in diabetic patients. In a population without diabetes, the Helsinki Policeman study and a sub-group analysis of the Quebec Cardiovascular study described fasting plasma insulin levels to be an independent predictor of stable angina and acute coronary syndromes among men without pre-existing cardiovascular or cerebrovascular disease or T2DM.8,9 Similarly, Yanase et al., 17 García et al. 18 and a subgroup analysis of the Trandolapril Cardiac Evaluation (TRACE) study found endogenous insulin levels to be an independent predictor for recurrent cardiovascular events among non-diabetic patients with established CAD. 19 A subgroup analysis of the Atherosclerosis Risk In Communities (ARIC) longitudinal cohort study demonstrated insulin resistance, as measured by the HOMA-IR, to be associated with risk of incident heart failure among patients without a diagnosis of T2DM, prior myocardial infarction or heart failure. 20 Conversely, the Paris Prospective Study, Caerphilly prospective study, Busselton study and several others have suggested that plasma insulin levels did not predict the risk of atherosclerotic vascular disease among non-diabetics independent of other cardiovascular risk factors.10–12
Several mechanisms have been proposed to explain the association of hyperinsulinaemia with atherosclerotic vascular disease. Reaven 21 introduced the concept of syndrome X, later renamed metabolic syndrome, in which resistance of peripheral tissues to insulin-mediated glucose disposal results in a cluster of risk factors including impaired glucose tolerance, elevated triglycerides, decreased HDL cholesterol, elevated blood pressure and central adiposity. This hypothesis remains controversial, and it is unclear whether the role of plasma insulin is causal versus correlative. Our observations in ACCELERATE suggest that hyperinsulinaemia may increase the risk of progression of atherosclerotic vascular disease through alterations in metabolic processes other than that mediated by dyslipidaemia as participants in our study were on statin therapy with optimal LDL levels at baseline. Excess insulin itself may directly predispose patients to cardiovascular events mediated by both an inflammatory prothrombotic state and direct effect on the arterial wall. 22 Insulin increases the synthesis of plasminogen activator inhibitor-1 which promotes thrombosis and is associated with vascular inflammation.23,24 Plasminogen activator inhibitor-1 can in turn accelerate development of atheroma within vessel walls that are prone to rupture and has been associated with an increased risk of myocardial infarction.25,26 In addition, plasminogen activator inhibitor-1 can increase proliferation of mural cellular elements and restenosis after percutaneous coronary intervention. 27 Other mechanisms have also been proposed to explain the close relationship between hyperinsulinaemia, endothelial dysfunction and hypertension, including altered cell membrane ion exchange, enhanced sympathetic and renin–angiotensin–aldosterone system activity and suppressed atrial natriuretic peptide activity. 28
Although investigation of the cardiovascular safety and clinical efficacy of newer anti-glycaemic medications is now mandated by the Food and Drug Administration, 29 the management of hyperinsulinism and insulin resistance itself in diabetic patients with vascular disease is not well studied. The Bypass Angioplasty Revascularization Investigation 2 Diabetes (BARI 2D) study was the first major study to investigate the optimal treatment for patients with T2DM and angiographically defined CAD, comparing outcomes associated with an insulin sensitizing strategy versus those with an insulin provision strategy. 30 BARI 2D found no significant difference between the two arms in terms of death or cardiovascular events; however, patients randomized to the insulin-sensitizing arm had less weight gain, higher HDL-C levels, decreased plasma insulin levels and changes in biomarker profiles suggestive of restricted fibrinolysis. 31
It is possible to treat insulin resistance with pharmacologic interventions that enhance insulin sensitivity (i.e. metformin, thiazolidinediones). As such, there was an initial enthusiasm for this treatment strategy given several studies suggesting a beneficial effect of these agents on cardiovascular outcomes.32–34 However, the ability of such an approach to improve clinical outcomes compared with weight reduction and exercise alone was tempered by data suggesting limited benefit and possible harm associated with the use of thiazolidinedione drugs. 35 Notably, our study population had very low rates of thiazolidinedione or alpha-glucosidase inhibitor usage. Furthermore, several clinical trials have demonstrated the failure of intensifying glucose control beyond the current recommendations of the American Diabetes Association to show reductions in cardiovascular events. 36 As such, aggressive lifestyle modification focusing on weight reduction, appropriate diet and increased physical activity is currently the primary therapy for the management of metabolic syndrome.37–39 Although our findings do not support routine use of baseline fasting plasma insulin level for risk stratification, further studies may be warranted regarding its utility in intensifying medical therapy and risk factor modification. In addition, it may be used as a tool for clinical trial design to identify high-risk patients with T2DM who may be more prone to cardiovascular events and thus reduce the number of patients which need to be enrolled.
Conclusion
In a contemporary population of patients with T2DM and high-risk vascular disease on optimum medical therapy, baseline fasting plasma insulin levels was an independent predictor for MACE and the need of future coronary revascularization suggesting a pathophysiological link between hyperinsulinaemia and progression of atherosclerotic vascular disease. Future studies investigating the use of fasting plasma insulin levels as a marker for risk stratification to guide use of adjunctive therapies and programmes to promote intensive lifestyle modifications among diabetic patients with high-risk vascular disease are warranted.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was sponsored by Eli Lilly and was coordinated by the Cleveland Clinic Coordinating Center for Clinical Research (C5Research) and Covance (Princeton, NJ).
