Abstract
Background:
Since several additional actions of bone bisphosphonates have been proposed, we studied the effect of the bisphosphonate alendronate (ALN) on the vascular response to environmental stress.
Methods:
Primary cultures of endothelial cells (EC) and vascular smooth muscle cells (VSMC) exposed to strained conditions were employed for experimental evaluation. After ALN treatment, cell migration, proliferation, and angiogenesis assays were performed. The participation of signal transduction pathways in the biochemical action of ALN was also assessed.
Results:
In VSMC cultures, ALN counteracted the stimulation of cellular migration elicited by the proinflammatory agent lipopolysaccharide (LPS) or by high levels of calcium and phosphorus (osteogenic medium). Indeed, ALN reduced the increase of VSMC proliferation evoked by the stressors. When LPS and osteogenic medium were added simultaneously, the enhancement of cell proliferation dropped to control values in the presence of ALN. The mechanism of action of ALN involved the participation of nitric oxide synthase, mitogen-activated protein kinase (MAPK), and protein kinase C (PKC) signaling pathways. The study revealed that ALN exhibits a proangiogenic action. On EC, ALN enhanced vascular endothelial growth factor (VEGF) synthesis, and induced capillary-like tube formation in a VEGF-dependent manner. The presence of vascular stress conditions (LPS or osteogenic medium) did not modify the proangiogenic action elicited by ALN.
Conclusion:
The findings presented suggest an extra-bone biological action of ALN, which could contribute to the maintenance of vascular homeostasis avoiding cellular damage elicited by environmental stress.
Introduction
Bisphosphonates have emerged as the first-line treatment for diseases such as postmenopausal osteoporosis, Paget’s disease, and osteolytic tumor metastasis. 1 Conventionally, the bone protective properties of bisphosphonates have been attributed to the induction of osteoclast apoptosis, leading to reduced bone resorption. 2 Indeed, a direct action on osteocytes and osteoblasts which preserve cell viability and increase the lifespan of cells has also been described. 3
Furthermore, there are many reports about extra-bone actions of bisphosphonates that provide evidence of their modulatory biochemical effects in several tissues.1,4 It has been reported that bisphosphonates exhibit an antiatherogenic action, evoked by the reduction of intima-media vessels thickening, and arterial calcification prevention.5–7
Osteoporosis and cardiovascular diseases such as atherosclerosis represent multifactorial entities frequently related to aging, which share pathophysiological similarities. 8 Notably, in atherosclerotic calcification, vascular tissue is replaced by osteogenic cells and extracellular matrix mineralization of the intima. 9 Under healthy conditions, vascular smooth muscle cells (VSMC) exhibit a contractile phenotype and low turnover rates. However, in an inflammatory microenvironment or in the presence of high calcium and phosphate levels, VSMC switch towards a synthetic phenotype, which leads the cells to express extracellular matrix proteins involved in cell proliferation and migration.10,11 This biochemical interplay between bone and vascular metabolism hypothesizes a bidirectional pharmacological intervention for the simultaneous treatment of bone and vascular diseases.
In adult vessels, revascularization represents a survival mechanism for ischemic tissue. 12 In response to stress conditions, blood vessels trigger angiogenesis as a mechanism of damaged tissue repair. 13
Studies from our laboratory have demonstrated that alendronate (ALN) lowers cellular and molecular events involved in atherosclerosis. ALN acutely enhances endothelial nitric oxide production, and prevents adhesion of platelets and monocytes to endothelial cells (EC) by down-regulation of cell adhesion molecules expression. The drug also exhibits the ability to decline the osteogenic transdifferentiation of VSMC. In contrast, on calvarial osteoblasts, ALN promotes osteoblastogenesis and increases mineralization. 14
In the present work, the effect of ALN on vascular response to extracellular environmental stress was investigated. We studied the impact of ALN on cellular proliferation and migration profile, and on the ability to vascularize when VSMC or EC were exposed to the stressful conditions. To that end, we selected bacterial lipopolysaccharide (LPS) treatment or cell incubation with high calcium and phosphate levels to mimic an inflammatory condition or vascular calcification induction, respectively.
Materials and methods
Materials
Fetal bovine serum (FBS) was obtained from Natocor (Villa Carlos Paz, Argentina). Phenol red-free Dulbecco’s modified Eagle’s medium (DMEM), β-glycerophosphate disodium, ALN, Nω-nitro-
Animals
We used young female (3–5 weeks old) Wistar rats fed with standard rat food, given water ad libitum, and maintained on a 12 h light/12 h darkness cycle. All procedures concerning animals and their care were achieved at the Unit of Animal Care belonging to the Biology, Biochemistry and Pharmacy Department of the National University of the South (UNS) in accordance with the guidelines published in the National Institutes of Health Guide for the Care and Use of Laboratory Animals. The protocol used in this work was approved by The Animal Care Use Committee of this Unit.
Vascular cells culture
Primary EC and VSMC cultures were obtained from aortic rings explants isolated from young Wistar rats (3–5 weeks old) as previously described. 15 Further details are available in the online supplementary content.
Measurements of protein levels of VEGF
Vascular endothelial growth factor (VEGF) levels were measured in the culture supernatants using a VEGF ELISA Kit (Sigma-Aldrich, St Louis, MO, USA) according to the manufacturer’s instructions, as previously described. 16 Further details are available in the online supplementary content.
In vitro tube formation assay
Tube formation assay was performed using three-dimensional fibrin-based matrices, by an adaptation of the technique described by Koolwijk et al. 17 Further details are available in the online supplementary content.
Wound healing assay
Cell migration was carried out using the wound healing assay, as previously described. 18 Further details are available in the online supplementary content.
Transwell VSMC migration assay
VSMC migration assay was carried out using a co-culture system, by adaptation of a transwell cell migration assay previously described. 19 Further details are available in the online supplementary content.
Methyl thiazolyl tetrazolium (MTT) cell proliferation assay
VSMC proliferation was evaluated by MTT proliferation assay. 20 Further details are available in the online supplementary content.
Statistics
Results were expressed as mean ± SD of five independent experiments (n = 5) where each experimental condition was carried out in sextuplicate. Comparisons between means were made using one-way ANOVA followed by Fisher’s least significant difference test (Fisher LSD). All statistical analysis was performed using IBM SPSS Statistics software for Windows, Version 23.0 (IBM Corp., Armonk, NY, USA). All p-values below 0.05 were considered statistically significant.
Results
In order to induce environmental stress, cell cultures were incubated with high levels of calcium and phosphorus or exposed to the proinflammatory agent LPS. To that end, cells were incubated in normal DMEM or osteogenic medium (DMEM plus 4 mM CaCl2 and 10 mM β-glycerophosphate). When the proinflammatory agent LPS was used (1 µg/mL), it was added 30 min prior to ALN treatment. As we reported previously, osteogenic medium represents a cellular stressor that prompts VSMC transdifferentiation into bone-like cells. 14
ALN prevents VSMC proliferation induced by stressors: mechanism of action
The effect of ALN on the stimulation of VSMC proliferation evoked by the stressors was evaluated. In Figure 1A it can be observed that after 96 h of treatment with 1, 5 or 10 µM ALN, no significant changes in cell proliferation were detected at all concentrations tested. When cells were exposed to LPS, VSMC proliferation was significantly increased (31% above control, p < 0.02). Pretreatment of VSMC with ALN (1–10 μM) completely abolished cell proliferation induced by LPS. Similar results were obtained when cells were cultured in osteogenic medium. The remarkable stimulation of VSMC proliferation induced by the osteogenic medium (108% vs control, p < 0.001) was partially reduced in the presence of ALN. Indeed, Figure 1A show that preincubation of the monolayers with ALN dropped to control values the enhancement on cell proliferation induced by simultaneous treatment with osteogenic medium and LPS.
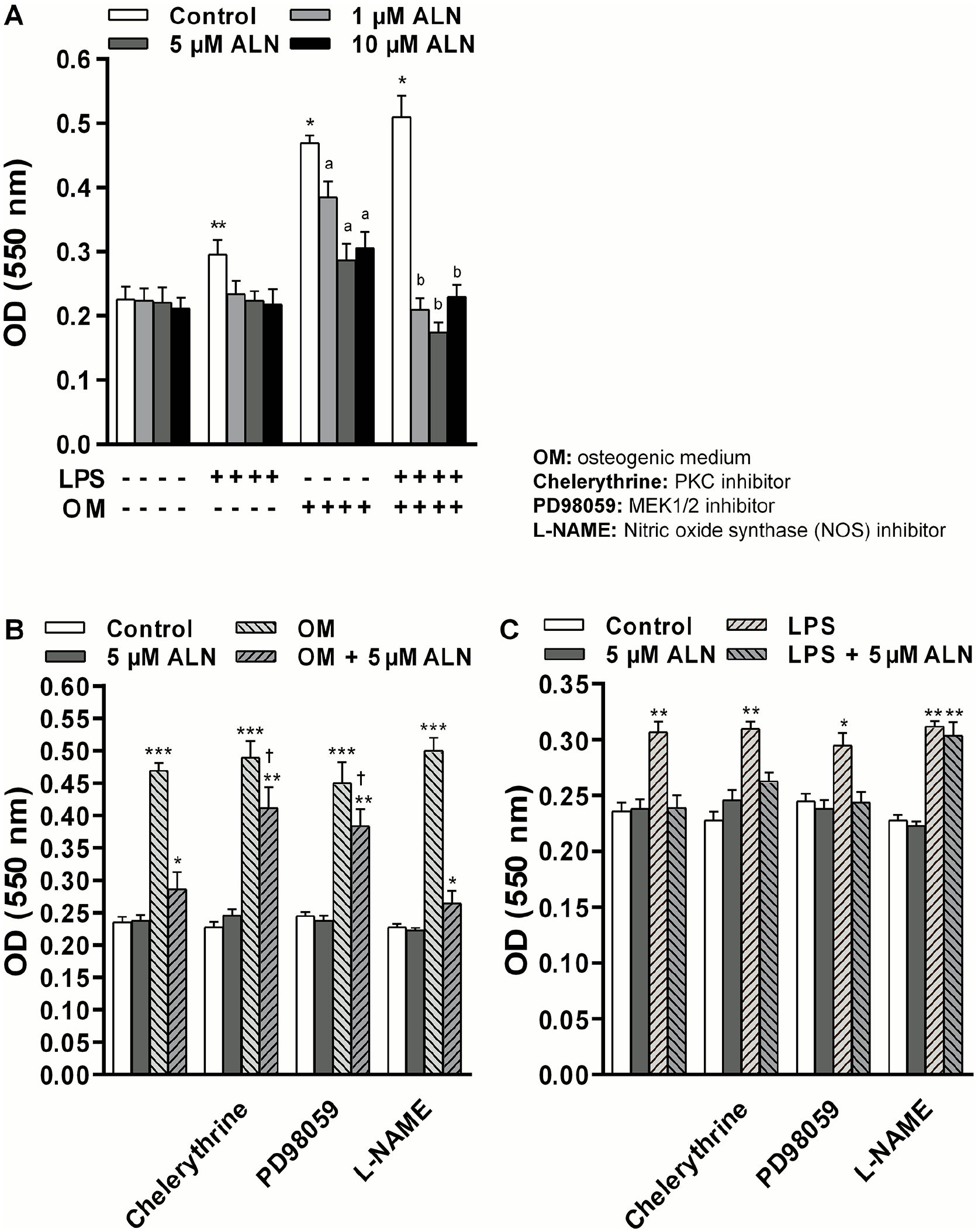
Effect of LPS and calcifying medium on the regulation of VSMC proliferation mediated by ALN: signal pathways involved. VSMC were treated with ALN, at the indicated concentrations, or vehicle (control) for 96 h in DMEM or OM (DMEM plus 4 mM CaCl2 and 10 mM β-glycerophosphate). LPS (1 µg/mL) was added 30 min before ALN treatment. When the indicated inhibitors were used, they were added to the incubation medium 1 h before ALN treatment. Cell proliferation was measured by MTT assay as described in the ‘Materials and methods’ section. Results represent the mean ± SD of five independent experiments (n = 5) in which each experimental condition was performed in sextuplicate. Comparisons between means were made using one-way ANOVA followed by Fisher’s least significant difference test.
The involvement of intracellular pathways on the inhibition of cell proliferation induced by ALN was tested. Specifically, we checked the participation of mitogen-activated protein kinase (MAPK), protein kinase C (PKC), and nitric oxide synthase signaling pathways on the mechanism of action elicited by ALN. The data presented in Figure 1B shows that in an osteogenic microenvironment, pretreatment of VSMC with the PKC inhibitor chelerythrine (1 μM), or with the MEK1/2 inhibitor PD98059 (5 μM), partially abrogated the inhibitory action of ALN (5 μM) on cell proliferation. In the presence of the inhibitors, cell proliferation rose towards 43% above osteogenic medium plus ALN (p < 0.05). However, when cell monolayers were preincubated with the nitric oxide synthase inhibitor
ALN suppresses VSMC migration elicited by the stressors
Since muscle cell could alter its migration profile as a response to vascular stress, the effect of ALN on VSMC migration was assessed. Figure 2 shows that ALN exposure did not modify the cell migratory pattern. In contrast, a marked increase in cell migration was detected when cells were incubated with norepinephrine, a well-known stimulatory agent of vascular cell motility (199% above control; p < 0.001). Simultaneous treatment with ALN (5 µM) markedly reduced the cell migration pattern evoked by norepinephrine. The incubation of cell monolayers with LPS or osteogenic medium enhanced cell migration (65% and 70% above control, respectively; p < 0.02), and the presence of ALN partially suppressed the stimulatory action elicited by the stressors (online Supplementary Figure 1).
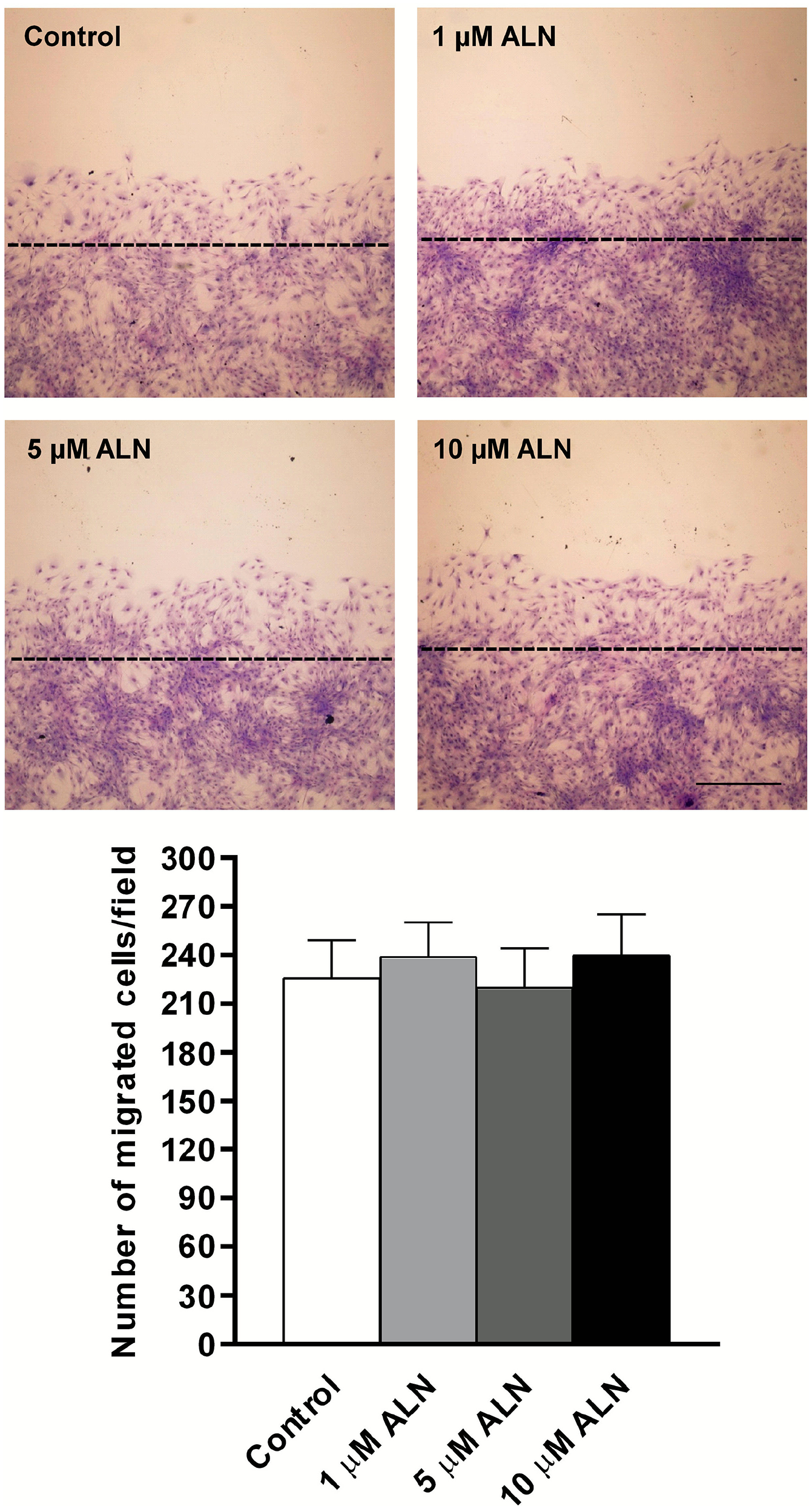
Effect of ALN on VSMC migration. Cell migration was carried out using the wound healing assay, as described in the ‘Materials and methods’ section. After scraping, the remaining monolayer was treated with the indicated concentrations of ALN for 48 h. Images show representative fields after Giemsa staining (40× magnification, scale bar = 30 μM). Results represent the mean ± SD of five independent experiments (n = 5) in which each experimental condition was performed in sextuplicate. Comparisons between means were made using one-way ANOVA followed by Fisher’s least significant difference test.
Having in mind that, in blood vessels, muscle cell motility could be influenced by tight interactions with EC, we tested whether the presence of EC would modulate VSMC migration. For this purpose, co-culture assays were performed using the transwell system. To that end, EC were seeded in the lower compartment of the chamber and VSMC in the upper one. After 48 h of treatment with ALN (1–10 µM), no significant differences were detected in the number of VSMC that migrated through the membrane with respect to the control group (online Supplementary Figure 2). LPS (1 μg/mL) markedly enhanced cell movement across the membrane (69% vs control, p < 0.001). Noteworthy, a complete suppression of VSMC migration induced by the proinflammatory agent was detected when ALN treatment was performed in the presence of EC (online Supplementary Figure 2). Similar results were obtained with osteogenic medium. These results suggest that the biochemical action of ALN requires the concomitant presence of EC and VSMC to completely counteract the effect of vascular stressors.
Proangiogenic action of ALN is sustained under vascular stress
To test the effect of ALN on angiogenesis, capillary tube formation assays were carried out. To that end, EC were seeded on a three-dimensional fibrin-based matrix and exposed to ALN. As can be observed in the photomicrographs and the quantitative data of Figure 3, a great number of tube-like structures were detected after 96 h of treatment with ALN (5.34 ± 0.48, 6.94 ± 0.58, 7.52 ± 0.53, and 7.2 ± 0.68 mm; control, 1, 5, and 10 μM ALN, respectively, p < 0.05). We found that the proangiogenic action of ALN was mediated by VEGF, since the presence of the VEGF receptor-2 (VEGFR-2) inhibitor, compound SU5416, completely suppressed the angiogenic stimulus of ALN (Figure 4A). Indeed, we provided evidence that ALN stimulated EC-VEGF synthesis, since a significant increase in the growth factor production compared to control group (35.7% above control, p < 0.05) was detected (online Supplementary Figure 3). Considering that hypoxia-inducible factor-1 (HIF-1) is a well-known stimulatory factor of VEGF expression, CoCl2 (HIF-1 inducer) was used as a positive control of VEGF synthesis. The data revealed that treatment with the bisphosphonate ALN increased VEGF synthesis in a similar quantity than CoCl2 (online Supplementary Figure 3).
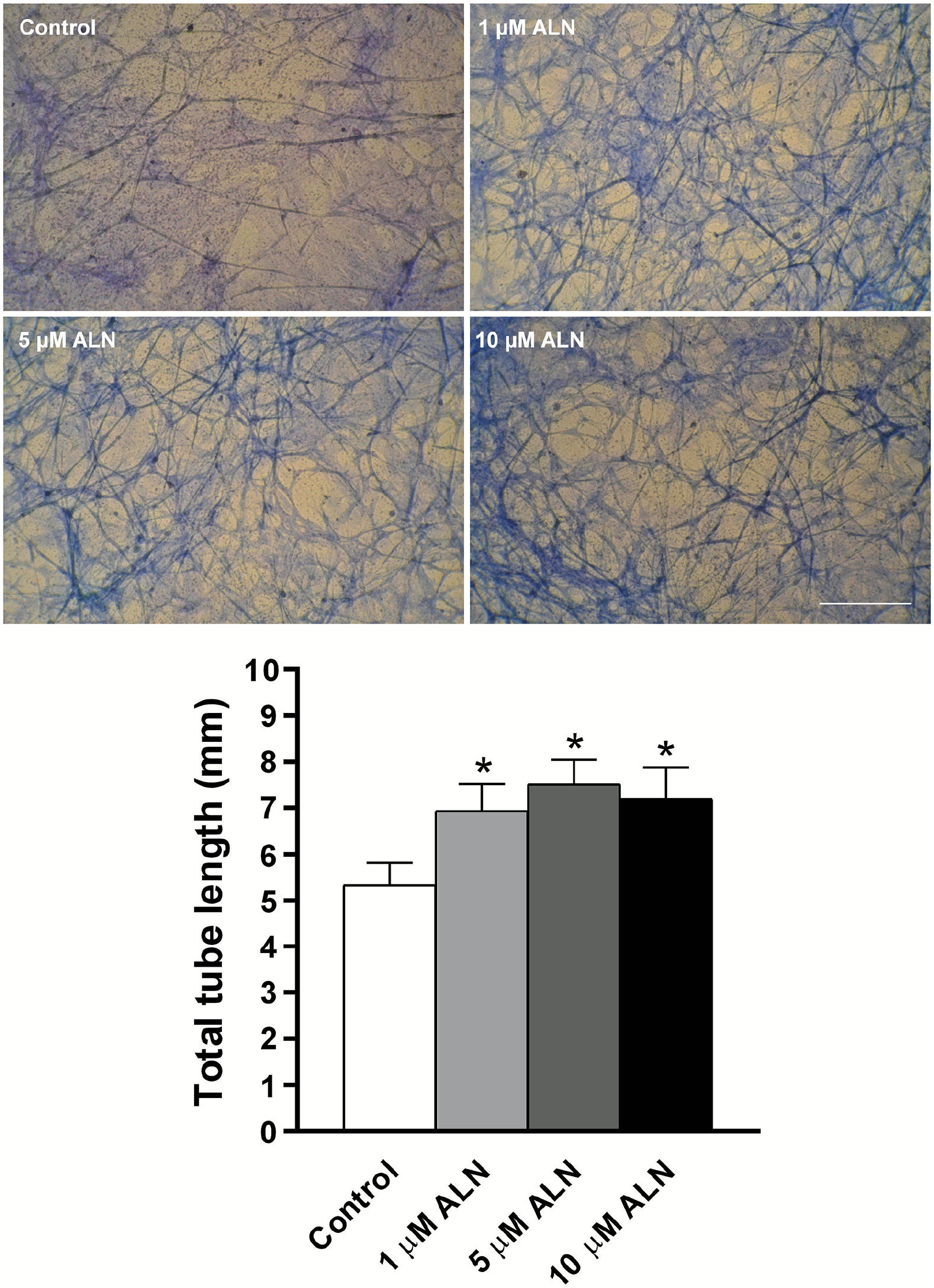
Effect of ALN treatment on capillary tubes formation. EC were seeded on each gel and exposed to ALN at the indicated concentrations for 96 h, or vehicle alone (control). Tube formation assay was carried out as described in the ‘Materials and methods’ section. Images show representative fields after Giemsa staining (40× magnification, scale bar = 30 μM). The total length of vessel segments was quantified using optical microscopy and ImageJ software. Results represent the mean ± SD of five independent experiments (n = 5) in which each experimental condition was performed in sextuplicate. Comparisons between means were made using one-way ANOVA followed by Fisher’s least significant difference test.
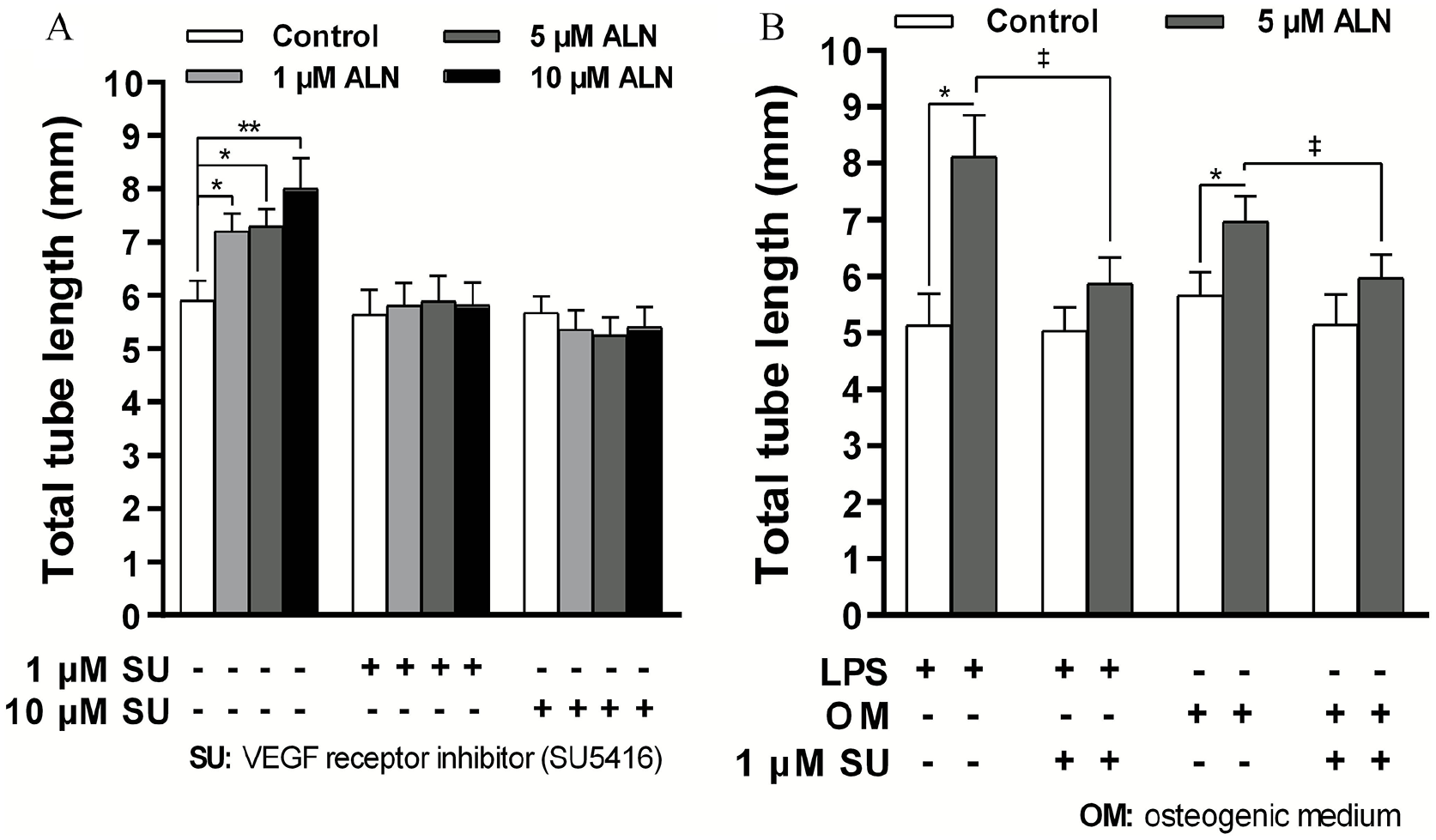
Effect of LPS and calcifying medium on capillary tubes formation induced by ALN: involvement of VEGF receptor. A: Endothelial cell suspensions in DMEM or in osteogenic medium were seeded on each gel and exposed and exposed to ALN at the indicated concentrations for 96 h, or vehicle alone (control). B: When LPS (1 µg/mL) was used, it was added 30 min prior ALN. The VEGF receptor inhibitor (compound SU5416) was added 1 h before ALN treatment. The total length of vessel segments was quantified using optical microscopy and ImageJ software. Results represent the mean ± SD of five independent experiments (n = 5) in which each experimental condition was performed in sextuplicate. Comparisons between means were made using one-way ANOVA followed by Fisher’s least significant difference test.
Finally, we tested whether the environmental stress could modify the angiogenic action exhibited by ALN. Figure 4B shows that in the presence of LPS or osteogenic medium the stimulatory action of ALN on capillary tube formation was sustained (58% or 23% above control, respectively). Indeed, the results provided evidence that the VEGF pathway was also involved in the angiogenic effect of ALN under vascular stress. The presence of SU5416 blunted the proangiogenic action of ALN elicited under LPS or osteogenic medium exposure.
Discussion
In the present work, we provided evidence that ALN exhibits a protective effect on the vascular system, thus avoiding cellular alterations triggered by external stressors. ALN preserves cell viability and prevents the enhancement of VSMC proliferation and migration induced by environmental stress. The mechanism of action involves MAPK, PKC, and nitric oxide synthase signal transduction pathways. Furthermore, ALN exhibits a proangiogenic action that is not affected by vascular stress.
The regulation of cellular behaviors, such as proliferation, differentiation, migration, or survival, plays an important role in many tissues. We provided evidence that ALN prevented the stimulation of VSMC proliferation induced by extracellular stress, through the activation of signaling cascades, and partially diminished VSMC migration elicited by inflammatory stress. Prevention of VSMC migration could contribute to reduce the risk of intima thickening. Moreover, the biochemical action of ALN on VSMC migration regulation requires the concomitant presence of EC, which suggests that the putative protective vascular effect of ALN involves a crosstalk between VSMC and EC. Other authors provided evidence that ALN induced osteoblastogenesis mediated by interactions between EC and mesenchymal stem cells. 22
Revascularization arises as a helpful mechanism for tissue regeneration. The repair of adult vessels exposed to inflammatory or oxidative stress can be aided by the angiogenesis process. 23 Here, we provide evidence that ALN stimulated endothelial VEGF synthesis, and that the activation of VEGF signaling prompted capillary tube formation. Moreover, our findings revealed that the stressors do not affect the ability of ALN to promote new vessel generation. The data available in the literature show that the bisphosphonates exert pro-24,25 or antiangiogenic actions. 26 This controversy would depend on several factors such as the type of bisphosphonate, time of exposure, and cellular source. Suppression of angiogenesis is of clinical importance in bone metastasis where high doses of bisphosphonates are employed to inhibit VEGF-induced angiogenesis and arrest tumor growth. 27 However, in osteoporotic patients treated with lower doses of bisphosphonates, angiogenesis may be desirable for an appropriate bone remodeling and adequate bone mass enhancement.
Conclusions
Overall, the findings presented in this work suggest an extra-bone biological action of ALN, which could contribute to the maintenance of vascular homeostasis, thus avoiding cellular damage elicited by environmental stress. However, since the physiological or pathophysiological interpretation of our results are limited to in vitro scenery, future investigations are required to fully comprehend whether the data presented mean a promising effect of ALN on vascular repair and remodeling.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X221112168 – Supplemental material for Vascular response to stress: Protective action of the bisphosphonate alendronate
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X221112168 for Vascular response to stress: Protective action of the bisphosphonate alendronate by Pablo H Cutini, Adrián E Campelo and Virginia L Massheimer in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X221112168 – Supplemental material for Vascular response to stress: Protective action of the bisphosphonate alendronate
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X221112168 for Vascular response to stress: Protective action of the bisphosphonate alendronate by Pablo H Cutini, Adrián E Campelo and Virginia L Massheimer in Vascular Medicine
Supplemental Material
sj-tif-3-vmj-10.1177_1358863X221112168 – Supplemental material for Vascular response to stress: Protective action of the bisphosphonate alendronate
Supplemental material, sj-tif-3-vmj-10.1177_1358863X221112168 for Vascular response to stress: Protective action of the bisphosphonate alendronate by Pablo H Cutini, Adrián E Campelo and Virginia L Massheimer in Vascular Medicine
Supplemental Material
sj-tif-4-vmj-10.1177_1358863X221112168 – Supplemental material for Vascular response to stress: Protective action of the bisphosphonate alendronate
Supplemental material, sj-tif-4-vmj-10.1177_1358863X221112168 for Vascular response to stress: Protective action of the bisphosphonate alendronate by Pablo H Cutini, Adrián E Campelo and Virginia L Massheimer in Vascular Medicine
Supplemental Material
sj-tif-5-vmj-10.1177_1358863X221112168 – Supplemental material for Vascular response to stress: Protective action of the bisphosphonate alendronate
Supplemental material, sj-tif-5-vmj-10.1177_1358863X221112168 for Vascular response to stress: Protective action of the bisphosphonate alendronate by Pablo H Cutini, Adrián E Campelo and Virginia L Massheimer in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by grants from the Secretaría General de Ciencia y Tecnología (SGCyT), Universidad Nacional del Sur, Bahía Blanca, Argentina (PGI 24/B203), Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, Argentina, PIP D-4061), and Agencia Nacional de Promoción Científica, Argentina (PICT-2017-1096). The funders were not involved in the study or manuscript preparation.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
