Abstract
Keywords
Introduction
Percutaneous coronary intervention (PCI) is regarded as a useful treatment for coronary heart disease (CHD) with atherosclerosis, but approximately 30–50% of patients have vascular restenosis (RS) within half a year after PCI.1,2 Among them, neointimal hyperplasia and vascular remodeling are important pathological mechanisms of RS. 3 Therefore, it is of great importance to study the mechanism of vascular neointimal hyperplasia after injury to identify effective intervention targets.
The renin-angiotensin system (RAS) is an important system for maintaining normal blood pressure, water and electrolyte balance, and cardiovascular homeostasis. 4 In recent years, a comprehensive understanding of RAS has increased with increasing research. For example, the renin (pro) receptor ([pro]renin receptor, [P]RR) was discovered in 2002. 5 There are two main functions of renin-specific receptors. First, they receive signals from the cell membrane of Ang II and trigger a cascade of biological responses, including vasoconstriction, facilitation of adrenal secretion of catecholamines, and augmentation of tubular reabsorption in the kidneys. Additionally, it engages in interactions with other molecules, thus eliciting certain biological effects unrelated to Ang II.6,7 As previously reported, after prorenin binds to renin-specific receptors, it activates the mitogen-activated protein kinase–extracellular signal-regulated kinase 1/2 (MAPK-ERK1/2) signal transduction pathway and further promotes the expression of transforming growth factor-β1 (TGF-β1) and plasminogen activator inhibitor-1 (PAI-1), which accelerates end-organ fibrosis. 6 Studies have shown that the combination of prorenin and (pro)renin receptor ([P]RR) can promote renal fibrosis and myocardial fibrosis in an Ang II-independent manner. 7
Vascular smooth muscle cells (VSMCs) play an indispensable role in the process of neointimal hyperplasia and vascular remodeling.8,9 VSMCs have been confirmed to have prorenin receptors, and renin and prorenin can promote the proliferation of VSMCs through (P)RR. 10 Thus, we inferred that the receptor-associated prorenin system (RAPS) plays an essential role in the occurrence of RS. However, it is not clear whether prorenin promotes the proliferation of VSMCs in a (P)RR-mediated Ang II-independent manner.
In this study, we investigated whether (P)RR can mediate inflammation, oxidative stress, and the ERK1/2-AKT pathway.
Methods
Balloon injury model and treatment
The Experimental Animal Center of Shanxi Medical University provided healthy male Sprague Dawley rats weighing 250–300 g. Sodium pentobarbital (2%, 50 mg/kg) was intraperitoneally injected for anesthesia. The skin and subcutaneous tissue were incised along the anterior midline of the neck, and the left common carotid artery and internal and external carotid arteries in the anterior cervical triangle were exposed. Afterwards, the distal end of the external carotid artery was ligated, and the blood flow at the proximal end of the common carotid artery was closed with a vascular clip. A wedge shape was incised inside the distal end of the external carotid artery, a 1.5 F balloon catheter was inserted from the external carotid artery to the beginning of the common carotid artery, and the balloon was inflated to block blood flow for 30 seconds. Subsequently, the balloon was slowly pulled back to the bifurcation of the internal and external carotid arteries three times, and then the balloon was withdrawn. Finally, the outside of the neck arteries were ligated, after which the subcutaneous tissue and skin were sutured layer by layer.
Rats were randomly divided into five groups: sham operation (sham), a common carotid artery on the noninjured side of model rats (control), a common carotid artery on the injured side of model rats (injury), PRO20, and losartan (Los) groups. The specific (P)RR inhibitor PRO20 (700 μg/kg/day, subcutaneously; Cayman Chemical, USA) was used to treat mice in the PRO20 group. The positive control group (Los) was intragastrically administered 100 mg/kg losartan (which is an antagonist of the angiotensin I receptor) (Sigma, USA). All these treatments were administered for 17 consecutive days beginning 3 days before surgery. All the surgical interventions and postoperative animal care were performed following the NRC’s Guide for the Care and Use of Laboratory Animals, Eighth Edition and the ‘Guidelines and Policies for the Care and Use of Laboratory Animals’ provided by Shanxi Medical University, with an associated permit number (Dr2008043).
Cell culture and treatment
Rat VSMCs were purchased from Procell (Wuhan, China) and were cultured in Dulbecco’s Modified Eagle Medium (DMEM) (Gibco, USA) supplemented with 10% fetal bovine serum (FBS), 100 units/mL penicillin, and 100 μg/mL streptomycin under 5% CO2 at 37 °C. Cells were treated with different concentrations of renin (Sigma‒Aldrich, USA), 10–6 mol/L Los, 10–5 mol/L PD123319 (which is an antagonist of angiotensin II receptor) (Sigma), 10 nmol/L platelet-derived growth factor-BB (PDGF-BB) (PeproTech, USA), or 10–5 mol/L diphenyleneiodonium chloride (DPI; which is an inhibitor of nicotinamide adenine dinucleotide phosphate [NADPH]) (Sigma‒Aldrich). Renin, PDGF-BB, and DPI were added after the cells were treated with Los and PD123319 for 30 min.
Construction of the miRNA interference plasmid for pro(renin) receptor, (P)RR
Four pairs of microRNA (miRNA) and negative control (NC) sequences were designed and synthesized according to the gene sequence (GenBank accession number: AB188298). Afterwards, the miRNAs and the NCs were cloned and inserted into the pcDNA™6.2-GW/EmGFP vector (Invitrogen, USA) to determine the most effective miRNA. Cells were transfected with miRNA-(P)RR using Lipofectamine™ 2000 reagent (Invitrogen).
Cell Counting Kit-8 (CCK-8) assay
The logarithmic phase VSMCs were collected, and the cell suspension concentration was adjusted to 1 × 104/well. Subsequently, the medium was replaced with DMEM (without FBS), and the cells were incubated for another 24 h. Cells that were treated or transfected with different reagents were incubated with 5% CO2 at 37 °C overnight until the cell monolayer covered the bottom of the 96-well plate. Afterwards, cells in each well were mixed with 10 μL of Cell Counting Kit-8 (CCK-8) assay (CCK-8 (Solarbio, Beijing, China) solution and continued to culture for another 4 h. A microplate reader was used to detect the optical density of each well at 450 nm wavelength.
Detection of the cell cycle
Cells were seeded into six-well plates at a density of 1 × 106/well and cultured overnight. Afterwards, the cells were cultured in DMEM (without FBS) for 24 h in a 37 °C, 5% CO2 incubator. The cells were then treated or transfected with the corresponding reagents for 12 h. We collected the cells, washed them three times with phosphate-buffered saline (PBS), and added 70% ethanol to fix the cells. Subsequently, the cells were collected, and RNase was added for incubation at 37 °C for 30 min. Afterwards, the cells were stained with 50 μL of propidium iodide (PI) and incubated at 4 °C for 30 min in the dark. Finally, the cell cycle distribution was detected via flow cytometry (FCM) and analyzed by using ModFit LT software (Verity Software House, Bedford, MA, USA).
Measurement of malondialdehyde and superoxide dismutase
VSMCs were seeded into plates and cultured overnight. Afterwards, the cells were collected for experiments. The production of malondialdehyde (MDA) and the antioxidant enzyme superoxide dismutase (SOD) in VSMCs was examined by using an MDA assay kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, Jiangsu, China) and a superoxide dismutase assay kit (Nanjing Jiancheng Bioengineering Institute), respectively, based on the manufacturer’s specifications.
Measurement of reactive oxygen species
The Reactive Oxygen Species Assay Kit (Beyotime, Haimen, Jiangsu, China) was used in this study. Briefly, VSMCs were inoculated in a six-well plate (1 × 104 cells/well) and cultured overnight until 70–80% confluence was reached, after which the experiments were performed. The positive control group was treated with H2O2 at a final concentration of 100 μmol/L for 1 h. At this point, the culture medium was discarded, and the cells were rinsed three times with PBS at room temperature. Subsequently, the DCFH-DA fluorescent probe was added to the cells at 10 μmol/L, and the cells were incubated at 37 °C in the dark for 20 min and rinsed three times with PBS to observe the intracellular fluorescence under a microscope. The cell suspension was collected and transferred to a flow tube, 400 μL of PBS was added, and the fluorescence intensity was measured with a flow cytometer.
Immunofluorescence staining assay
VSMCs in a 12-well plate were treated with different reagents for 24 h. After washing with PBS, the cells were fixed with 4% paraformaldehyde, permeabilized with 0.2% Triton X-100 (Sigma) in PBS for 15 min, and washed in PBS again. Afterwards, 5% nonfat milk was added to the desired wells for 30 min at 37 °C. The cells were then washed and stained with primary antibodies against (P)RR (Sigma‒Aldrich) and alpha-smooth muscle actin (α-SMA) (Abcam, UK) at 4 °C overnight. After six washes, the cells were incubated with fluorescein isocyanate (FITC)-conjugated goat antimouse (KPL, USA) secondary antibody at 37 °C for 1 h. Next, 4′,6-diamidino-2-phenylindole (DAPI; Solarbio) was added for incubation in the dark for 5 min, and the excess DAPI was removed by washing with PBS for 5 min. Finally, images were acquired by using a fluorescence microscope (Nikon, Tokyo, Japan) at 400× magnification.
Hematoxylin and eosin (H&E) staining
Common carotid artery tissues from each group were fixed in 4% paraformaldehyde at room temperature, embedded in paraffin, sectioned at a thickness of 5 μm, and stained with hematoxylin and eosin (H&E). Vascular intimal hyperplasia and histopathological changes were observed under a light microscope (Olympus, Tokyo, Japan) at 100× and 400× magnification and quantified by using the intima/media area ratio (I/M).
Enzyme-linked immunosorbent assay
Arterial blood from mice and VSMCs in each group were collected, and the levels of renin (Abcam) and PDGF-BB (PeproTech) in the mice, as well as the levels of proinflammatory cytokines, including tumor necrosis factor-α (TNF-α; Beyotime), interleukin-6 (IL-6; Abcam), IL-1β (Beyotime), and inducible nitric oxide synthase (iNOS; Abcam), were determined via enzyme-linked immunosorbent assay (ELISA based on the manufacturer’s instructions. The optical density (OD) value of each well was immediately read at 450 nm.
Real-time polymerase chain reaction
Total RNA from common carotid artery wall tissues or VSMCs was isolated by using TRIzol reagent according to the manufacturer’s protocol (Invitrogen, Thermo Fisher Scientific, USA). The expression levels of NOX1, cyclin D1, proliferating cell nuclear antigen (PCNA) in tissues and (P)RR, NOX1, cyclin D1, and PCNA in cells were determined by using the SYBR Premix Ex Taq II Kit (Takara, Tokyo, Japan) based on the manufacturer’s protocol. β-Actin served as a control. The 2–ΔΔCt method was used to quantify the relative expression levels of (P)RR, NOX1, cyclin D1, and PCNA.
Western blot analysis
To confirm the expression levels of ERK1/2, p-ERK1/2, AKT, p-AKT, cyclin D1, PCNA, (P)RR, and NOX1 in common carotid artery wall tissues and VSMCs, HTR8/SVneo cells were lysed with Radio-Immunoprecipitation Assay (RIPA) buffer (Beyotime, China). Lysates were separated on a 10% SDS‒polyacrylamide gel, and proteins were transferred to polyvinylidene difluoride (PVDF) membranes. Afterwards, the membranes were blocked with 5% nonfat milk for 1 h at room temperature and incubated with the corresponding primary antibodies at 4 °C overnight, followed by incubation with a horseradish peroxidase-conjugated secondary antibody at room temperature for 1 h. Protein expression was detected with a high-sensitivity ECL chemiluminescence detection kit (Vazyme, China). The band intensity was expressed by normalizing the data to the values of β-actin by using ImageJ (USA) software.
Statistical analysis
Statistical analysis was performed with IBM SPSS Statistics 22.0 (Armonk, NY, USA) and GraphPad (USA) Prism 9 software. The data are shown as mean values ± SD. One-way analysis of variance (ANOVA) or two-way ANOVA followed by post hoc tests were used to compare the differences among multiple groups. Furthermore, p < 0.05 was considered to indicate statistical significance.
Results
Renin improved VSMC proliferation
To explore the effects of renin on VSMC proliferation, VSMCs were treated with Los and PD123319 or further supplemented with different concentrations of renin after 30 min. A CCK-8 assay showed that the OD at different concentrations of renin increased markedly in a concentration-dependent manner compared with that in the control group (Figure 1A). The FCM results demonstrated that the proportion of VSMCs in the G1 phase decreased with increasing renin concentration, but the percentage of VSMCs in the S phase and the proliferation index increased (Figures 1B, 1C). RT‒PCR and WB assays indicated that both the mRNA and protein levels of cyclin D1 and PCNA were markedly upregulated with increasing concentrations of renin (Figures 1D, 1E). Furthermore, to observe the expression of (P)RR following renin treatment, an immunofluorescence staining assay was conducted. The results showed that a high concentration of renin enhanced the mean fluorescence intensity (MFI) of (P)RR (Figure 1F). These data suggested that the level of (P)RR increased after renin treatment and that renin promoted the proliferation of VSMCs in a concentration-dependent and Ang II-independent manner.
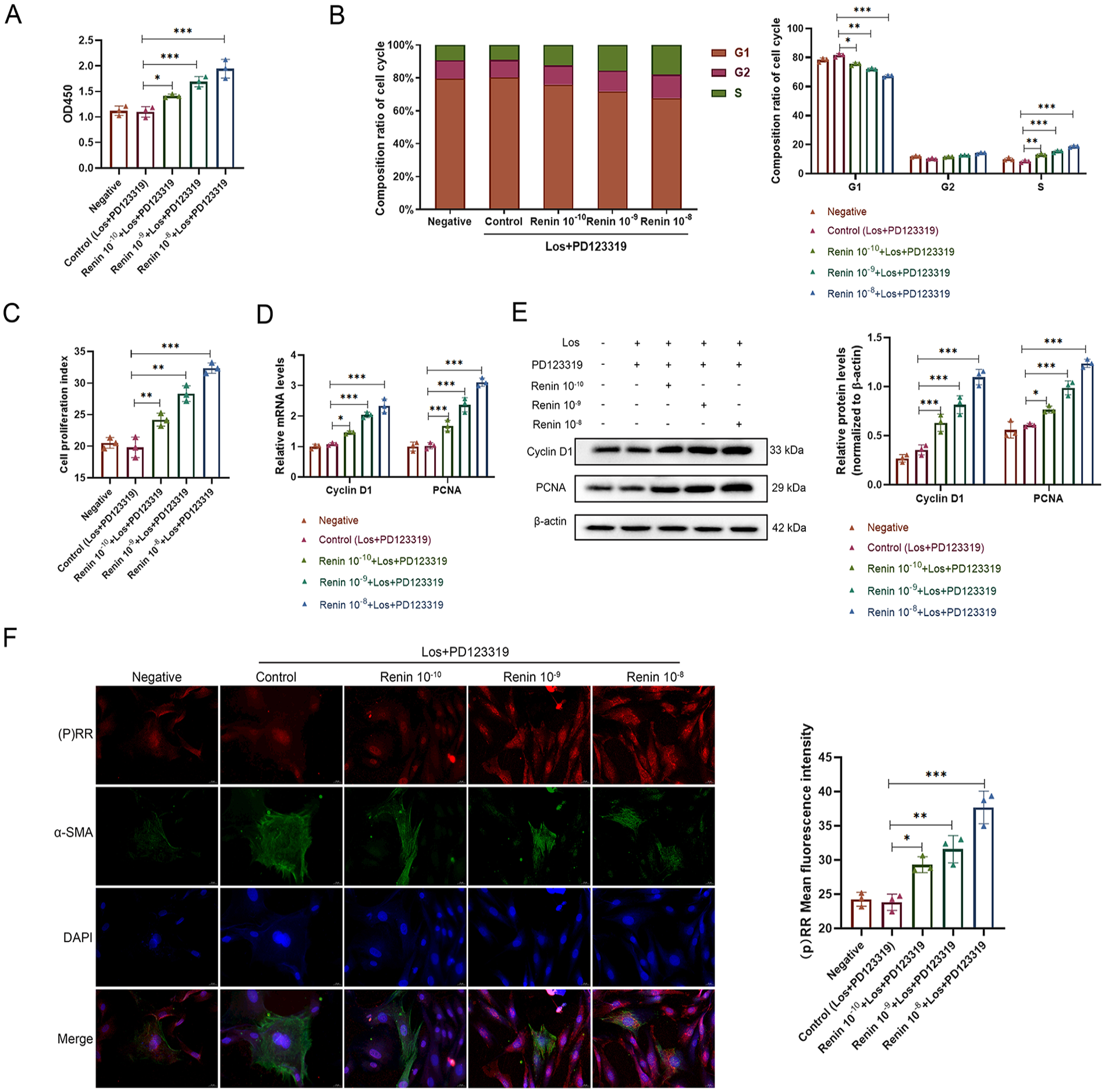
Renin improved the proliferation of VSMCs.
Silencing (P)RR restrained VSMC proliferation
To determine whether the function of renin was associated with (P)RR, we designed and synthesized four pairs of miRNA and NC sequences to silence (P)RR, and the resulting construct was termed RNAi-(P)RR. The sequences are shown in Table 1. The most effective miRNA was RNAi-(P)RR-2 (Figures 2A, 2B). Among the growth factors that are released by damaged cells in the early stage of arterial injury, PDGF-BB can promote the proliferation of VSMCs. 11 Therefore, the addition of PDGF-BB can serve as a positive control. First, the mRNA and protein levels of (P)RR were both significantly downregulated by RNAi-(P)RR treatment, as determined via RT‒PCR and WB assays (Figures 2C, 2D). In the miRNA-(P)RR + PDGF-BB or miRNA-(P)RR + renin group, the proportion of cells in the G1 phase increased, whereas the proportion of cells in the S phase and the proliferation index decreased (Figures 2E, 2F). This was further demonstrated by the markedly reduced mRNA and protein levels of cyclin D1 and PCNA in the RNAi-(P)RR-treated groups (Figures 2G, 2H). These results suggested that the proliferative function of renin in VSMCs was mediated by (P)RR in an Ang II-independent manner.
MicroRNA oligomeric single-stranded DNA sequence of (pro)renin receptor.

miRNA, microRNA.
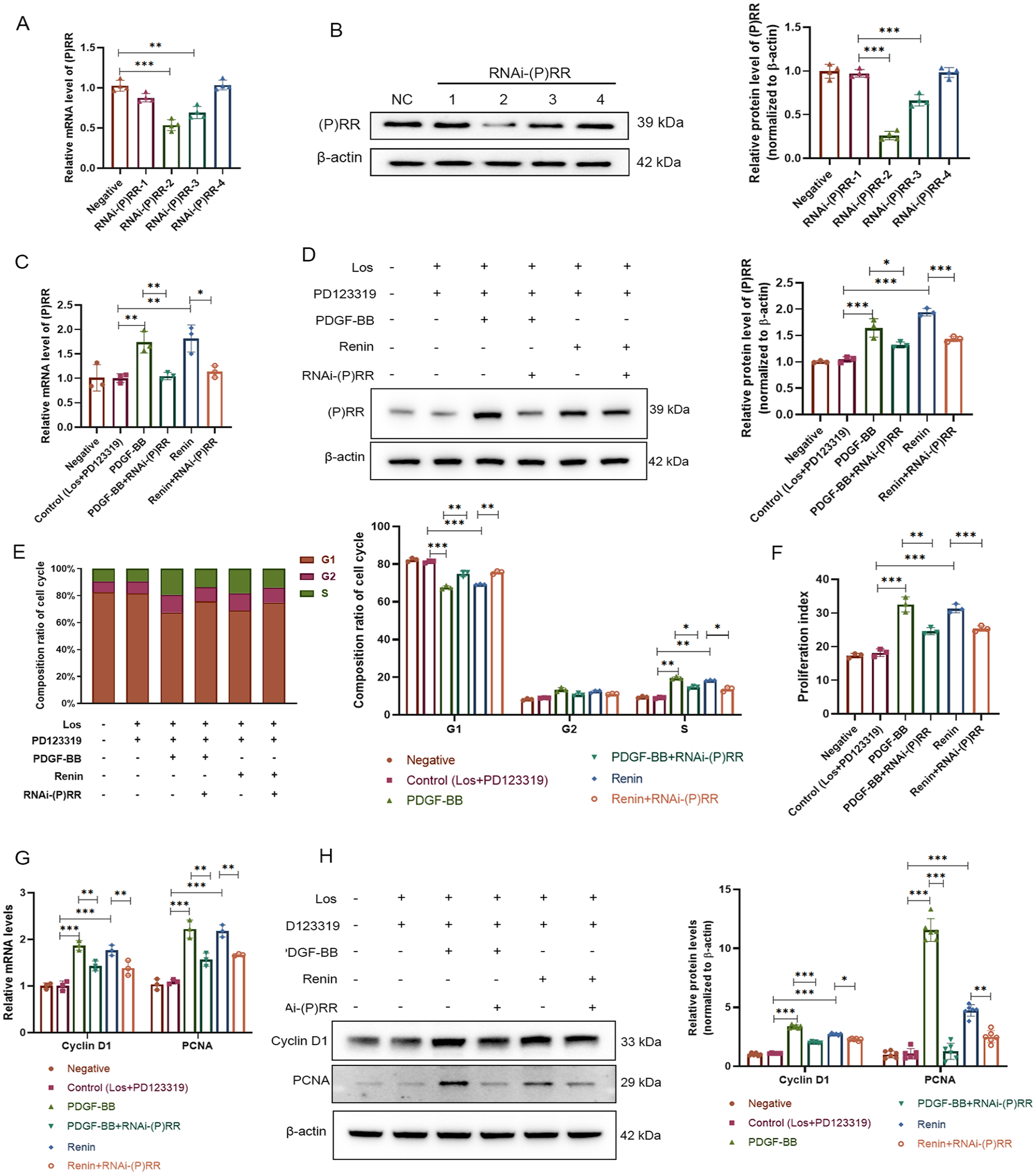
Silencing (P)RR restrained VSMC proliferation.
Renin induced VSMC proliferation by activating oxidative stress and the inflammatory response
To elucidate the mechanism of action of renin, we focused on oxidative stress and the inflammatory response. FCM and ELISA showed that renin treatment could significantly increase the levels of ROS and MDA while reducing SOD expression (Figures 3A, 3B). However, compared with those in the control group, the levels of ROS, MDA, and SOD in VSMCs treated with DPI (which is an NADPH oxidase inhibitor) decreased or increased, respectively, and renin + DPI treatment reversed the function of renin (Figures 3A, 3B). Additionally, the levels of proinflammatory cytokines, including IL-6, TNF-α, IL-1β, and iNOS, were markedly increased by renin treatment compared with those in the control group; however, they were decreased after treatment with DPI (Figures 3C, 3D). These findings suggested that renin treatment could activate oxidative stress and inflammation. FCM indicated that the renin + DPI group had an increase in the G1 phase constituent ratio but a decrease in the S phase constituent ratio compared with the renin group (Figure 3E). The same phenomenon was observed regarding the reduced protein levels of proliferation index (Figure 3F), cyclin D1 and PCNA in the renin + DPI group compared with those in the renin group (Figure 3G). Finally, the protein levels of p-ERK1/2 and p-AKT were both significantly increased in the renin-treated group and were partially reduced by DPI supplementation (Figure 3H). These data indicated that renin could enhance VSMC proliferation by increasing oxidative stress and the inflammatory response and that the ERK1/2-AKT signaling pathway may be involved in this process.
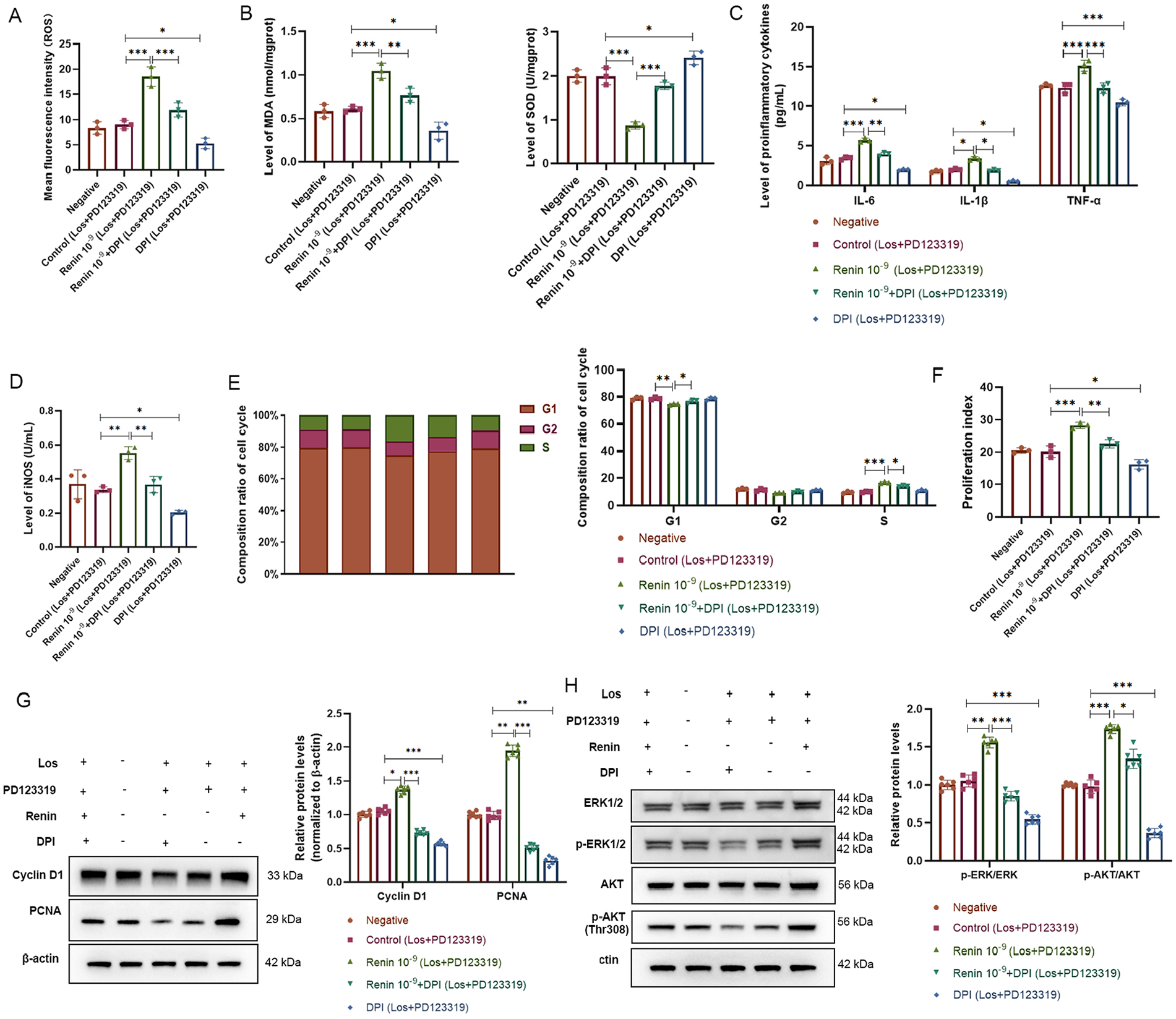
Renin induced VSMCs proliferation via activating oxidative stress and inflammatory response. VSMCs treated with Los, PD123319, renin, or DPI.
Silencing (P)RR decreased the levels of oxidative stress and inflammation
When considering that silencing (P)RR could reverse the proliferative effect of renin, we explored whether silencing (P)RR could reverse the oxidative stress and inflammation induced by renin. The results showed markedly increased MDA and decreased SOD levels in VSMCs supplemented with PDGF-BB or renin; however, the MDA and SOD levels were significantly reduced and enhanced, respectively, by RNAi-(P)RR treatment (Figures 4A, 4B). IL-6, TNF-α, IL-1β, and iNOS expression was markedly greater in the PDGF-BB and renin groups than in the control group but decreased after the addition of RNAi-(P)RR (Figure 4C). It has been demonstrated that ROS induced by NOX1 could promote the proliferation of VSMCs; 12 therefore, the expression of NOX1 was also determined. The RT‒PCR and WB results indicated that the mRNA and protein levels were both markedly increased in the PDGF-BB and renin groups; in contrast, the overexpression of NOX1 could be reversed by RNAi-(P)RR (Figures 4D, 4E). Interestingly, we found that RNAi-(P)RR inhibited the ERK1/2-AKT pathway by inhibiting the expression of p-ERK1/2 and p-AKT (Figure 4F). These data indicated that (P)RR, which is regulated by renin, could promote the proliferation of VSMCs by regulating NOX1-mediated oxidative stress and inflammation to activate the ERK1/2-AKT signaling pathway.
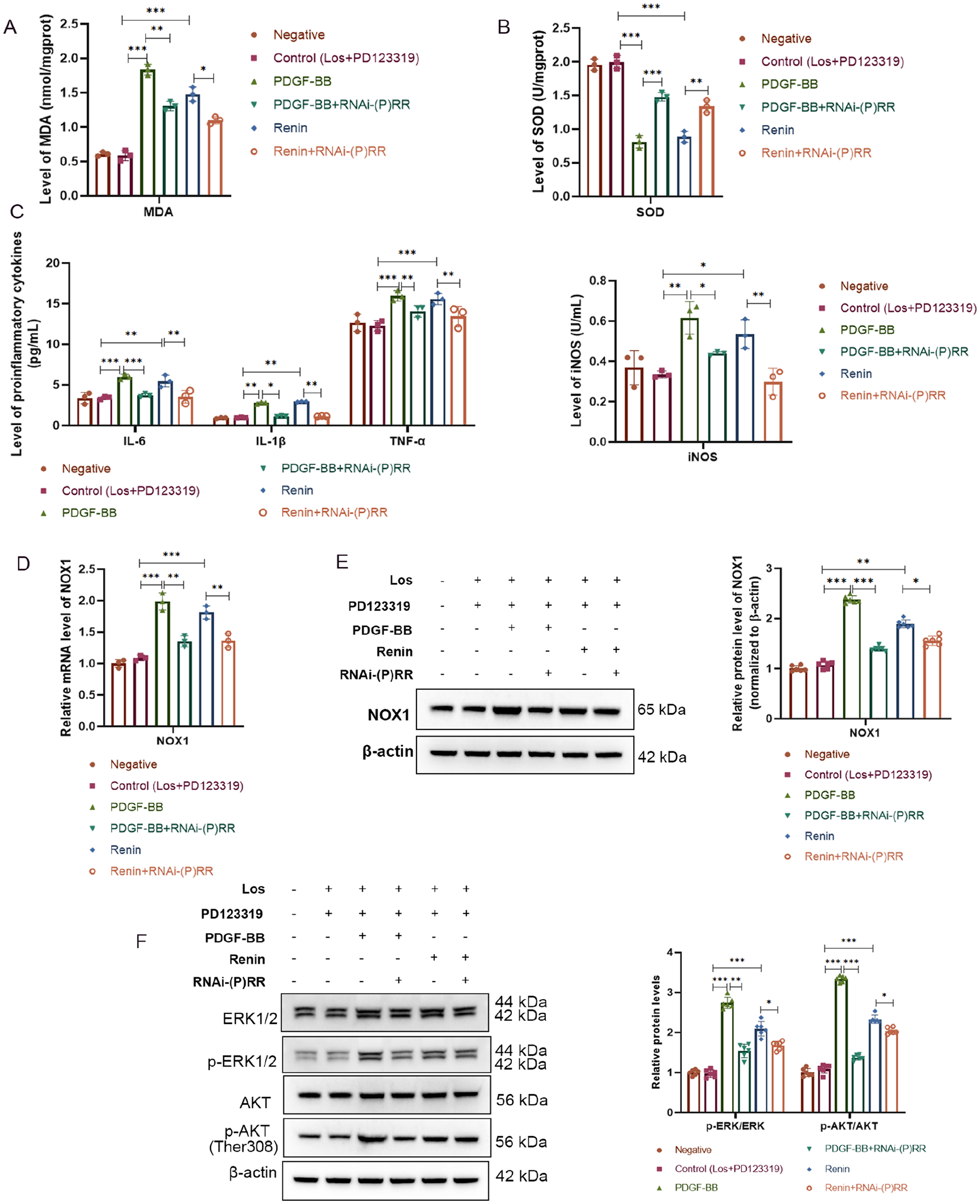
Silencing (P)RR decreased the levels of oxidative stress and inflammation. VSMCs treated with Los, PD123319, PDGF-BB, renin, or RNAi-(P)RR.
Inhibition of (P)RR restrained neointimal hyperplasia after common carotid artery injury in rats
To elucidate the function of the (P)RR in vivo, a balloon injury model was constructed to assess common carotid artery injury. HE staining showed that neointimal hyperplasia was distinctly observed in the injury group. However, these phenomena were alleviated after PRO20 and losartan treatment (Figure 5A). Importantly, compared with the sham group, the control group had no obvious pathological changes. Compared with the injury group, the PRO20 and losartan treatment groups exhibited slight neointimal hyperplasia and a markedly reduced I/M ratio (Figure 5B). In addition, the ELISA results indicated that the levels of renin and PDGF-BB in arterial blood were both increased after injury but decreased by losartan and PRO20 treatment, with a marked difference in the sham and control groups being observed (Figure 5C). Furthermore, the mRNA and protein expression levels of cyclin D1 and PCNA in the vascular wall of the injury group were both significantly greater than those in the control group; moreover, these levels were markedly decreased in the losartan and PRO20 groups after injury (Figures 5D, 5E). These results showed that (P)RR could improve neointimal hyperplasia after common carotid artery injury in rats.
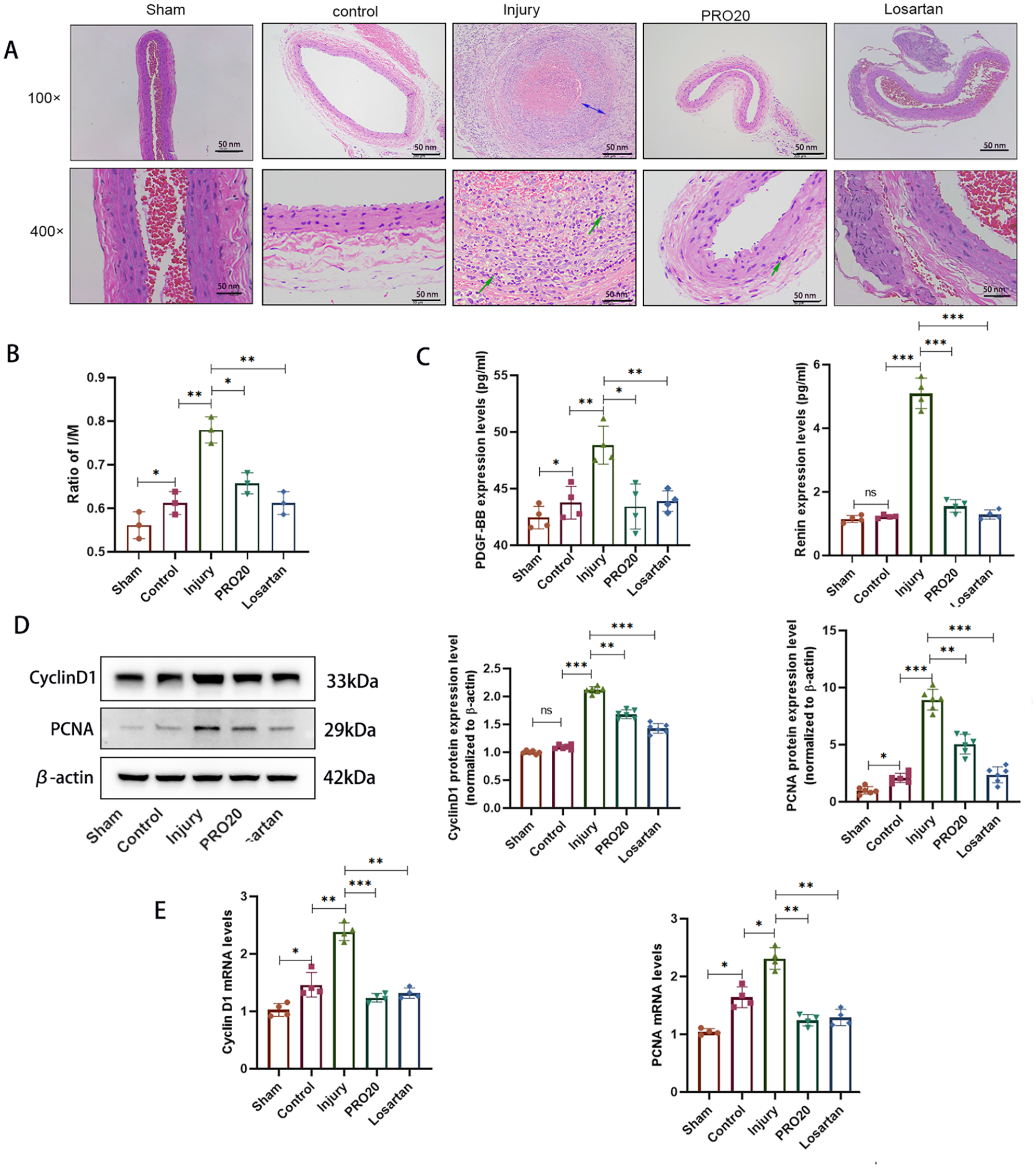
Inhibition of (P)RR restrained neointimal hyperplasia after common carotid artery injury of rats. Each group of rats was treated with PRO20 or losartan.
Inhibition of (P)RR restrained NOX1-mediated oxidative stress by downregulating the ERK1/2-AKT pathway
Subsequently, we aimed to explore whether the specific mechanism of (P)RR was related to oxidative stress and the ERK1/2-AKT pathway. By detecting SOD and MDA levels in the vascular wall, we found that the levels of MDA and SOD in the injury group were greater than those in the sham operation group. In contrast, the MDA and SOD levels in the losartan and PRO20 groups were markedly reduced and increased, respectively (Figures 6A, 6B). Afterwards, the RT‒PCR results indicated that the NOX1 mRNA level was markedly upregulated in the injury group compared with the control group but decreased due to losartan and PRO20 treatment (Figure 6C). Finally, WB analysis demonstrated that the levels of p-ERK1/2 and p-AKT were increased after injury and decreased in the losartan and PRO20 groups (Figure 6D). These data demonstrated that the inhibition of (P)RR restrained NOX1-mediated oxidative stress by downregulating the ERK1/2-AKT pathway.
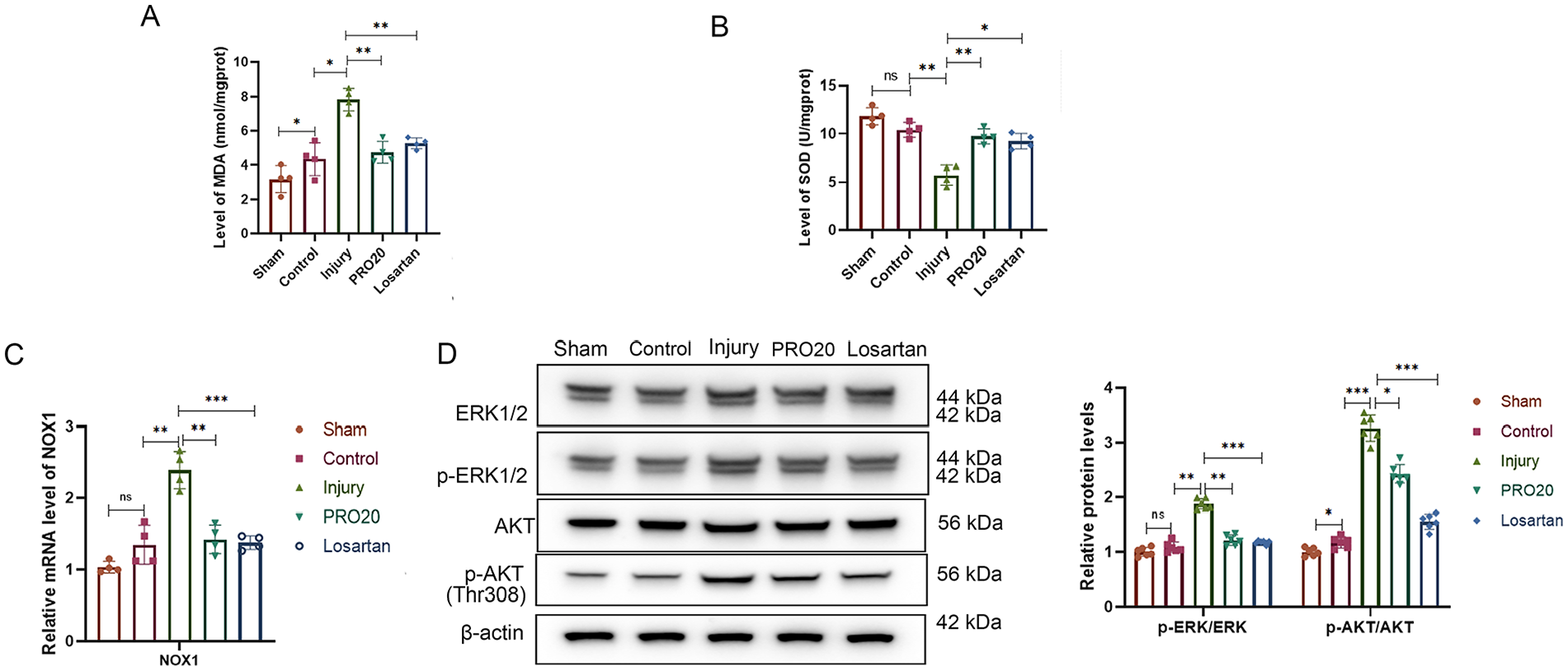
Inhibition of (P)RR restrained NOX1-mediated oxidative stress by downregulating the ERK1/2-AKT pathway. Each group of rats was treated with PRO20 or losartan.
Discussion
At present, it is widely believed that the migration and proliferation of VSMCs after vascular endothelial injury are the pathological basis of atherosclerosis and RS after PCI.13,14 The proliferation of VSMCs leads to a series of pathological changes in the blood vessel wall. 15 The RAS serves as a classic cardiovascular regulatory system, and it plays an indispensable role in the proliferation of VSMCs.16,17 As a newly discovered member of the RAS system, (P)RR plays an essential role in the pathogenesis of cardiovascular diseases and diabetic nephropathy. 18 In the past, it was believed that renin relies on Ang II to exert its biological activity. However, in recent years, many studies have shown that the biological role of renin precursors may not depend on traditional RAS activation but may be related to (P)RR. 19
Researchers have confirmed that prorenin can promote the increase in and hypertrophy of VSMCs. 19 However, whether renin promotes the proliferation of VSMCs through a (P)RR-mediated Ang II-independent mechanism remains unclear. Moreover, the division and proliferation of VSMCs are mainly regulated by the eukaryotic cell cycle. 20 Cyclin D1 is an important cyclin that accelerates cells to the S phase through the G1/S restriction point and plays a positive regulatory role in cell proliferation. 21 PCNA was shown to be expressed only in proliferating cells that increased in the late G1 phase, peaked in the S phase, and decreased in the G2 and M phases. It is a specific marker of the S phase; thus, it can be used as an indicator of cell proliferation. 22 In this study, VSMCs were treated with different concentrations of renin and transfected with the RNAi-(P)RR plasmid to explore the effect of (P)RR. Based on these results, we found that the level of (P)RR markedly increased after renin treatment. Renin significantly promoted VSMC proliferation, the S phase composition ratio, the proliferation index, and the expression of cyclin D1 and PCNA in a concentration-dependent and Ang II-independent manner. However, inhibition of (P)RR reversed the proliferation of renin, thus suggesting that the proliferative function of renin in VSMCs was mediated by (P)RR in an Ang II-independent manner.
Oxidative stress-induced local vascular damage is considered an important factor for RS after PCI. After local vascular injury, the concentration of oxidatively active substances is significantly increased, which plays an important role in stimulating the proliferation of VSMCs. 23 ROS generation in VSMCs is significantly increased by certain stimulants. 24 The application of NADPH oxidase inhibitors, such as DPI, has confirmed that ROS are generated by NADPH oxidase. In addition, NOX1, which is a member of the NOX family, was upregulated in rat carotid arteries after balloon injury. 25 More importantly, the inflammatory process remains the predominant major cause of atherosclerosis in CHD patients worldwide. 26 Cytokines such as IL-1β or TNF-α are secreted during all phases of atherosclerotic lesion progression and induce the expression of ‘messenger’ or secondary signaling cytokines, such as IL-6. 27 Furthermore, iNOS activation can enhance inflammatory processes to contribute to atherosclerosis progression. 28 ROS participate in a variety of signal transduction pathways, such as activating ERK1/2, MAPK, and Janus kinase/signal transducer and activator of transcription (JAK/STAT) signaling,29,30 thus promoting cell proliferation and vascular remodeling. Our research demonstrated that renin treatment significantly enhanced the levels of ROS and MDA while reducing SOD expression; however, DPI treatment reversed this effect. Additionally, the levels of proinflammatory cytokines, including IL-6, TNF-α, IL-1β, and iNOS, were increased by renin treatment but decreased after treatment with DPI. Subsequently, FCM and WB assays indicated that renin promoted the proliferation of VSMCs, whereas DPI inhibited this proliferation. In addition, the protein levels of p-ERK1/2 and p-AKT were increased in the renin-treated group and were partially reduced by DPI supplementation. These results suggested that renin promoted VSMC proliferation by upregulating oxidative stress, the inflammatory response, and the ERK1/2-AKT signaling pathway.
Previous experimental results have shown that (P)RR plays an important role in the proliferation of VSMCs, and the proliferation and differentiation of VSMCs are the main pathological basis of RS. Therefore, we further investigated the role and mechanism of (P)RR in the restenosis of the carotid artery injury model. Studies have shown that the proliferation and migration of VSMCs after balloon injury are key factors in the development of restenosis after PCI.31,32 After arterial injury, VSMCs migrate from the medial membrane to the luminal surface through the internal elastic plate. After migrating to the luminal surface, VSMCs begin to proliferate and form new intima. An earlier study demonstrated that losartan, which is a nonpeptide Ang II receptor antagonist, inhibited neointima formation following balloon injury to rat carotid arteries. 33 PRO20 is a novel P(RR) decoy peptide consisting of 21 amino acids that selectively obstructs the interaction between renin/prorenin and the P(RR) with high efficacy. 34 Therefore, losartan and PRO20 were used in this study to observe the role of (P)RR in restenosis after carotid artery injury. It has been reported that the superoxide anion (O2–) and lipid peroxidation products can be produced by local vascular wall cells in a very short period of time after PTCA, and the MDA level in coronary veins was also significantly greater than that before PTCA. 35
In this study, HE staining and the I/M ratio showed that neointimal hyperplasia was distinctly observed after injury, but these effects were alleviated after PRO20 and losartan treatment. The expression levels of cyclin D1 and PCNA in the injury group were both significantly greater than those in the sham group and were markedly decreased in the losartan and PRO20 groups. Furthermore, the ELISA results indicated that the levels of renin and PDGF-BB in arterial blood were both increased after injury but were reduced by losartan and PRO20 treatment, thus further suggesting the proliferative effect of renin. The MDA, NOX1, p-ERK1/2, and p-AKT levels in the injury group were clearly greater than those in the control group after losartan and PRO20 treatment, whereas SOD expression exhibited the opposite trend. These results showed that (P)RR can improve neointimal hyperplasia after common carotid artery injury in rats by regulating NOX1-mediated oxidative stress and the ERK1/2-AKT pathway.
In conclusion, our data indicated that renin and (P)RR can enhance VSMC proliferation and neointimal hyperplasia by upregulating oxidative stress, the inflammatory response, and the ERK1/2-AKT signaling pathway in an Ang II-independent manner. These results may provide a new perspective for targeted therapy of coronary heart disease with RS.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241261368 – Supplemental material for Renin and (pro)renin receptors induce vascular smooth muscle cell proliferation and neointimal hyperplasia by activating oxidative stress and inflammation
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241261368 for Renin and (pro)renin receptors induce vascular smooth muscle cell proliferation and neointimal hyperplasia by activating oxidative stress and inflammation by Nana Zhang, Xiaosu Song, Yunfei Bian, Rui Bai, Huiyu Yang, Gang Wang, Hong Li and Chuanshi Xiao in Vascular Medicine
Footnotes
Data availability statement
The datasets that were used and/or analyzed during the present study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Research Funding Project for Returned Overseas Students in Shanxi Province (No. 2011-110).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
