Abstract
This study examines the effect and mechanism of action of Netrin-1 on bone marrow mesenchymal stem cells in angiogenesis. Tube formation and migration of bone marrow mesenchymal stem cells were observed in cell culture. Bone marrow mesenchymal stem cells or Netrin-1–bone marrow mesenchymal stem cells were injected into the ischaemic area of the rat hind limb on the first day after surgery. Laser Doppler perfusion imaging was performed to analyse the levels of vascular endothelial growth factor in plasma and muscles, and immunohistochemistry and immunofluorescence were used to analyse angiogenesis. Bone marrow mesenchymal stem cells in medium containing Netrin-1 markedly increased the number of tubes formed and the migration of bone marrow mesenchymal stem cells compared with the untreated control group. The function of Netrin-1 in tube formation and migration is similar to vascular endothelial growth factor, and combined with vascular endothelial growth factor, Netrin-1 has more enhanced effect than in the other three groups. The Netrin-1–bone marrow mesenchymal stem cell group had better augmented blood-perfusion scores and vessel densities, as well as improved function of the ischaemic limb than that of the group injected with bone marrow mesenchymal stem cells (treated with bone marrow mesenchymal stem cells individually) or the control group (treated with medium). These results suggest that Netrin-1 has the ability to augment the angiogenesis of bone marrow mesenchymal stem cells and improve the function of the ischaemic hind limb by increasing the level of vascular endothelial growth factor.
Introduction
Therapeutic angiogenesis has been developed as a method of treating patients with vascular diseases.1–3 Myeloid cells play crucial roles in immune defence and angiogenesis.4,5 Compared with other stem cells, bone marrow mesenchymal stem cells (BMSCs) have many advantages. They are easily accessible through aspiration of the bone marrow from patients (thus avoiding any serious ethical problems) and can be readily expanded to a large-scale culture for autotransplantation and have low immunogenicity. Additionally, they have the ability to secrete multiple angiogenic cytokines6–8 and differentiate into endothelial cells (ECs) or smooth muscle cells (SMCs),9,10 inducing angiogenesis in ischaemic lesions. 11 Therefore, BMSCs are a promising source for inducing therapeutic angiogenesis. Administration of either exogenous angiogenic factors or stem cells alone to induce angiogenesis could improve function; however, these therapies have little effect on the repair of ischaemic tissue. Combining cell therapy with the administration of angiogenic factors might overcome the shortcomings of these treatment strategies.
Accumulating evidence has indicated that crosstalk between vascular and neural systems occurs at the molecular level in both development and injury response. 12 Vascular endothelial growth factor (VEGF) drives angiogenesis in a wide variety of tissues and lesions, and it plays an important role in neural development, axonal outgrowth, cell survival and neural progenitor attraction.13–15 Conversely, neuroregulatory signals and neuronal guidance factors also influence vascular development. Netrin-1 is a survival factor, also known as a novel angiogenic factor, 16 which promotes the migration, proliferation, differentiation, adhesion and survival of ECs. 17 Additionally, it plays a major role in local angiogenesis and progenitor cell migration. 18 In vivo, Fan et al. 19 demonstrated that Netrin-1 could promote blood vessel formation in the adult rodent central nervous system, and it had some ameliorating effects in cerebrovascular development and remodelling. We have shown that pretreating BMSCs with a recombinant Netrin-1 protein markedly augmented the blood-perfusion score and vessel density, as well as improved function of the ischaemic limb. 20
On the basis of these findings, we constructed a novel recombinant adenovirus Netrin-1 vector (Ad5-EGFP-Netrin-1) and successfully infected BMSCs that can express Netrin-1. This study tested the effects of Netrin-1 on the implantation of BMSCs in angiogenesis using the model of hind-limb ischaemia and examined the possible functional mechanism. Our results may provide a new therapeutic strategy for the treatment of ischaemic diseases.
Materials and methods
Animals
Adult male Sprague-Dawley rats weighing 250–300 g were used for this study (Shanghai Laboratory Animal Center, Chinese Academy of Sciences). All animals received care in compliance with the Principles of Laboratory Animal Care formulated by the National Society for Medical Research and the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (NIH publication 85-23, revised 1996), and all protocols were approved by the Institutional Animal Care and Use Committee of Nanjing Medical University. Rats were housed at a constant temperature (20°C−22°C) and humidity (45%–55%) on a 12-h light:12-h dark cycle. They received standard laboratory rat chow and had free access to tap water.
Isolation and culture of BMSCs
BMSC expansion was performed as previously described. 21 In brief, BMSCs were flushed out of the tibias and femurs of 100–150 g male Sprague-Dawley rats with L-Dulbecco’s modified Eagle’s medium (L-DMEM; Hyclone, Thermo Scientific, Logan, UT). Bone marrow cells were plated on 100-mm dishes and cultured in L-DMEM supplemented with 10% fetal bovine serum (FBS; Gibco, Invitrogen, Grand Island, NY). A small number of cells developed into visible symmetric colonies by days 5–7. Non-adherent haematopoietic cells were removed by replacing the medium. Adherent BMSCs were passaged and diluted 1:4 once confluent. Passage 4 BMSCs were used for the experiments.
The quality of BMSCs was ensured by flow cytometry. BMSCs were stained with CD105-PE, CD73-PE, CD90-FITC, CD34-PE and CD45-PE and analysed using a Beckman Coulter FACS instrument. BMSCs were identified as CD105, CD73, CD90 triple-positive, CD34 and CD45 double-negative cells (data not shown).
Ad5-EGFP-Netrin-1 vector construction and production
The construction of the Netrin-1 recombinant adenovirus was completed in three phases. First, the Netrin-1 gene was cloned by RT-PCR and then subcloned into the shuttle vector, pDC316-CMV, which carries the reporter gene, EGFP. Next, this newly constructed plasmid, pDC316-Netrin-1, was identified by nuclease digestion sequencing analysis and transfected into a human embryonic kidney cell line, HEK293, using Lipofectamine 2000 mediation with the adenovirus-packaging plasmid pBHGlox_E1.3Cre. Based on the homologous recombination of the two plasmids within HEK293 cells, the recombinant adenovirus vector carrying Netrin-1, that is, Ad5-EGFP-Netrin-1, was created. Finally, Ad5-EGFP-Netrin-1 was subsequently identified by PCR, purified using repeated plaque passages, amplified by the freezing and thawing of HEK293 cells and titrated using 50% tissue culture infective dose (TCID50). Rat BMSCs were infected with Ad5-EGFP-Netrin-1, and the expression of Netrin-1 was confirmed by western blotting and enzyme-linked immunosorbent assay (ELISA).
BMSC migration assay
BMSC migration was evaluated using the transwell migration technique (8-mm transwell plates; Corning Inc., Corning, NY). BMSCs transfected with Ad5-EGFP-Netrin-1 or Ad5-EGFP (5 × 104) in 100 µL of medium were placed in the upper chamber. The lower chamber contained phosphate-buffered saline (PBS) or 10 ng/mL VEGF-containing medium. After 4, 8, 12 and 24 h of culture, we observed EGFP fluorescence to locate the position of BMSCs on the Matrigel (Becton Dickinson, Franklin Lakes, NJ) using laser scanning copolymerization microscope (LSCM). We calculated the distance of the EGFP from the membrane to quantify the migration of BMSCs. Migration activity was evaluated in four samples per group.
EC and BMSC network formation assay
BMSCs were cultured in L-DMEM supplemented with different growth factors (control medium, Netrin-1 supernatant or 10 ng/mL VEGF). Passage 4 BMSCs (3 × l04) or human umbilical vein endothelial cells (HUVECs, 3 × l04) were plated on 24-well plates coated with Matrigel and incubated at 37°C in 5% CO2 for 12 or 18 h. Images were captured with a microscope. The extent of tube formation was assessed in three random high-power fields (magnification × 100) per group.
Rat ischaemic hind-limb model and injection of transfected BMSCs
Isogenic rats served as donors and received allogeneic implantations of BMSCs. The rat model of hind-limb ischaemia has been previously described. 22 Right hind-limb ischaemia was induced by dissecting, ligating and cutting the right femoral artery and its branches under anaesthesia with 0.35 mL/100 g of 10% chloral hydrate administered intraperitoneally. The left hind limb was kept intact and used as a non-ischaemic control limb.
Approximately 24 h after surgery, rats in the Netrin-1 group (n = 12) were intramuscularly injected at 10 points within the ischaemic area (0.05 mL each) using a 1-mL syringe filled with 0.5-mL L-DMEM containing 1 × 106 BMSCs that had been transfected with Ad5-EGFP-Netrin-1. The Null-BMSC group (n = 12) was injected with 1 × 106 BMSCs that had been transfected with Ad5-EGFP, and the control group (n = 10) was injected with 0.5-mL L-DMEM only. All rats with limb ischaemia survived until the end of the experiment.
In vivo assessment of limb function
The functional assessment of the ischaemic limb was performed using a modified clinical standard score. 23 A semi-quantitative assessment of ambulatory impairment of the ischaemic limb was serially performed. The scoring was as follows: 0: flexing the toes to resist gentle traction on the tail, 1: plantar flexion, 2: no dragging but no plantar flexion and 3: dragging of foot. A blinded observer assigned all scores.
Laser Doppler perfusion imaging measurement of blood flow
Superficial blood flow (BF) of the ischaemic and contralateral foot was analysed at different time points (days 0, 7, 14 and 28) after ischaemia induction using colour laser Doppler perfusion imaging (LDPI; Lisca PIM3 Perimed, jarfalla, Sweden) as previously described. 24 Hair was removed from the limbs, and the animals were kept warm at 37°C before examination. Then, the ratio between BF in the ischaemic foot and BF in the contralateral foot was calculated and used as an index of % BF recovery (i.e. right ischaemic/left normal limb perfusion). A minimum of n = 6 rats per group were studied unless stated otherwise.
Western blotting analysis
Samples containing 50 µg of total protein from the injured gastrocnemius muscle were subjected to electrophoresis using 10% sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE) and electroblotted onto nitrocellulose membranes (Bio-Rad, Hercules, CA). Membranes were then blocked with 5% fat-free milk for 2 h at 37°C, incubated with rabbit anti-Netrin-1 and anti-VEGF primary antibodies (1:500; Santa Cruz Biotechnology, Santa Cruz, CA) and incubated with horseradish peroxidase–conjugated goat anti-rabbit immunoglobulin (IgG, 1:2000) as the secondary antibody. The chemiluminescence reaction was carried out with an enhanced chemiluminescence (ECL) kit (Bio-Rad) for 1 min and was followed by exposure using Kodak X-Omat radiographic film. Similar procedures were carried out with an anti-β-actin antibody (1:500; Santa Cruz Biotechnology), which was used for actin blotting as the loading control.
Histological analysis of vessel formation
Blood vessels were detected in histological sections by immunohistochemistry. Tissue specimens were obtained from the adductor and semi-membranous muscles on day 28. Frozen tissue sections (5 µm thick) were prepared and incubated with a polyclonal rabbit anti-rat CD31 antibody (dilution 1:500; Santa Cruz Biotechnology) overnight at 4°C. After washing in PBS, sections were incubated with biotinylated anti-rabbit IgG (Santa Cruz Biotechnology) for 1 h at room temperature. The binding reaction was detected using an avidin–biotin–peroxidase complex (Santa Cruz Biotechnology) according to the manufacturer’s instructions and visualized with diaminobenzidine (Sigma–Aldrich, Vienna, Austria) in a 0.03% H2O2 solution of Tris-buffered saline (pH 7.4). The sections were counterstained with Mayer’s haematoxylin, dehydrated, and mounted in a non-aqueous medium. The number of capillary profiles was used to calculate the capillary density per mm2 of muscle, as previously described. 25
Assessment of BMSCs incorporated in the vasculature
Immunofluorescence staining on an additional five rats assessed whether the implanted BMSCs had been incorporated into the vasculature. Approximately 28 days after the injection of BMSCs that had been transfected with virus or L-DMEM, the injured muscle was harvested, frozen and cut into 8-µm sections. The survival of BMSCs in vivo was confirmed by the identification of EGFP spots using a fluorescence microscope. Differentiation of the BMSCs into ECs or SMCs was determined by immunofluorescence staining using the following antibody sets: primary antibodies were rabbit anti-rat CD31 (for ECs; Santa Cruz Biotechnology) and anti-Smooth Muscle Antibody (aSMA) (for smooth muscle; Abcam, Cambridge, MA), and the secondary antibody was goat anti-rabbit CY3-conjugated-IgG (Abcam). Differentiation of ECs or SMCs from BMSCs was confirmed by identification of cells fluorescing with both EGFP and CY3 with a LSCM.
Blood levels of VEGF
Blood samples were collected on days 7, 14 and 28 after injection with Netrin-1-BMSCs, Null-BMSCs or L-DMEM alone. The blood samples were stored at 4°C for 30 min and then centrifuged at 3000 r/min for 15 min. Plasma was frozen at −80°C until the VEGF assay by ELISA.
Statistical analysis
All data were expressed as means ± standard deviations (SDs). Comparisons between groups were analysed by one-way analysis of variance (ANOVA) followed by Bonferroni’s post hoc test. Statistical significance was determined at a level of p < 0.05.
Results
Adenovirus vector mediated–Netrin-1 expression in BMSCs
To identify whether Ad5-EGFP-Netrin-1 was capable of mediating Netrin-1 expression, we examined Netrin-1 expression in the BMSCs transfected with Ad5-EGFP-Netrin-1 at different multiplicity of infections (MOIs). After the BMSCs were transfected with Ad5-EGFP-Netrin-1, Netrin-1 was detected by western blotting, and EGFP expression in BMSCs was detected by fluorescence microscopy (Figure 1(a) and (b)).
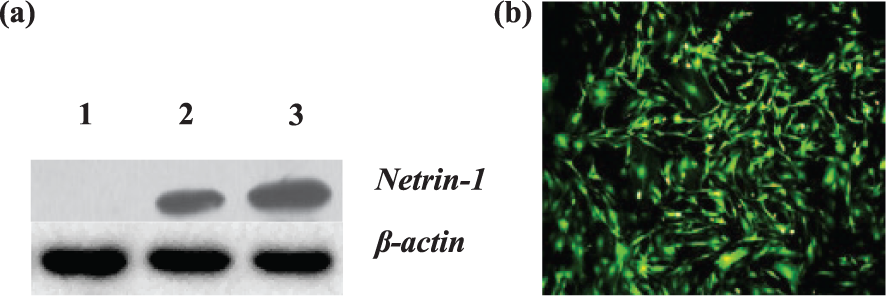
Netrin-1 protein expression by western blotting and the characterization of BMSCs by fluorescence microscopy. (a) Netrin-1 protein expression in BMSCs transfected with Ad5-EGFP-Netrin-1. The Netrin-1 band is approximately 67 kDa. Lane 1: BMSCs transfected with Ad5-EGFP; Lane 2: BMSCs transfected with Ad5-Netrin-1-EGFP (50 particles/cell); Lane 3: BMSCs transfected with Ad5-Netrin-1-EGFP (100 particles/cell). (b) BMSCs transfected with Ad5-EGFP-Netrin-1 at 24 h; EGFP expression can be observed in most BMSCs by fluorescence microscopy.
Release of Netrin-1 in vitro
Figure 2 depicts the time course of Netrin-1 release over 2 weeks after transduction with Ad5-EGFP-Netrin-1 in vitro. The concentration of Netrin-1 is highest at day 7, approaching 1000 pg/mL. After day 7, its expression is gradually reduced.
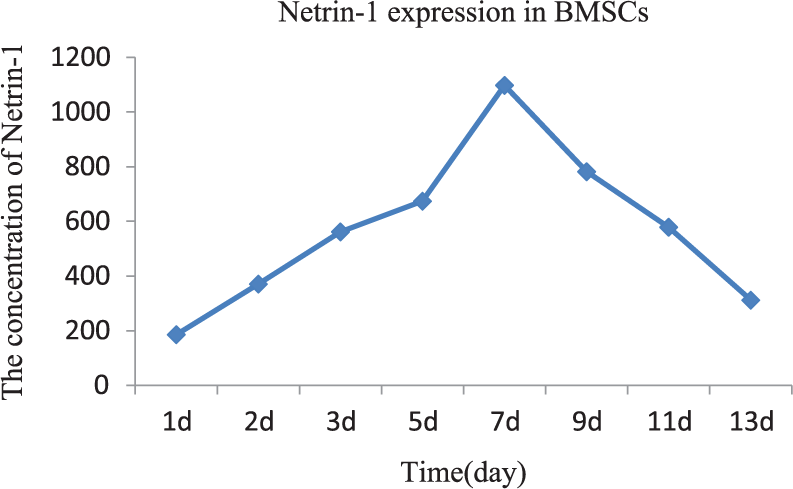
Time course of the Netrin-1 protein expression in BMSCs transfected with Ad5-EGFP-Netrin-1 analysed by ELISA.
Netrin-1 promotes BMSC migration in vitro
Using the transwell migration assay, we assessed the migratory response of ex vivo expanded BMSCs towards Netrin-1. We also compared the effects with those induced by VEGF (positive control group, 10 ng/mL), which is known to induce the migration of BMSCs. Both Netrin-1 and VEGF stimulated the migration of BMSCs compared with the untreated control group, but there were no significant differences between the groups (p > 0.05). Netrin-1, in combination with VEGF, stimulated migration of BMSCs to a greater degree than that of the other three groups (p < 0.01, Figure 3).
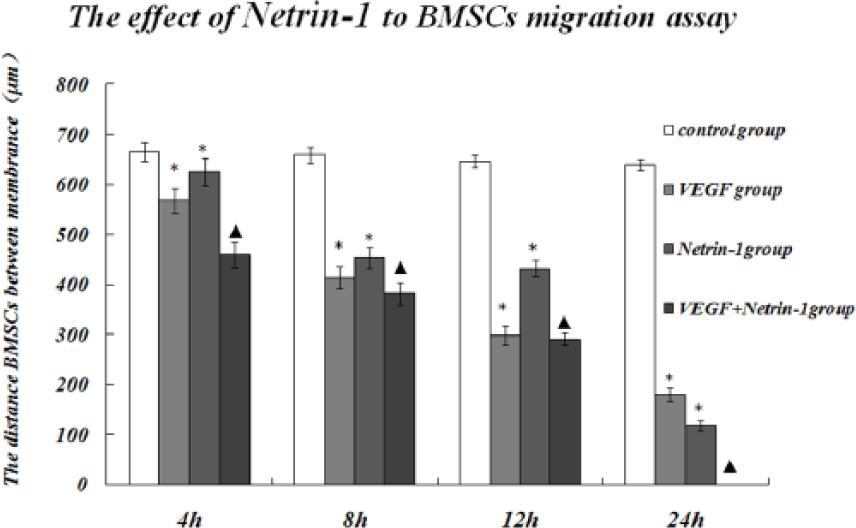
Effects of Netrin-1 on BMSCs migration.
Netrin-1 facilitates EC tube formation
To elucidate the effects of Netrin-1 on BMSC incorporation into tube formation by ECs, we quantified the tubular networks formed by human umbilical vein endothelial cells (HUVECs) on Matrigel. Netrin-1 facilitated tube formation by HUVECs, with a function comparable to VEGF (10 ng/mL). Netrin-1, in combination with VEGF, markedly increased the number of tubular networks formed by HUVECs compared with HUVECs cultured with Netrin-1 supernatant or VEGF (10 ng/mL) alone (p < 0.01) No tube formation was observed in cultures of HUVECs treated with culture medium alone in this assay (Figure 4(a) and (b)).
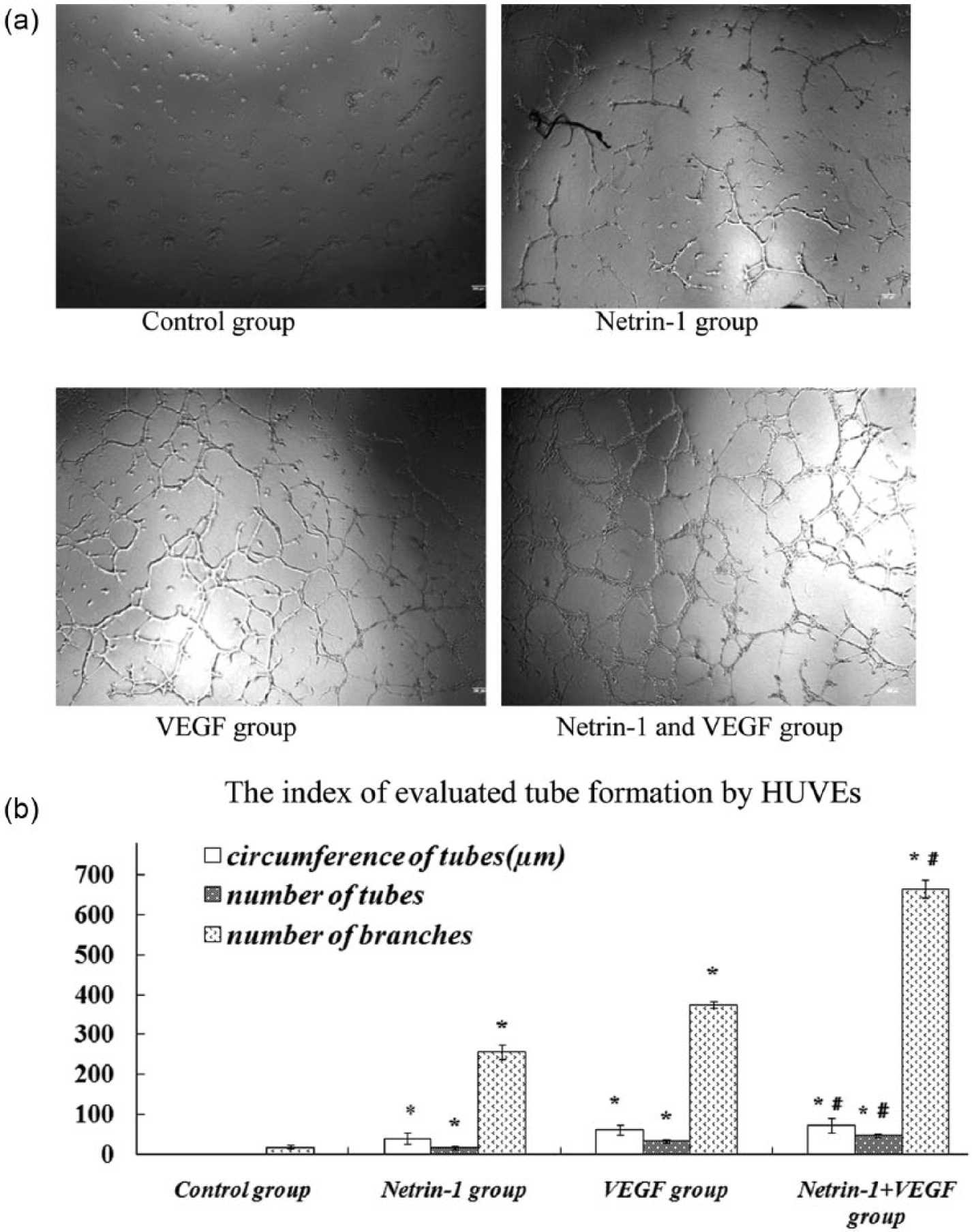
HUVECs tube formation assay. (a) Tube formation in each group with the different treatments after 18 h (magnification × 40). (b) Bar graph showing network formation by HUVECs seeded on Matrigel with the different treatments.
Netrin-1 facilitates BMSC tube formation
Similar to the findings of the HUVEC tube formation assay, after 12 h of treatment, tube formation was not observed in the control group. However, BMSCs formed tubes in the other three groups. Netrin-1 facilitated BMSC tube formation, with a function comparable to VEGF (10 ng/mL). Netrin-1 supernatant in addition to VEGF markedly enhanced the formation of tubular networks by BMSCs compared with BMSCs cultured in either Netrin-1 supernatant or VEGF (10 ng/mL) alone (p < 0.01; Figure 5(a) and (b)).
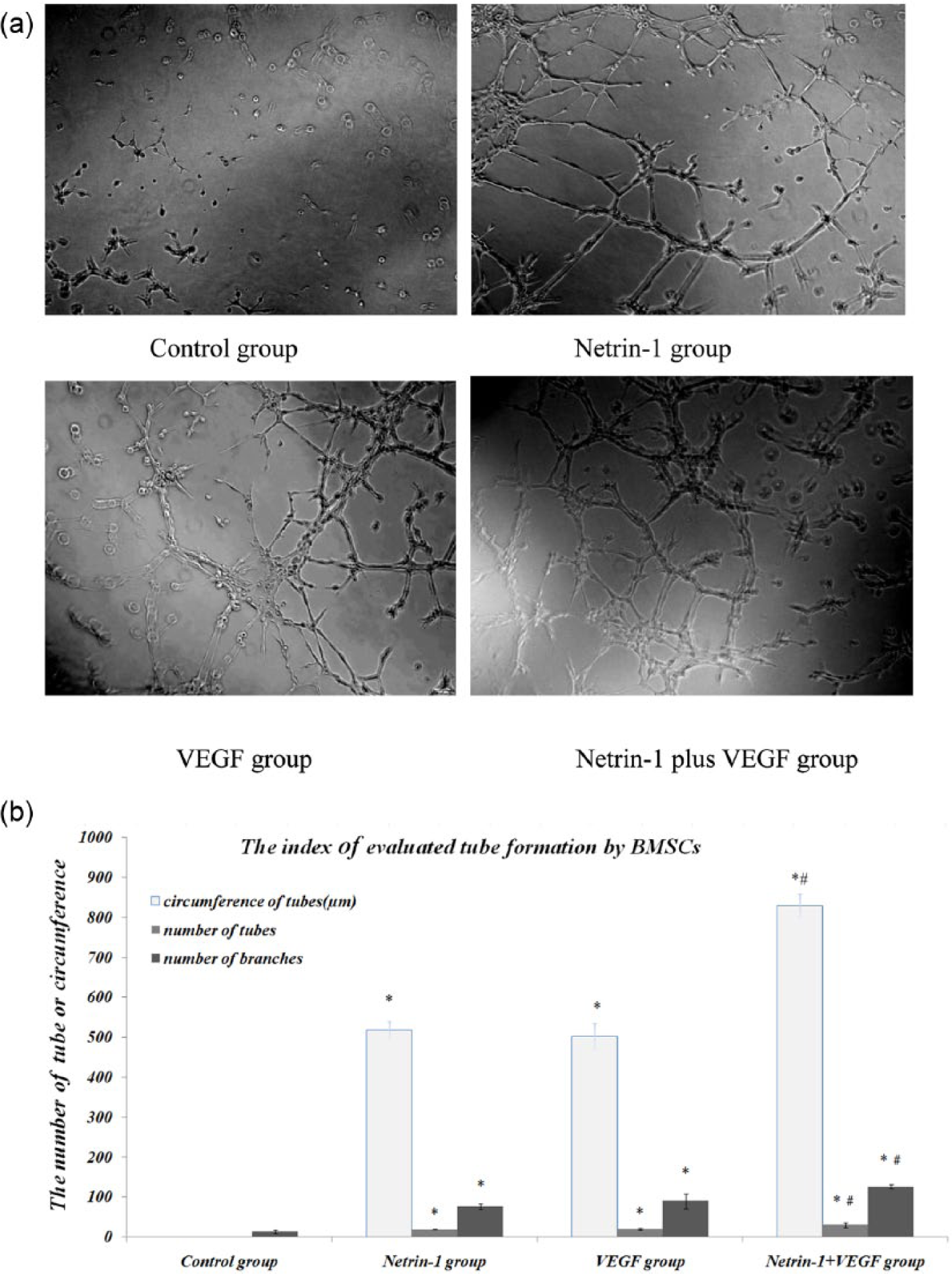
BMSCs tube formation assay. (a) Tube formation with different treatments in each group after 12 h (magnification × 100). (b) Bar graph showing network formation by BMSCs seeded on Matrigel with different treatments.
Limb recovery
To observe the recovery of hind-limb ischaemia, we used modified clinical standard scores. We discovered that there were no significant differences among the groups at postoperative day 7 or 14. On postoperative day 28, the recovery in the BMSC transplantation group was superior to that of the control group (p < 0.001), and the Netrin-1-BMSC group further out-performed the Null-BMSC group (p < 0.05; Figure 6).
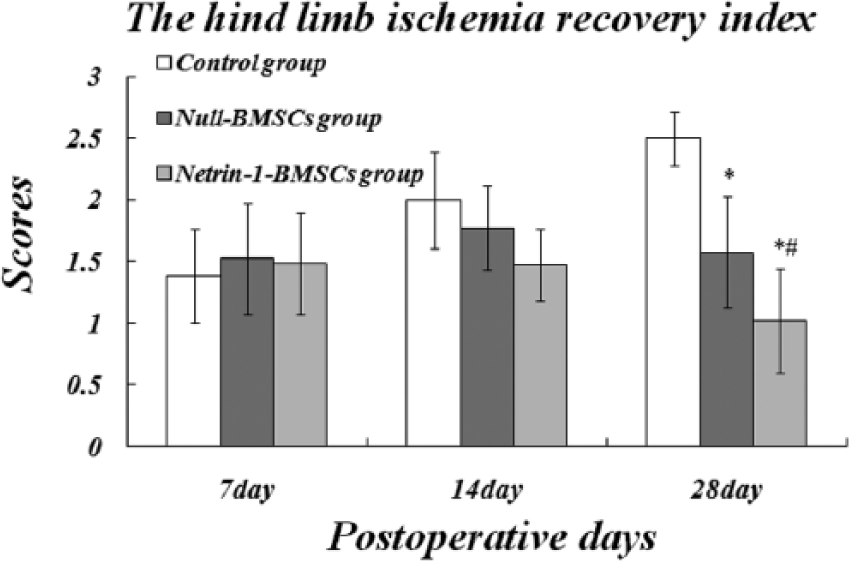
Results of the hind-limb ischaemia assay at different postoperative times.
Blood perfusion
Tissue BF in the ischaemic limbs was measured using LDPI. The time course of blood perfusion is depicted in Figure 7. The images show that there is a larger area of strong blood-perfusion signal in the hind ischaemic limb of the Null-BMSCs group and the Netrin-1-BMSCs group than in the control group, but the signal is strongest in the Netrin-1-BMSCs group at postoperative days 14 and 28. Quantitative analyses with LDPI PeriScan PIM 3 System image analysis software showed that the tissue blood recovery in the Null-BMSCs group and the Netrin-1-BMSCs group is better than in the control group, with significantly higher blood perfusion in the Netrin-1-BMSCs group than the other two groups (p < 0.05; Figure 7(a) and (b)).
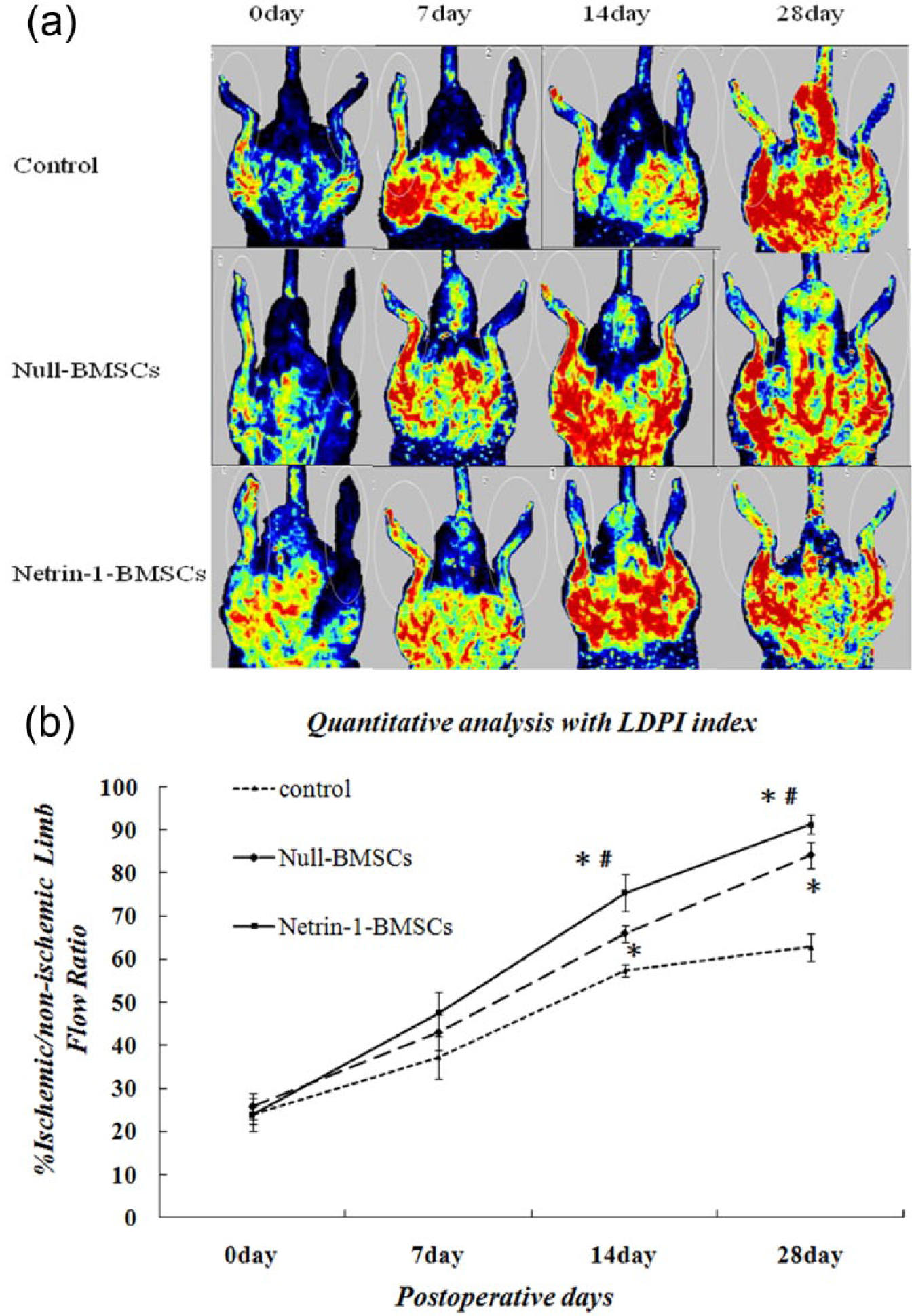
Tissue blood flow in the ischaemic limbs was measured using LDPI. (a) Representative LDPI colour images of each group at different points after surgery. (b) Time course of ischaemic/non-ischaemic blood-perfusion ratio in the different groups of treated rats. Quantitative analysis of the LDPI index showed a more significant improvement in the Netrin-1-BMSCs group compared with all other groups.
Histological analysis of vessel formation
To evaluate the role of BMSCs or/and Netrin-1 in vessel formation in response to limb ischaemia, we used immunohistochemistry to observe vessel formation by anti-CD31 staining to detect ECs and anti-aSMA staining to detect SMCs of the blood vessel. The results are shown in Figure 8(a).
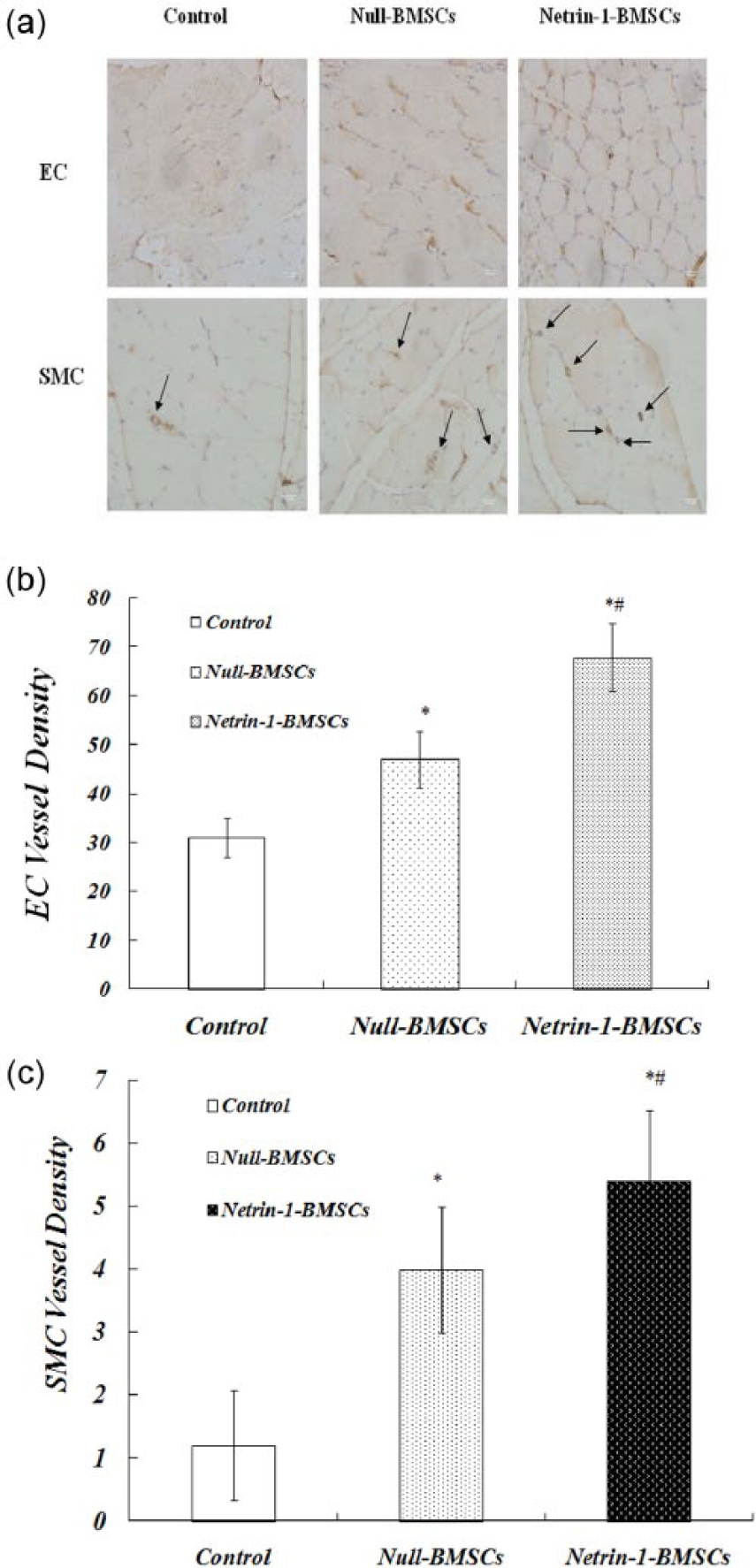
Anti-CD31 and anti-aSMA staining of vessel formation in limb ischaemia. (a) Representative immunohistochemistry images of vessels stained to show ECs and SMCs within the hind ischaemic limb 4 weeks after transplantation (×100). (b) Density of vessels stained positive for ECs. (c) Density of vessels stained positive for SMCs (values are the means ± SDs of data from six separate experiments).
The density of vessels that stained positive for ECs was significantly increased in the Netrin-1-BMSCs group and the Null-BMSCs group than in the control group, with the staining density being highest in the Netrin-1-BMSCs group. Similarly, the density of vessels that were positively stained for SMCs demonstrated a larger increase in the Netrin-1-BMSCs group and the Null-BMSCs group than in the control group, while the density of vessels in the Netrin-1-BMSCs group was higher than that in the Null-BMSCs group (Figure 8(b) and (c)).
Assessment of BMSC participation in angiogenesis and arteriogenesis
Immunofluorescence staining was carried out on six rats to assess whether the implanted BMSCs had been incorporated into angiogenesis and arteriogenesis. The ischaemic thigh muscles were harvested, frozen and cut into 8-µm sections 28 days after injection with BMSCs that had been transfected with adenovirus or L-DMEM. Survival of BMSCs in vivo was confirmed by the identification of EGFP spots using fluorescence microscopy. The differentiation of the BMSCs into ECs or SMCs was determined by immunofluorescent staining using the following antibody sets: primary antibodies were rabbit anti-rat CD31 (Santa Cruz Biotechnology) or anti-aSMA (Abcam) to stain for ECs and SMCs, respectively, and the secondary antibody was goat anti-rabbit CY3-conjugated-IgG (Abcam). Differentiation of ECs or SMCs from BMSCs was confirmed by identifying cells that were labelled with both EGFP and CY3 using an LSCM (Figure 9(a) and (b)).
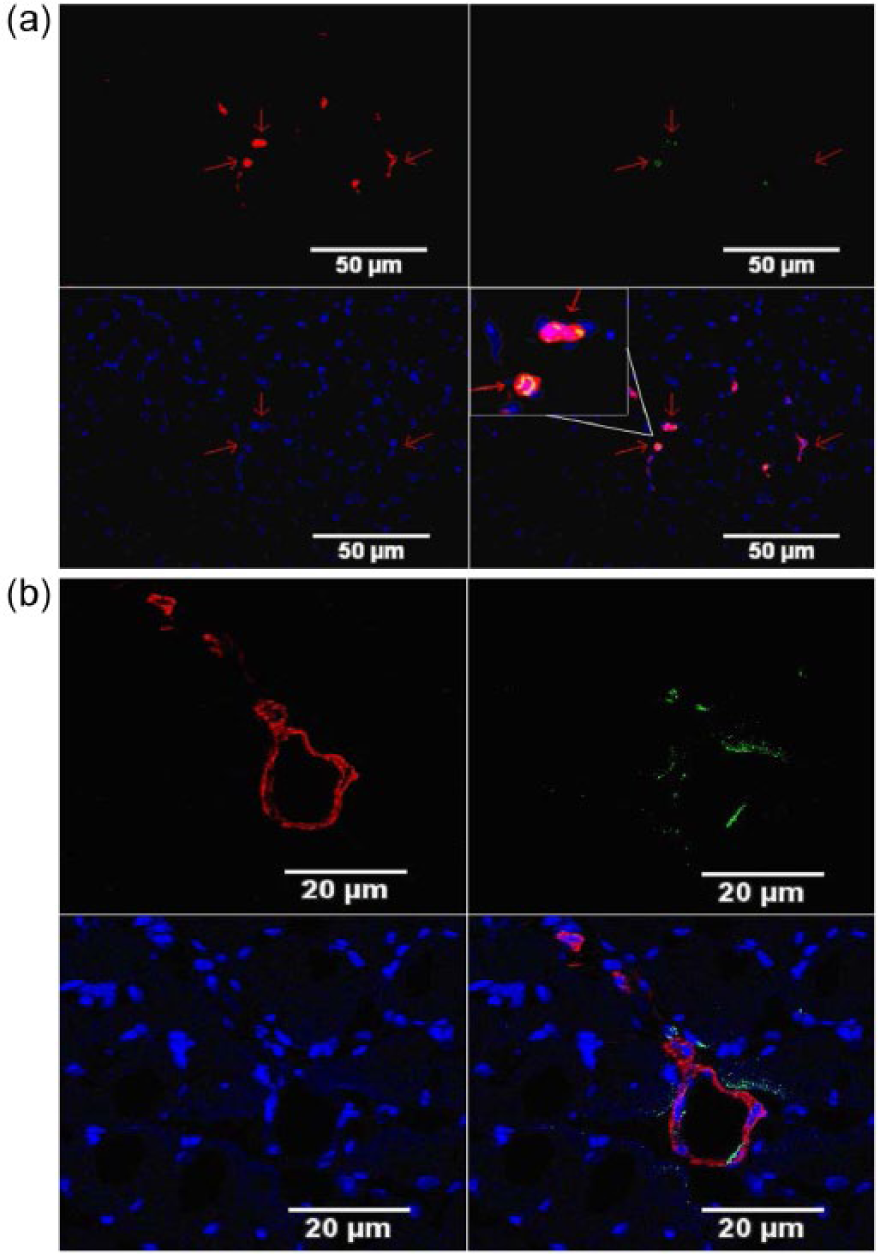
Transplanted EGFP-labelled BMSCs that survived and were incorporated into the vasculature by differentiating into ECs and SMCs. (a) Transplanted BMSCs that survived and were incorporated into vessels in ECs. (b) Transplanted BMSCs that survived and were incorporated into the vasculature into SMCs. Blue fluorescence signifies DAPI, red indicates CD31 or aSMA and yellow represents the ECs and SMCs differentiated from the implanted BMSCs.
VEGF expression is markedly increased in the Netrin-1-BMSCs treated group
To examine the level of VEGF protein expression, we measured the plasma level of VEGF in the three treatment groups at days 7, 14 and 28. At day 7 post surgery, the blood level of VEGF increased to 149.73 ± 38.26 pg/mL in the Netrin-1-BMSCs group, which was the highest level among the three groups (p < 0.05). In the Null-BMSCs group, the level of VEGF was significantly increased (108.62 ± 24.25 pg/mL) compared with the control group (48.67 ± 18.12 pg/mL). At day 14, in the Netrin-1-BMSCs and the Null-BMSCs groups, the level of VEGF was 131.35 ± 22.06 pg/mL and 107.16 ± 15.37 pg/mL, respectively, and both levels were significantly higher than that in the control group (48.04 ± 11.87 pg/mL; p < 0.05). However, at day 28, the levels of VEGF were not significantly different between all groups (Figure 10(a)).
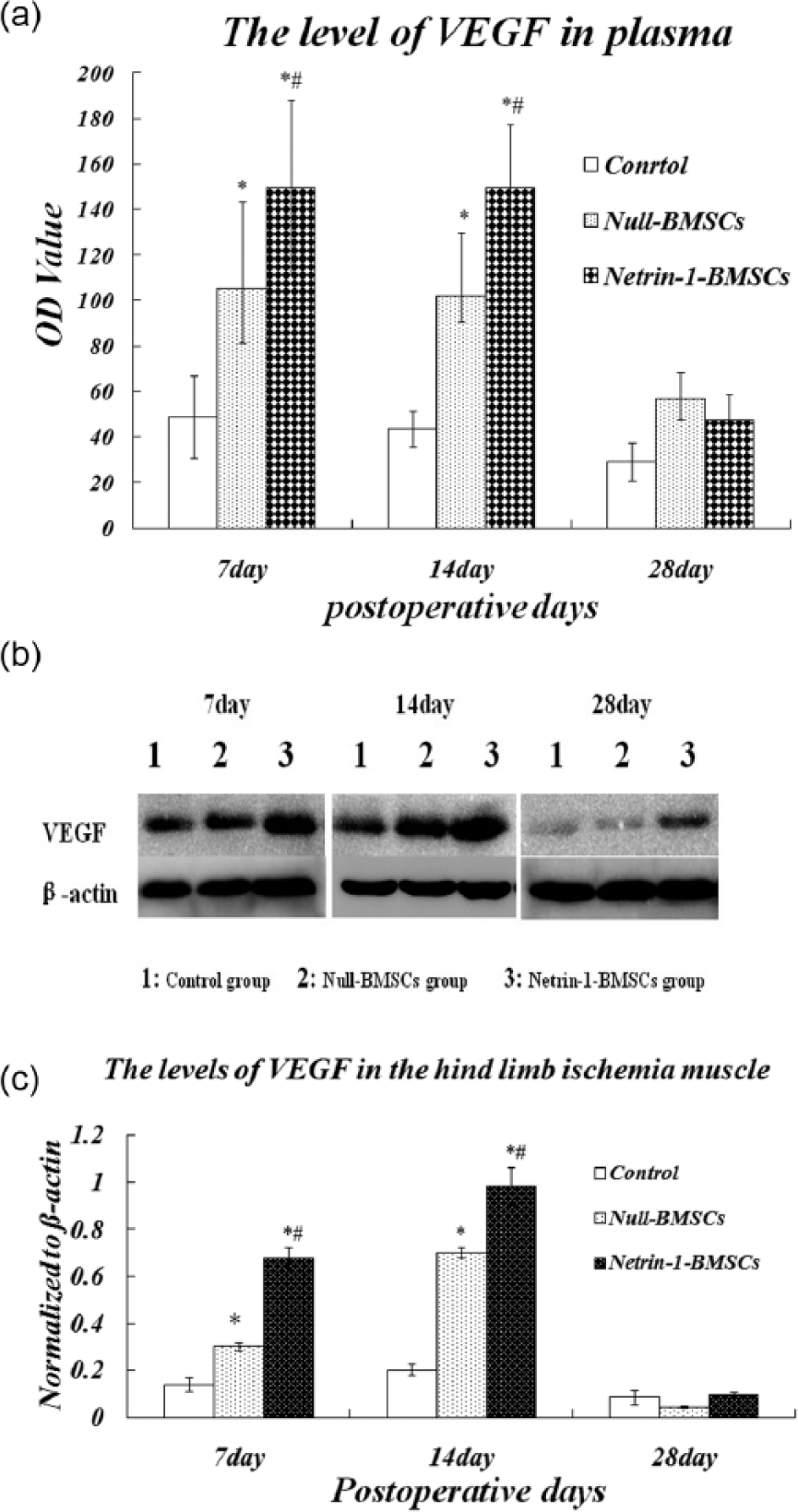
Levels of VEGF were measured by ELISA and western blotting. (a) Levels of VEGF in rat serum. Compared with the control group, the VEGF levels were significantly higher in the treated group at days 7 and 14 (b and c). The levels of VEGF in the hind-limb ischaemic muscle were measured by western blotting at days 7, 14 and 28. A greater amount of VEGF protein was observed in BMSC-treated ischaemic limbs compared with the control group at days 7 and 14. There was no significant difference among the groups at day 28 (values are the means ± SDs of data from six separate experiments).
Similarly, we performed western blotting analysis of the protein lysate derived from the treated ischaemic tissue. We found a higher amount of VEGF protein in BMSC-treated ischaemic limbs compared with the control group at both day 7 and day 14 (p < 0.05). However, the level of VEGF protein in the treated ischaemic limbs was not significantly different among the groups at day 28 (Figure 10(b) and (c)).
Discussion
BMSCs are considered a comparatively suitable source of cells to induce therapeutic angiogenesis in vascular disease. Because BMSCs can secrete multiple angiogenic cytokines, differentiate into ECs and SMCs that incorporate into the vessels and be amplified in vitro to generate a considerable number of high-quality cells for transplantation 26 ; BMSC transplantation has been considered a fitting treatment for ischaemic diseases with the absence of immunosuppression and ethical problems.27,28 Although therapeutic transplantation of BMSCs alone in ischaemic disease can achieve some improvements, the therapeutic effect is not marked in all patients and even failed in some. The major reason for failure is the low survival rate of transplanted cells. Therefore, reprogramming BMSCs with angiogenic factors can promote the therapeutic effect. One reason for this increased effect is that angiogenic factors facilitate angiogenesis; another reason is that BMSCs can cause massive mobilization and homing of stem cells from peripheral circulation to protect the transplanted stem cells.29–31
Netrins are important in axonal guidance and the regulation and maintenance of central nervous system, and they are also involved in the development of the mammary gland, lung, pancreas and blood vessels. 32 However, recent studies suggested that Netrin-1 functions as a survival factor or pro-angiogenic factor. 33 It promoted the migration of ECs, as well as their proliferation, differentiation, adhesion and anti-apoptosis.34–38
In this in vitro study, we found that the genetic modification of BMSCs with Netrin-1 can promote BMSCs migration and facilitate HUVEC or BMSC tube formation in a similar manner to the function of VEGF. In the presence of Netrin-1, its receptors in colorectal cancer, (DCC) and UNC5H transduce signalling cascades that lead to the proliferation, migration and differentiation of the cells. 28 Using a Matrigel angiogenesis assay, we also discovered augmented angiogenesis and arteriogenesis (blood vessels ⩾20 µm in diameter) in the Netrin-1-BMSCs group compared to Null-BMSCs, while in the control group, virtually no angiogenesis was observed. New angiogenesis consisted of ECs, which were mostly derived from the soma, with only a few ECs differentiated from BMSCs. However, the number of ECs differentiated from BMSCs in the Netrin-1-BMSCs group was greater than that in the Null-BMSCs group (data not shown).
We know that vascular regeneration occurs in three stages: vasculogenesis, angiogenesis and arteriogenesis. Of them, arteriogenesis is the key mechanism of enhancing perfusion and is, therefore, critical for the rescue of ischaemic organs. In this study, LDPI showed that in the Netrin-1-BMSCs and Null-BMSCs group, blood recovery was better than in the control group, and optimal recovery was observed in the Netrin-1-BMSCs group post-implantation days 14 and 28. Histological analysis showed that on post-implantation day 28, the density of microvessels and arteriogenesis in the Netrin-1-BMSCs group was the highest, while in the Null-BMSCs group, it was also higher than in the control group. Immunofluorescence staining analysis showed that some BMSCs had differentiated into ECs and SMCs and had been incorporated into vasculogenesis, angiogenesis and arteriogenesis. The number of ECs that differentiated from BMSCs in the Netrin-1-BMSCs group was greater than in the Null-BMSCs group. We therefore concluded that the recovery effect in the Netrin-1-BMSCs group was the best.
Our result showed that the expression of VEGF in plasma and hind-limb ischaemic gastrocnemius muscle in the Netrin-1-BMSCs group was higher than in the Null-BMSCs group or the control group at the early stage, that is, post-implantation days 7 and 14. The ischaemic hind-limb model produces an ischaemic and hypoxic milieu for BMSCs transplantation. We know that a reduced oxygen tension milieu may provide the stimulus required to promote VEGF production by BMSCs through the stimulation of hypoxia inducible factor expression.39–41 We therefore propose that Netrin-1 may promote VEGF expression by BMSCs and other tissue cells in the hind limb. The reason may be because Netrin-1 promotes the survival rate of transplanted BMSCs in the ischaemic muscle, allowing them to generate more VEGF and differentiate into more ECs and SMCs, 42 which are then incorporated into vasculogenesis, angiogenesis and arteriogenesis. Thus, the resulting vessel density is higher than in the other groups. Ahmed et al. 43 implanted BMSCs with a modified Sonic Hedgehog gene into an ischaemic heart. The result showed that these cells maximized their survival and angiogenic potential in the infracted heart. In this process, NO production increased, which was associated with higher levels of VEGF and Netrin-1 in a protein kinase C-dependent manner. Therefore, we postulate that more VEGF produced by mesenchymal stem cells with Netrin-1 can result in a multifold increase in NO production, induce significant angiogenesis, improve regional BF and significantly preserve the ischaemic lesion function. Of course, the mechanism of their signalling pathway should be further studied.
In summary, Netrin-1-BMSCs may have increased cellular survival, cause an increase VEGF expression, enhance the effect of VEGF and augment vessel density. Transplantation of Netrin-1-BMSCs can enhance the angiogenic effect and achieve a better therapeutic effect. Netrin-1 gene overexpression combined with stem cell transplantation could be a promising approach for ischaemic diseases of the limbs.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Jiangsu Planned Projects for Postdoctoral Research Funds, Nanjing Medical Technology Development Project (no. ZKX08014), Nanjing Medical Science and Technique Development Foundation (QRX11083) and National Natural Science Foundation of China (no. 81200594).
