Abstract
Introduction:
We aimed to investigate deep femoral artery (DFA) status during endovascular treatment (EVT) and the patency of the femoropopliteal (FP) artery and DFA using current stent devices for ostial FP lesions.
Methods:
A multicenter, retrospective study analyzed data from 457 patients who underwent EVT with stent deployment for de novo ostial FP lesions between April 2018 and December 2021 at eight centers in Japan. Propensity score-matched analysis was performed to compare the clinical impacts of DFA coverage for ostial FP lesions with stent deployment. The prognostic value was analyzed based on DFA status during EVT, primary patency of the FP artery, clinically driven-target lesion revascularization (CD-TLR) of the FP artery, and incidence of DFA occlusion/major amputation/acute limb ischemia (ALI) at 3 years.
Results:
A total of 132 matched pairs of patients was analyzed using propensity score matching. The percentage of DFA occlusion and stenosis was significantly higher in the DFA coverage group. However, there were no significant differences in terms of primary patency of the FP artery, CD-TLR, DFA occlusion, major amputation, or ALI after 3 years.
Conclusions:
Stent deployment in DFA coverage significantly led to the DFA stenotic events of EVT during the procedure, but patency of the FP artery and DFA, major amputation, and ALI up to 3 years did not differ according to stent deployment for ostial FP lesions.
Introduction
The development of endovascular treatment (EVT) devices and techniques authorizes clinicians to perform catheter intervention for the revascularization of lower-extremity artery disease (LEAD). EVT for aorto-iliac artery revascularization is recommended as a first-line treatment. However, the treatment of the femoropopliteal (FP) artery is controversial. 1
Increasing evidence supports better outcomes from EVT for FP artery disease.2–5 The emergence of drug-eluting stents (Eluvia stent; Boston Scientific, Marlborough, MA, USA) has improved 1-year primary patency outcomes compared with drug-coated stents (Zilver PTX; Cook Medical, Bloomington, IN, USA). 6 According to the Japan data registry, spot stenting using Eluvia stents was significantly associated with an increased risk of 1-year restenosis, which would require a full metal stenting procedure. 7 In clinical settings, upon encountering ostial FP lesions, we sometimes use stents as a final device. This is done because another Japanese registry demonstrated ostial FP lesions as a restenosis predictor in the balloon strategy using drug-coated balloons. 8
To date, only two studies have examined the clinical outcomes of stenting for ostial FP lesions coverage with deep femoral artery (DFA) coverage. First, Yamawaki et al. reported the results of ostial FP artery stenting using Luminexx (C.R. Bard, Murray Hill, NJ, USA), S.M.A.R.T. (Cordis Endovascular, Warren, NJ, USA), or WALLSTENT (Boston Scientific) and the outcome of DFA. 9 The following year, Hong et al. reported the clinical outcomes of EVT for the ostial FP artery and DFA with 171 lesions. 10 Although these studies have important clinical implications, they are not clinically applied in the use of old devices and are limited by small patient samples and single-center data.
Therefore, the present study was conducted to investigate the clinical outcomes of stenting for an ostial FP artery with DFA coverage using current EVT devices and an increased patient number in propensity-matched populations with a multicenter registry data.
Methods
Study design and patients
Retrospectively, we reviewed the records of patients with LEAD who underwent revascularization with EVT for FP lesions between January 2018 and December 2021 at eight cardiovascular centers in Japan. This study is a multicenter retrospective chart review of data from 2173 patients. The inclusion criteria were as follows: (1) age ⩾ 20 years; (2) symptomatic lower-limb ischemia with atherosclerotic lesions; and (3) presence of de novo lesions. Of these, data from a total of 457 consecutive patients (457 lesions), who had ostial FP lesions and who underwent revascularization with EVT, were extracted.
We divided them into two groups: stent with and without DFA coverage. A 1:1 propensity score-matched analysis of populations was performed to adjust the baseline and lesion characteristics differences.
Demographic, angiographic, and procedural data were collected from each hospital or the database by independent researchers. Follow-up clinical evaluations were carried out in months 1, 6, 12, 24, and 36 using the ankle–brachial index (ABI), duplex ultrasound, computed tomography (CT), or angiography. The follow-up data were obtained from hospital charts or by contacting patients, family members, or referring physicians.
The study protocol was approved by the Ethics Committees or Review Boards of the eight centers and was conducted following the tenets of the Declaration of Helsinki. Owing to retrospective enrollment, the requirement for obtaining written informed consent from the patients was waived.
Intervention
All procedures were performed under local anesthesia, which was supplemented with intravenous anesthesia depending on each institution. Depending on the complexity of the lesion, an ipsilateral antegrade or a contralateral crossover approach was chosen with the operator’s discretion. For the ipsilateral antegrade approach, a 6-Fr sheath, 6-Fr destination (Terumo, Tokyo, Japan), or a 6-Fr parent sheath (Medikit, Tokyo, Japan) was introduced through the ipsilateral common femoral artery (CFA). For the contralateral crossover approach, a 6-Fr Destination sheath (Terumo, Tokyo, Japan), 6-Fr parent sheath (Medikit, Tokyo, Japan), or the 6-Fr SheathLess PV (Asahi Intec, Aichi, Japan) was inserted through the contralateral femoral artery. After insertion of the sheath, unfractionated heparin was administered to achieve an activated clotting time of > 250 s. For the treatment of the FP lesions, a 0.014/0.018/0.035-inch guidewire was used, depending on the lesion characteristics and the operator’s preference. If the target lesion was chronic total occlusion (CTO), the procedure was performed by passing the guidewire through the intraplaque route as far as possible with an angiography, intravascular ultrasound, or extravascular sound guide. All lesions were treated with angioplasty using stents (Zilver PTX stent; Eluvia stent; S.M.A.R.T. stent; Supera stent [Abbott, Chicago, IN, USA]). Depending on the lesion and the patient’s background, we deployed the stents with or without DFA coverage; however, our routine practice was to avoid DFA coverage when possible.
Antiplatelet therapy with aspirin (100 mg daily), clopidogrel (75 mg daily), prasugrel (3.75 mg daily), or cilostazol (100 mg twice daily) was started at least 1 week before EVT and were continued for at least 1 month following the use of bare metal stents and for 3 months for drug-coated and eluting stents. At least one antiplatelet drug was maintained after EVT in all patients.
Endpoints
The following were analyzed as endpoints: percentage of DFA stenosis and occlusion during EVT, predictors of DFA occlusion immediately after EVT, primary patency of the FP artery at 3 years after revascularization, clinically driven-target lesion revascularization (CD-TLR) of the FP artery at 3 years, and cumulative incidence of DFA occlusion/major amputation/acute limb ischemia (ALI) until 3 years.
Definitions
Procedure success was defined as residual stenosis of < 30% without a suboptimal result. In the current study, cases with DFA occlusion prior to EVT were excluded and only patent or stenotic DFA status before EVT were analyzed. The Medina classification consisted of the distal CFA (1 cm proximal from the carina), the proximal FP artery (1 cm distal from the carina), and the ostial DFA. If stenosis ≥ 50% is present in an artery, it is classified as stenotic. Only the DFA was assessed as either patent or stenotic, as shown in Figure 1. Stenting with DFA coverage was considered to cover the DFA if it covered more than 50% of the artery.
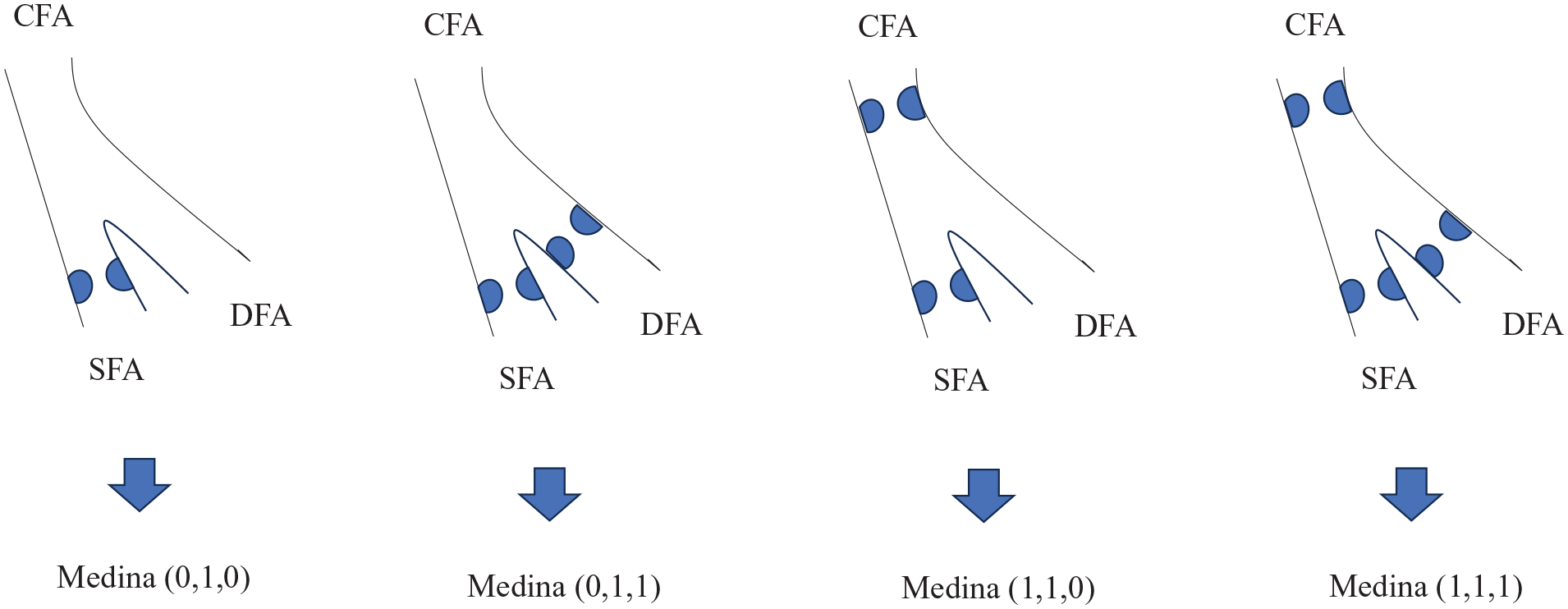
Medina classification of the common femoral artery. Blue circles represent stenotic lesions. Medina classification scores are presented as CFA, SFA, and DFA, respectively; 0 indicates no significant stenotic lesions and 1 indicates significant stenotic or occluded lesions.
Chronic limb-threatening ischemia (CLTI) includes a broader and more heterogeneous condition with varying degrees of ischemia that may delay wound healing and increase amputation risk. 11 Severe calcification was defined as the peripheral artery calcification scoring system 3–4, which was used to categorize the degree of lesion calcification on angiography. 12 Poor run-off was defined as having less than one patent infrapopliteal artery. Primary patency was defined as no restenosis of the treated vessel and revascularization that remained patent. CD-TLR was defined as re-EVT at an initial target lesion of ≥ 50% stenosis found at follow-up. Recurrence was defined as a peak systolic velocity ratio of > 2.4, ≥ 50% stenosis on angiography, duplex ultrasound, CT, or a decrease in the resting ABI of 0.2. 13 The recurrence pattern was defined according to the Tosaka classification. 14
The patency of DFA was not routinely assessed with duplex; therefore, we determined its patency using follow-up duplex ultrasound, angiography, or CT. In this study, we focused solely on whether the DFA was patent, and the Kaplan–Meier curve for DFA does not account for the presence of stenosis.
Statistical analysis
Statistical analyses were performed using JMP version 14.0.2 software (SAS Institute Inc., Cary, NC, USA). To minimize the potential influence of the nonrandomized study design, a propensity score was calculated using a multivariable logistic regression model with stent deployment position (stent with vs stent without DFA coverage) as the dependent variable. All baseline characteristics, age, sex, body mass index, hypertension, diabetes mellitus, dyslipidemia, hemodialysis, coronary artery disease, cerebral vascular disease, atrial fibrillation, current smoking, CLTI, reference diameter, lesion length, CTO, severe calcifications, poor run-off, crossover approach, pre-ABI, and Medina classification, were set as covariates. Matching was performed using the logit of the propensity score. A caliper cut-off of 0.20 was used to obtain a satisfactory balance. 15 Data are presented as numbers and percentages, mean ± SD, or median (IQR). Categorical variables were compared between groups using the chi-squared (χ2) or Fisher’s exact tests, as appropriate. Continuous variables were compared between groups using the t-test or Mann–Whitney U-test. Univariable analysis was performed to detect predictors of DFA occlusion immediately after EVT. A probability (p) value of less than 0.05 was considered statistically significant.
Results
Figure 2 depicts a patient flowchart process. Of the 457 patients, five patients were excluded due to DFA occlusion before EVT. As a result, 228 and 224 patients underwent stent deployment with and without DFA coverage, respectively. After propensity score matching, 132 matched pairs of patients who underwent EVT were included for analysis. In this study, follow-up was conducted for up to 3 years. During this period, 46 patients died, 13 underwent major amputations, and 79 were lost to follow-up. Thus, 126 patients were available for evaluation at 3 years. The baseline patient and lesion characteristics are summarized in Tables 1 and 2. Before matching, several significant differences were observed in patient and lesion characteristics, including: a lower percentage of patients undergoing hemodialysis (25.0% vs 44.6%; p < 0.001), and a higher prevalence of CTO (81.6% vs 53.1%; p < 0.001) in the group with stent deployment and DFA coverage. In the group with stent deployment and DFA coverage, the lesion involving CFA was significantly observed. In the matched populations, no significant differences were observed in the baseline patient and lesion clinical data between the two groups.
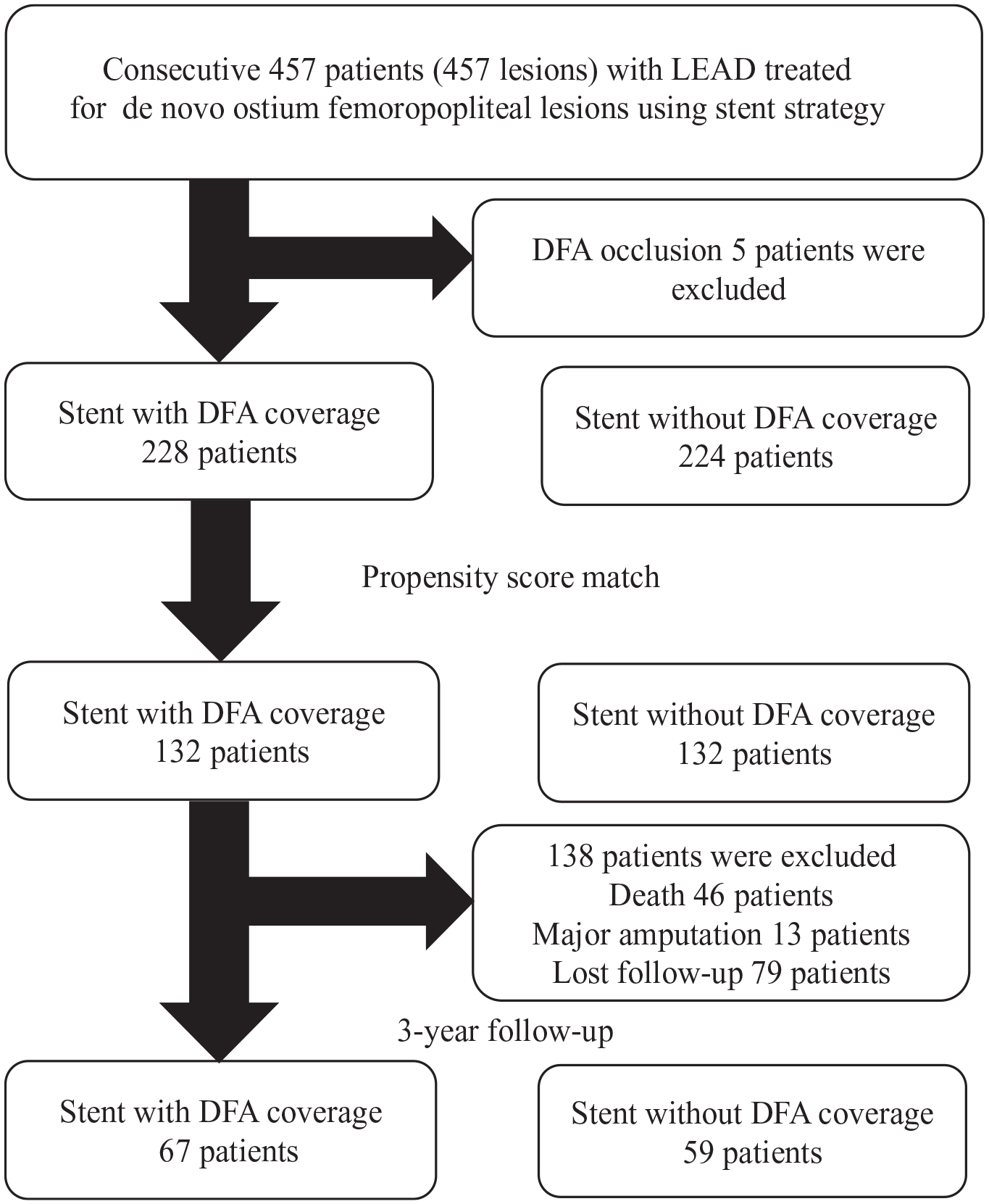
Patient flow chart.
Baseline characteristics.
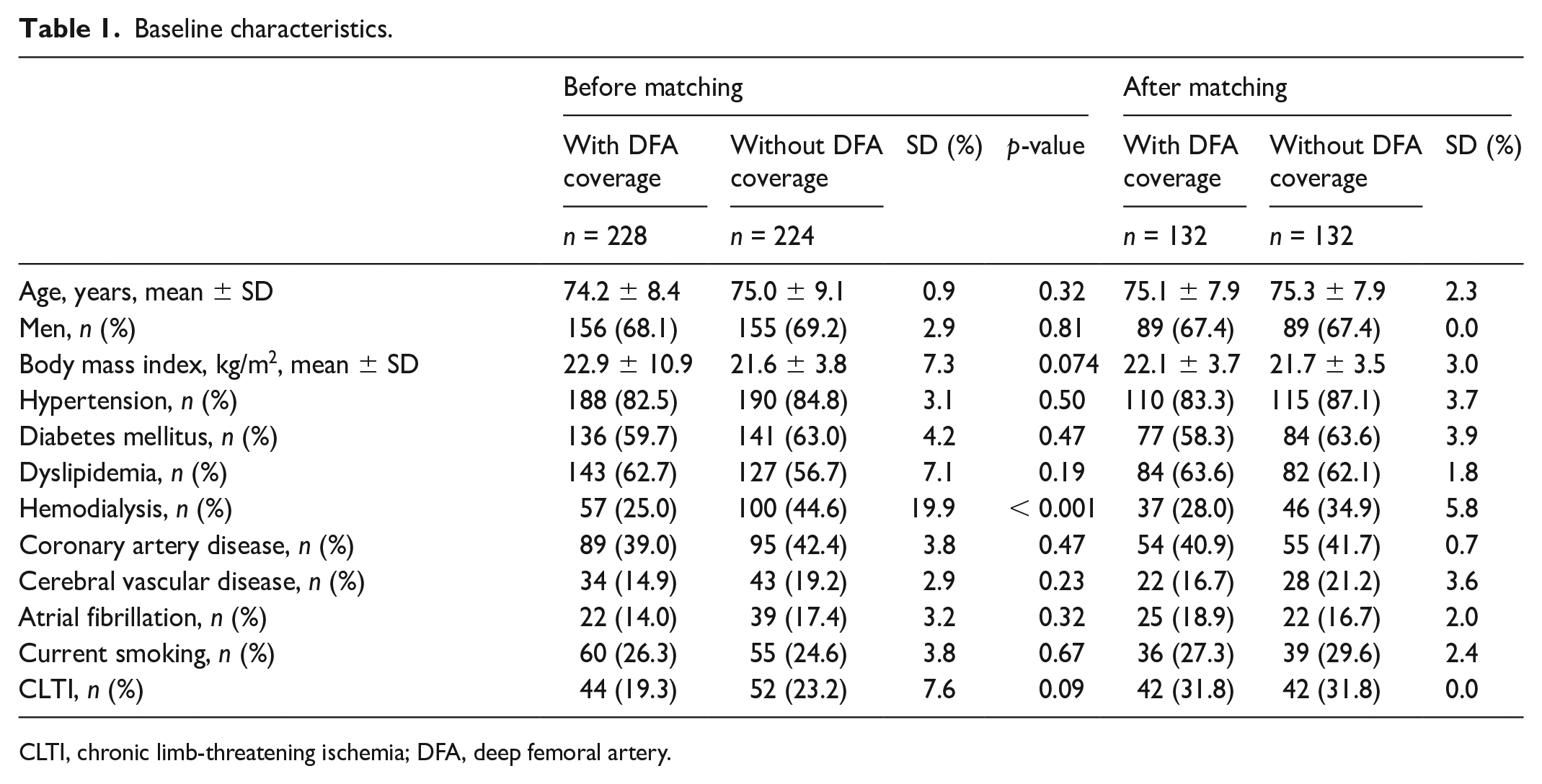
CLTI, chronic limb-threatening ischemia; DFA, deep femoral artery.
Lesion and procedural characteristics.
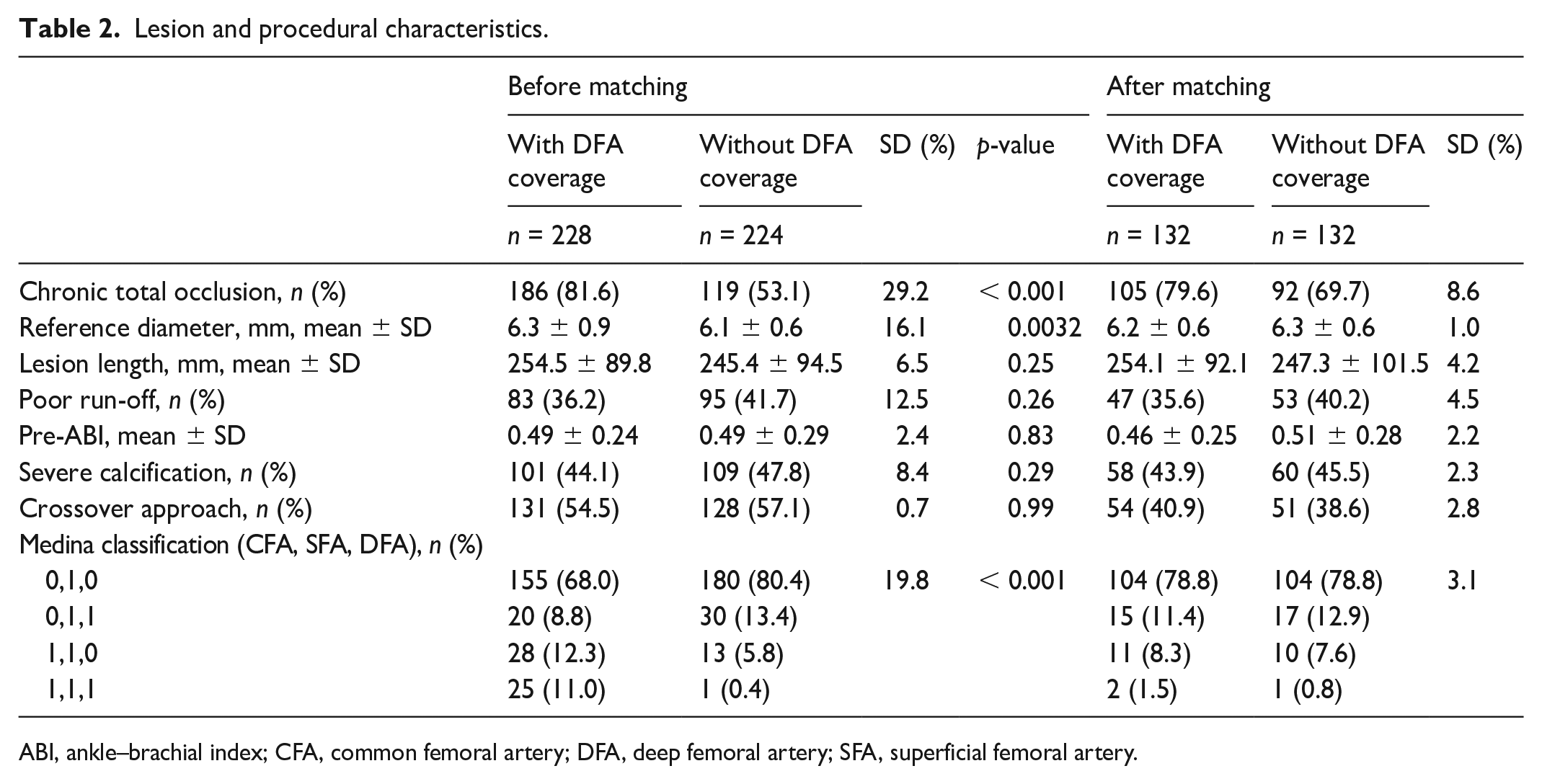
ABI, ankle–brachial index; CFA, common femoral artery; DFA, deep femoral artery; SFA, superficial femoral artery.
During the EVT procedure, only three DFA occlusion cases occurred in the stent group with DFA coverage. In these cases, additional EVT was performed, where the operators crossed the DFA occlusion and balloon angioplasty was performed to address the occluded DFA. Finally, the DFA flow improved. Subsequently, ≥ 50% stenosis of the DFA was frequently observed in the stent group with DFA coverage (Table 3).
Intervention results of deep femoral artery status after endovascular treatment.
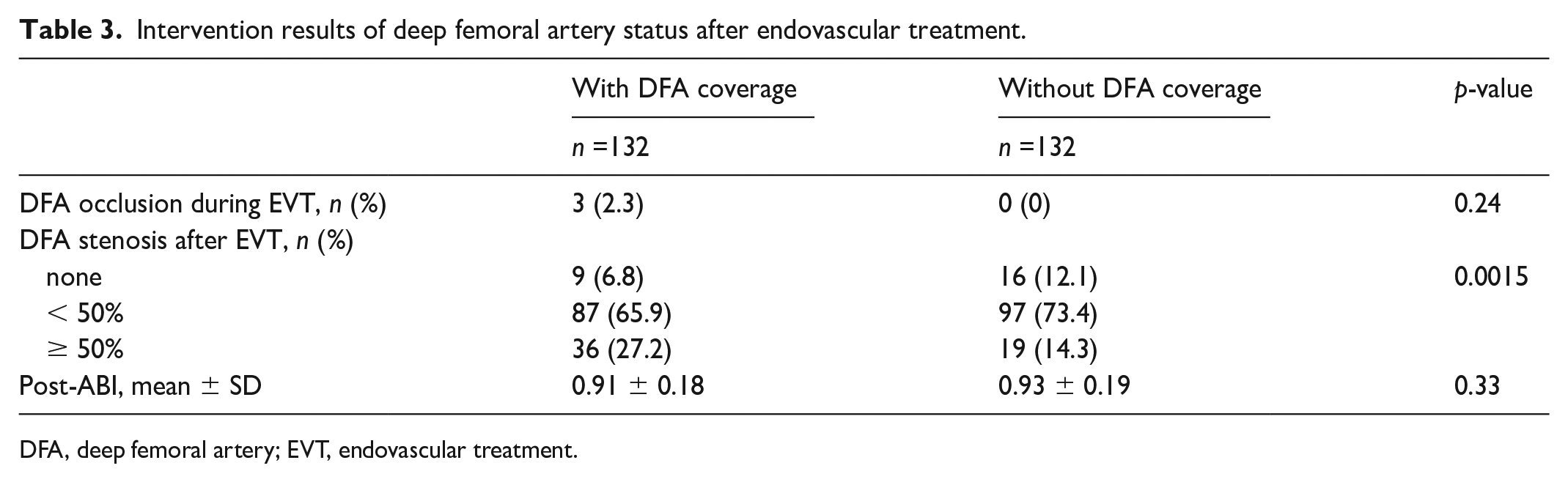
DFA, deep femoral artery; EVT, endovascular treatment.
In a univariable analysis, stenting with DFA coverage using Medina classifcations (0,1,1), (1,1,0), and (1,1,1), where 0 indicates no significant stenotic lesions and 1 indicates significant stenotic or occluded lesions in the CFA, SFA, and DFA, respectively, were predictors of DFA occlusion during EVT (Table S1).
No significant differences were observed between the two groups in primary patency of the FP artery, CD-TLR, incidence of DFA occlusion, major amputation, or ALI (Figures 3, 4, and S1–S3). During the study period, patients with intermittent claudication did not progress to CLTI or undergo any surgical conversions.
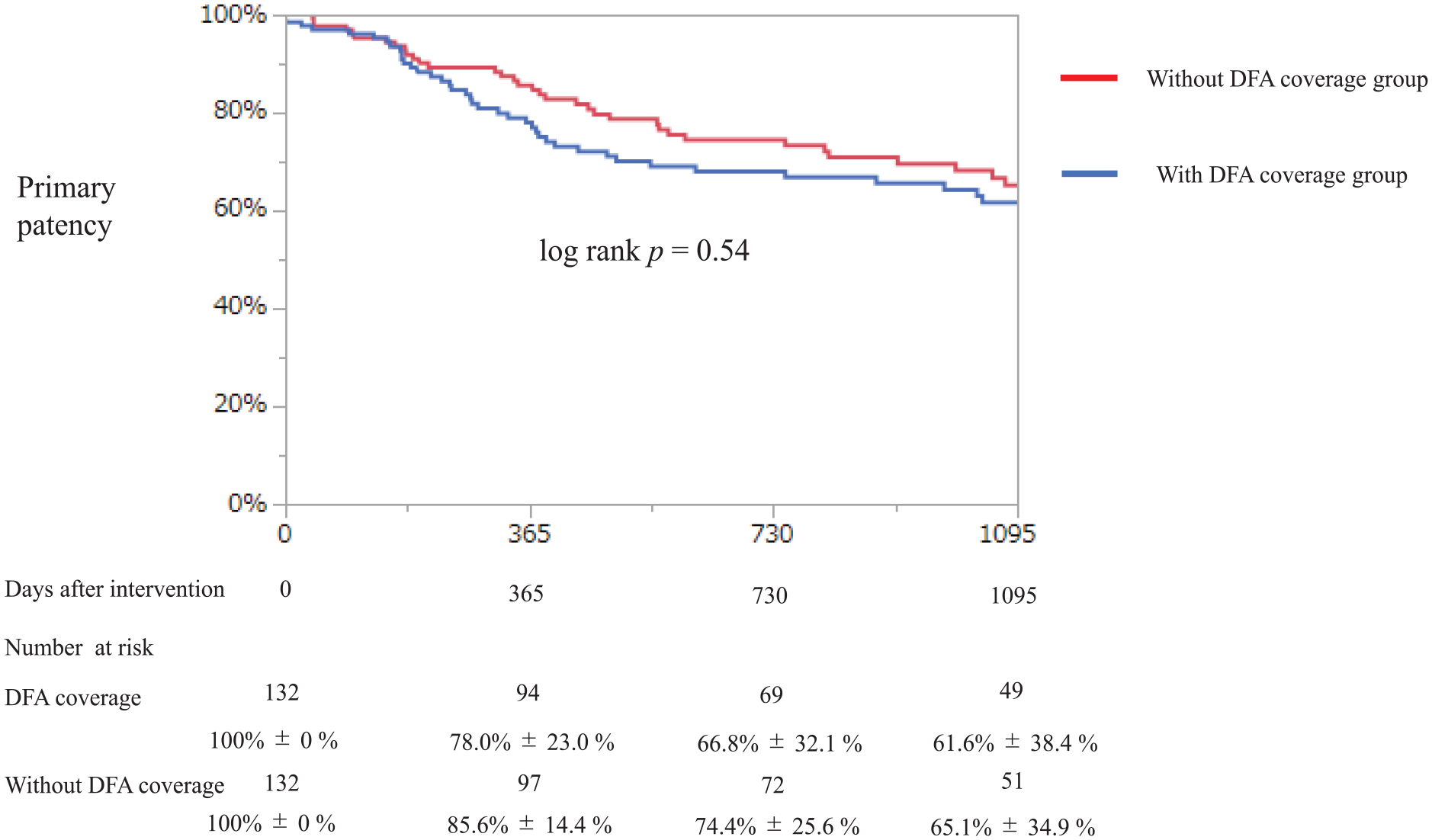
Kaplan–Meier curve for primary patency of femoropopliteal artery at 3 years.
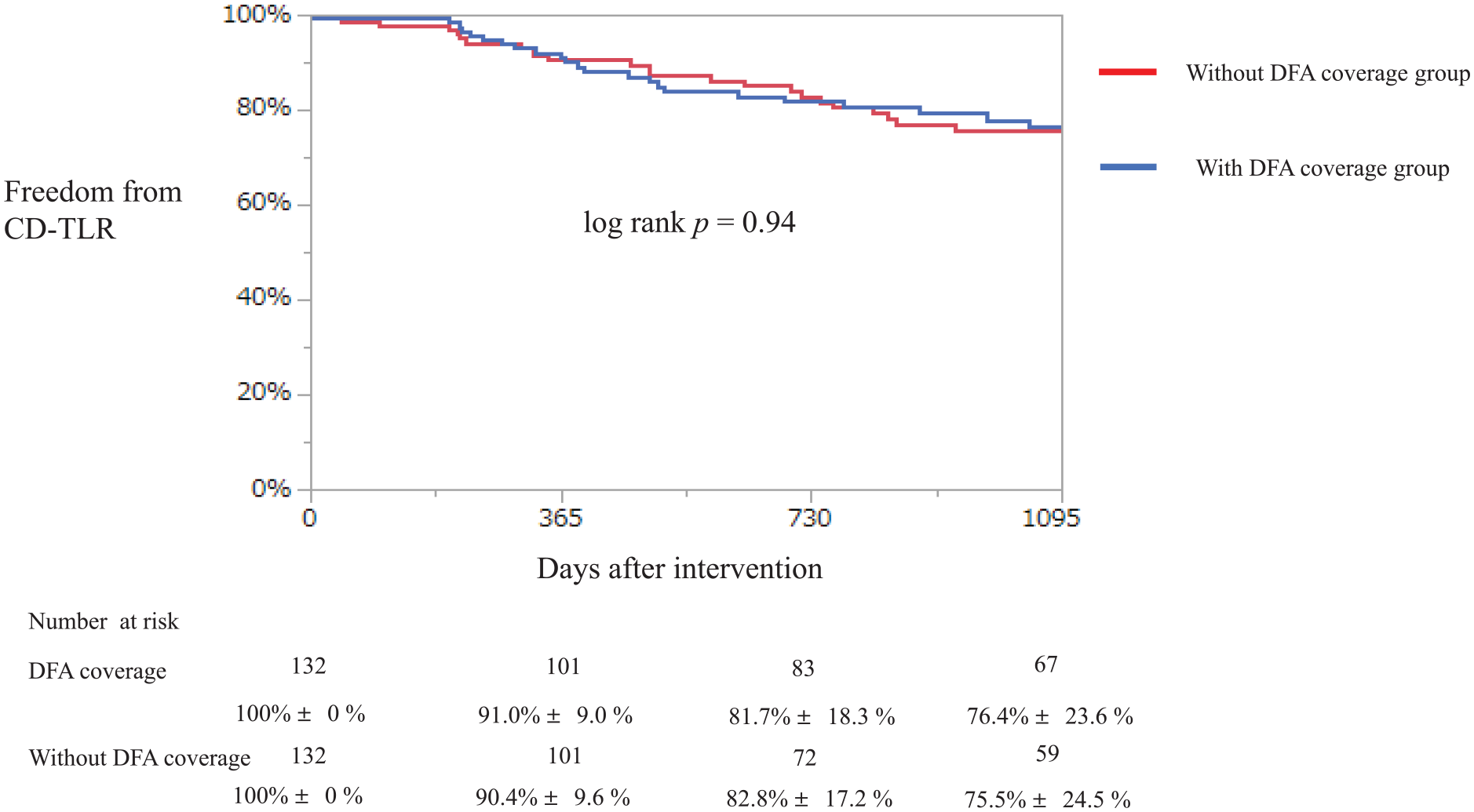
Kaplan–Meier curve of freedom from CD-TLR at 3 years.
Recurrence patterns are presented in Table S2, with complicated lesions observed more frequently in the stent group without DFA coverage. Tosaka III was significantly higher in the stent group without DFA coverage (62.5% vs 33.3%, p = 0.013).
Discussion
This study has three important findings: (1) stenting with DFA coverage led to immediate DFA stenosis after EVT; (2) CFA lesion complexity may contribute to DFA occlusion or stenosis immediately after DFA coverage stenting; and (3) no significant differences were observed in primary patency, CD-TLR of the FP artery, DFA occlusion, major amputation, and ALI between both groups during the chronic phase.
The role of DFA is important in maintaining collateral flow to the FP artery and limb viability if FP artery stenosis or occlusion occurs. Thus, obtaining sufficient blood flow from DFA is required even when stenting with DFA coverage is needed.
Two studies on the clinical outcomes of stent with DFA coverage stated that stenting with DFA coverage led to comparable FP artery patency. Importantly, these studies discussed the outcome of DFA after stenting with DFA coverage. Yamawaki et al. showed that ostial FP artery stenting was one of the predictors for loss of DFA patency. 9 Hong et al. demonstrated that the existence of DFA stenosis might lead to DFA flow limitation immediately after EVT. 10 However, the studies had several important limitations. For example, the procedure was done by an old device, which cannot match the current clinical practice and single-center, retrospective, and small patient numbers. To deal with these problems, we conducted a multicenter, large number, and current device data analysis. As patients were not randomized into the two groups, there may be baseline patient and lesion differences; therefore, a propensity score-matched analysis was performed. The results showed no significant differences in baseline and lesion characteristics between the groups. An important consideration in propensity score matching of the current study was whether the CFA lesion distribution was accounted for. In this study, we adjusted the Medina classification to balance the lesion background. After adjustment, no significant differences were observed, suggesting that the key points were appropriately addressed. After propensity score matching, DFA stenosis immediately after EVT was frequently observed in the stent group with DFA coverage. DFA occlusion or stenosis after stent deployment may result from the stent itself covering the DFA, or from intimal detachment causing obstruction. Additionally, the anatomical characteristics or plaque distribution of the CFA may also contribute to DFA occlusion or stenosis, or these mechanisms may interact. In percutaneous coronary intervention (PCI), longitudinal plaques or carina shifts are considered as the main cause of bifurcation occlusion. 16 The expert consensus document on PCI showed that side branch diameter stenosis is a clinical risk factor for a side branch occlusion, which was similar to one of the predictors of DFA occlusion in the current study. However, this discussion is specific to PCI and may not directly apply to EVT. Hence, close attention should be given to DFA occlusion if we deploy stents with DFA coverage due to DFA stenosis’s original existence. To avoid DFA occlusion immediately after EVT, dilation of DFA before stenting could be a solution. Similarly, jailed wire or balloon technique could be useful, as reported in the review paper of coronary intervention area. 17 Similar to the previous study, we demonstrated that primary patency and CD-TLR of the FP artery did not differ in the current study. 10 The explanation of residual plaque existence, balloon injury, or weak radial forces on a residual plaque at the edge of the stent could apply to the other study, but recent intervention techniques or knowledge including intravascular ultrasound sonography use or balloon angioplasty might improve the patency of the FP artery, even in the stent without DFA coverage group. 9
In addition, we investigated the frequency of major amputation and ALI. In this study, we demonstrated that DFA stenosis immediately after EVT was frequently observed in the stent deployment with DFA coverage group. Thus, though the possibility of an increased risk of major amputation or ALI should be considered, no significant difference in the occurrence of major amputation or ALI was observed in this study. This may be because DFA occlusion during follow-up did not differ between the two groups. This study holds significant importance because, although stent occlusion in the SFA may not result in ALI, occlusion extending to the DFA can lead to ALI. Therefore, if stenosis persists in the DFA, there is a potential risk of ALI. No significant differences were found between the two groups regarding ALI or major amputations, but the available data covers only up to 3 years, and longer-term data may yield different results. We believe that routine stent deployment with DFA coverage should be avoided, as DFA stenosis immediately after EVT was more frequently observed in the stent group with DFA coverage, posing a potential risk of compromised future collateral flow. However, we must consider careful deployment of stents with DFA coverage if lesions extended to the CFA exist. Our findings need further validation through prospective studies and long-term clinical outcomes.
Study limitations
There are some important limitations to consider in the present study. First, the number of patients was low, and propensity score-matched analysis was performed to overcome baseline differences. Second, the study was retrospective and not evaluated in a core laboratory. Additionally, the angle of the DFA could not be measured and was not analyzed. Third, DFA patency was evaluated only with angiography or CT; hence, we could not demonstrate the stenosis percentage of the DFA. Fourth, we must consider recall bias. Although this study was retrospective and we attempted to verify cases directly whenever possible, there were instances where this was not feasible.
Finally, the study cohort was limited to Japanese patients. Therefore, further prospective and increased studies are warranted to confirm these results in other populations.
Conclusion
Stent deployment with deep femoral artery (DFA) coverage significantly led to stenotic events immediately after endovascular treatment. However, in the chronic phase, there were no differences in the patency of the femoropopliteal (FP) artery and DFA, major amputation, and ALI based on stent deployment for ostial FP lesions.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241311936 – Supplemental material for Outcomes of contemporary stents with deep femoral artery coverage
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241311936 for Outcomes of contemporary stents with deep femoral artery coverage by Takahiro Tokuda, Naoki Yoshioka, Akiko Tanaka, Shunsuke Kojima, Kohei Yamaguchi, Takashi Yanagiuchi, Kenji Ogata, Tatsuro Takei and Tatsuya Nakama in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X241311936 – Supplemental material for Outcomes of contemporary stents with deep femoral artery coverage
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X241311936 for Outcomes of contemporary stents with deep femoral artery coverage by Takahiro Tokuda, Naoki Yoshioka, Akiko Tanaka, Shunsuke Kojima, Kohei Yamaguchi, Takashi Yanagiuchi, Kenji Ogata, Tatsuro Takei and Tatsuya Nakama in Vascular Medicine
Supplemental Material
sj-pdf-3-vmj-10.1177_1358863X241311936 – Supplemental material for Outcomes of contemporary stents with deep femoral artery coverage
Supplemental material, sj-pdf-3-vmj-10.1177_1358863X241311936 for Outcomes of contemporary stents with deep femoral artery coverage by Takahiro Tokuda, Naoki Yoshioka, Akiko Tanaka, Shunsuke Kojima, Kohei Yamaguchi, Takashi Yanagiuchi, Kenji Ogata, Tatsuro Takei and Tatsuya Nakama in Vascular Medicine
Supplemental Material
sj-pdf-4-vmj-10.1177_1358863X241311936 – Supplemental material for Outcomes of contemporary stents with deep femoral artery coverage
Supplemental material, sj-pdf-4-vmj-10.1177_1358863X241311936 for Outcomes of contemporary stents with deep femoral artery coverage by Takahiro Tokuda, Naoki Yoshioka, Akiko Tanaka, Shunsuke Kojima, Kohei Yamaguchi, Takashi Yanagiuchi, Kenji Ogata, Tatsuro Takei and Tatsuya Nakama in Vascular Medicine
Supplemental Material
sj-pdf-5-vmj-10.1177_1358863X241311936 – Supplemental material for Outcomes of contemporary stents with deep femoral artery coverage
Supplemental material, sj-pdf-5-vmj-10.1177_1358863X241311936 for Outcomes of contemporary stents with deep femoral artery coverage by Takahiro Tokuda, Naoki Yoshioka, Akiko Tanaka, Shunsuke Kojima, Kohei Yamaguchi, Takashi Yanagiuchi, Kenji Ogata, Tatsuro Takei and Tatsuya Nakama in Vascular Medicine
Footnotes
Acknowledgements
A commentary by Dubosq-Lebaz and Secemsky accompanies this article. 18
Data availability statement
The datasets of the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tatsuya Nakama is a consultant of Asahi Intecc, BD, Boston Scientific, COOK Medical, Cordis, Kaneka Medix, NIPRO, and OrbusNeich. All other authors have no conflicting interests.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
