Abstract
Ascertainment bias is a well-recognized source of bias in research, but few studies have systematically analyzed sources of ascertainment bias in randomized trials in which blinding is not possible and endpoint assessment is not protocolized. In the current study, we sought to evaluate differences in the clinical practice patterns of trial investigators with respect to bias in the ascertainment of pre-revascularization patient risk and the incidence of secondary endpoints post-revascularization. We conducted a cross-sectional survey of active investigators (n = 936) from the Best Endovascular Versus Best Surgical Therapy for Patients with Critical Limb Ischemia (BEST-CLI) trial. The total survey response rate was 19.6% (183/936). Vascular surgeons were more likely than nonsurgical interventionalists to order tests for cardiac complications after both surgical bypass (p < 0.001) and endovascular revascularization (p = 0.038). Post-procedure, investigators were more likely to order additional testing for cardiac complications in open surgery versus endovascular cases (7% vs 16% never, 41% vs 65% rarely, 43% vs 17% sometimes, 9% vs 2% always, respectively; p < 0.0001). Significant variation in practice patterns exist in the pre- and post-procedure assessment of cardiac risk and events for patients with CLI undergoing revascularization. Variation in the ascertainment of risk and outcomes according to the type of revascularization procedure and physician specialty should be considered when interpreting the results of clinical studies, such as the BEST-CLI trial.
Introduction
Critical limb ischemia (CLI) is the most advanced manifestation of lower extremity peripheral artery disease (PAD) and is characterized by pain at rest in the ankle or foot and/or the presence of ischemic ulcerations or necrotic tissue. Approximately 20–40% of patients with CLI will require amputation and greater than 20% of patients will die without successful lower extremity revascularization.1,2 Equipoise exists in the optimal technique (open vs endovascular) for revascularization in patients with CLI.3,4
The Best Endovascular Versus Best Surgical Therapy for Patients With Critical Limb Ischemia (BEST-CLI) trial is a prospective randomized study comparing the safety and efficacy of open surgical versus endovascular lower extremity revascularization in patients with CLI. 5 Blinding of treatment strategy is often not feasible in interventional trials such as BEST-CLI where a surgical operation is compared to a minimally invasive catheter-based procedure. 6 This lack of blinding introduces the possibility of ascertainment bias, a well-recognized source of bias even in randomized controlled trials. 7 Ascertainment bias is the systematic distortion of the assessment of outcome measures by the investigators or trial participants because they are aware of treatment allocation. This is particularly problematic for trial endpoints that do not have protocol-mandated testing for their ascertainment, as may be the case for observational studies comparing minimally invasive to open surgical interventions.8 –10 The detection of such endpoints is therefore subject to the practice patterns of the individual investigators or study sites.
In the current study, we sought to characterize the clinical practices of trial investigators with respect to pre- and post-procedure testing for cardiac events using a specially designed survey to assess ascertainment bias. The primary purpose of the survey was to determine whether the potential for ‘missed’ secondary endpoints in BEST-CLI, such as myocardial infarction (MI), and other cardiovascular events, might differ between patients randomized to open surgery versus endovascular revascularization due to dissimilarities in individual or institutional practice patterns. We also compared practice patterns between vascular surgeons and investigators from other specialties.
Methods
Survey and study design
We conducted a cross-sectional survey of BEST-CLI investigators. The survey was distributed via Survey Monkey to the 936 active investigators as of June 2016. We analyzed practice patterns according to (1) the type of revascularization (endovascular vs open surgical); and (2) medical subspecialty. All ethical and institutional review boards of the participating BEST-CLI centers approved this study.
Questionnaire
The survey tool was developed by the principal investigators of the BEST-CLI trial. The survey evaluated operator background and demographics, self-reported primary specialty, practice location (zip code), and individual volume of CLI cases. The survey then explored the individual and institutional practice patterns with respect to pre- and/or post-procedure cardiac testing. Finally, survey participants were asked a series of questions to characterize which circumstances (patient subgroup or particular symptoms/signs) would trigger pre-procedure cardiac testing for risk assessment. Furthermore, post-procedure evaluation for MI for both endovascular and open procedures were used to determine potential differences in operator practice between modes of intervention (see the supplementary online material for the complete survey).
Statistical analysis
Fisher’s exact test was used to compare response rates between different investigator specialties. Descriptive statistics for continuous outcomes were presented using medians and IQR range. Dot plots were used to present data rather than box-and-whisker plots because the latter can be uninformative for extremely skewed data.
Comparisons of practice patterns for patients undergoing open surgery versus endovascular revascularization took into account the paired nature of these data (i.e., one response for open surgery patients and one response for endovascular revascularization patients). For yes/no responses, McNemar’s test was used. For continuous outcomes, we used the Wilcoxon signed rank test. For ordered categorical outcomes, we assigned consecutive scores for the possible responses (e.g., if the possible responses were never, rarely, sometimes, and always, the scores were 0, 1, 2, and 3, respectively), and then used the Wilcoxon signed rank test to compare differences between the investigator’s response for open surgery patients and endovascular revascularization patients. Missing data were rare. Investigators who were missing one or both of the paired responses were dropped from that particular analysis.
Comparisons of responses from vascular surgeons versus other specialties used Fisher’s exact test for binary outcomes, the Wilcoxon test for continuous outcomes, and an exact version of the Mantel–Haenszel chi-squared test for ordered categorical outcomes. All analyses were done with SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Provider characteristics and response rate
Overall demographic characteristics are shown in Table 1. A majority of respondents were high volume operators including 76% of respondents who reported caring for over 25 patients with CLI per year. The vast majority were vascular surgeons (77%); interventional cardiologists (13%) were the next largest group of respondents (online Supplemental Figure 1). Most respondents had approximately 20 years of clinical service. The total response rate for the survey was 19.6% (183/936), with some variation according to specialty: 19.9% (140/702) of vascular surgeons, 21.4% (24/112) of interventional cardiologists, 13.9% (16/115) of interventional radiologists, 100% (3/3) of vascular medicine physicians, and 0% (0/4) of other specialties.
Demographics of respondents.
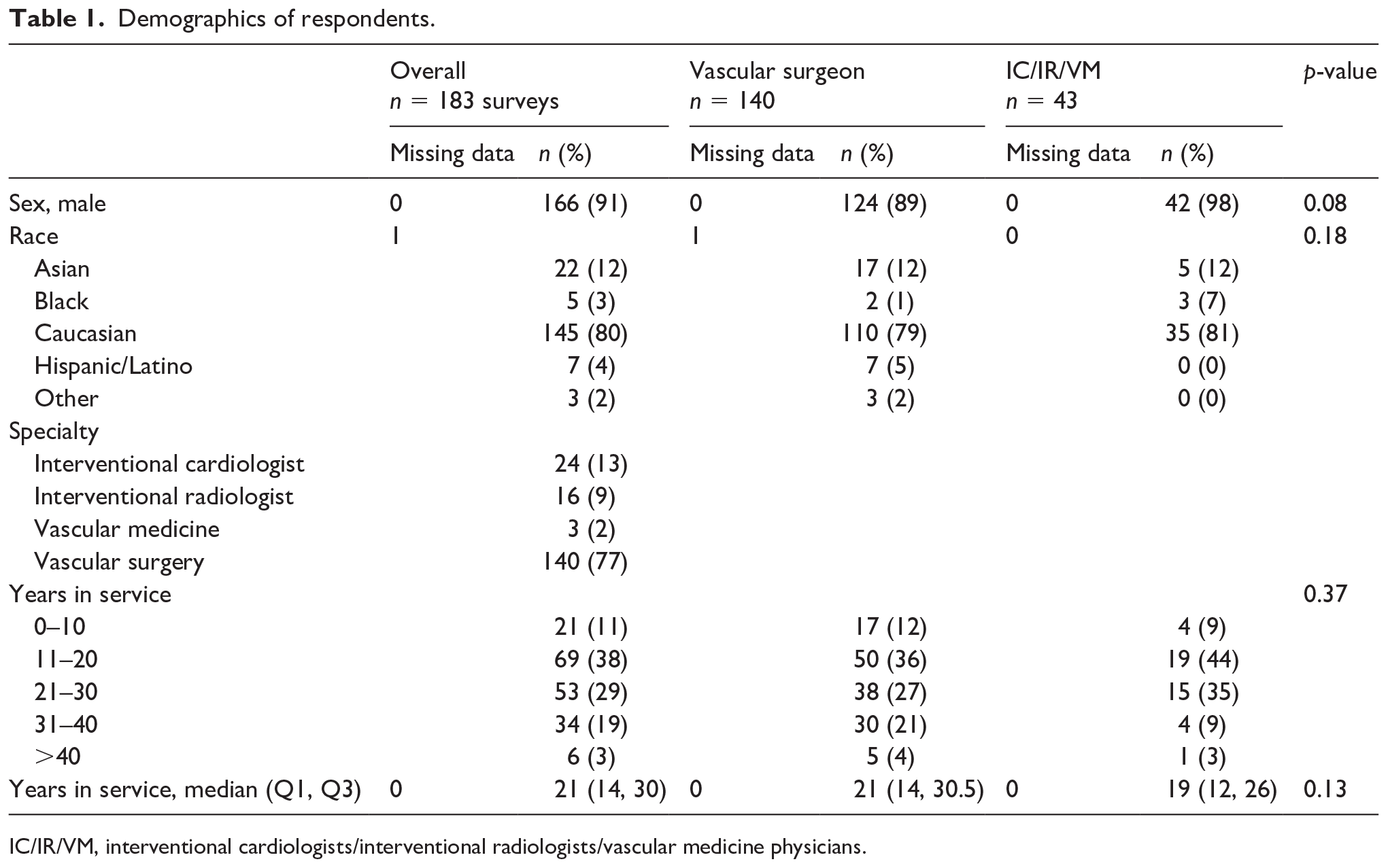
IC/IR/VM, interventional cardiologists/interventional radiologists/vascular medicine physicians.
Comparison of practice patterns for patients treated with open surgical vs endovascular revascularization
Standardized pre-procedural practice patterns
In their personal practice, 65% of investigators reported following a standardized algorithm for the cardiac evaluation of patients prior to open surgical bypass, compared to 32% who followed a standardized algorithm prior to endovascular revascularization (p < 0.0001) (online Supplemental Figure 2A). Institutional algorithms for pre-revascularization cardiac evaluation were reported by 34% of investigators for open surgery, and by 12% for endovascular procedures (p < 0.0001). Pre-procedure, 90% of investigators ordered electrocardiograms (ECGs) for all open surgery patients, and 63% ordered them for all endovascular revascularization patients (Figure 1). Cardiac stress tests, cardiac catheterizations, and cardiac biomarker tests were less commonly ordered (medians of 50%, 10%, and 0% of open surgery patients and 5%, 0%, and 0% of endovascular patients, respectively) (Figure 1). All four types of pre-procedure cardiac evaluation were ordered more often for patients undergoing open surgery (p < 0.0001 for each comparison) (Figure 1).
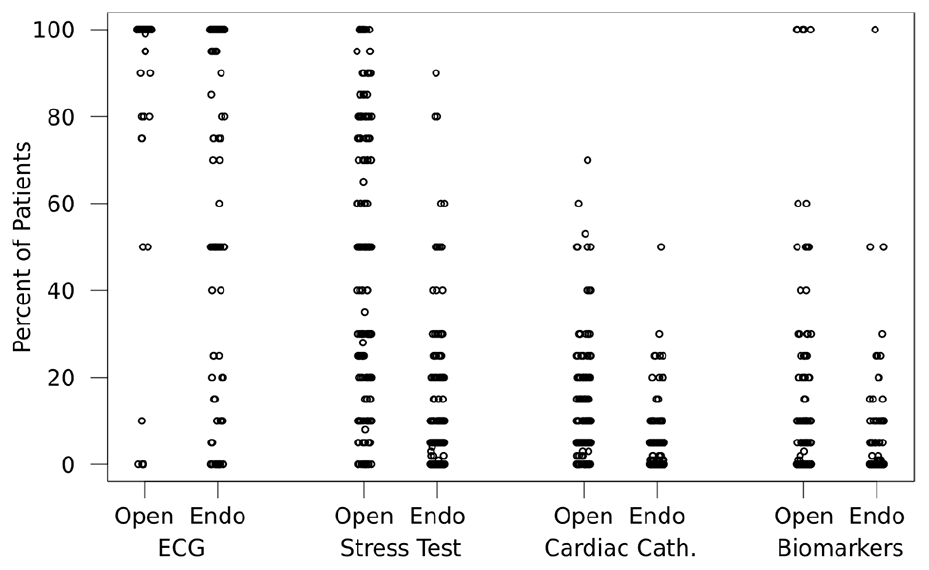
Reported percentage of patients with CLI undergoing pre-procedure cardiac evaluations before open surgical and endovascular revascularization procedures.
Post-procedural practice patterns
Following revascularization, investigators were more likely to order additional testing for cardiac complications in open surgery cases (7% never, 41% rarely, 43% sometimes, 9% always) than endovascular cases (16%, 65%, 17%, and 2%, respectively; p < 0.0001) (Figure 2B). In their personal practice, 29% of investigators claimed to follow a standardized algorithm for cardiac evaluation of open surgery patients, compared to 21% for endovascular patients (p = 0.0004). Institutional algorithms for cardiac evaluation were reported by 15% of investigators for open surgery and 9% for endovascular procedures folllowing revascularization (p = 0.0008) (online Supplemental Figure 2B). Post-procedure ECG, creatinine phosphokinase (CPK), creatinine phosphokinase-myocardial band (CPK-MB), and troponin tests were all ordered more often for open surgery patients than for endovascular patients (p < 0.0001 for each comparison) (Figure 3).
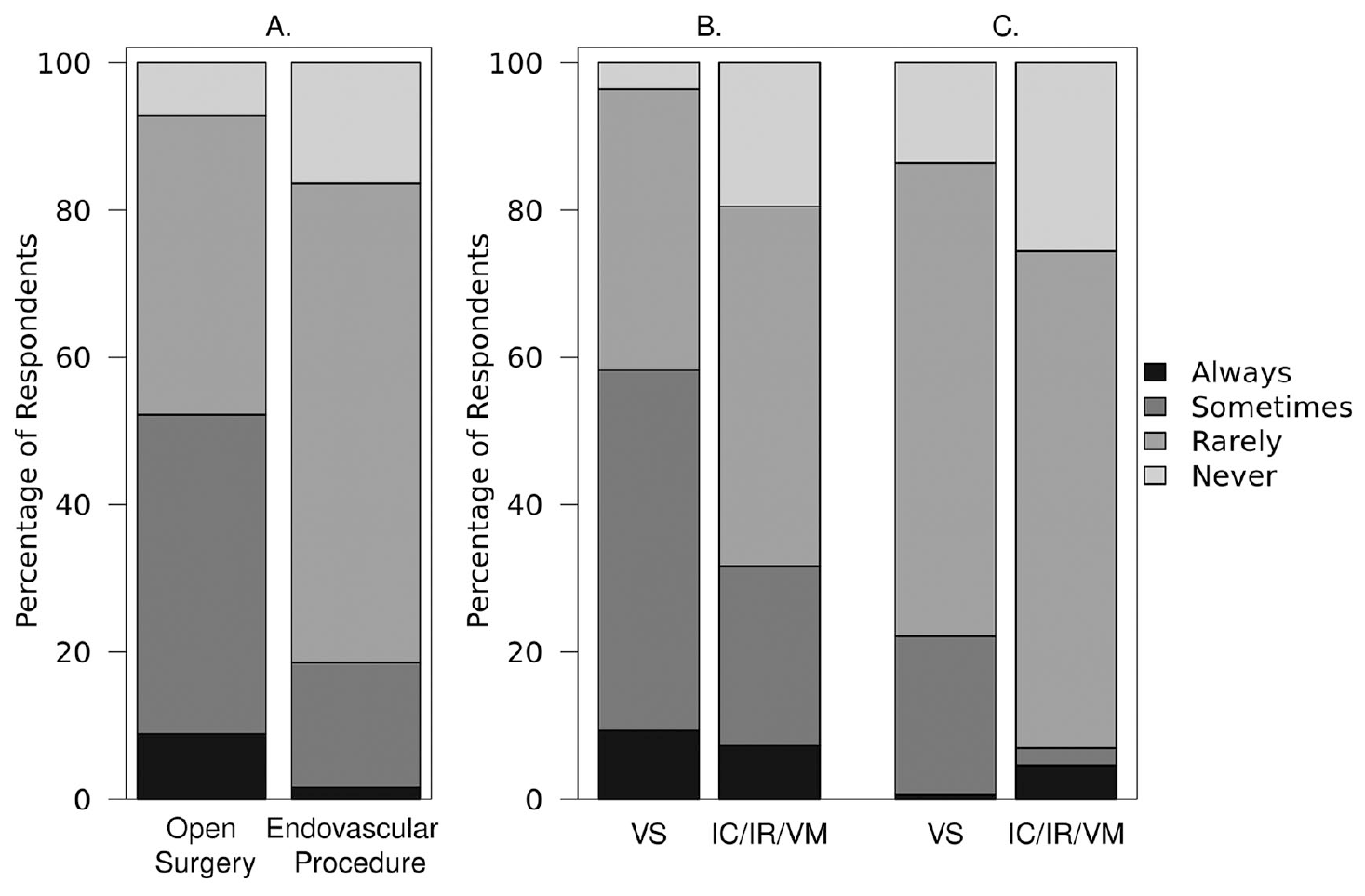
Frequency in which additional testing is ordered to assess for cardiac complications following revascularization in patients with CLI (A) by procedure type and by investigator specialty for (B) open surgery and (C) endovascular revascularization subjects.
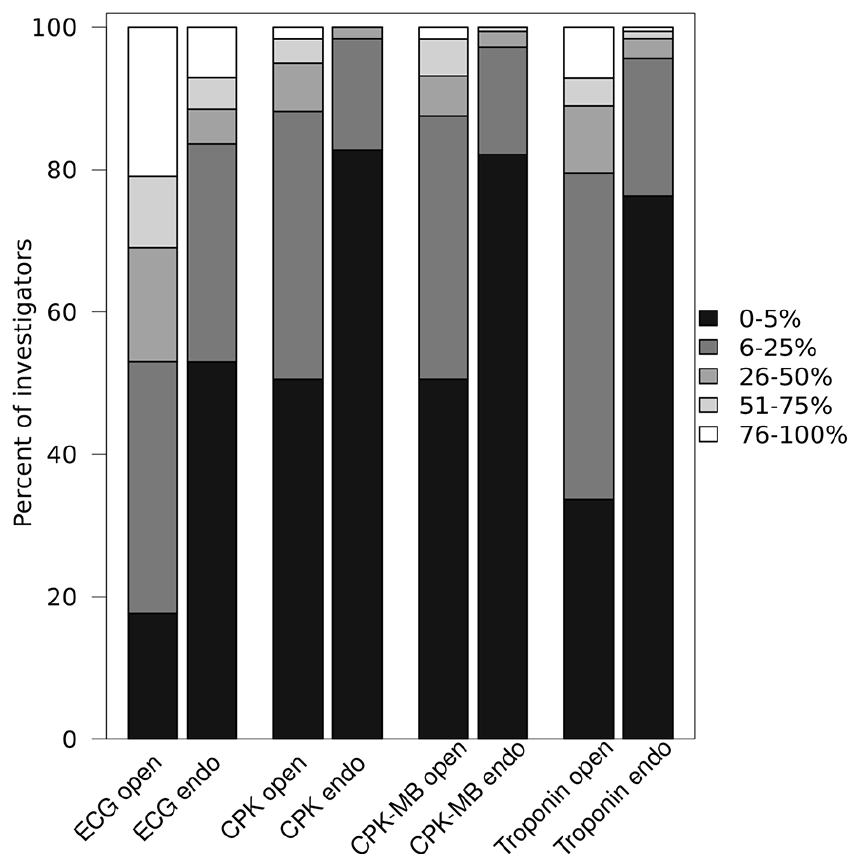
Reported percentage of patients with CLI in which the specified test is collected following revascularization, by procedure type.
Comparison of practice patterns of vascular surgeons vs nonsurgical investigators
Revascularization techniques
Vascular surgeons reported the following breakdown of revascularization techniques for patients under their care with CLI: a median 50% (IQR: 40–62.5%) receive endovascular revascularization, 30% (5–40%) receive open surgical revascularization, and 10% (7.5–20%) undergo hybrid revascularization (endovascular + open surgical) (online Supplemental Figure 3). Nonsurgical interventionalists reported a median of 85% (75–95%), 10% (0–20%), and 5% (0–10%) of their patients with CLI receive endovascular, open surgical, and hybrid revascularization, respectively (online Supplemental Figure 3). Vascular surgeons were more likely to report performance of open surgical and hybrid revascularization, whereas nonsurgical interventionalists more commonly reported performing endovascular revascularization (p < 0.001 for all comparisons) (online Supplemental Figure 3).
Pre-procedural cardiac assessment of patients undergoing open surgical revascularization
For patients undergoing open surgical revascularization, 67% of vascular surgeons and 56% of other investigators reported a personal algorithm for pre-procedure cardiac evaluation (p = 0.20); 33% of vascular surgeons and 37% of other investigators reported institutional algorithms (p = 0.71) (online Supplemental Figure 2A).
Vascular surgeons were more likely than other investigators to order ECGs before open surgical revascularization (p = 0.02) (Figure 2). Cardiac stress tests and cardiac biomarker testing before open surgical revascularization did not differ by investigator type. Regarding invasive evaluation, there was a trend towards fewer cardiac catheterizations among vascular surgery versus nonvascular surgeon investigators (10% (IQR: 5–20%) vs 15% (IQR: 5–25%), p = 0.06) (Figure 1).
Pre-procedural cardiac assessment of patients undergoing endovascular revascularization
There was no statistical difference between the percentage of vascular surgeons versus other investigators reporting a personal algorithm for pre-procedure cardiac evaluation for patients scheduled for endovascular procedures (34% vs 28%, p = 0.58); similarly, there was no difference between these groups reporting institutional algorithms (12% vs 12%, p = 1.00) (online Supplemental Figure 2A).
ECGs were commonly performed by both vascular surgeons and nonsurgical interventionalists (100% (IQR: 65%–100%) vs 100% (40%–100%), p = 0.08) before endovascular revascularization. Cardiac stress tests, cardiac catheterization, and cardiac biomarkers were rarely ordered for these patients, and vascular surgeons were less likely to order cardiac catheterization compared to their nonsurgical counterparts (0% (0–5%) vs 5% (0–10%), p = 0.02) (Figure 2).
Pre-procedural triggers for cardiovascular assessment
A minority of both vascular surgeons and other investigators reported performing pre-procedure cardiac evaluation in every patient before revascularization (15% and 19%, p = 0.63). Specific triggers for cardiac evaluation for vascular surgeons compared to nonsurgical interventionalists included history of congestive heart failure (CHF) (69% vs 53%), history of coronary artery disease (CAD) (56% vs 67%), presence of cardiac risk factors (52% vs 53%), history of arrhythmia (34% vs 26%), and older age (28% vs 19%). Pre-procedural triggers did not significantly differ by investigator type.
Post-procedure cardiovascular assessment
A personal standardized algorithm for cardiac evaluation following open surgical bypass was reported by 31% of vascular surgeons and 25% of other investigators (p = 0.56); 14% of vascular surgeons and 20% of other investigators reported the existence of an institutional algorithm (p = 0.46). A personal standardized algorithm for cardiac evaluation after endovascular intervention was reported by 20% of vascular surgeons and 23% of other investigators (p = 0.67); 7% of vascular surgeons and 16% of other investigators reported the existence of an institutional algorithm (p = 0.13) (Figure 2B).
Figures 2B and 2C show how often vascular surgeons and nonsurgical interventionalists reported performing post-procedure testing for cardiac complications in patients who have undergone open surgical and endovascular revascularization. Vascular surgeons were more likely than nonsurgical interventionalists to order tests for cardiac complications after both surgical bypass (p < 0.001) and endovascular revascularization (p = 0.0381).
Figure 4A shows the frequency by which vascular surgeons and nonsurgical interventionalists order various types of cardiac evaluations after open surgical bypass. There was no difference in frequency of ECGs ordered by specialty type (p = 0.71); however, following open surgical revascularization, vascular surgeons were more likely than nonsurgical interventionalists to order CPK, CPK-MB, or troponin (p = 0.05, 0.004, and 0.004, respectively).
Figure 4B depicts the practice patterns of vascular surgeons and nonsurgical interventionalists regarding ordering cardiac tests after endovascular revascularization. The specialty groups did not differ in how often CPK and CPK-MB were ordered (p = 0.23 and 0.16, respectively), but vascular surgeons were less likely to order ECGs (p = 0.02) and more likely than nonsurgical interventionalists to order troponin (p = 0.02).
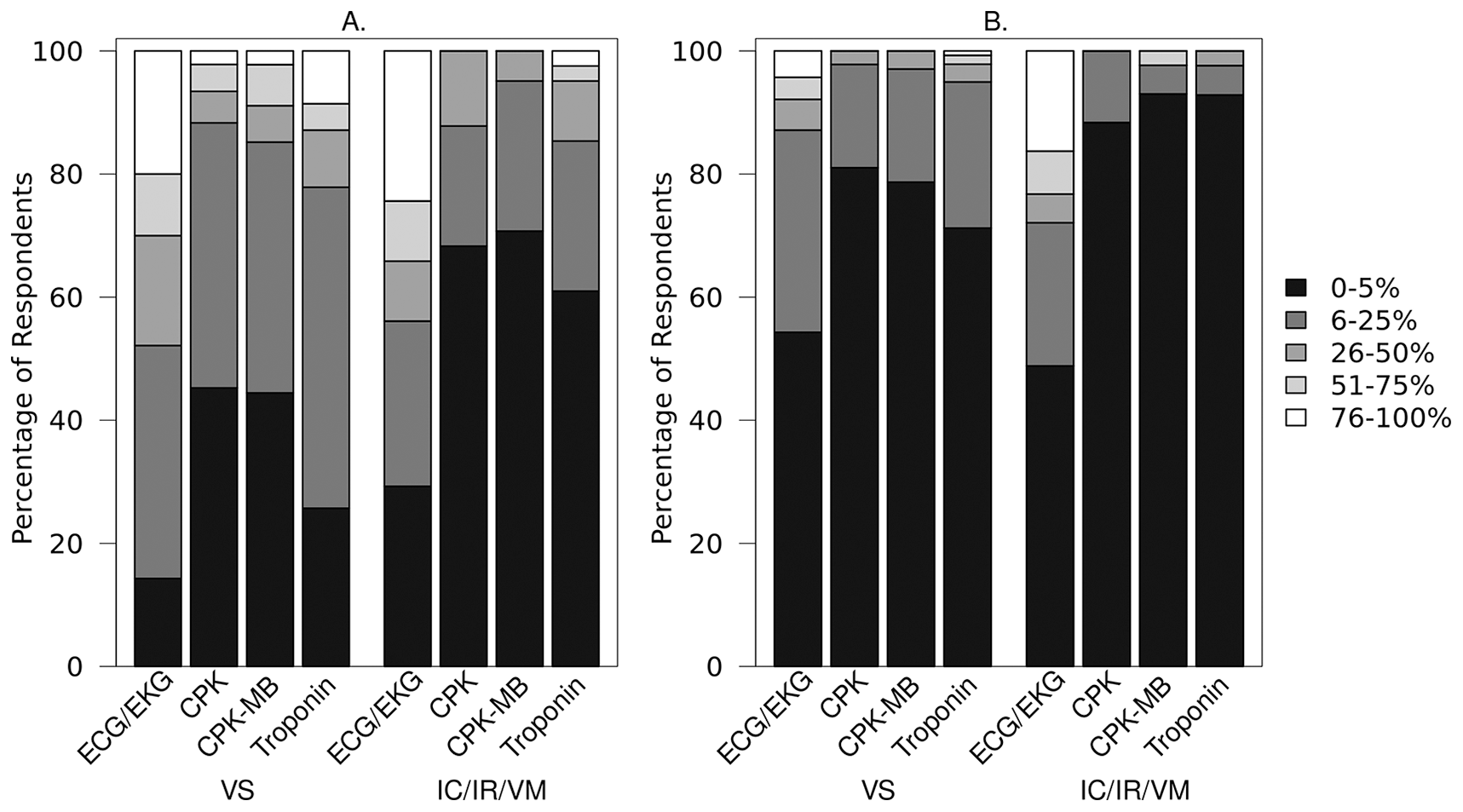
Reported percentage of patients with CLI in which specified test is collected following revascularization, by investigator specialty for (A) surgical bypass and (B) endovascular revascularization subjects.
Post-procedural triggers for cardiovascular assessment
Indications that trigger post-procedure cardiac evaluation, testing, or referral included clinical signs of ischemia (98% of vascular surgeons and 93% of other investigators (p = 0.14)), ECG changes during or after the procedure (94% vs 84%, p = 0.0499), clinical signs of decompensated heart failure (96% vs 81%, p = 0.01), hemodynamic instability during or after the procedure (89% vs 79%, p = 0.13), and arrhythmia during or after the procedure (89% vs 72%, p = 0.01). Only 2% of vascular surgeons and 5% of other investigators routinely evaluate all patients for cardiac complications (p = 0.34).
Discussion
The validity and relevance of clinical trial results may be critically affected by bias. Ascertainment bias is an important type of bias that may be difficult to detect and appraise, particularly in interventional trials comparing invasive and minimally invasive therapies.11,12 To our knowledge, this survey is the first study to systematically examine perioperative cardiac and neurologic assessments of patients by investigators in a clinical intervention research study. In the current study, we sought to explore how nonblinded detection of secondary outcomes may be affected by ascertainment bias in the BEST-CLI trial. The principal findings of our study are that, both pre- and post-procedure, assessment of cardiac risk and events is significantly more intense for CLI patients scheduled for open surgery compared to endovascular revascularization. Furthermore, vascular surgeons are more likely to perform post-procedure cardiac biomarker assessment than their nonsurgical interventional colleagues.
Pre-procedure practice patterns
We observed that a minority of investigators reported the existence of an institutional algorithm for cardiac risk assessment prior to either surgical or endovascular revascularization. However, both surgical and nonsurgical investigators reported following a personal protocol for preoperative cardiac risk assessment (e.g., cardiac stress tests, cardiac catheterizations, and cardiac biomarker tests) more commonly before open surgical compared to endovascular revascularization. Although vascular surgeons reported obtaining an ECG more frequently before surgical revascularization, pre-surgical cardiac stress testing did not differ by investigator type.
The American Heart Association guidelines recommend additional cardiovascular testing in patients at elevated risk of a perioperative cardiovascular event with poor or unknown functional capacity. 13 One major predictor of such perioperative events is suprainguinal vascular surgery. 14 Although most patients with CLI tend to have infrainguinal and infrapopliteal PAD, 15 many of these patients have poor functional capacity 16 that may prompt clinicians to pursue additional testing, as we observed among BEST-CLI investigators. Various forms of stress testing have been shown to improve risk stratification in patients at elevated risk for a perioperative event; however, no study has shown that interventions (i.e., coronary revascularization) 17 subsequent to such testing improves outcomes. Although uncommon for all specialties, pre-revascularization (both open and endovascular) cardiac catheterization was more frequently reported to be performed on patients cared for by nonsurgical investigators than their surgical colleagues. Current guidelines do not recommend routine coronary angiography and only recommend such testing in the presence of high-risk noninvasive findings that would otherwise prompt angiography irrespective of surgery. 13
Post-procedure practice patterns
No differences were reported in the presence of an institutional or personal standardized algorithm for post-procedure cardiac assessment by investigator-type. However, post-procedure assessment for cardiac complications was more common after open surgery than endovascular revascularization. The most common post-procedure test was the ECG. According to existing guidelines, the utility of postoperative ECG or cardiac biomarkers in patients at high risk for perioperative MI is ‘uncertain’. 13 Although no data exist describing the frequency of ECG testing following open bypass versus endovascular revascularization, our survey findings seem consistent with the common clinical practice of obtaining an ECG following surgery as opposed to minimally invasive therapeutic counterparts.
Vascular surgeons were more likely than nonsurgical interventionalists to order CPK, CPK-MB, or troponin after bypass surgery, and less likely to order ECG but more likely to order troponin after endovascular procedures. The pattern of increased testing following open surgery by vascular surgeons may reflect the possibility that surgeons are more frequently the responding clinicians for patients in the immediate postoperative period following surgical bypass. Nonsurgical investigators may not routinely be involved in the immediate postoperative care of their patients referred for surgical bypass and their responses may be more theoretical than reflective of their clinical practice. The reasons underlying increased utilization of cardiac biomarkers by surgeons following endovascular revascularization are unclear and require further study. Although it is well established that postoperative elevations in cardiac biomarkers is associated with a worse prognosis, 18 in the absence of any demonstrated therapeutic approach to elevated biomarkers in this setting, the benefit of routine biomarker screening remains circumspect.
Study limitations
We acknowledge our study has several limitations. Only 20% of BEST-CLI investigators responded to the survey. However, the relative proportion of investigator specialists who did respond was relatively balanced. Given that survey response rates in the 20% range have been considered adequate in providing a representative sample, particularly when total responses exceed 100, 19 our results are likely to reflect the general practice patterns of the investigators in the BEST-CLI trial. Indeed, the approximate 20% response rate was observed among vascular surgeons and interventional cardiologists who represent the majority of investigators in the BEST-CLI trial. Regarding bias in the ascertainment of outcomes between revascularization strategies, the survey did not collect data on triggers for cardiac evaluation separately for open surgery and endovascular revascularization. Finally, our survey was administered exclusively to investigators in the BEST-CLI trial, which may not reflect the clinical practice patterns of the broader vascular community.
Conclusions
Our findings have important implications for the clinical contextualization of the main results of the BEST-CLI trial, including the possibility that: (a) more intense pre- and post-procedure evaluation for patients with CLI undergoing open revascularization may create the ‘appearance’ of greater baseline procedural risk, in spite of the prospective randomized trial design; and (b) the detection of post-procedure myocardial injury or infarction may be increased among patients cared for by vascular surgeons due to a greater tendency to perform post-procedure troponin testing among these providers. Findings from this study indicate the potential for ascertainment bias in the important secondary endpoint of MI, wherein patients undergoing endovascular revascularization could be diagnosed less frequently with an MI, based on variations in testing that reflect biases and differences in routine clinical practice patterns.
Supplemental Material
sj-docx-2-vmj-10.1177_1358863X21995897 – Supplemental material for Clinical practice patterns and ascertainment bias for cardiovascular events in a randomized trial: A survey of investigators in the BEST-CLI trial
Supplemental material, sj-docx-2-vmj-10.1177_1358863X21995897 for Clinical practice patterns and ascertainment bias for cardiovascular events in a randomized trial: A survey of investigators in the BEST-CLI trial by Mazen S Albaghdadi, Michael N Young, Mohammed M. Chowdhury, Susan Assmann, Taye Hamza, Sandra Siami, Maria Villarreal, Michael Strong, Matthew Menard, Alik Farber and Kenneth Rosenfield in Vascular Medicine
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X21995897 – Supplemental material for Clinical practice patterns and ascertainment bias for cardiovascular events in a randomized trial: A survey of investigators in the BEST-CLI trial
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X21995897 for Clinical practice patterns and ascertainment bias for cardiovascular events in a randomized trial: A survey of investigators in the BEST-CLI trial by Mazen S Albaghdadi, Michael N Young, Mohammed M. Chowdhury, Susan Assmann, Taye Hamza, Sandra Siami, Maria Villarreal, Michael Strong, Matthew Menard, Alik Farber and Kenneth Rosenfield in Vascular Medicine
Footnotes
Acknowledgements
We would like to thank Dr Eric G Campbell PhD, formerly of the Massachusetts General Hospital Institute for Health Policy and now Director of Research at the University of Colorado’s Center for Bioethics and Humanities, for his assistance in developing the survey tool used in this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Institutes of Health (1U01HL107407).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
