Abstract
Early-phase clinical trials in patients with critical limb ischemia (CLI) have shown positive results of mononuclear cell therapy. The current meta-analysis investigated whether cluster of differentiation (CD) 34+ mononuclear cell therapy (CD34+MCT) is effective for no-option CLI. Ten randomized controlled clinical studies of CD34+MCT for no-option CLI with 479 patients were identified and analyzed for pooled results. Compared to control groups, the CD34+MCT was associated with lower total amputation (odds ratio (OR): 0.45, p=0.01; 95% confidence interval (CI): 0.24–0.85) and a higher complete ulcer healing rate (OR: 2.80, p=0.008; 95% CI: 1.31–6.02), but showed no advantage in major amputation (OR: 0.58, p=0.11; 95% CI: 0.29–1.14) and all-cause mortality (OR: 0.82, p=0.62; 95% CI: 0.36–1.83) . Studies with a high CD34+ cell dosage showed significant results in major amputation (OR: 0.38, p=0.002; 95% CI: 0.21–0.70), total amputation (OR: 0.31, p=0.0002; 95% CI: 0.17–0.57) and complete ulcer healing (OR: 7.58, p=0.0005; 95% CI: 2.40–23.88), which were not observed in the low-dose studies. However, inclusion of placebo-controlled studies showed no improvement of the CD34+MCT in total amputation (OR: 0.67, p=0.42; 95% CI: 0.25–1.79), major amputation (OR: 1.31, p=0.43; 95% CI: 0.67–2.54) or complete ulcer healing (OR: 1.52, p=0.27; 95% CI: 0.72–3.21), which were extremely significant in non-placebo-controlled studies (p<0.001). In conclusion, the significant results of CD34+MCT might not support its therapeutic benefit due to high placebo-effect risk and considerable heterogeneity caused by distinct cell doses. More sizable double-blinded, randomized, placebo-controlled trials with higher CD34+ cell dosage are needed in the future.
Keywords
Introduction
Critical limb ischemia (CLI) is the terminal stage of peripheral artery disease (PAD), with a prevalence of 500–1000/million/year, a 1-year mortality rate of 25%, and a 1-year above-ankle amputation rate of 30%.1,2 Bypass surgery and endovascular therapy are currently the main therapeutic methods. 3 However, up to 25% of patients with CLI are not suitable candidates for the main therapies due to distal run-off deficiency or inflammatory factors. Six-month mortality and major amputation rates are as high as 40% and 20%, respectively, in these no-option CLI cases.1,4–6 Atherosclerosis/diabetes mellitus (DM)-related PAD is widely regarded as the main cause of CLI due to its relatively high prevalence. 1 Thromboangiitis obliterans (TAO), on the other hand, is characterized by an invasion of distal small arteries below the knee and a high risk of restenosis after bypass surgery or endovascular therapy, which always leads to no-option CLI, 7 although its prevalence is relatively low.
Early-phase clinical trials of CLI treatment by autologous implantation of mononuclear cells (MNCs) – either from granulocyte colony-stimulating factor mobilized peripheral blood (PB) or bone marrow (BM) – showed improved blood perfusion and limb salvage, 8 which has been discussed in previous systematic reviews.6,9,10 Cluster of differentiation (CD) 34+ (CD34+) cells have been isolated as an endothelial progenitor cell (EPC)-enriched fraction which functions in both vasculogenic and paracrine angiogenic pathways.11–15 The total number of implanted CD34+ cells is considered as a significant long-term prognostic factor for CLI mononuclear cell treatment.16,17 Furthermore, several pilot studies have demonstrated the safety and feasibility and have indicated positive trends on efficacy of purified CD34+ cell therapy in patients with CLI.18–20 In the present meta-analysis of randomized controlled trials (RCTs), we evaluated whether CD34+ mononuclear cell therapy (CD34+MCT) leads to a reduction in amputation rate, all-cause mortality, and rest pain score, and improves ulcer healing, ankle–brachial index (ABI), and transcutaneous oxygen pressure (TcPO2) in CLI patients as compared to control groups. Note that, the CD34+MCT included both unselected BM- or PB-derived MNCs and selected CD34+ cells.
Materials and methods
The meta-analysis was carried out following the recommendations of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 21
Inclusion criteria
The inclusion criteria were as follows. (1) The clinical trial type was an RCT; texts were not limited to English. (2) Patients were diagnosed with CLI (Rutherford 4–6, Fontaine III–IV, or other equivalent clinical scales) 22 and unsuitable for surgical or endovascular interventional vascular reconstruction, with no limit in terms of age, sex, race, or country. (3) Participants had undergone intervention involving unselected autologous BM- or PB-derived MNC or selected CD34+ cell transplantation, with the exact total number of implanted CD34+ cells reported; the control group received a placebo or standard care. (4) Primary outcomes included major/total amputation and all-cause mortality; secondary outcomes included complete ulcer healing, rest pain score, ABI, and TcPO2.
Literature search strategy
A literature search was carried out for RCTs comparing MNC therapy with placebo or standard care in CLI patients in PubMed, Cochrane Library, and Embase medical databases (from 1 January 2000 to 1 August 2017 in any language). We used the following search terms in the PubMed and Embase databases: (“critical limb ischemia” OR “critical limb ischaemia” OR “peripheral artery disease” OR “peripheral arterial disease” OR “peripheral vascular disease” OR “peripheral arteriosclerosis” OR “arteriosclerosis obliterans” OR “peripheral atherosclerosis” OR “atherosclerosis obliterans” OR “thromboangiitis obliterans” OR “Buerger’s disease” OR “Buerger disease”) AND (“cell therapy” OR “stem cell” OR “endothelial progenitor cell” OR “circulating angiogenic cell” OR “outgrowth endothelial cell” OR “hematopoietic stem cell” OR “haematopoietic stem cell” OR “bone marrow cell” OR “bone marrow derived mononuclear cell” OR “peripheral blood derived mononuclear cell” OR “peripheral blood mononuclear cell” OR “mesenchymal stromal cell” OR “mesenchymal stem cell” OR “granulocyte colony stimulating factor” OR “GCS” OR “cluster of differentiation 34” OR “CD34”). The same search strategy was used in Cochrane Library.
Quality assessment and data collection
Two reviewers independently searched the literature and assessed the eligibility of studies for inclusion. They also separately performed quality evaluation and data extraction. A third reviewer made the final decision in cases of disagreement between the two initial reviewers. Data collection included demographic and disease characteristics (sample size, age, sex, comorbidity, main cause of CLI and disease severity), characteristics of intervention (CD34+ cell dosage, administration route, CD34+ cell origin, and follow-up duration), and outcomes (major amputation, total amputation (major and minor amputation), all-cause mortality, number of complete ulcer healing, final value or changed value of rest pain score, ABI and TcPO2). Dichotomous outcomes were extracted in the form of event number with a sample size, while continuous variables were extracted in the form of mean ± standard deviation (SD) with a sample size according to the Cochrane Handbook for Systematic Reviews of Interventions, Version 5.1.0. 23
Statistical analysis
To address possible clinical and methodological heterogeneity between studies, we used the random effects model to synthesize and calculate the effects of included RCTs, despite statistical homogeneity. The combined effects of dichotomous data were represented by odds ratio (OR) and 95% confidence interval (CI), while continuous data were represented by mean difference (MD) or standard mean difference (SMD) plus 95% CI. A two-sided p-value < 0.05 was considered statistically significant. Statistical heterogeneity across studies was estimated using I2 tests, supplemented with χ2 tests. I2 index ≤25%, 50%, and ≥75% presented low, medium, and high inconsistency, respectively, and the heterogeneity was considered significant when a p-value of χ2 was <0.05. Publication bias was assessed with visual inspection of Begg’s funnel plot 24 and quantitatively analyzed with Egger’s test 25 if a sufficient number of trials were available. Subgroup analyses by dosage of implanted CD34+ cells were carried out, and an outcome comparison of placebo and non-placebo trials was performed. All analyses were carried out using Review Manager (RevMan), Version 5.3 (The Nordic Cochrane Centre, Copenhagen, Denmark) and Stata SE 12.0 (StataCorp LP, College Station, TX, USA).
Results
Quality of studies
Results of the literature search are shown in the flow diagram of Figure 1. A total of 11 RCTs were included in the qualitative analysis.26–36 Tateishi-Yuyama et al.’s trial was excluded from quantitative synthesis due to its inappropriate placebo administration using PB-MNCs with low CD34 expression. 26 Skóra et al.’s trial was also excluded because its intervention of BM-MNCs plus vascular endothelial growth factor (VEGF) 165 DNA plasmid intramuscular injection was not limited to cell therapy. 35 All the included trials were designed as two-armed except that Losordo et al. 32 reported a dose-escalation three-armed trial, in which the low-dose and the high-dose CD34+ cell intervention group were independent of each other, sharing the same placebo group. Therefore, nine RCTs (10 studies) were ultimately quantitatively analyzed, wherein the two dosage groups with equally divided control groups of Losordo et al.’s trial were separated into two studies.27–34,36 A total of 479 CLI patients (498 limbs) were included in the meta-analysis, with 234 patients (243 limbs) in the CD34+MCT groups and 245 patients (255 limbs) in the control groups. The sample size ranged from 20 to160 patients, with a follow-up duration of 3–6 months (Table 1).
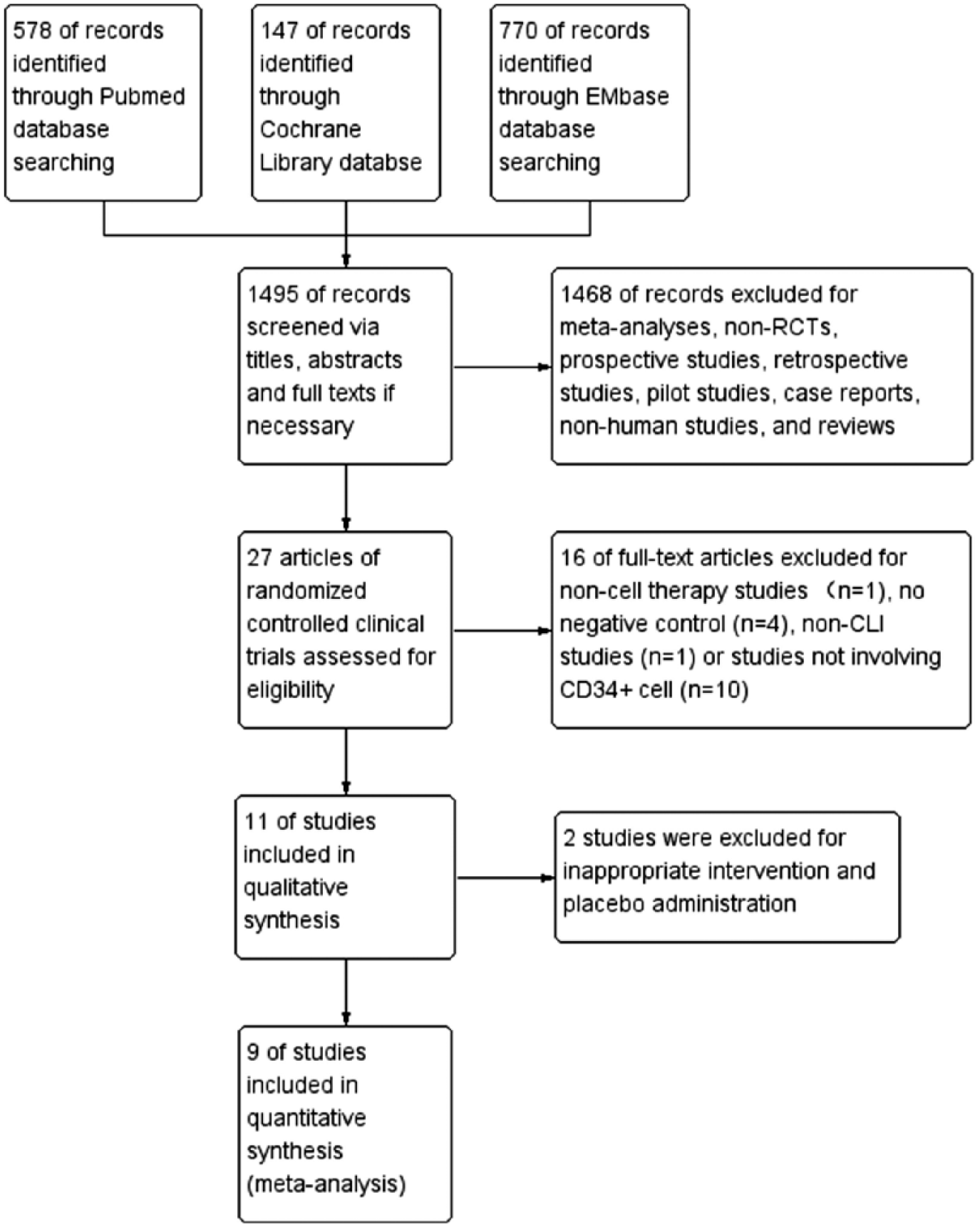
Flow diagram of literature search and study selection. RCT, randomized controlled trial; CLI, critical limb ischemia.
Characteristics of RCTs included in the qualitative analysis.
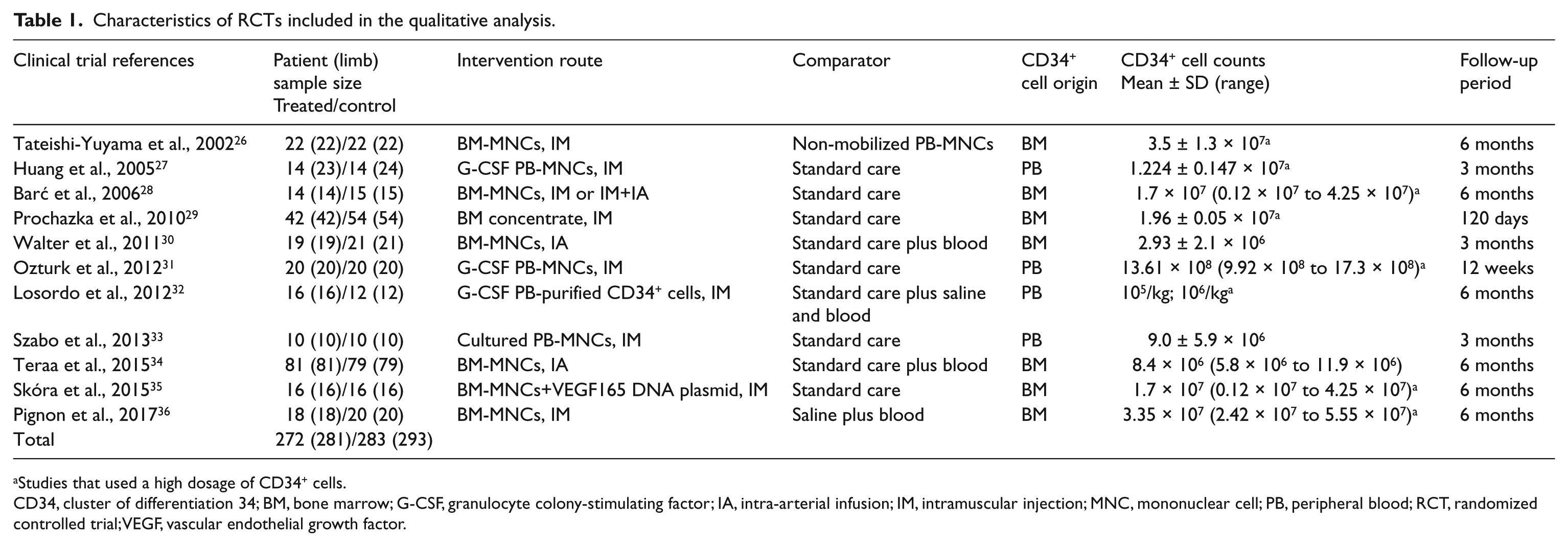
Studies that used a high dosage of CD34+ cells.
CD34, cluster of differentiation 34; BM, bone marrow; G-CSF, granulocyte colony-stimulating factor; IA, intra-arterial infusion; IM, intramuscular injection; MNC, mononuclear cell; PB, peripheral blood; RCT, randomized controlled trial; VEGF, vascular endothelial growth factor.
Patient and disease characteristics
Based on the analyzable data, the mean age of the study population ranged from 60.6 to 72 years, with a median number of 66.7 and the male patients accounted for a greater proportion (median: 70.7%; range: 50–90%) than the females. Most of the included patients had smoking histories (median: 66.6%; range: 10–100%), with DM (median: 57.8%; range: 35–100%), hypertension (median: 83.3%; range: 55–91%), and hyperlipidemia (median: 62.5%; range: 20–91%) as the most frequent comorbidities. Atherosclerosis/DM-related PAD was common and accounted for the main etiology of the study population. Besides, 10 patients in three trials were diagnosed with TAO.30,32,33 Regarding the disease severity, all the patients included in the analysis were scored within the range of Rutherford 4–6 or Fontaine III–IV (Table 2).
Baseline information of RCTs included in the meta-analysis.
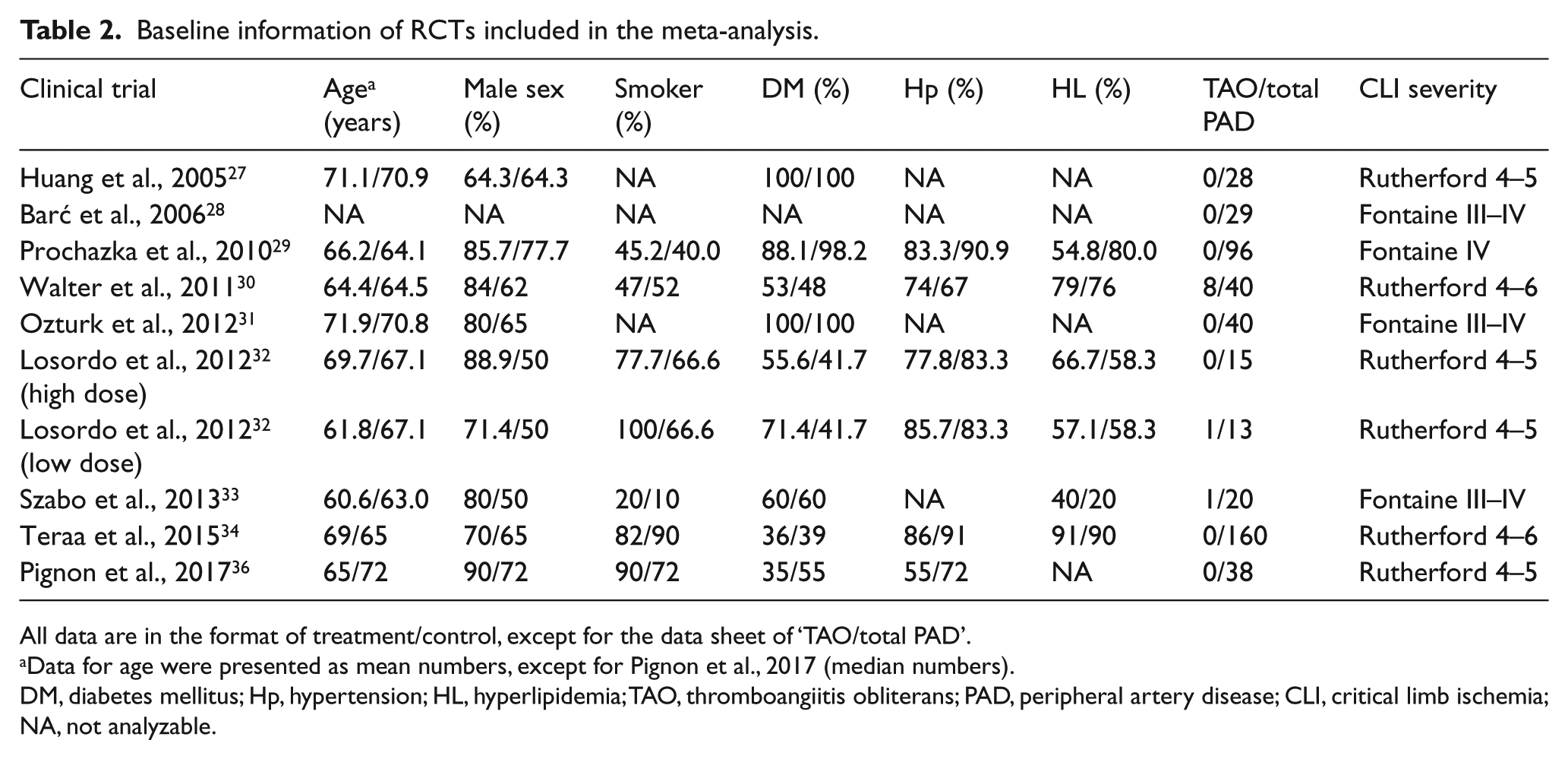
All data are in the format of treatment/control, except for the data sheet of ‘TAO/total PAD’.
Data for age were presented as mean numbers, except for Pignon et al., 2017 (median numbers).
DM, diabetes mellitus; Hp, hypertension; HL, hyperlipidemia; TAO, thromboangiitis obliterans; PAD, peripheral artery disease; CLI, critical limb ischemia; NA, not analyzable.
Characteristics of cell transplantation
The included studies reported mean total implanted CD34+ cell numbers ranging from 2.93×106 to 13.61×108, indicating a large difference of cell dosages across the studies. The cut-off value for subgroup analysis was described as cell dosage of whole body weight. As mentioned in two pilot dose-escalation clinical trials, the low CD34+ cell dosage was defined as 1×105/kg and the high dosage was defined as 1×106/kg.19,20 Hence, 1×107 was chosen as the cut-off value, which was definitely higher than the low dosage and lower than the high dosage. Accordingly, four studies were divided into the low-dose subgroup and six studies were divided into the high-dose subgroup, wherein the two cell dosage groups (1×105/kg and 1×106/kg) of Losordo et al.’s trial were included in the low- and high-dose subgroups, respectively. 32 PB- or BM-MNCs were used in the analyzed studies. Among the five BM-MNC trials, one used concentrated BM-MNCs 29 while the others used MNCs obtained directly from marrow aspirate. Among the four PB-MNC trials, one used dose escalation-purified CD34+ cells, while another used in vitro-expanded autologous PB-MNCs;32,33 the others collected and used the PB-MNCs directly after granulocyte colony-stimulating factor mobilization for 4–5 consecutive days. Routes of cell transplantation were intramuscular injection in six trials, intra-arterial infusion in two trials, and a combination of both in one trial (Table 1).
Outcomes
Primary outcomes
Primary outcomes including major amputation, total amputation (major and minor amputation) and all-cause mortality were extracted from all of the 10 studies. Complete data were measured and reported from each patient who experienced the initial randomization and allocation, except for two patients in Pignon et al.’ s study, wherein one in the BM-MNC group was excluded for difficulty of BM harvest and the other in the placebo group was excluded for a technical failure in cell collection. 36 Since these two events seemed not to correlate with the outcomes and were evenly distributed in two groups, both of the patients were excluded from the meta-analysis with a low risk of bias. Combined results revealed the CD34+MCT groups had a trend toward lower major amputation compared to the control groups but the difference was not statistically significant (OR: 0.58, p=0.11; 95% CI: 0.29–1.14). The CD34+MCT lowered the major amputation significantly relative to the control in the high-dose subgroup (OR: 0.38, p=0.002; 95% CI: 0.21–0.70), while no significant difference was observed in the low-dose subgroup (OR: 1.03, p=0.97; 95% CI: 0.23–4.56) (Figure 2A). On the other hand, the CD34+MCT groups significantly lowered the incidence of total amputation compared to the control groups (OR: 0.45, p=0.01; 95% CI: 0.24–0.85). The high-dose but not low-dose subgroup yielded a statistically significant result (OR: 0.31, p=0.0002; 95% CI: 0.17–0.57 and OR: 0.81, p=0.71; 95% CI: 0.26–2.48, respectively) (Figure 2B). Considering the all-cause mortality, there was no difference between the CD34+MCT intervention and the control groups in the overall (OR: 0.82, p=0.62; 95% CI: 0.36–1.83) or subgroup analysis (high-dose, OR: 0.88, p=0.82; 95% CI: 0.28–2.71 and low-dose, OR: 0.76, p=0.64; 95% CI: 0.24–2.42) (Figure 2C). Sensitivity analysis was performed to evaluate the impact of exclusion of two patients from Pignon et al.’s study. The significance of all the outcomes remained unchanged when adding either two hypothesized negative or positive events to the total sample. Re-evaluation after removing each single study was also performed to test its impact on the overall effect. All the outcomes remained stable except that the overall difference of major amputation between the CD34+MCT and control became statistically significant (OR: 0.44, p=0.01; 95% CI: 0.24–0.84), and I2 dropped from 41% to 13% when Teraa et al.’s study was removed.
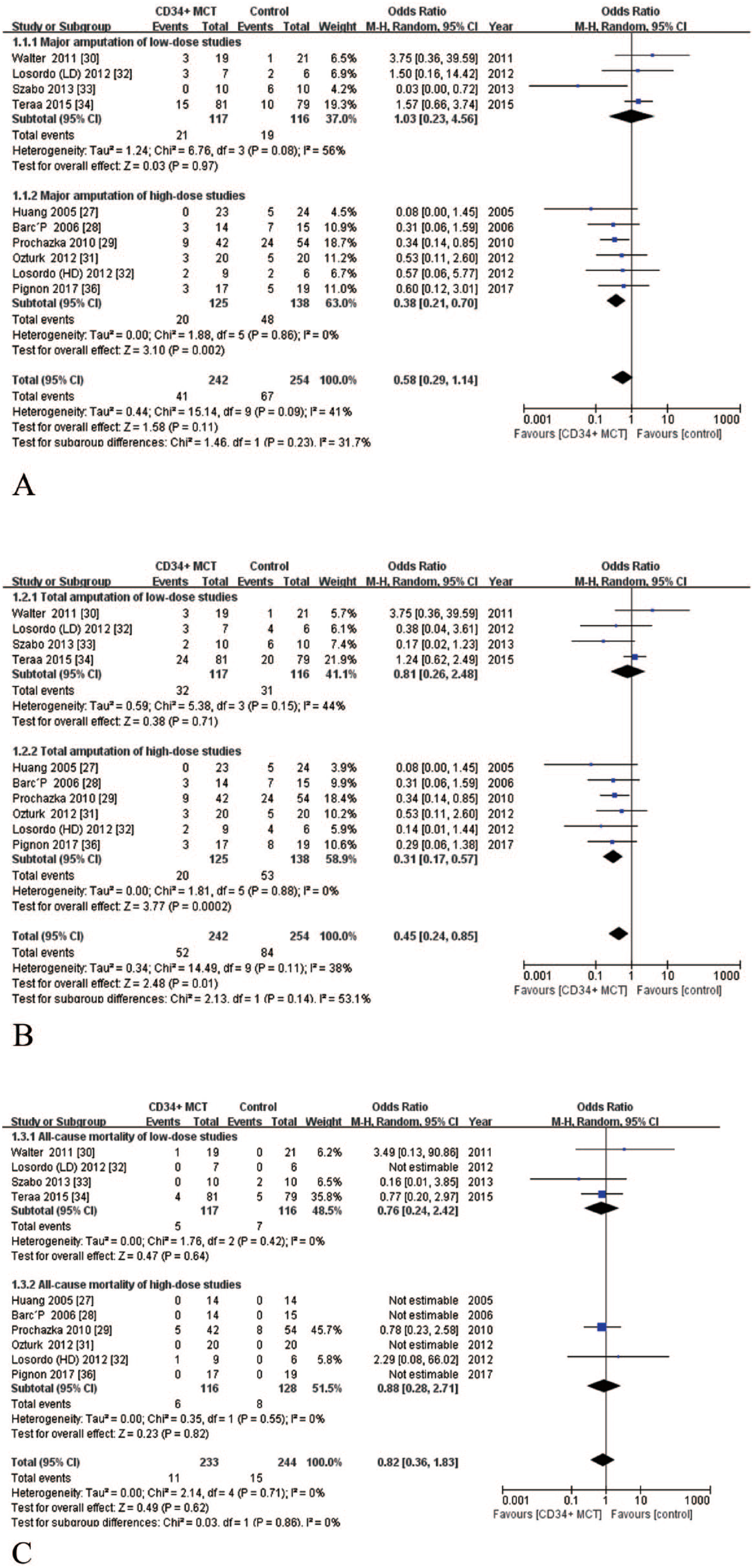
Forest plots showing odds ratios of primary outcomes including (A) major amputation, (B) total amputation, and (C) all-cause mortality.
Secondary outcomes
Eight studies reported the exact number of patients with healed ulcer in both groups with no missing data, while those in the other two studies29,36 were measured but not properly reported. Based on the available studies, we found that the CD34+MCT significantly improved complete ulcer healing compared to the control (OR: 2.80, p=0.008; 95% CI: 1.31–6.02) (Figure 3). In the high-dose subgroup, the CD34+MCT significantly improved ulcer healing relative to the control (OR: 7.58, p=0.0005; 95% CI: 2.40–23.88), which was not observed in the low-dose subgroup (OR: 1.56, p=0.23; 95% CI: 0.76–3.18). The rest pain score was not measured in two studies,29,33 and was presented in an obviously skewed distribution and inappropriate for data conversion in another study. 30 In the remaining seven studies27,28,31,32,34,36, 28% of data were missing and could hardly be estimated. The values of ABI were measured and reported in eight studies,27,30–34,36, four of which presented outcomes in obviously skewed distribution and failed to report the mean with SD or 95% CI.30,32,36 Data conversion was abandoned due to the small sample sizes and a lack of individual data for these studies. Besides, around 35% of the individual data were missing in the remaining four studies reporting the qualified mean with SD.27,31,33,34 As for TcPO2, 30% of the individual data were missing in four studies which measured and properly reported outcomes in both groups,30,31,33,34 while another study failed to measure the data in the control group. 29 Considering the unacceptable high risks of reporting bias and attrition bias, the pooled-analyses of rest pain score, ABI and TcPO2 were aborted.
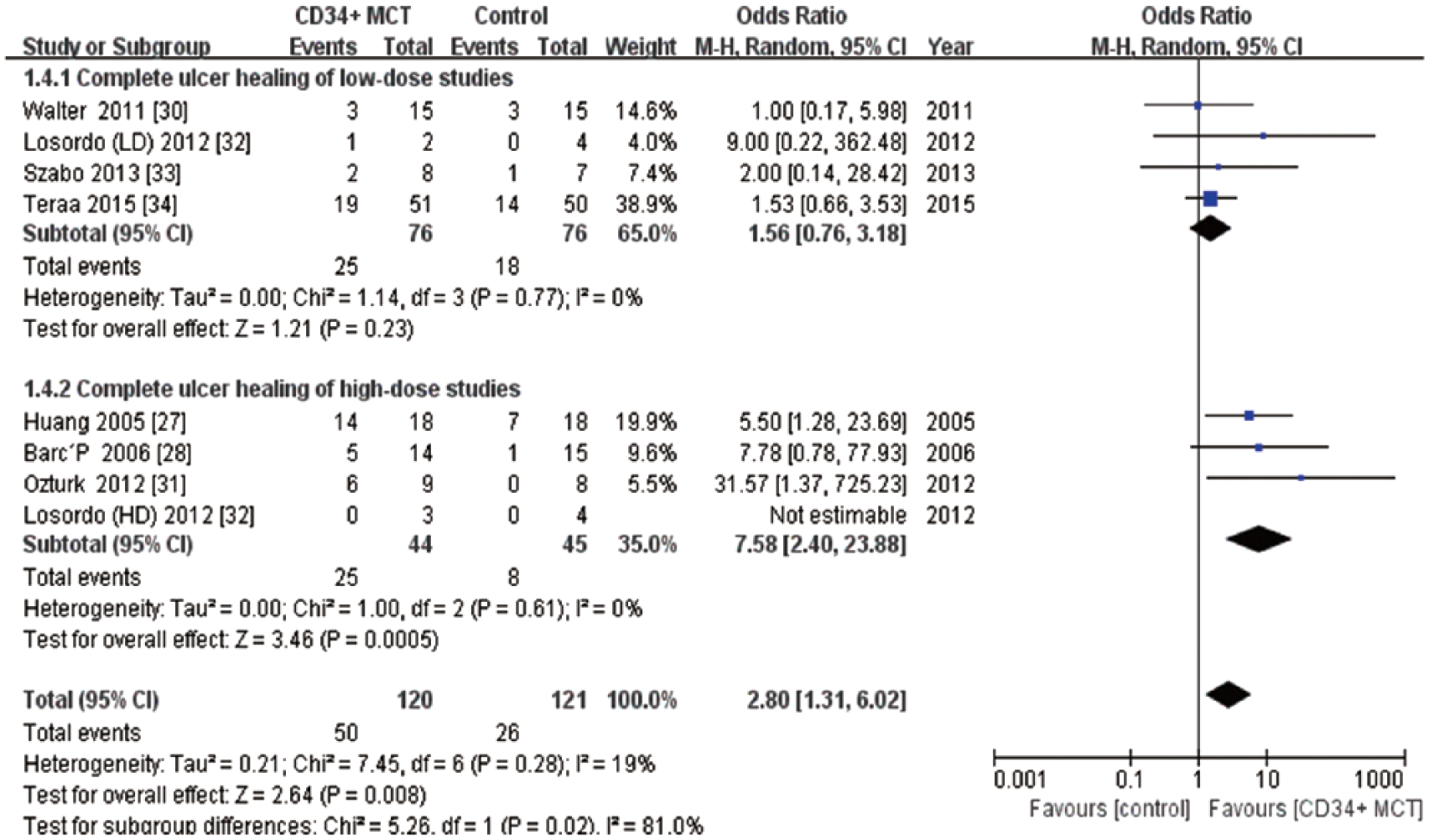
Forest plots showing odds ratios of complete ulcer healing.
Results of placebo and non-placebo controlled studies
Five placebo-controlled studies involved 134 patients in the CD34+MCT groups and 132 patients in the control groups.30,32,34,36 The CD34+MCT showed no statistically significant improvements in major amputation (OR: 1.31, p=0.43; 95% CI: 0.67–2.54), total amputation (OR: 0.67, p=0.42; 95% CI: 0.25–1.79), all-cause mortality (OR: 1.07, p=0.91; 95% CI: 0.33–3.44), and complete ulcer healing (OR: 1.52, p=0.27; 95% CI: 0.72–3.21). As for the non-placebo studies, all the outcomes showed statistically significant differences except for all-cause mortality (Table 3).
A comparison of pooled results between placebo- and non-placebo trials.
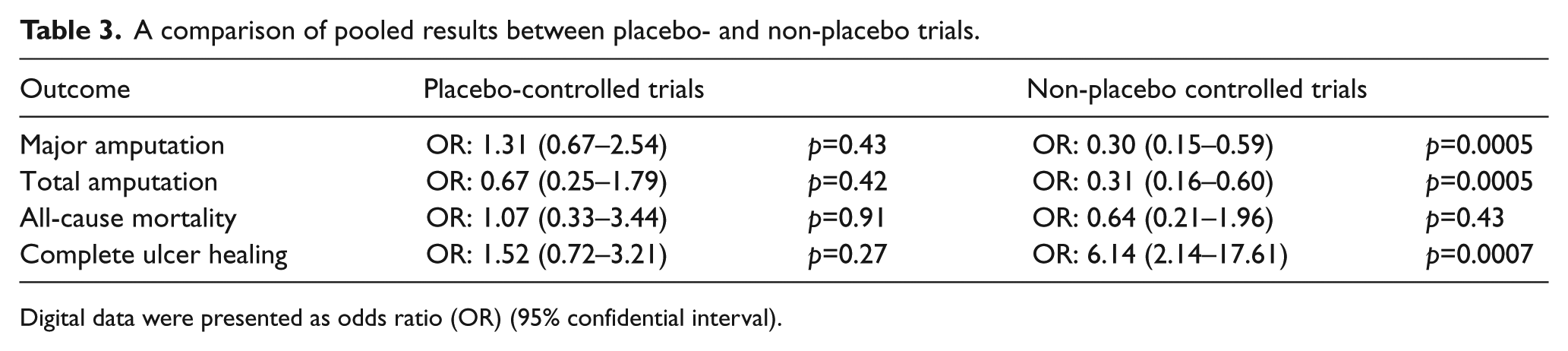
Digital data were presented as odds ratio (OR) (95% confidential interval).
Inter-study heterogeneity and risk of bias
The I2 indexes indicated low risks of inter-study heterogeneity in all-cause mortality and complete ulcer healing, while those in major and total amputation indicated the risks of heterogeneity between low and medium grade (Supplemental Table 1). The risk of bias for each included study was summarized in Figure 4. High risks of performance bias were observed in Ozturk et al.’s study and Szabo et al.’s study for unblinding of both investigators and participants.31,33 Prochazka et al.’s study was considered to be high risk for reporting bias because the researchers failed to measure the outcomes of rest pain score, ABI, and TcPO2, and did not report the ulcer healing number in the control group. 29 High risks of attrition bias were observed in Walter et al., Teraa et al. and Szabo et al.’s study because a considerable amount of individual data for rest pain scores, ABI and TcPO2 were missing.30,33,34 Begg’s funnel plots for major amputation and all-cause mortality were almost symmetrical in shape, while those for total amputation and complete ulcer healing were visually asymmetrical (Figure 5). However, the quantitative analysis with either Begg’s test or Egger’s test detected no statistically significant publication bias in each outcome (Supplemental Figures 1–4 and Table 2).
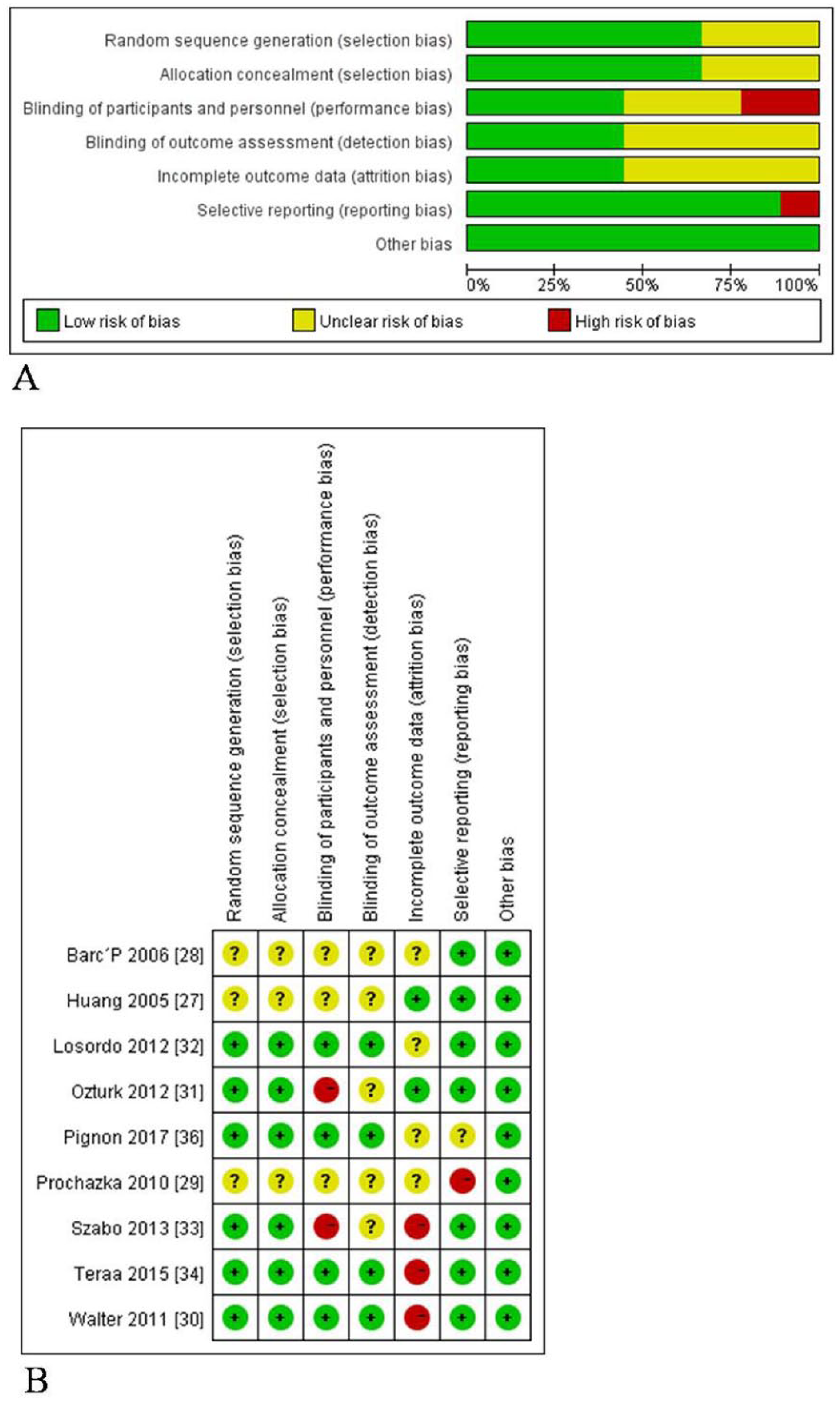
Risk of bias shown as percentages across all included studies (A) and authors’ judgements about risk of bias for each item in each included study (B). (‘+’, low risk; ‘–’, high risk; ‘?’, unclear risk.) (Note: Figure is in color online.)
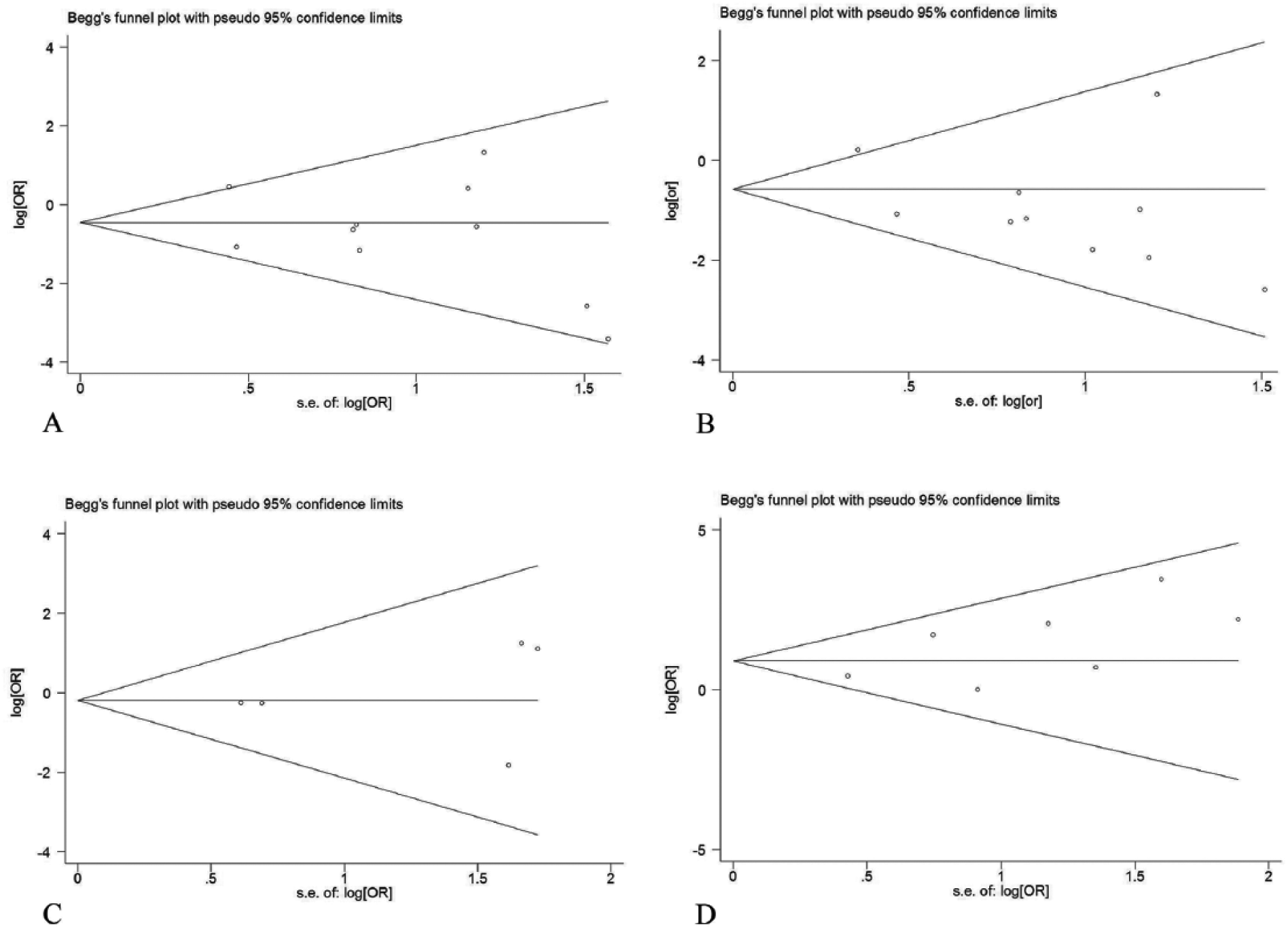
Begg’s funnel plots for the outcomes of (A) major amputation, (B) total amputation, (C) all-cause mortality, and (D) complete ulcer healing. s.e., standard error; OR, odds ratio.
Discussion
CD34+ cells were recognized as a main EPC-enriched fraction in BM- or PB-MNCs and the total CD34+ cell number was used to assess the quality of graft products in the included trials. The combined results of all the included studies revealed that the CD34+MCT was associated with a lower risk of total amputation and a higher rate of complete ulcer healing in patients with no-option CLI. The CD34+MCT groups also had an insignificant trend towards lower major amputation compared to the control, and no between-group difference of all-cause mortality was observed. In the subgroup analysis, a high dosage of CD34+ cells was associated with significant improved major amputation, total amputation, and complete ulcer healing compared to the control groups, which were not observed in the low-dose studies. Noteworthy, the inclusion of only placebo control designed studies showed moderate insignificant differences in all the outcomes, while the results of major/total amputation and ulcer healing were all extremely positive in the non-placebo-controlled trials, which might indicate a placebo effect in the CD34+MCT.
All the included studies were designed as randomized control and should be considered as a high grade of evidence initially, according to the GRADE approach. 37 However, only five studies with 266 patients used placebo as control in a double-blinded way and revealed insignificant combined results of all the outcomes.30,32,34,36 As a contrast, the other five non-placebo-controlled studies, including two open-label trials, showed significant results of major amputation, total amputation, and complete ulcer healing in favor of CD34+MCT.27–29,31,33 Hence, the non-placebo designs and unblinding of researchers or participants might cause a high risk of performance bias and induce a degradation of evidence for the combined effect. 37 Besides, the sample sizes of most of the included studies were relatively low and might not be powered enough to detect superiority of the CD34+MCT. In this context, the qualities of the primary outcomes were considered to be moderate or low grade, although the data for major/total amputation and all-cause mortality were complete and at low risk of attrition bias, reporting bias, and publication bias. As a secondary outcome, complete ulcer healing was considered to be a risk of informative censoring because the status of ulcer was not independent of amputation or death. However, none of the studies took the competing risk of mortality or amputation into the assessment of ulcer healing, which might cause confounding bias. Furthermore, two studies failed to properly report the exact ulcer healing number, which might cause a high risk of reporting bias.29,36 In this context, the combined result of complete ulcer healing was considered to be of low quality for evidence grade.
Several previous meta-analyses included randomized or non-randomized trials to investigate an overall efficacy of stem-cell based treatment for CLI,6,10,38–40 while others focused on autologous BM-derived mononuclear or mesenchymal cells.41–44 Most of them suggested that MNC therapy could bring beneficial efficacy to CLI patients. However, pooled results based on placebo-controlled trials in several analyses did not support the therapeutic efficacy to lower the major amputation rate or improve amputation-free survival.10,40,42,43 It was consistent with our results that the major/total amputation and complete ulcer healing rate were all significantly in favor of the CD34+MCT in the non-placebo-controlled trials and completely insignificant in the placebo-controlled studies. These findings suggested that the control design of placebo versus non-placebo might be a major source of heterogeneity among studies. This point was supported by the sensitivity analysis showing that the combined result of major amputation became significantly in favor of CD34+MCT, with I2 dropping from 41% to 13%, after removing a well-designed, double-blinded, placebo-controlled study with a large sample size (Teraa et al.’s study).
Dosage of implanted cells was assumed as a main factor affecting therapeutic efficacy. Recently, an updated review assessing autologous cell therapy for PAD found a significant inverse correlation between MNC dose and amputation based on BM-MNC studies, 10 implying a dose–response relationship between implanted cells and probability of tissue repair. Nevertheless, a relationship between CD34+ cell dosage and therapeutic efficacy was not observed in previous meta-analyses owing to a lack of high-quality RCTs, although the potency of CD34+ cells for therapeutic neovascularization has been demonstrated in preclinical studies.45–47 Our meta-analysis showed that the studies using a high dose of implanted CD34+ cells (total number >107 cells or >106/kg) achieved more significant results for major amputation, total amputation rate and complete ulcer healing. One included study found that the absolute number of CD45+/CD34+ cells was higher in patients who exhibited complete ulcer healing than in those who did not. 30 Several other studies have also reported better prognosis for amputation and survival at higher CD34+ cell doses.16,17 These findings might be explained by an observation that the CD34+ cell counts were positively correlated to endothelial progenitor cell levels in both BM- and PB-MNCs. 48 Accordingly, the dosage of implanted CD34+ cells was considered a main source of heterogeneity across studies and might positively influence the therapeutic efficacy of MNC for CLI. However, we were unable to draw a clear conclusion on the dose–response relationship because well-designed placebo-controlled trials with purified CD34+ cells were lacking.
Limitations
There were several limitations to the present meta-analysis. First, as mentioned above, the estimates of only the placebo-controlled studies showed much less significant outcomes than the inclusion of all the studies or non-placebo-controlled studies, which might suggest a placebo effect in the pooled results. Moreover, administration of distinct CD34+ cell dosages across studies was considered to be another major source of heterogeneity. Second, although the included studies had similar average age and male sex ratio, suggesting similar demographic characteristics of the study population, clinical heterogeneity might still exist due to various frequencies of comorbidities among studies. Considering the etiology of CLI, TAO differed from other PAD, with regard to epidemiology and pathology. One included study suggested that patients with TAO respond more favorably to BM-MNC cell therapy than those with atherosclerosis or DM, in terms of ulcer healing and alleviation of resting pain. 30 However, only 10 patients were related to TAO in our analysis,30,32,33 which might exert a limited impact on the overall disease background. Third, apart from the CD34+ cell lineage, some other specific cell types might be considered as possible effector fractions in PB or BM, such as CD133+ or c-kit+/CD45– progenitor cells.49,50 Various cell fractions might have overlapping or synergistic mechanisms in neovascularization and tissue repair. The therapeutic effects of cells with different immunophenotypes are yet to be determined. Finally, the inclusion of only RCTs reporting the dosage of implanted CD34+ cells might introduce a selection bias because some non-randomized or non-controlled pilot studies also suggested a positive efficacy for CD34+MCT in limb ischemia. However, the inclusion of these studies would have introduced a greater heterogeneity. In addition, some negative outcomes were likely not published and might cause potential publication bias.
Implications for future clinical trials
Stem cell therapies for CLI tend to be more precise in terms of cell phenotype, dose, and function. CD34+MNC is a main EPC-enriched fraction that should be examined more extensively for their therapeutic potential in NO-CLI treatment. In the present meta-analysis, a paucity of trials with placebo control design and administrations of distinct CD34+ cell dosages might bring notable bias and heterogeneity to the pooled results, limiting the investigation toward the true therapeutic efficacy of CD34+MCT for CLI. Thus, we argue that future trials should use placebo as the control rather than standard care and administrated cell therapies with higher dose of CD34+ cells.
Conclusions
Pooled results of 10 randomized controlled studies with 479 patients in our meta-analysis seemed to imply a potential therapeutic benefit of the CD34+MCT for NO-CLI in terms of limb salvage and ulcer healing. However, these results might not be powered enough to draw a definite therapeutic conclusion due to the relative small sample sizes of most studies and a risk of informative censoring of ulcer status. In addition, subgroup analysis of lower CD34+ cell dosages seemed to achieve much more insignificant results than that of higher CD34+ cell dosages, leading to heterogeneity across the studies. More importantly, estimates of placebo-controlled studies versus non-placebo-controlled studies revealed contradictory outcomes, suggesting a placebo effect in the CD34+MCT. Hence, more well-designed, sizable, double-blinded, randomized, placebo-controlled trials using selected or non-selected MNC with a higher CD34+ cell dose are needed to test the true therapeutic efficacy in the future.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Funds (grant no. 30801122), Shanghai Talent Development Funds (grant no. 2010017), and Excellent Core Member Training Program of Zhongshan Hospital, Fudan University (grant no. 2015ZSYXGG02). The authors declared no commercial, proprietary, or financial interest.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
