Abstract
This study investigated cross-sectional associations of peripheral artery disease (PAD) severity (defined by the ankle–brachial index (ABI)) and amounts of daily sustained physical activity (PA) (defined as > 100 activity counts per minute lasting 5 consecutive minutes or more). This study also investigated associations of amounts of daily sustained PA with 6-minute walk (6MW) distance and the Short Form-36 physical functioning domain (SF-36 PF) score in cross-sectional analyses and with serious adverse events (SAEs) in longitudinal analyses of people with PAD. PA was measured continuously for 10 days using a tri-axial accelerometer at baseline in 277 participants with PAD randomized to the LITE clinical trial. In regression analyses, each 0.15 lower ABI value was associated with a 5.67% decrease in the number of daily bouts of sustained PA (95% CI: 3.85–6.54; p < 0.001). Every additional bout of sustained PA per day was associated with a 4.56-meter greater 6MW distance (95% CI: 2.67–6.46; p < 0.0001), and a 0.81-point improvement in SF-36 PF score (95% CI: 0.34–1.28; p < 0.001). Participants with values of daily bouts of sustained PA below the median had higher rates of SAEs during follow-up, compared to participants above the median (41% vs 24%; p = 0.002). In conclusion, among participants with PAD, lower ABI values were associated with fewer bouts of daily sustained PA. A greater number of bouts of daily sustained PA were associated with better 6MW performance and SF-36 PF score, and, in longitudinal analyses, lower rates of SAEs.
Introduction
Lower-extremity peripheral artery disease (PAD) affects at least 8.5 million men and women in the United States. 1 Among people with PAD, lower physical activity (PA) levels during daily life are associated with worse disease severity and functional performance,2,3 and higher rates of cardiovascular mortality, all-cause mortality, and cardiovascular events.2–6 Prior studies investigating associations of daily PA with health and functional outcomes in people with PAD have evaluated total volume of daily PA such as total steps, blocks walked, or minutes per day.2–6
Recent evidence suggests that daily PA comprised of lower proportions of continuous activity bouts lasting more than 5 minutes is associated with greater mortality risk 7 and reduced functional performance 8 among older adults without PAD. This pattern of sustained PA appeared more strongly associated with mortality risk than total volume of daily PA. 7 Little is known about patterns of daily sustained (uninterrupted) PA in people with PAD, defined as continuous PA of 5 minutes or more without rest. Daily sustained PA may be associated with health and functional outcomes among people with PAD, making it a potential important target for intervention.
Using baseline data from the LITE trial (NCT02538900) 9 , this study investigated cross-sectional associations of participants’ PAD severity (defined by the ankle–brachial index (ABI)) with daily amounts of sustained PA. It was hypothesized that lower ABI values would be associated with fewer bouts of sustained PA per day and fewer total minutes of sustained PA per day. It was further hypothesized that a greater number of bouts and total minutes of sustained PA per day would be cross-sectionally associated with a better 6-minute walk (6MW) performance and health-related quality of life. Finally, using longitudinal data across the 12 months of treatment, we hypothesized that a greater number of bouts and total minutes of sustained PA per day would be associated with lower rates of serious adverse events (SAEs).
Methods
Identification of potential participants
Participants were identified from among all participants randomized into the Low InTensity Exercise Intervention in PAD (LITE) trial, a 1-year randomized clinical trial designed to determine whether a home-based low-intensity walking intervention improved 6MW distance at 12-month follow-up compared to a home-based high-intensity intervention and an attention control group, respectively, among participants with PAD. All participants were followed for 12 months. PA, 6MW, and health-related quality of life data in this report were collected at baseline from participants in the LITE trial, prior to randomization. SAE data in this report were collected during follow-up via monthly telephone calls to study participants. Four medical centers participated (University of Minnesota, University of Pittsburgh, Tulane University, and Northwestern University). Participants provided written informed consent and the Institutional Review Board at each site approved the study.
People with PAD were recruited through newspaper or radio advertisements, physician referrals, or postcards mailed to individuals aged 55 years or older in the cities where the trial took place. In addition, individuals with PAD who had previously participated in research and expressed interest in future research with the principal investigator at the recruitment site were contacted.
Inclusion/exclusion criteria
The inclusion criteria for the LITE trial were an ABI of 0.90 or less, 10 an ABI of 0.90–1.00 with an ABI decline of 20% after a heel-rise test, 11 or evidence of PAD from vascular laboratory or angiographic testing. However, only participants with an ABI < 1.00 were included in these analyses. Participants also had evidence of ischemic leg symptoms, defined by one of the following criteria: (a) ischemic leg symptoms (assessed with the San Diego Claudication Questionnaire 12 ); (b) reported ischemic leg symptoms at the end of the 6MW test or baseline treadmill stress test.
Participants were excluded if they had an amputation below or above the knee, were confined to a wheelchair, had a foot ulcer, or had undergone major surgery or a revascularization procedure during the 3 months prior to baseline testing or was planned in the next 12 months. Potential participants were excluded if they had a New York Heart Association class III or IV heart failure or angina, an abnormal baseline stress test, or a major medical illness including kidney disease requiring dialysis, or lung disease requiring oxygen. Participants treated for cancer within the past 3 years (other than non-melanoma skin cancer) were excluded, unless they had an early-stage cancer for which the prognosis was excellent. Potential participants whose walking was primarily limited by a reason other than lower extremity ischemia were excluded. Patients with a Mini-Mental State Examination score of less than 23 were also excluded. 13
Ankle–brachial index (ABI) measurement
A handheld Doppler probe (Nicolet Pocket-Dop II; Nicolet Biomedical Inc., Golden, CO, USA) was used to obtain systolic pressures in the right and left brachial, dorsalis pedis, and posterior tibial arteries. 14 Each pressure was measured twice. The ABI was calculated in each leg by dividing the mean of the dorsal pedis and posterior tibial pressures in that leg by the mean of the four brachial pressures. 14
Accelerometer-measured physical activity
Free-living PA, defined as PA during daily life, was measured prior to randomization as part of baseline testing for participants in the LITE trial using a GT3X activity monitor (ActiGraph, Pensacola, FL, USA). This device contains a tri-axial accelerometer that processes movement data into 1-minute intervals of activity counts (unitless quantities of movement). 15 The GT3X activity monitor has been widely used in clinical research16,17 and has been shown to be an accurate measure of free-living PA.18,19 Participants were instructed to wear the device on the right side of their beltline below the axilla for 10 consecutive days, removing it only for bathing or sleeping. Participants returned the accelerometer in a stamped, pre-addressed envelope. All wearing time, up to 10 days of PA data, was used in the analyses. ActiGraph data were uploaded using ActiLife software, version 6.13.4 (ActiGraph).
Participants with at least one valid wearing day were included in the analyses. 20 Valid wearing days were defined as ⩾ 8 hours of data in 1 day (00:00–23:59) . To determine active versus sedentary time, each accelerometer minute interval was labelled as ‘active’ if activity counts reached a threshold of 100 or more counts per minute, or as sedentary if activity counts were fewer than 100 counts per minute. This cut-point was commonly used in prior studies21–23 using accelerometry to measure PA in older adults and is based on previous evidence suggesting that sitting and standing typically results in fewer than 100 counts per minute.24–26 Total PA per day was calculated by summing the number of active minutes in each valid wearing day and calculating the mean across the number of valid wearing days for each participant. The number of bouts of sustained PA per day was calculated by summing the number of PA bouts lasting 5 or more consecutive active minutes in each valid wearing day and calculating the mean across the number of valid wearing days for each participant. Total sustained PA per day was calculated by summing the minutes of all PA bouts that lasted 5 or more consecutive active minutes in each valid wearing day and calculating the mean number of sustained PA minutes across the number of valid wearing days for each participant.
Active minutes (⩾ 100 activity counts) were also categorized by intensity (i.e. 100–760 counts/min was categorized as low-intensity activity, 760–2019 counts/min was categorized as moderate-intensity activity, and > 2020 counts/min was categorized as vigorous-intensity activity). Total PA time per day in minutes, the number of bouts of sustained PA per day, and total sustained PA time per day in minutes were also categorized into low, moderate, and vigorous-intensity activity.
Six-minute walk (6MW) test
Baseline 6MW performance data were used for analyses. The 6MW is a well-validated 27 measure of walking endurance in people with PAD and predicts mobility loss, all-cause mortality, and cardiovascular mortality in people with PAD.5,28,29 Participants were asked to walk up and down a 100-foot (30.5 m) hallway for 6 minutes after instruction to cover as much distance as possible. All participants received identical verbal encouragement at each minute during the test via a standardized script read by study coordinators. The total distance covered after 6 minutes was recorded.
Health-related quality of life
Health-related quality of life data were collected at baseline with the Short Form-36 physical functioning domain (SF-36 PF). 30 The SF-36 PF documents quality of life on a scale from 0 (severe limitation) to 100 (no limitation).
Serious adverse events (SAEs)
SAEs reported during the 12-month LITE trial were defined as hospitalizations, death, and new events associated with significant disability (i.e. hip fracture). Deaths were obtained from next of kin or family members or medical records. SAEs were ascertained three ways. First, systematically during monthly adverse event telephone calls in which participants were asked about new hospitalizations or new significant medical events. Second, a questionnaire was administered at 6-month follow-up and at 12-month follow-up visits by study staff to ascertain SAEs. Third, SAEs were ascertained during weekly telephone calls as part of the interventions for both exercise groups and the attention control group. For the purposes of these analyses, three categories of SAEs were defined: (a) all SAEs; (b) cardiovascular SAEs (any ischemic of arrhythmic event; i.e. myocardial infarction, unstable angina, coronary revascularization, coronary artery disease, arrythmias, transient ischemic attacks, stroke, congestive heart failure, or cardiovascular-related death); and (c) lower-extremity SAEs (an SAE due to lower extremity revascularization, critical limb ischemia, or amputation).
Additional measures
Age, sex, self-identified race/ethnicity, smoking history, and comorbid diseases (myocardial infarction, heart failure, stroke, angina, pulmonary disease, cancer, diabetes mellitus, knee or hip arthritis) were collected using questionnaires administered by trained and certified study coordinators. Leg symptoms were characterized using the San Diego claudication questionnaire. 12 Body mass index (BMI) was determined by measuring weight and height and calculating the weight in kilograms divided by height in meters squared.
Statistical analyses
We assessed differences in baseline characteristics and PA measures by ABI categories using analysis of variance and trend tests. For these analyses, each participant’s lowest calculated ABI was categorized in one of five groups: less than 0.40; 0.40 to less than 0.60; 0.60 to less than 0.80; 0.80 to less than 0.90; 0.90 to less than 1.00. We performed Poisson regression analyses to estimate the association between ABI (continuous variable) and the number of bouts of sustained PA per day (count variable). The Poisson regression includes number of days with valid data as an offset. Linear regression analyses were performed to estimate the association between ABI (continuous variable) and total sustained PA time per day, and between ABI (continuous variable) and total PA time per day.
Differences in PA were compared across quartiles of the 6MW distance and SF-36 PF score, using trend tests. Linear regression analyses were also used to estimate the associations between all PA variables of interest and 6MW distance and SF-36 PF score as a continuous variable. All regression analyses were adjusted for age, sex, and race in model 1, and, additionally, BMI, current smoking status, comorbid conditions, and leg symptoms in model 2.
Fisher’s exact tests were performed to determine whether presence versus absence of SAEs reported during the LITE trial differed among participants with baseline PA measures above the median compared to participants with baseline PA measures below the median. All analyses were conducted with SAS software, version 9.4 (SAS Institute, Inc., Cary, NC, USA). The test result was considered significant if p < 0.05.
Results
Characteristics of participants
Among 305 participants with PAD who completed baseline testing for the LITE trial, 293 (96%) completed actigraphy testing, and 287 (94%) had at least one valid wearing day. Of these, 10 were excluded because their baseline ABI was > 1.00, resulting in 277 in the final analyses. Lower ABI values at baseline were associated with lower BMI values, a higher proportion of participants with classical symptoms of intermittent claudication, and shorter 6MW distance. Valid ActiGraph wearing days and total ActiGraph wearing minutes per day were not different across ABI categories (Table 1).
Characteristics of study participants with PAD according to ABI.
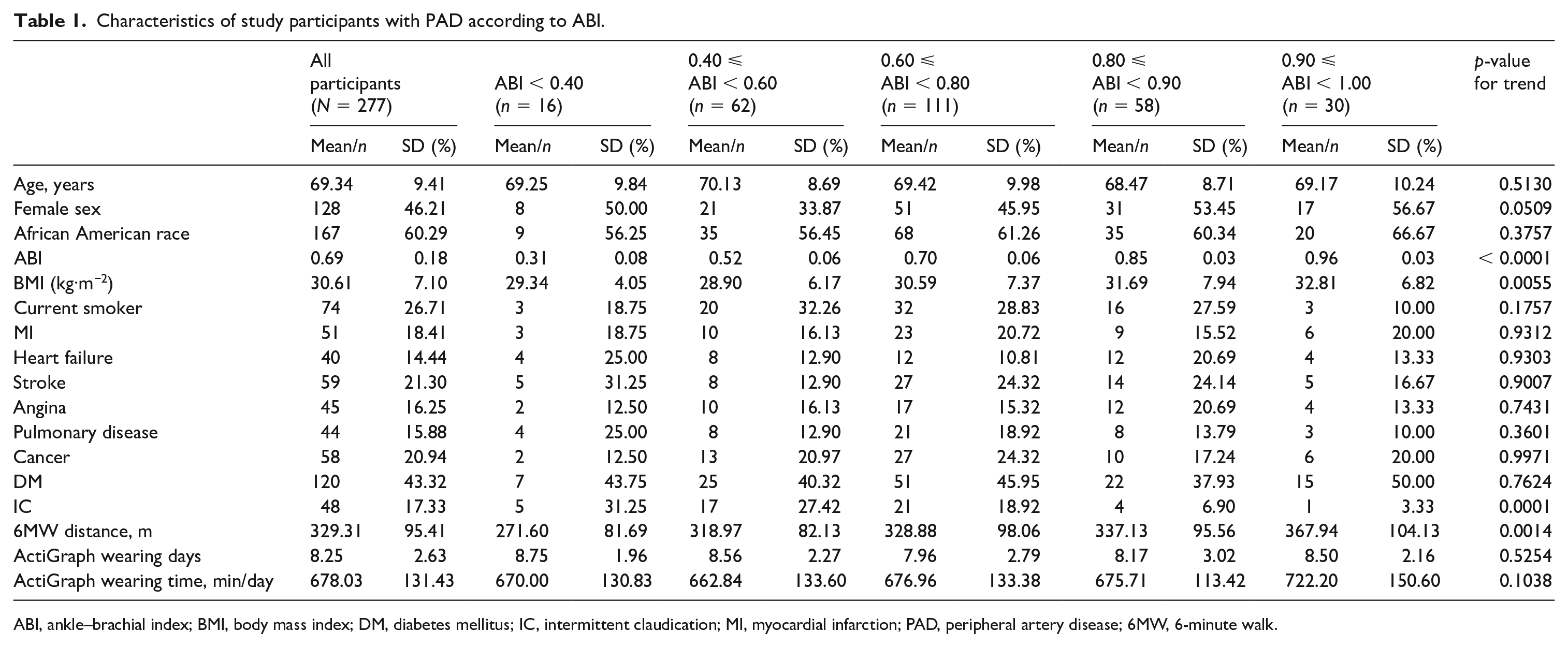
ABI, ankle–brachial index; BMI, body mass index; DM, diabetes mellitus; IC, intermittent claudication; MI, myocardial infarction; PAD, peripheral artery disease; 6MW, 6-minute walk.
ABI and physical activity
Overall, the number of bouts of sustained PA per day was greater in participants with a higher ABI, compared to those with lower ABI (p < 0.0001). Higher ABI values were associated with a greater total minutes of sustained PA per day, compared to lower ABI values (p = 0.02). There were no associations of ABI with total PA minutes per day (Table 2).
Variables of daily PA by ABI category for overall activity and each category of activity intensity.
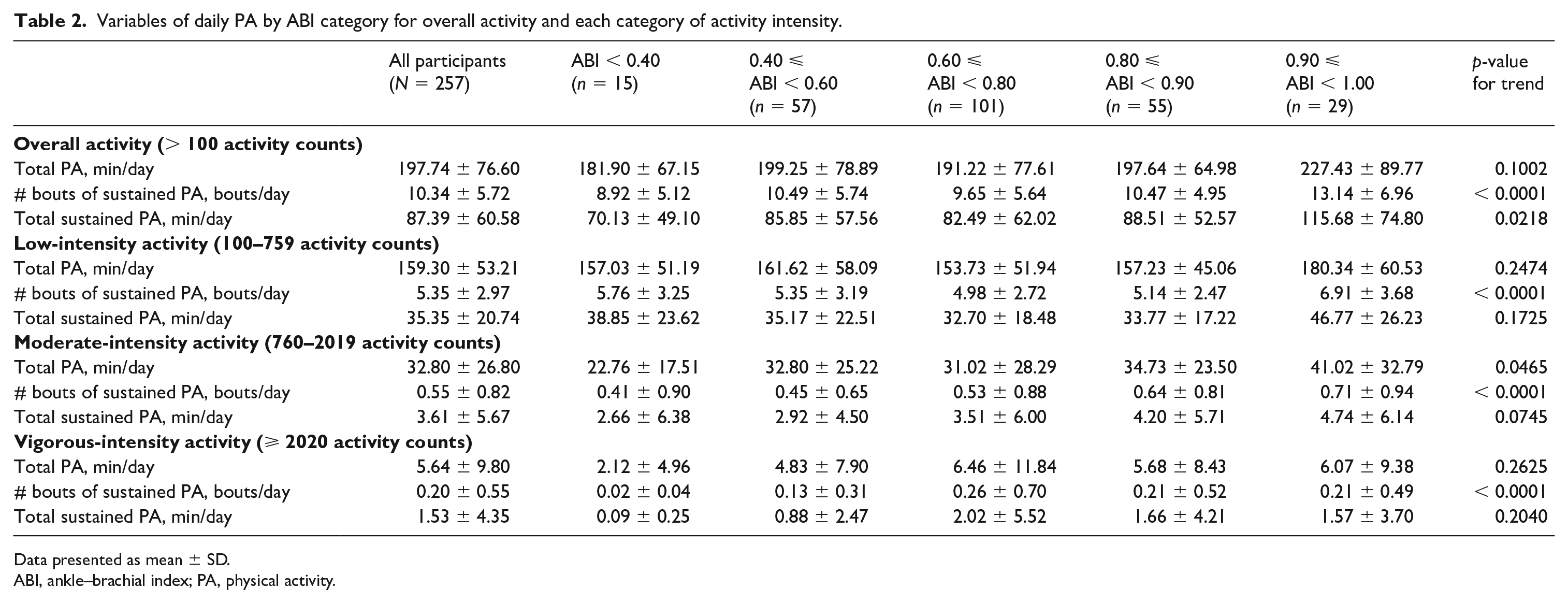
Data presented as mean ± SD.
ABI, ankle–brachial index; PA, physical activity.
After adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, every 0.15 lower ABI value was associated with a 6% lower number of sustained bouts per day (exp(β) = 1.06; 95% CI: 1.04–1.07; p < 0.0001; model 2). In analyses adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, every 0.15 lower ABI value was associated with 6.65 fewer total minutes of sustained PA per day (β = 6.65; 95% CI: 0.87–12.44; p = 0.02; model 2). After adjusting for age, sex, race, BMI, smoking, and comorbidities, there were no associations of ABI with total PA minutes per day (Table 3).
Associations between numerical ABI and variables of daily PA for overall activity and at each category of activity intensity.
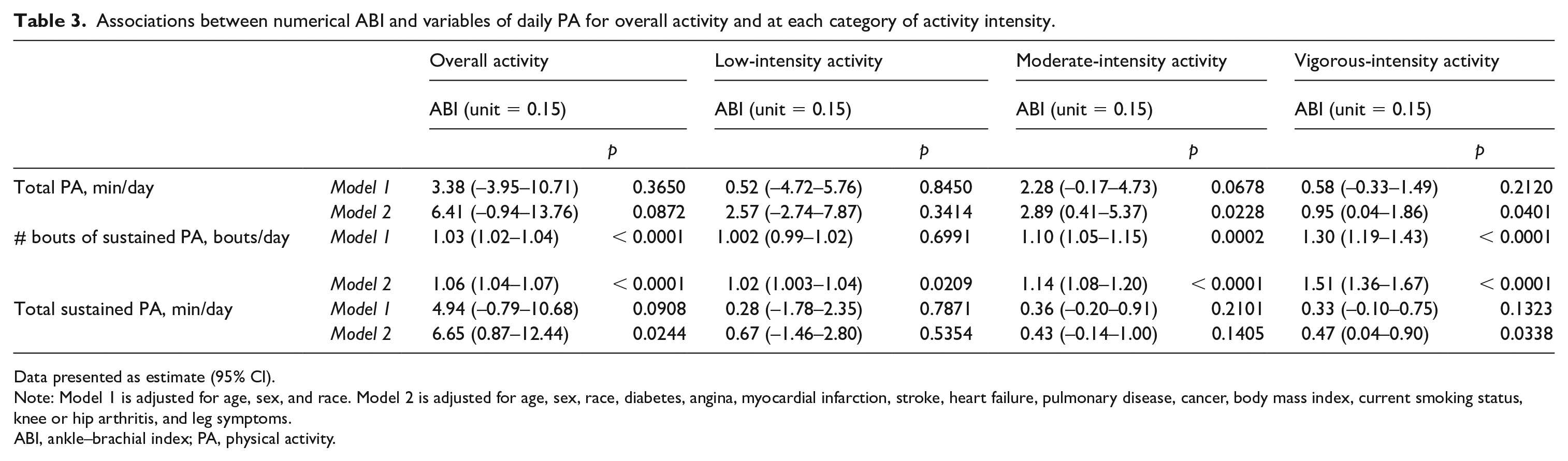
Data presented as estimate (95% CI).
Note: Model 1 is adjusted for age, sex, and race. Model 2 is adjusted for age, sex, race, diabetes, angina, myocardial infarction, stroke, heart failure, pulmonary disease, cancer, body mass index, current smoking status, knee or hip arthritis, and leg symptoms.
ABI, ankle–brachial index; PA, physical activity.
Physical activity and 6MW performance
Compared to fewer sustained PA bouts per day, a greater number of sustained PA bouts per day was associated with better 6MW distance (p < 0.001). A greater number of total minutes of sustained PA per day was associated with better 6MW distance, compared to fewer total minutes of sustained PA per day (p < 0.001). PAD participants with greater total PA per day overall had longer 6MW distances (p < 0.001; Table 4).
Variables of daily PA by 6MW distance quartiles for overall activity and each category of activity intensity.
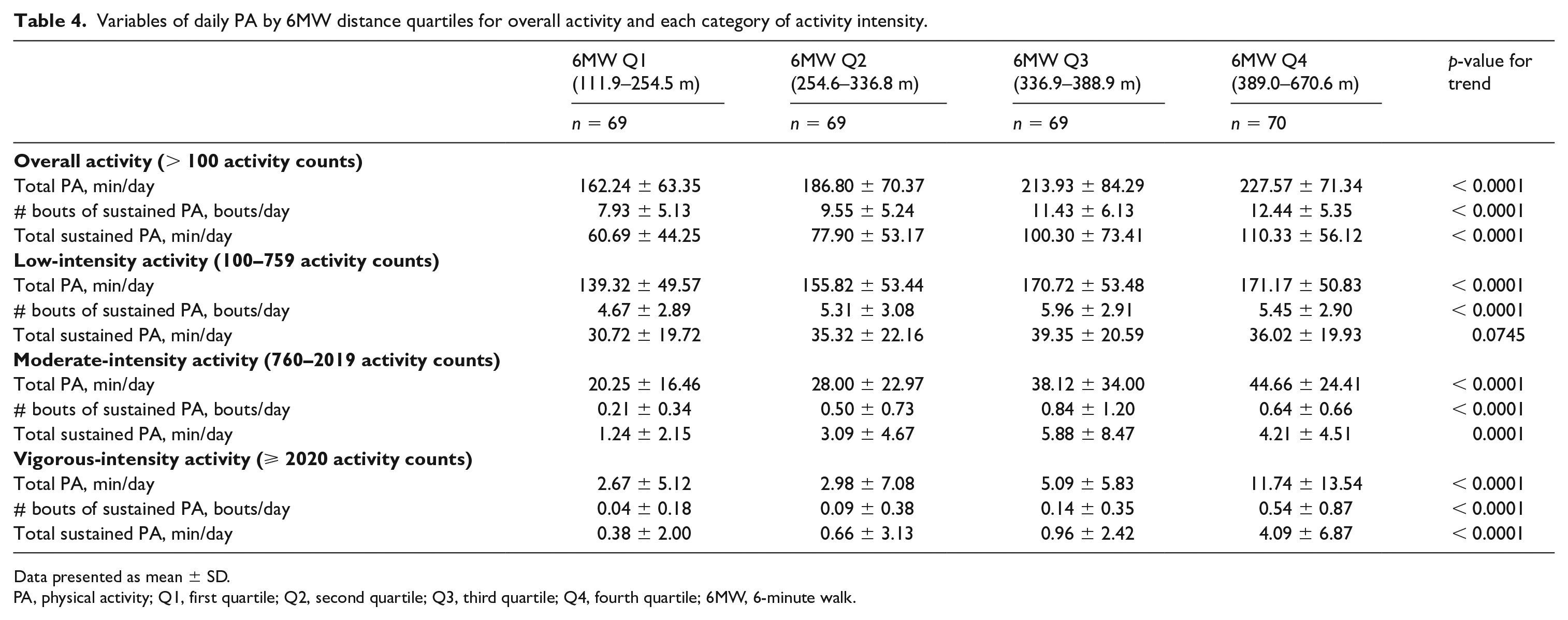
Data presented as mean ± SD.
PA, physical activity; Q1, first quartile; Q2, second quartile; Q3, third quartile; Q4, fourth quartile; 6MW, 6-minute walk.
In analyses adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, every additional bout of sustained PA per day was associated with a 4.56-meter increase in 6MW distance (β = 4.56; 95% CI: 2.67–6.46; p < 0.0001). Adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, every additional 30 minutes of sustained PA per day was associated with a 12.73 higher 6MW distance (β = 12.73; 95% CI: 7.39–18.06; p < 0.0001). In analyses adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, every additional 30 minutes of total PA per day was associated with a 9.90-meter greater 6MW distance (β = 9.90; 95% CI: 5.70–14.4; p < 0.0001; Table 5).
Associations between variables of PA and 6MW distance for overall activity and at each category of activity intensity.
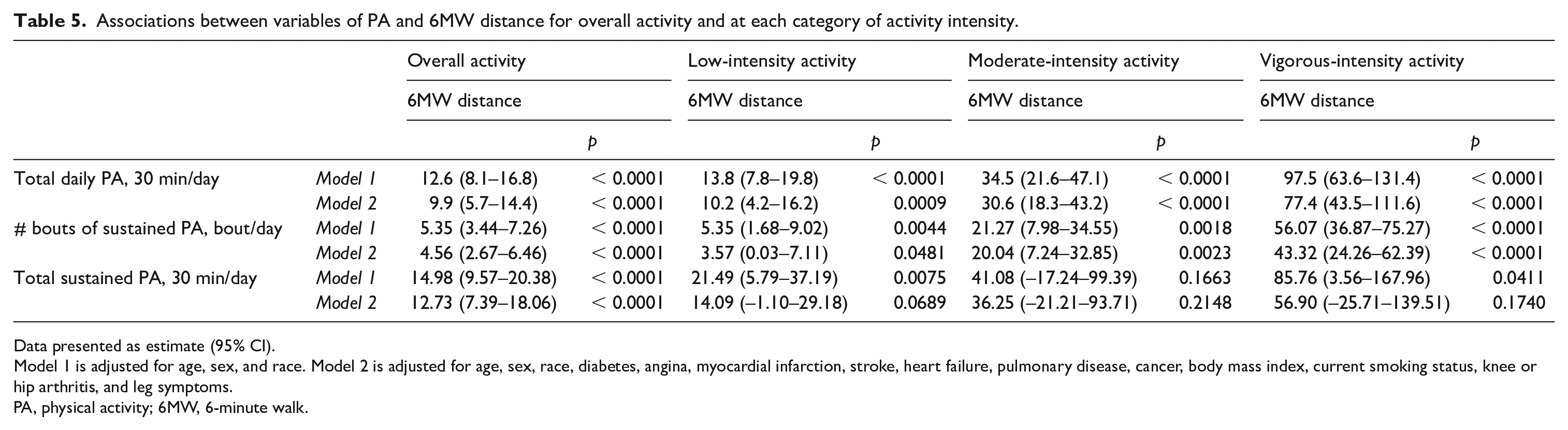
Data presented as estimate (95% CI).
Model 1 is adjusted for age, sex, and race. Model 2 is adjusted for age, sex, race, diabetes, angina, myocardial infarction, stroke, heart failure, pulmonary disease, cancer, body mass index, current smoking status, knee or hip arthritis, and leg symptoms.
PA, physical activity; 6MW, 6-minute walk.
Physical activity and SF-36 PF score
Compared to fewer sustained PA bouts per day, a greater number of sustained PA bouts per day was associated with a better SF-36 PF score (p < 0.001). A greater number of total minutes of sustained PA per day was associated with a better SF-36 PF score, compared to fewer total minutes of sustained PA per day (p < 0.001). PAD participants with greater total PA per day overall had higher SF-36 PF scores (p < 0.001; Supplemental Table 1).
In analyses adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, an additional bout of sustained PA per day was associated with a 0.81-point improvement in SF-36 PF scores (β = 0.81 ; 95% CI: 0.34–1.28; p < 0.001). Adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, an additional 30 minutes of sustained PA per day was associated with a 2.09-point improvement in SF-36 PF scores (β = 2.09; 95% CI: 0.77–3.41; p = 0.002). In analyses adjusting for age, sex, race, BMI, smoking, comorbidities, and leg symptoms, an additional 30 minutes of total PA per day was associated with a 1.95-point improvement in SF-36 scores (β = 1.95; 95% CI: 0.92–2.98; p < 0.001; Supplemental Table 2).
Physical activity and SAEs
Among the 277 PAD participants included in these analyses, there were 159 reported SAEs during a mean follow-up of 6.2 months. Compared to participants with number of sustained bouts per day below the median value of sustained bouts per day, participants with number of sustained bouts per day above the median value had significantly fewer SAEs during the LITE trial (p = 0.002) (Supplemental Table 3). Participants with total minutes of sustained PA per day above the median value of total minutes of sustained PA per day had significantly fewer SAEs during the LITE trial (p = 0.030), compared to participants with total minutes of sustained PA per day below the median value (Supplemental Table 3). There were no associations between total PA per day and SAEs reported during the LITE trial (Supplemental Table 3).
Cardiovascular SAEs
There were no associations between sustained PA per day and cardiovascular SAEs. There was also no association between total PA per day and cardiovascular SAEs (Supplemental Table 3).
Lower extremity SAEs
Compared to participants with total minutes of sustained PA per day below the median value of total minutes of sustained PA per day, participants with total minutes of sustained PA per day above the median had significantly fewer lower extremity SAEs (p = 0.025). There were no associations between number of bouts of sustained PA and total PA per day and lower extremity SAEs (Supplemental Table 3
Discussion
In cross-sectional analyses of participants with PAD, lower ABI values were associated with fewer bouts and fewer total minutes of sustained PA per day. A greater number of bouts and total minutes of sustained PA per day were each associated with greater distance in the 6MW test and better SF-36 PF scores. In longitudinal analyses, those participants with greater amounts of sustained PA per day reported fewer SAEs during the 12-month intervention of the LITE trial. In contrast, among participants with PAD, higher ABI values were not associated with greater total PA per day.
ABI and physical activity
Results reported here demonstrating no association of ABI with total PA per day differ from prior work. For example, McDermott et al. reported that lower ABI values were associated with lower total activity counts after 7 days among 460 people with PAD and 280 without PAD. 2 In a separate study, Sieminski and Gardner reported that in 85 PAD patients, ABI was significantly correlated with free-living PA, measured by accelerometry (r = 0.41, p < 0.001), and that per 0.10 lower ABI value, the decline in free-living daily activity was 42 kcal/day and 612 steps/day. 31
The reason for the lack of association of ABI with total daily PA reported here is unclear. However, many prior studies2,32 reporting an association of ABI with total daily PA typically included participants without PAD (i.e. ABI 1.1–1.5), which may have been responsible for the statistically significant difference in PA across the ABI, since people with PAD have significantly lower PA than those without PAD 2 and differences among people with PAD who have higher versus lower ABI values are smaller in magnitude than differences in PA between people with versus without PAD. 2
This study was not able to discern why lower ABI values were associated with fewer sustained bouts of PA and fewer overall minutes of sustained PA but not with fewer total minutes of PA. However, PAD is specifically characterized by reduced walking endurance since sustained bouts of walking activity result in increasing lower extremity ischemia and associated leg symptoms requiring frequent rest. Prior work shows that lower ABI values are associated with shorter maximal treadmill walking distance and shorter 6MW distance,2,33 consistent with the finding that PAD is associated with fewer bouts and fewer minutes of sustained PA. Interestingly, the current data show that although those with lower ABIs had fewer bouts and fewer minutes of sustained PA than those with higher ABIs, these individuals were able to accrue similar amounts of total daily PA as people with less severe PAD by undergoing more frequent but shorter bouts of PA (i.e. bouts lasting less than 5 minutes) and fewer sustained bouts of PA (i.e. fewer PA bouts lasting at least 5 minutes). Thus, although cross-sectional in nature, these data suggest that the pattern in PA behavior across the day may be critical to clinical outcomes in PAD. Future work should consider holding total PA constant across the data yet distribute the activity differently. 34 This study was also unable to discern why lower ABI values were associated with lower BMI values. It is possible that the association is due to the fact that people with lower ABI were frailer and had lower ABI values.
Physical activity and 6MW performance
Our study shows that a greater number of sustained PA bouts and total minutes of sustained PA per day were associated with a better functional performance. Gommans et al. 35 showed associations between the number of PA bouts per day (irrespective of bout duration) and functional performance. Here, it was reported that in 46 people with PAD, a greater number of PA bouts per day was associated with improved treadmill measurements of performance, but not improved 6MW performance. 35 Other studies have shown associations of lower total PA and impaired functional performance. McDermott et al. reported that lower total activity counts over 7 days were associated with poorer lower extremity performance among 225 people with PAD and 121 without PAD, 36 while Garg et al. reported that in 203 people with PAD, greater total activity counts after 7 days were associated with lower rates of functional decline. 3
It is known that continuous exercise induces physiological adaptations, such as increases in the number of capillaries per muscle fiber and in the number and size of mitochondria in skeletal muscle.37,38 Therefore, bouts of sustained PA in daily life, particularly at vigorous intensity, may induce favorable physiological changes that may contribute to better 6MW distance. However, in this study, data were not available on ischemic leg symptom severity during periods of sustained PA. Furthermore, the finding that even sustained PA at low intensity was associated with better 6MW distance suggests that intensity may not be as important as simply sustaining PA for longer periods before rest. However, further study is needed.
Study limitations
This study has limitations. First, data were cross-sectional, and no causal inferences can be made. Second, this study did not include participants without PAD, which may have reduced the ability to demonstrate a statistically significant association of PA across the ABI. Third, although analyses adjusted for confounding variables, it is possible that statistically significant findings are due to unmeasured or residual confounding. Fourth, some participants had several wearing days that were excluded from the analyses due to incomplete data, and these missing data may have affected the overall results. Finally, results may not be generalizable to people with PAD who were not eligible or interested in the LITE trial.
Conclusion
Among participants with PAD, lower ABI values were associated with fewer bouts of sustained PA per day and fewer total minutes of sustained PA per day. A greater number of bouts and total minutes of daily sustained PA were associated with better 6MW performance and health-related quality of life at baseline and fewer SAEs during follow-up. Future study is needed to determine whether the total number of sustained PA bouts per day and the total minutes of sustained PA per day are associated with outcomes such as mobility loss and mortality in people with PAD.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X21989430 – Supplemental material for Sustained physical activity in peripheral artery disease: Associations with disease severity, functional performance, health-related quality of life, and subsequent serious adverse events in the LITE randomized clinical trial
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X21989430 for Sustained physical activity in peripheral artery disease: Associations with disease severity, functional performance, health-related quality of life, and subsequent serious adverse events in the LITE randomized clinical trial by Joshua T Slysz, W. Jack Rejeski, Diane Treat-Jacobson, Lydia A Bazzano, Daniel E Forman, Todd M Manini, Michael H Criqui, Lu Tian, Lihui Zhao, Dongxue Zhang, Jack M Guralnik, Luigi Ferrucci, Melina R Kibbe, Tamar S Polonsky, Bonnie Spring, Robert Sufit, Christiaan Leeuwenburgh and Mary M McDermott in Vascular Medicine
Footnotes
Declaration of conflicting interests
Dr. McDermott reports a research grant from Regeneron and other research support from Helixmith, Chromadex, Mars, ReserveAge, Hershey, and ArtAssist. The remaining authors have nothing to disclose.
Funding
This study was funded by the National Heart Lung and Blood Institute R01-HL122846. Supported by the American’s Heart Association’s strategically focused vascular research network (18SFRN339700097), National Institute on Aging (intramural division), AG P30AG059988, and by the Jesse Brown VA Medical Center.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
