Abstract
Keywords
Introduction
Right heart thrombi generally fall into two categories, either the mobilization of intravascular thrombi from another source, termed clot in transit (CIT), or the in situ formation of thrombus on a catheter or lead. Both instances are associated with an increased risk of the development of pulmonary embolism (PE). 1 Right heart thrombi occur in 3–18% of cases of acute PE and are associated with an up to 45% mortality rate.2–5
There is no current consensus or data supporting the optimal treatment for right heart thrombus, with treatment options including anticoagulation, systemic or catheter-directed thrombolysis, or surgical removal of the thrombus.6–8 However there is evidence that favors a clot removal strategy, such as surgical thrombectomy or pharmacologic thrombolysis over conservative anticoagulation alone. 9 The risk of bleeding and hemorrhage associated with the use of anticoagulation or thrombolysis and inherent risk of surgical complications in unstable patients has led to efforts to identify minimally invasive percutaneous endovascular treatment options.10,11
The AngioVac System (AngioDynamics, Latham, NY, USA) represents an alternative, minimally invasive approach for the removal of right heart thrombi.10,11 The device is comprised of a large bore venous drainage cannula and extracorporeal venovenous bypass for extracting thrombi from the right heart. Given the relatively larger luminal diameter of the AngioVac’s aspiration cannula (22 F) compared to other percutaneous thrombectomy devices, there is the potential to rapidly remove large volumes of material with decreased risk of fragmentation and distal embolization.
In order to further understand the safety and procedural efficacy of vacuum-assisted thrombectomy using the AngioVac System to remove right heart thrombi, we conducted a subanalysis of the Registry of AngioVac Procedures in Detail (RAPID) multicenter registry. 12
Methods
The RAPID registry was conducted as a prospective multicenter registry to assess the safety and procedural outcomes associated with the use of the AngioVac System (
The AngioVac System is indicated for use as a venous drainage cannula and for removal of fresh, soft thrombi or emboli during extracorporeal bypass for up to 6 hours. All procedures utilized the Generation 2 AngioVac System consisting of a 22 F aspiration cannula and combined with a veno-venous bypass circuit and a reinfusion cannula.13–16 The use of the system required two venous access points using a combination of the femoral or jugular veins, with one for the aspiration cannula and the other for the reperfusion cannula. An extracorporeal bypass circuit was created outside the body consisting of an outflow line, centrifugal pump, a filter, and an inflow line. Once access was obtained, the centrifugal pump was activated creating a one-way flow that provided suction at the tip of the cannula. The device has a balloon-activated tip, which augmented the venous flow to facilitate removal of the thrombus. The circuit reinfused filtered blood back into the body through the reperfusion cannula, minimizing blood loss.
Removal of clot or mass was reported as 0%, 1–50%, 50–70%, and 70–100%, as determined by the physician performing the procedure. Given the heterogeneity of imaging techniques used during thrombus or mass removal procedures (e.g., venography, intravascular ultrasound, transesophageal echocardiogram [TEE]), a semiqualitative metric of 70–100% thrombus or mass removal was decided by the study design committee as the definition of tech-nical success based on the previous use of this criteria inassociation with the AngioVac System. 11 This allowed for the differing imaging modalities used at each site.
Statistical analysis
Descriptive statistical analyses were conducted for adverse events. Quantitative variables were summarized by frequency counts, percentages, means, SDs, minimums and maximums. Categorical variables were summarized by frequencies and percentages. Unless explicitly stated, percentages utilized a denominator corresponding to the number of unique patients who contributed to the endpoint. The statistical analysis was run using Microsoft Excel for Microsoft 365 MSO (16.0.13029.20232).
Results
Patient population
A total of 47 patients out of 234 (20.1%) included in the RAPID registry who underwent AngioVac procedures for the removal of right heart thrombi were included in the present subanalysis. Clinical characteristics of the patient population are listed in Table 1. Mean patient age was 46.7 ± 18.6 years (range 19–98 years). A total of 41 (87.2%) patients had one or more risk factors for venous thromboembolism (VTE). A majority of patients were significantly ill at the time of the procedure as evidenced by their American Society of Anesthesiologists (ASA) scores. This included 10.6% of patients with an ASA score of 5 (moribund patient who is not expected to survive), 29.8% of patients with an ASA score of 4 (severe systemic disease that is a constant threat to life), and 34.0% with an ASA score of 3 (severe systemic disease).
Clinical characteristics for patients enrolled in the RAPID registry with right heart thrombi (n = 47).

Data are presented as n (%) or mean ± SD.
ASA score, American Society of Anesthesiologists score; BMI, body mass index; IVC, inferior vena cava; VTE, venous thromboembolism.
Systemic anticoagulation was being utilized in 95.8% of patients (45 of 47) prior to the AngioVac procedure. This included 36 (76.6%) patients receiving unfractionated heparin (UFH) alone, one (2.1%) patient each receiving UFH in combination with either fondaparinux (Arixtra; GlaxoSmithKline, London, UK) or enoxaparin (Lovenox; Sanofi, Paris, France) (2.1%), three (6.4%) patients receiving enoxaparin alone, three (6.4%) patients receiving argatroban (Acova; Pfizer, New York, NY, USA) alone, and one (2.1%) patient receiving bivalirudin (Angiomax; Baxter, Deerfield, IL, USA). One (2.1%) patient received systemic thrombolysis therapy with alteplase (Activase [tPA]; Genentech, South San Francisco, CA, USA) and underwent catheter-directed thrombolysis with tPA prior to the AngioVac procedure. Systemic thrombolysis therapy was contraindicated for six (12.8%) patients.
Among the patients presenting with right heart thrombi, 42 (89.4%) had thrombi located in the right atrium alone, three (6.4%) in the right ventricle alone, and two (4.3%) in both the right atrium and ventricle. Four (8.5%) patients had concomitant caval thrombi, three (6.4%) also had catheter-related thrombi, and one (2.1%) patient had both caval and catheter-related thrombi in combination with their right heart thrombi.
Procedural data
Table 2 reports the percentage of procedures performed by differing physician specialties. Aspiration cannulas were placed in a femoral vein alone for 20 (42.6%) procedures, the jugular vein only for 20 (42.6%) procedures, directly in the right atrium for one (2.1%) procedure, with six (12.8%) procedures using both the femoral and jugular vein following an intraoperative change of the aspiration cannula. At the discretion of the operator, off-label insertion of the aspiration cannula directly into the open right atrium was performed for one patient. The reinfusion cannula was inserted in a femoral vein alone for 40 (85.1%) procedures, a jugular vein alone for three (6.4%) procedures, the subclavian vein or the femoral artery each for one (2.1%) procedure respectively. Two (4.3%) patients had an intraoperative change of the location of the reperfusion cannula with one procedure using both the femoral and jugular vein and another procedure using two different femoral veins.
Procedural data (n = 47).
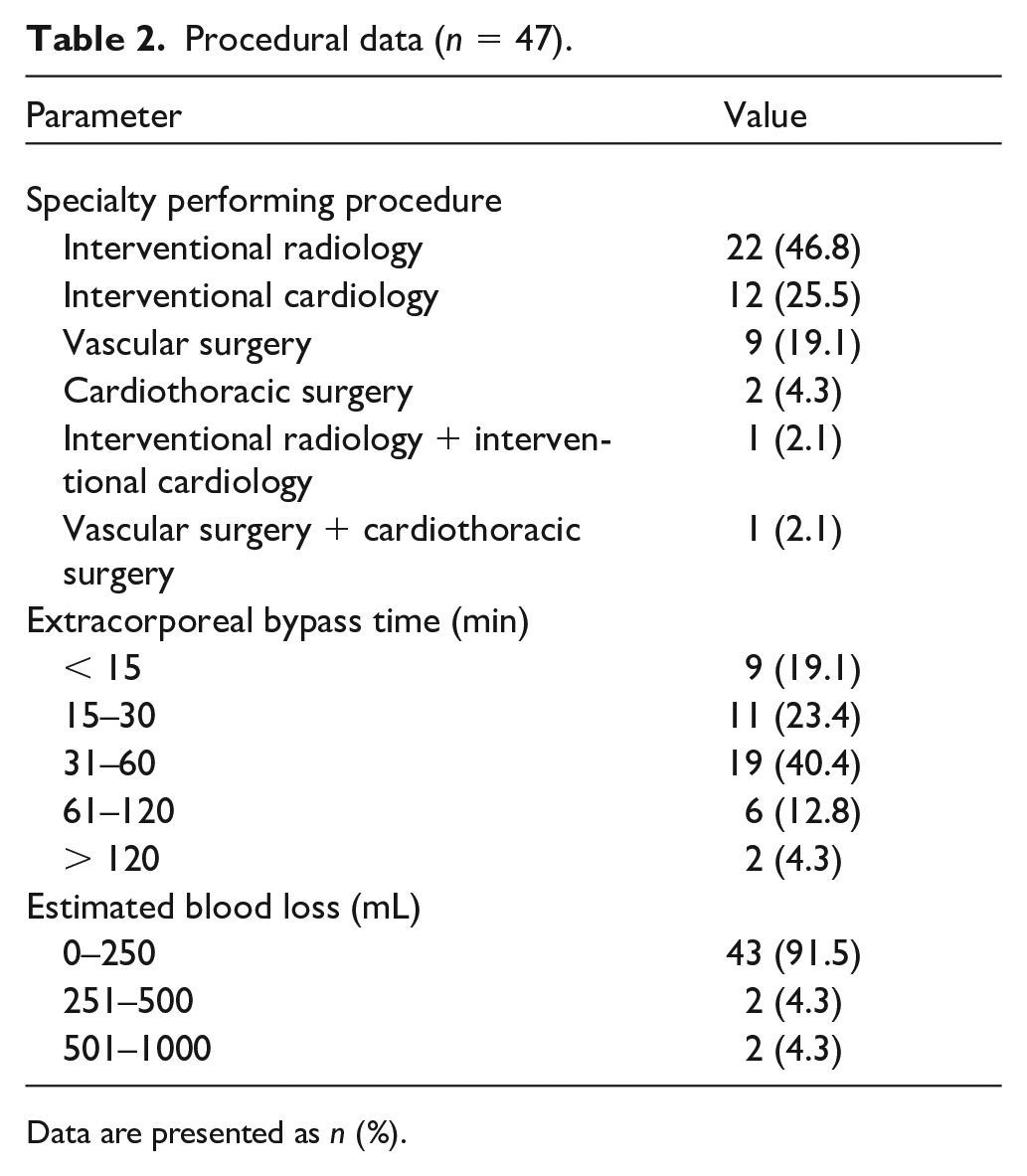
Data are presented as n (%).
The majority of procedures (39 of 47; 83.0%) required less than 1 hour of bypass time (Table 2). Mean fluoroscopy time was 19.3 ± 17.1 minutes, with a mean contrast volume of 23.3 ± 51.9 cc. The majority of patients (91.5%) also had less than 250 cc of estimated blood loss (Table 2). The mean change in hemoglobin was −1.1 ± 0.5 g/dL, decreasing from 10.7 ± 2.2 g/dL preoperatively to 9.6 ± 1.6 g/dL postoperatively. Transfusion of red blood cells (RBCs) was required for eight (17.0%) procedures, with one (2.1%) patient receiving a single unit, six (12.8%) patients receiving 2 units, and one (2.1%) patient receiving 3 units. Systemic anticoagulation was used for all of the procedures, with heparin being the primary anticoagulant used for the majority of patients (43 of 47; 91.5%). Argatroban (Acova; Pfizer) and bivalirudin were used as anticoagulants for three (6.4%) and one (2.1%) procedures, respectively.
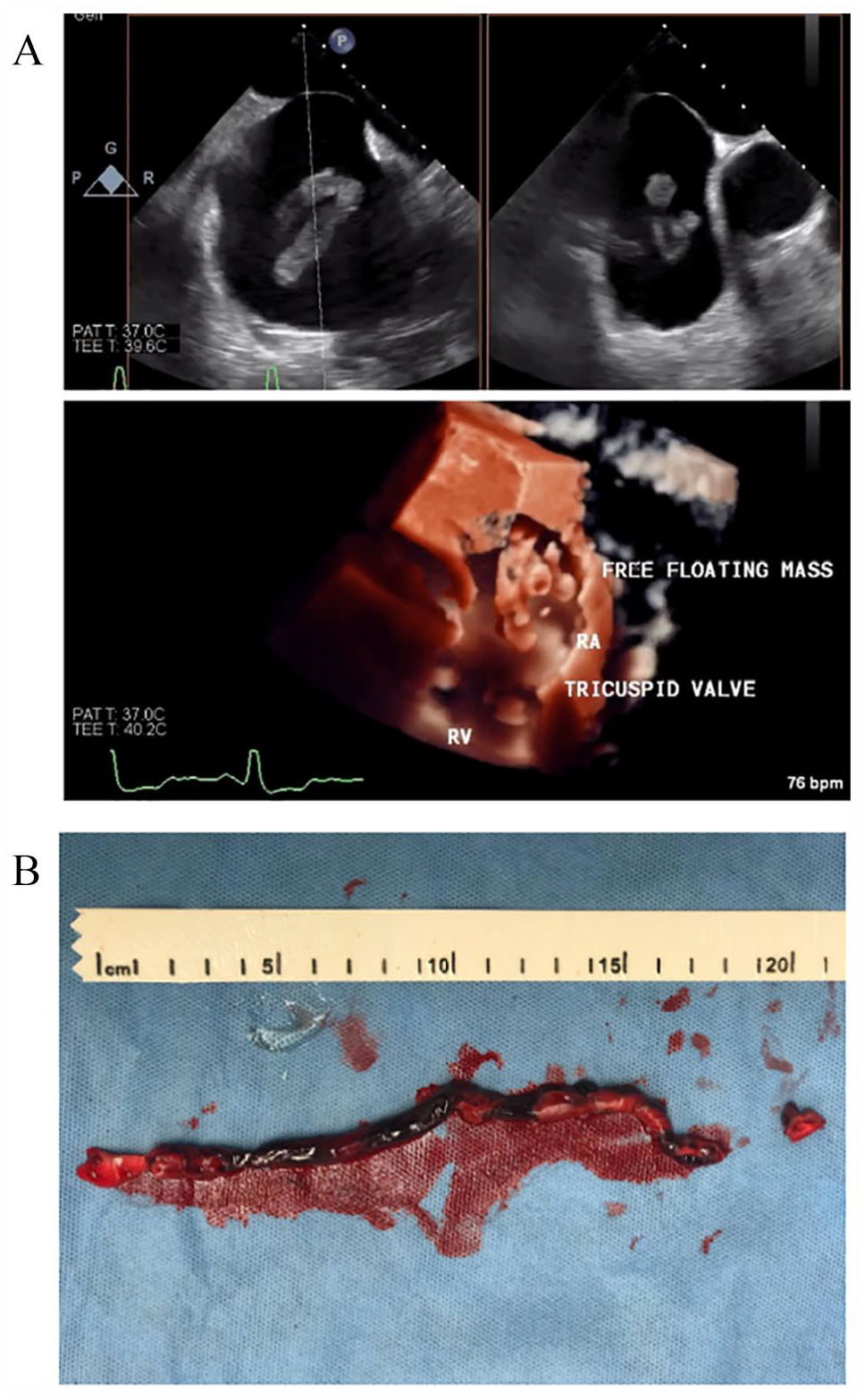
TEE was utilized in conjunction with the AngioVac System during 43 (91.5%) of the procedures (Figure 1). An additional veno-veno or veno-arterial extracorporeal membrane oxygenation (ECMO) circuit was used in four (8.5%) and three (6.4%) procedures, respectively. Adjuvant devices were utilized during 19 (40.4%) of the procedures, with the most commonly utilized adjuvant devices being a snare (21.2% of procedures) or an angioplasty balloon (6.4% of procedures). Placement and removal of inferior vena cava (IVC) filters were each performed during two (4.2%) procedures, respectively.
Procedure outcomes
Seventy to 100% removal of thrombus was achieved in 28 (59.6%) patients following vacuum-assisted thrombectomy. Figure 2 reviews physician-reported ratings of procedural success, with the majority of procedures (80.9%) rated as being somewhat to completely successful.
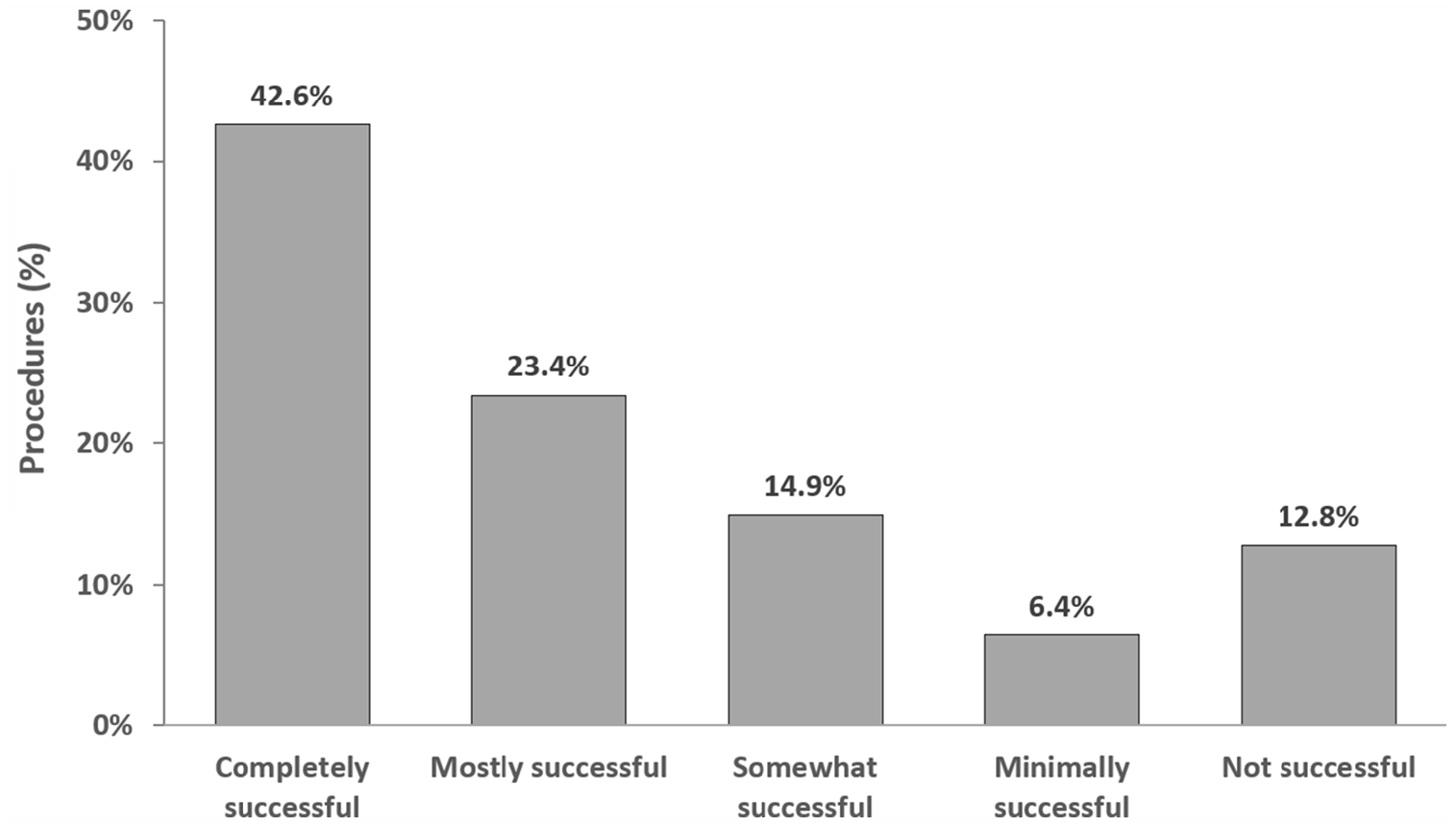
Physician-reported assessment of outcomes following use of AngioVac for right heart thrombi removal.
Adverse events
One patient (2.1%) developed a small hematoma at the reinfusion cannula access site which resolved after compression and no invasive treatment. No other adverse events associated with trauma to the aspiration or reinfusion site were reported. As reported previously, in the analysis of the entire RAPID registry database, 11 one (2.1%) patient experienced a major hemorrhage associated with caval rupture, pericardial effusion, and a substantial decrease in hemoglobin. The patient required a pericardial drain, which was removed the day following the procedure, with the patient discharged on postprocedural day two. One (2.1%) patient experience a minor hemorrhage, which was treated with a transfusion of 1 unit of packed RBCs.
Distal embolization to the pulmonary arteries was identified to have occurred in association with three (6.4%) procedures. All were managed with continued anticoagulation, and none required additional mechanical, aspirational, or surgical therapy. A single patient of the three remained intubated until the fourth postoperative day, when they were extubated and discharged in a stable condition.
There was one (2.1%) patient death, which was not deemed to be procedure related. This occurred in a 43-year-old female with a recent heart-liver transplant who developed a right ventricular thrombus and stroke with the family electing to withdraw care due to the patient’s heart failure.
Discussion
Despite being rare, CIT represents a significant clinical complication associated with venous thromboembolism with an associated mortality rate of up to 100% if left untreated. 7 The prevalence of CIT in association with PE has been shown to range from 3% to 18%, with a higher rate in patients with massive PE and are hemodynamically unstable.2,3 This high rate of mortality necessitates the rapid and immediate removal of right heart thrombi to reduce the risk of PE and the associated morbidity and mortality.
Although there are a number of options for reducing the risk associated with the embolization of right heart thrombi, including new pharmacotherapeutic therapies, endovascular devices, and surgical techniques, there is no clear consensus for the treatment of CIT.6,7,17,18 Differing clinical characteristics upon patient presentation, the presence of comorbidities, and specific procedural-related risk can also influence treatment approach. Since the surgical management of cardiac thrombi is associated with significant blood loss and high rates of morbidity and mortality, the use of fibrinolytic therapy to treat CIT has increased. Unfortunately, the use of fibrinolytic therapy is associated with high rates of major and clinically relevant bleeding.19–21 Hence, the ability to endovascularly aspirate thrombus without exposing the patient to either open surgery or thrombolytic medications is a potentially transformative therapeutic option.
The RAPID registry is a prospective, multicenter registry which collected safety and procedural outcome data associated with the real-world application of the AngioVac System for the treatment of various clinical pathologies. The present subanalysis of data from this registry focusing on 47 patients with right heart thrombi represents the largest reported experience with the device reported to date in this patient population. Unlike previous reports associated with the use of the AngioVac System across a range of uses including removal of right heart thrombi, the registry enabled the ability to obtain and analyze select safety and procedural outcomes data specific to this patient population.
Use of the AngioVac System and outcomes for right heart thrombus removal has been previously reported.10,11 Al Badri et al. described the percutaneous removal of right heart thrombus using vacuum aspiration in seven patients undergoing right atrial thrombectomy. 10 Two of the patients in their series were diagnosed with submassive PE. The procedure was successful for six (85.7%) patients; treatment was successful with the remaining patient who developed cardiogenic shock requiring brief extracorporeal membranous oxygenation. The authors reported no device or procedure-related complications. Additionally, no patients experienced a postprocedural drop in hematocrit or required a transfusion. They concluded that vacuum-assisted thrombectomy was a potential treatment option for hemodynamically stable patients who were not surgical candidates who had right-sided intracardiac thrombus. For patients with pulmonary emboli in the presence of right heart thrombi, vacuum-assisted thrombectomy can be used as an adjuvant to the management of the PE.
Donaldson et al. retrospectively reported on 15 procedures performed with the use of the AngioVac System. 16 Eleven (73.3%) of these cases were for the removal of an intracardiac mass located in the right atrium, with three procedures also involving a mass from the right ventricle. Complete removal of the mass was achieved in 11 of the 15 (73.3%) procedures. The authors did not specify the percentage of these successful procedures performed in patients presenting with cardiac masses. No dissections or perforations of the vasculature occurred during the procedures. Eleven patients (73.3%) had a postprocedure hematocrit drop, with six (54.5%) patients requiring a postoperative transfusion.
The present analysis suggests that vacuum-assisted thrombectomy for the treatment of CIT has a low rate of adverse events and procedural complications. The ability to maximize clot removal on an emergent basis with the device could potentially reduce the risk of PE and avoid potential complications which contribute to increase morbidity, mortality, and healthcare costs.
Although the use of the AngioVac System for VTE has previously been shown to result in a decrease in postprocedural hemoglobin and the need for blood transfusions,16,22 estimated blood loss and use of transfusions appear to be less when the device is used for the removal of right heart thrombi. The percentage of patients requiring less than 1 hour of bypass time was similar between patients being treated for removal of right heart thrombi compared to the other patients in the Rapid registry (82.9% vs 78.9%), but there was a much higher percentage of patients with an estimated blood loss of less than 250 cc (91.5% vs 69.3%), and a reduced need for transfusions by almost 50% (12.8% vs 25.2%) in the right heart thrombi group. There were no significant differences in the rates of hematoma formation, hemorrhage or distal embolization between the subset of patients being treated for removal of right heart thrombi in the RAPID registry versus the entire RAPID registry population. Overall, with the AngioVac System, we believe that the large bore aspiration cannula assists in the en bloc removal of the material and the simultaneous reinfusion can minimize blood loss during the procedure and the need for transfusions.
Study limitations
There are some limitations to the present subanalysis. This includes the lack of a control population where alternative approaches or therapies were utilized during the same time interval and the lack of rigorous monitoring compared to a randomized controlled study with the potential for an underreporting of adverse events. Also, since the decision to utilize the AngioVac System was dependent upon each individual physician and site, there is a potential for selection bias for use of the device during the study period. Another limitation was the lack of detailed information on some clinical parameters and patient characteristics as a part of the RAPID registry and the patient’s clinical course prior to the use of the AngioVac System. There was also no attempt to collect information on the clinical course of patients who did not achieve procedural success or postdischarge outcomes data. These elements may have an impact on the utility of the device in this setting. Lastly, although the presence of right heart thrombi increases the risk of PE, there were no patients with concomitant PE included in the present analysis.
Conclusion
The ideal treatment approach to patients with right heart thrombus is not yet defined due to the lack of data. A subanalysis of data from the RAPID registry demonstrated both the safety and clinical utility of the AngioVac System relative to treating patients with right heart thrombi. Although the present data represent the largest series of cases performed to date with the device for the removal of right heart thrombi, further studies are needed to identify longer-term outcomes associated with the use of vacuum-assisted thrombectomy in this patient population.
Footnotes
Acknowledgements
The institutions and associated investigators who participated in the RAPID registry are as follows: UCLA Medical Center (John Moriarty, Adam Plotnik), Eastern Virginia Medical School/Sentara Healthcare (David Dexter), University of Southern California (David Shavelle), University of Colorado (Paul J Rochon), Allegheny General Hospital (Mithun Chakravarthy, David Lasorda), Washington University (Mohamed Zayed, Gerald Fortuna, John Ohman), Cedars Sinai Medical Center (Danny Ramzy), Franciscan Health (Charles Kiell), Montefiore Hospital (Yosef Golowa, Jacob Cynamon), Swedish Heart and Vascular Institute (Eric Lehr), Ochsner Clinic (Zola N’Dandu, Stephen Jenkins), Charleston Area Medical Center (Christopher Adams), Northwestern University (Kush Desai, Daniel Schimmel), Mt Sinai Medical Center (Aaron Fischman, Robert Lookstein, Rahul Patel), Mayo Clinic (Harladur Bjarnason), Mt Carmel Hospital (Noah Jones), Greenville Memorial Hospital (Bruce Gray), Integris Health (Bryan Cogar, Jeffrey Sparling), Baptist Health (John Craig), Central Iowa Hospital (Nicholas Southard), and Oklahoma Heart Institute (Eugene Ichinos). Medical writing assistance was provided by Larry Yost from The Atticus Group, LLC (Portsmouth, NH, USA).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: John M Moriarty has received personal fees from AngioDynamics, Boston Scientific, and Thrombolex. Grace Hyun J Kim reports receiving a research consulting fee from MedQIA. Kush Desai has received personal fees from W.L. Gore, Medtronic, Becton Dickinson, Boston Scientific, Cook Medical, Penumbra, Tactile Medical, Cordis, Shockwave Medical, and Asahi Intecc.
Funding
Funding for the RAPID registry, including data collection, monitoring statistical analysis, and medical writing, was provided by AngioDynamics, Inc. (Latham, NY, USA).
