Abstract
Introduction:
Acute, laboratory-based bouts of prolonged sitting attenuate lower-limb arterial endothelial-dependent vasodilation. However, the impact of habitual sedentary patterns on popliteal artery endothelial health is unclear. We tested the hypothesis that greater habitual total sedentary time, more time spent in prolonged sedentary bouts, and fewer sedentary breaks would be associated with worse popliteal flow-mediated dilation (FMD) responses.
Methods:
This cross-sectional study used 98 healthy participants (19–77 years, 53 females) that wore an activPAL monitor on the thigh for 6.4 ± 0.8 days to objectively measure sedentary activity and completed a popliteal ultrasound assessment to determine FMD. Both relative (%baseline diameter) and absolute (mm) FMD were calculated. Using bivariate correlation and multiple regression analyses, we examined if there were relationships between sedentary outcomes and FMD while statistically controlling for any potential confounders.
Results:
In the multiple regression model, age (p = 0.006, β = −0.030, 95% CI = −0.051, −0.009) and total time in sedentary bouts > 1 hour (p = 0.031, β = −0.005, 95% CI = −0.009, −0.001) were independent predictors of relative FMD. Age (β = −0.002, 95% CI = −0.003, −0.001), mean blood flow (β = 0.013, 95% CI = 0.002, 0.024), moderate-intensity physical activity (β = 155.9E−5, 95% CI = 22.4E−5, 289.4E−5), sedentary breaks (β = 0.036, 95% CI = 0.007, 0.066), and total time spent in sedentary bouts > 1 hour (β = −25.02E−5, 95% CI = −47.67E−5, −2.378E−5) were predictors of absolute FMD (all, p < 0.047). All independent outcomes remained significant after partially controlling for all other predictor variables (all, p < 0.031).
Conclusions:
Habitual prolonged sedentary bouts and sedentary breaks, but not total sedentary time, were predictors of popliteal endothelial-dependent vasodilatory function. The patterns by which sedentary time is accumulated may be more important than the total sedentary time on lower-limb arterial health.
Introduction
A predominant independent risk factor for peripheral artery disease is the excessive engagement in sedentary behaviours,1,2 defined as any waking behaviour characterized by an energy expenditure ⩽ 1.5 metabolic equivalents while in a sitting, reclining, or lying posture. 3 Existing research in laboratory settings have demonstrated that acute bouts of prolonged sitting (i.e., ⩾ 3 h) attenuate lower-limb FMD responses.4–6 Furthermore, Thosar et al. 5 demonstrated that periodically breaking up an uninterrupted bout of sitting preserved FMD. Although these studies have provided insight into the vascular implications following a single bout of sitting in lab-based settings only, our understanding regarding the impact of habitual sedentary time or patterns on popliteal endothelial health in a free-living environment is unknown. The purpose of this study was to determine whether sedentary time and patterns measured in a free-living environment were associated with popliteal endothelial-dependent vasodilatory function. It was hypothesized that greater total habitual sedentary time, an increased number of prolonged sedentary bouts, and fewer sedentary breaks would be associated with lower popliteal FMD responses.
Methods
Participants
Based on a moderate effect size (f2 = 0.2) and eight predictor variables, a sample size calculation estimated that 84 participants were needed using a multiple regression model assuming a two-tailed, α = 0.05 and β = 80% power (G*Power, v3.1). 7 To minimize the effects of fluctuating hormonal levels, naturally menstruating younger females were tested 1–5 days following the start of menstruation, and those using oral contraceptives were assessed during the placebo pill phase. Postmenopausal females were not using hormonal replacement therapy. Participants’ activPAL™ data have been previously presented. 8 However, the current purpose and statistical analyses were independent from this previous report, which did not examine peripheral vascular function. Prior to testing, verbal and written informed consent were acquired. All protocols and procedures conformed to the Declaration of Helsinki and were approved by the Dalhousie University Health Sciences Research Ethics Board.
Habitual activity monitoring
The FMD assessments (see below) were completed in a thermoneutral environment (21°C), 6 hours postprandially, after participants had refrained from vigorous physical activity for 24 hours and caffeine and alcohol for 12 hours. Participants were equipped with an activPAL inclinometer (Pal Technologies Ltd, Glasgow, UK), a valid and reliable measure of habitual sedentary patterns and physical activity. 9 Using standardized procedures, the activPAL was waterproofed and secured using Tegaderm™ transparent medical dressing (3M, London, ON, Canada) to the midline of their right anterior thigh. 10 Participants wore the activPAL 24 hours per day for a minimum of 5 days (6.4 ± 0.8 d).10–12
The activPAL data were analyzed using a customized LabVIEW program (LabVIEW 2018; National Instruments, Austin, TX, USA) that estimated waking hours and summarized daily averages of waking hours spent in sedentary postures. Since FMD responses are attenuated following 1 hour of sitting, 5 we defined prolonged sedentary bouts as ⩾ 1 hour in duration. Participants self-reported their waking hours to accommodate activPAL analysis. Sedentary breaks were calculated per waking hour. Physical activity intensity was determined using step rate thresholds for younger 13 and older adults. 14 All habitual sedentary and physical activity data were analyzed by a researcher blinded to participant popliteal outcomes. Our analysis program has previously demonstrated excellent inter-observer reliability. 8
Systemic hemodynamics
Heart rate (HR) was determined from a lead II electrocardiogram configuration. Beat-by-beat systolic (SBP) and diastolic blood pressure (DBP) were measured via finger photoplethysmography (Portapres®; Finapres Medical Systems, Amsterdam, The Netherlands). The Portapres height correction unit accounted for deviations in the vertical distance between the heart and the pressure cuff. Intermittent brachial SBP and DBP were determined using an automated vital signs monitor (Carescape V100; General Electric Healthcare, Mississauga, ON, Canada) to calibrate the Portapres waveform (Figure 1). The electrocardiogram and Portapres waveforms were sampled at 1000 Hz and 200 Hz, respectively, using a PowerLab data acquisition system (PL3508 PowerLab 8/53; ADInstruments, Sydney, Australia). LabChart software (Version 8; ADInstruments) was used to view recorded signals in real-time and for offline analysis. At least 5 minutes of supine data were averaged to represent resting systemic hemodynamic outcomes. Mean arterial pressure (MAP) was calculated using the equation: ⅓SBP + ⅔DBP.
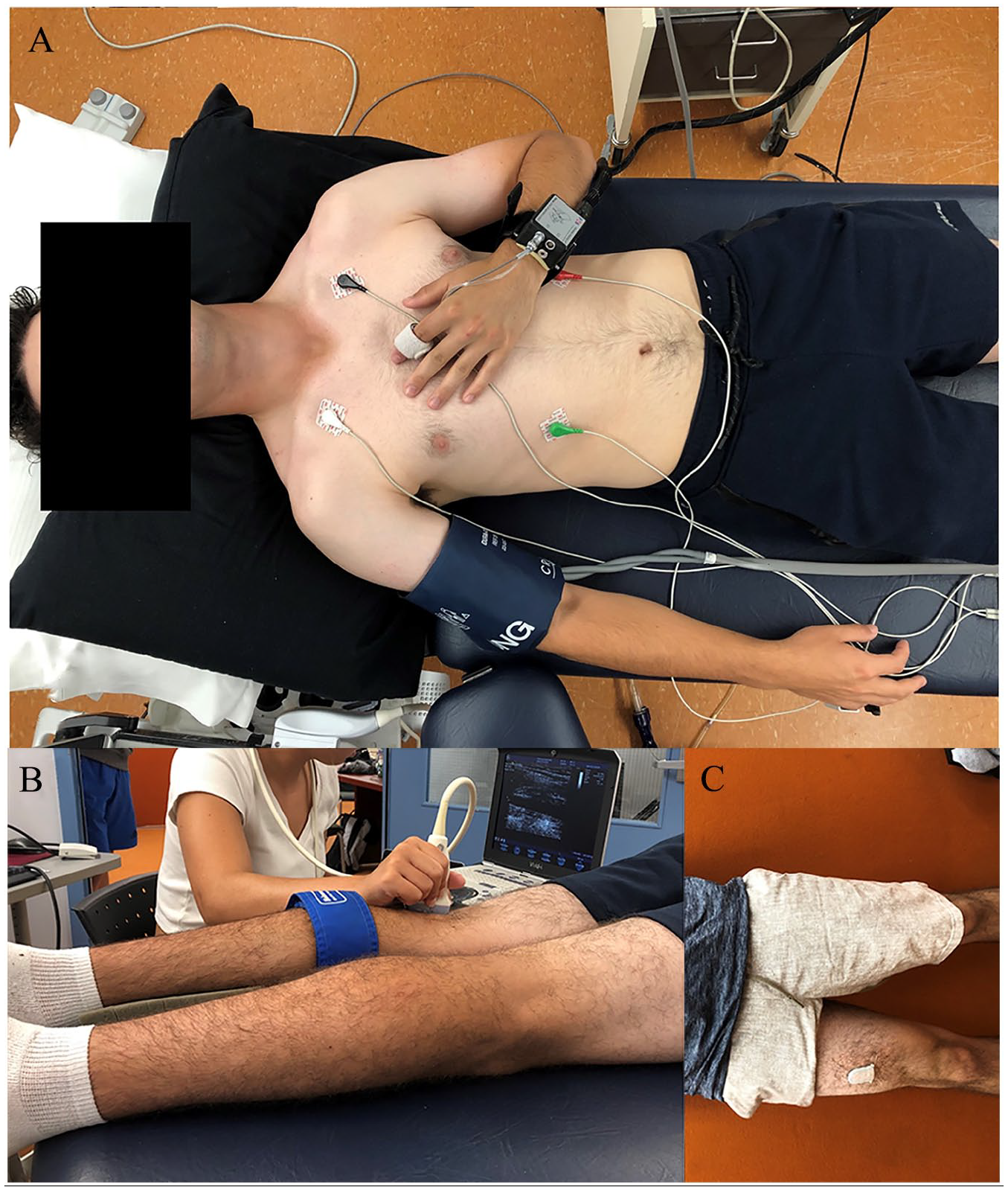
Hemodynamic data collection was performed in the supine position and featured a lead II electrocardiogram configuration, finger photoplethysmography (Portapres; Finapres Medical Systems, Amsterdam, The Netherlands), and automated brachial blood pressure assessments (Carescape V100; General Electric Healthcare, Mississauga, ON, Canada) (A). With participants in the prone position, a pressure cuff was secured around the widest circumference of the calf. The endothelial-dependent vasodilatory function was assessed via duplex ultrasonography using a 12-MHz multifrequency linear array probe (Vivid i, General Electric Healthcare) (B). Participants were equipped with a waterproofed activPAL inclinometer on the midline of their right anterior thigh using Tegaderm medical dressing to quantify sedentary and physical activity patterns (C).
Popliteal artery endothelial-dependent assessments
Popliteal assessments were conducted following published guidelines. 15 With participants in the prone position, a pressure cuff was secured around the widest circumference of the calf and attached to a rapid inflation system (E20 and AG101; Hokanson, Bellevue, WA, USA). The cuff was inflated to the supra-systolic pressure of 250 mmHg for 5 minutes to ensure distal ischemia was maintained. Endothelial-dependent vasodilatory function was assessed via duplex ultrasonography using a 12-MHz multifrequency linear array probe (Vivid i; General Electric Healthcare). The left popliteal artery was imaged proximal to the bifurcation at, or slightly above, the popliteal fossa by experienced operators (MWO, JAJ, JLP). Our lab has demonstrated intra-tester coefficients of variation of 2.2% and 4.2% for baseline diameter and relative popliteal FMD, respectively. 16 Specifically, we blindly analyzed the same ultrasound recording for 20 participants on two different occasions, and the variation was calculated for each recording as: [(difference between 2 measurements/average value) × 100%]. Red blood cell velocity (RBCv) was continuously recorded using a pulsed frequency of 5 MHz and an insonation angle of 60° that was maintained across all participants. 15 Superior and inferior edges of the pulsed-wave sample volume were adjusted to encompass the entire arterial lumen, as recommended in published guidelines. 15
Artery lumen diameter, RBCv, and shear rate (SR) were analyzed using an automated, commercial edge-detection and wall-tracking software (FMD Studio, Cardiovascular Suite; Quipu srl, Pisa, Italy) that has been previously demonstrated to have high reproducibility with the analyses of popliteal FMD using this software.4,16,17 Measures of popliteal blood flow, SR, shear rate area under curve (SRAUC), and absolute and relative FMD were determined using standardized calculations.18–20 The statistical assumptions required to conduct allometric scaling or SRAUC-normalization of FMD were not met. Specifically, the regression between relative FMD (β = 8.33E−5, 95% CI = 1.81E−5, 14.85−5; y-intercept = 3.566, 95% CI = 2.821, 4.311) and absolute FMD (β = 4.19E−6, 95% CI = −0.01E−6, 8.38E−6; y-intercept = 0.175, 95% CI = 0.175, 0.271), with SRAUC did not have an intercept of zero.
Statistical analysis
All participant descriptive characteristics (sex via independent samples t-test), habitual physical activity and sedentary patterns, systemic hemodynamic, and popliteal data were entered into a bivariate correlation to determine if they were univariately related to relative or absolute FMD. All univariately related (p < 0.05) predictor variables were entered into a multiple regression model. Measures exhibiting high multicollinearity (variance inflation factor ⩾ 2.5 and condition index > 15) were removed from the model. Primary models included moderate physical activity (MPA), mean RBCv, and total time in sedentary bouts > 1 hour in lieu of daily step counts, resting blood flow, and number of sedentary bouts > 1 hour due to multicollinearity, respectively. The primary model was selected based on the strongest R2 value. Alternate models are presented in online Supplemental Table 1. The predicted residuals of all models were confirmed normal via a Shapiro–Wilk test. Partial correlations were conducted for all significantly related predictor variables identified from the multiple regressions. All statistical analyses were completed in IBM SPSS, Version 26.0 (IBM Corp., Armonk, NY, USA). Statistical significance was accepted as p < 0.05. All data are presented as means ± SD.
Results
Ninety-eight healthy, normotensive adults (53 females) were included in the present study (n = 34 older than 55 years). Table 1 includes the descriptive characteristics, systemic and popliteal hemodynamics, and physical activity monitoring outcomes. All predictor variables in Table 2 were univariately associated with popliteal FMD (all, p < 0.047). For absolute popliteal FMD, there was no univariate relationship with total sedentary time (Figure 2A). The final multiple regression model accounted for ~44% of the variance in absolute FMD (R = 0.666, p < 0.001). Age, peak diameter, MPA, RBCv, total time in sedentary bouts > 1 hour (Figure 2B), and sedentary breaks (Figure 2C) remained as independent predictors (Table 2). Partially controlling for all independent predictors simultaneously did not alter this relationship (all, p < 0.031).
Participant descriptive characteristics, systemic hemodynamics, and habitual and sedentary activity.
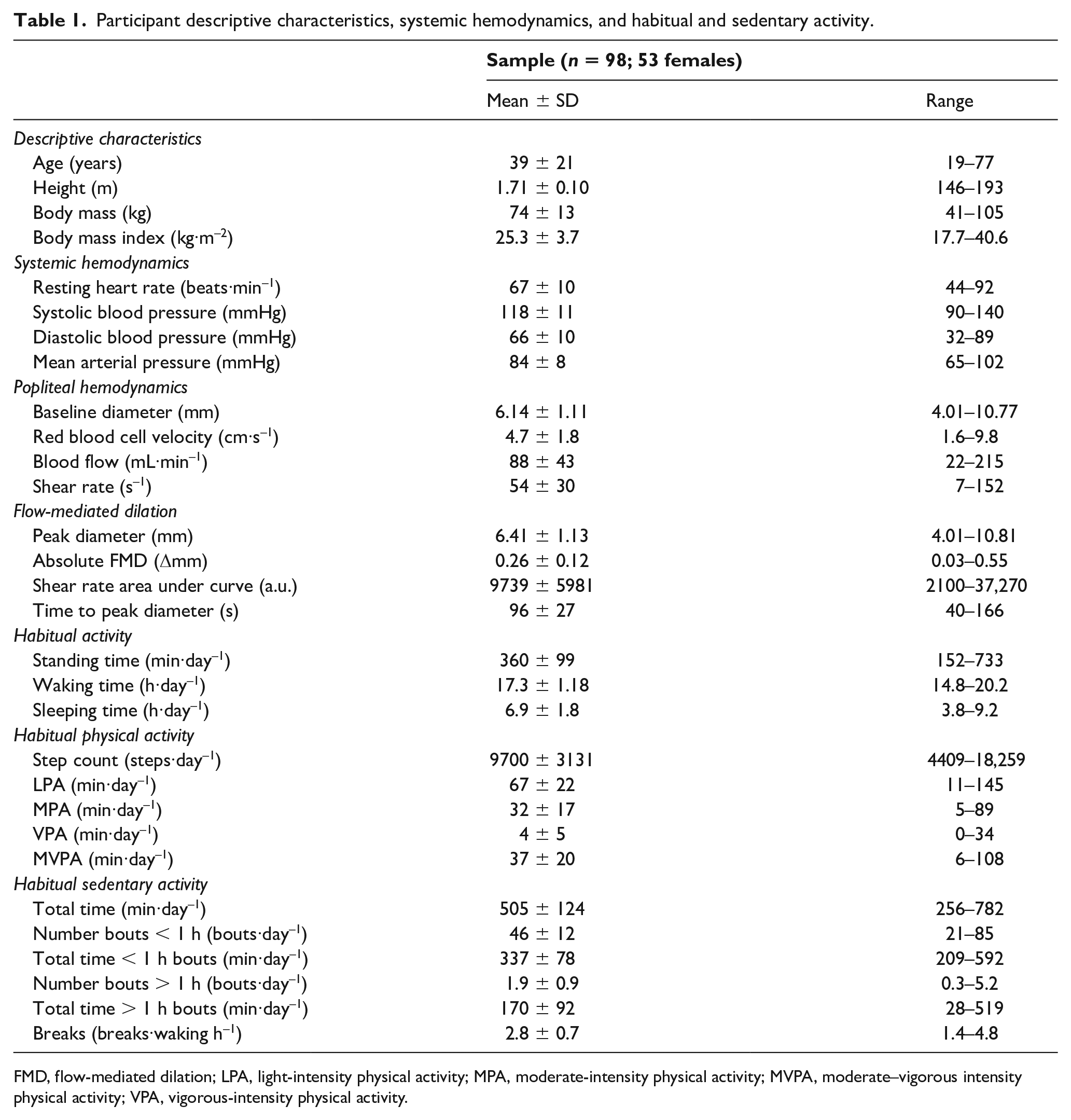
FMD, flow-mediated dilation; LPA, light-intensity physical activity; MPA, moderate-intensity physical activity; MVPA, moderate–vigorous intensity physical activity; VPA, vigorous-intensity physical activity.
Univariate and multivariate regression analyses examining the determinants of popliteal relative and absolute FMD..
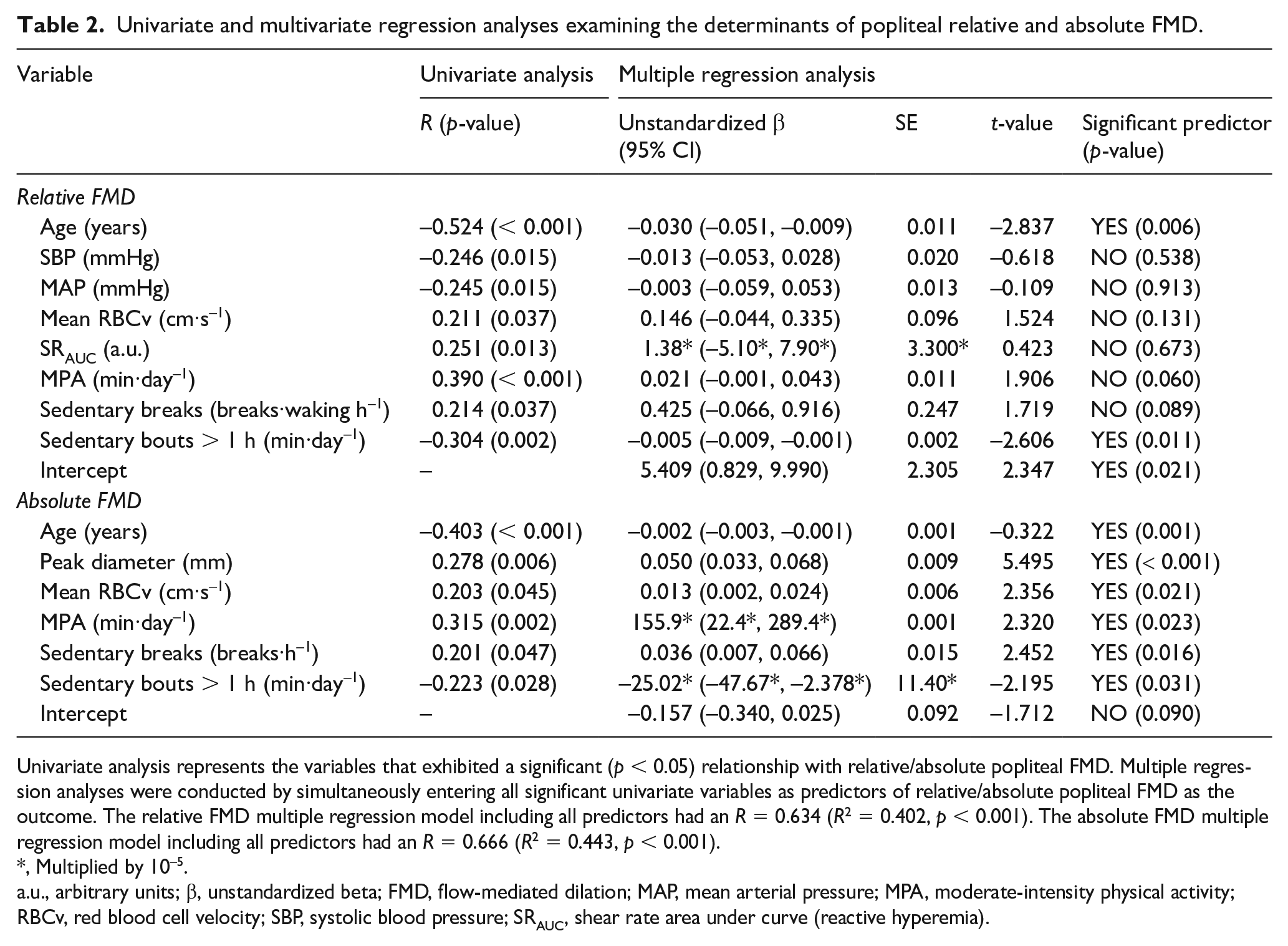
Univariate analysis represents the variables that exhibited a significant (p < 0.05) relationship with relative/absolute popliteal FMD. Multiple regression analyses were conducted by simultaneously entering all significant univariate variables as predictors of relative/absolute popliteal FMD as the outcome. The relative FMD multiple regression model including all predictors had an R = 0.634 (R2 = 0.402, p < 0.001). The absolute FMD multiple regression model including all predictors had an R = 0.666 (R2 = 0.443, p < 0.001).
, Multiplied by 10–5.
a.u., arbitrary units; β, unstandardized beta; FMD, flow-mediated dilation; MAP, mean arterial pressure; MPA, moderate-intensity physical activity; RBCv, red blood cell velocity; SBP, systolic blood pressure; SRAUC, shear rate area under curve (reactive hyperemia).
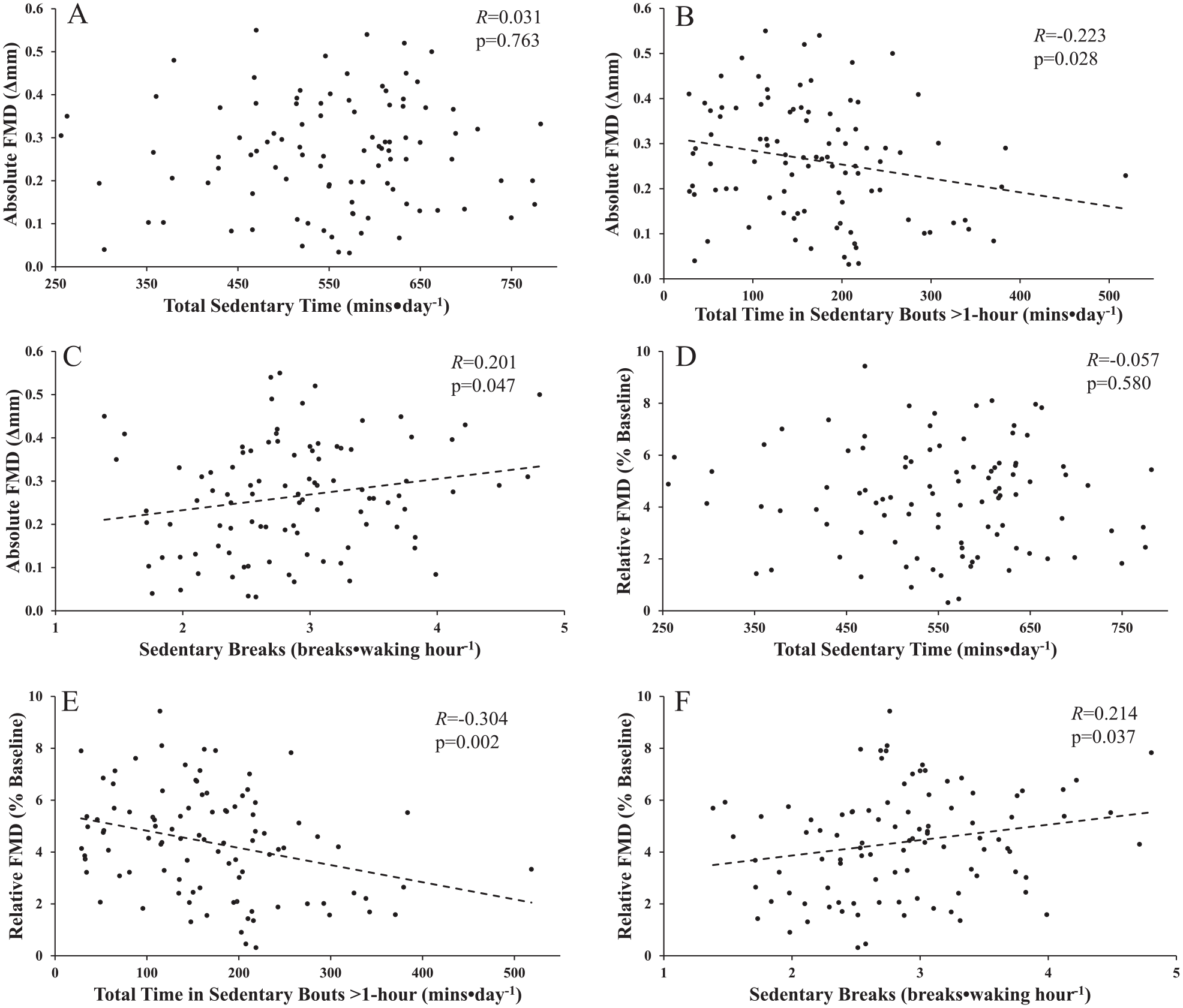
Pearson correlation between absolute popliteal flow-mediated dilation (FMD) versus total sedentary time (A), total time in sedentary bouts > 1 hour (B), and sedentary breaks (C); and relative popliteal FMD versus total sedentary time (D), total time in sedentary bouts > 1 hour (E), and sedentary breaks (F). A positive correlation was observed with sedentary breaks whereas a negative correlation was observed versus the number of daily prolonged sedentary bouts. The relationship absolute FMD and sedentary breaks (R = 0.250, p = 0.016) and total time in sedentary bouts > 1 hour (R = −0.225) remained following partial correlation. Similarly, the relationship between relative FMD and total time in sedentary bouts > 1 hour (R = −0.279, p = 0.006) also remained following partial correlation.
Total sedentary time was not univariately related with relative FMD (Figure 2D). The model explained ~40% of the variance of relative FMD (R = 0.634, p < 0.001), with age and total time in sedentary bouts > 1 hour (Figure 2E) remaining as the only independent predictors. Partially controlling for all independent predictors simultaneously did not alter this relationship (p < 0.01). Sedentary breaks were univariately (positively) correlated with relative FMD (Figure 2F).
Discussion
The purpose of this study was to test the hypothesis that higher total habitual sedentary time, fewer breaks in prolonged sedentary bouts, and an increase in the number of prolonged sedentary bouts would be associated with worse (i.e., lower) popliteal FMD responses. Our results support that increased engagement in habitual sedentary bouts > 1 hour and fewer sedentary breaks were associated with poorer popliteal endothelial-dependent vasodilation. In contrast to our hypothesis, total sedentary time was not associated with popliteal FMD responses.
Our results demonstrate that lower popliteal FMD was related to total time in prolonged bouts of sedentary time > 1 hour (Figure 2B/E). This observation is consistent with laboratory studies, which demonstrated that acute prolonged bouts of sedentary time decreased lower-limb FMD responses.4,5,21 As a lower-limb vessel, the popliteal artery is highly susceptible to decreases in blood flow and subsequent shear stress that accompanies bouts of sedentary activity.16,22,23 Restaino et al. 24 reported that sitting-induced popliteal endothelial dysfunction is mediated by a reduction in local shear stress which explains how habitual prolonged bouts of sedentary time may attenuate FMD responses. Specifically, habitual exposure of the popliteal artery to reduced local shear stress during prolonged sedentary bouts creates a pro-atherosclerotic environment that promotes endothelial dysfunction and increases the risk of atherosclerosis. 25 Although we cannot determine the exact mechanism responsible for our cross-sectional observations, more total time in prolonged bouts of sedentary time was associated with poorer lower-limb endothelial health. Interestingly, total sedentary time was not associated with popliteal endothelial-dependent vasodilation (Figure 2A/D). This highlights the importance of investigating the effects of sedentary patterns in addition to total sedentary time on lower-limb vascular function.
Our results also align with laboratory-based studies that observed that breaking up prolonged sitting bouts mitigated robust declines in FMD.5,21 Carter et al. 21 and Thosar et al. 5 recommend that 0.5 breaks/waking hour and one break/waking hour, respectively, was effective in preventing reduction in superficial femoral FMD. In contrast, this study demonstrated that participants engaged in 2.8 breaks/waking hour and that each additional break was associated with a 0.43% increase in popliteal relative FMD (Table 2). However, in comparison to the superficial femoral artery, the popliteal artery is subject to a robust distal deformation (i.e., kinked) in a knee-bent sitting posture, which may further attenuate blood flow/shear stress and endothelial function. 26 Sedentary breaks were an independent (positive) predictor of absolute FMD (Table 2), suggesting that breaking up sedentary bouts may offer protective benefits to popliteal health. This suggests that sedentary breaks may be important to include in a future version of the World Health Organization’s sedentary behaviour guidelines. 27
Study strengths and limitations
The present study is strengthened by the use of thigh-worn inclinometry rather than subjective questionnaires or nonthigh worn accelerometers that cannot distinguish standing time from sedentary time and thus cannot truly quantify sedentary time or patterns. Examination of sedentary breaks and time spent engaged in prolonged bouts, versus only total sedentary time, in a relatively large heterogeneous sample also adds to the impact of our findings. However, we acknowledge that this study is limited by its cross-sectional design and is unable to establish cause and effect. Lastly, our pooled sample was composed of mostly healthy younger (< 30 years: n = 64) and older adults (> 55 years: n = 34). As such, our findings cannot be extrapolated to middle-aged adults or persons with vascular diseases.
Conclusion
In conclusion, more time spent engaged in prolonged sedentary time, but not total sedentary time, was associated with worse popliteal endothelial-dependent vasodilator function. This provides support for the current sedentary behaviour guidelines from the World Health Organization that recommend minimizing the amount of time spent in prolonged sitting. 27 Also, it presents evidence for the addition of recommending breaking up long periods of sitting as often as possible to the existing guidelines. Decreasing habitual time spent engaged in prolonged sedentary bouts and implementing more sedentary breaks could be an effective strategy to promote better lower-limb arterial health.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211065494 – Supplemental material for Impact of habitual sedentary patterns on popliteal artery endothelial-dependent vasodilation in healthy adults
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211065494 for Impact of habitual sedentary patterns on popliteal artery endothelial-dependent vasodilation in healthy adults by Madeline E Shivgulam, Myles W O’Brien, Jarrett A Johns, Jennifer L Petterson, Yanlin Wu, Ryan J Frayne and Derek S Kimmerly in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Support provided by Canadian Foundation for Innovation: Leader’s Opportunity Fund (Derek S Kimmerly) and Faculty of Health Research Development Award (Derek S Kimmerly). Myles W O’Brien and Jarrett A Johns were supported by a Heart & Stroke Foundation of Nova Scotia BrightRed Scholarship. Myles W O’Brien was supported by a Nova Scotia Graduate Scholarship, a Research Nova Scotia – Scotia Scholars Award, a Killam PreDoctoral Scholarship, and CIHR Fredrick Banting and Charles Best Doctoral Award.
Funding
Dr. Derek S Kimmerly’s work was supported in part by the Canadian Foundation for Innovation: Leader’s Opportunity Fund, Dalhousie University Faculty of Health Research Development, and Nova Scotia Health Research Foundation (NSHRF) Development/Innovation.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
