Abstract
Background:
Combining low-dose statin and ezetimibe reduces the low-density lipoprotein cholesterol (LDL-C) similar to high-dose statin. However, whether there is a difference in the effect of these 2 lipid-lowering regimes on endothelial function is still controversial.
Methods:
We performed a systematic search of databases (MEDLINE [1950 to September 2011], EMBASE [1966 to September 2011]) and references of identified studies. Completely published randomized controlled trials comparing the effect of high-dose statin with low-dose stain plus ezetimibe on endothelial function (flow-mediated dilation [FMD] method) were included in this study.
Results:
Six trials with a total of 213 participants were included in the meta-analysis. The pooled weighted mean difference of FMD did not differ between the 2 lipid-lowering regimes (0.22%; 95% confidence interval [CI]: −0.85%-1.29%,
Conclusions:
Based on the currently available evidence, combining a low-dose statin with ezetimibe may provide similar beneficial effects on endothelial function as high-dose statin.
Introduction
The use of 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (statin) to reduce the levels of low-density lipoprotein cholesterol (LDL-C) is the standard of care for the routine management of hypercholesterolemia in a wide range of patients at risk of secondary cardiovascular (CV) events. 1 The established linear relationship between LDL-C and the risk of a CV event suggests that for every 1 mmol/L reduction in LDL-C, there is a one-fifth reduction in the risk of major coronary and vascular events. 2 Recent meta-analysis by Cholesterol Treatment Trialists’ Collaboration concluded that intensive regimens produced a highly significant 15% further reduction in major vascular events. 3 However, the Study of the Effectiveness of Additional Reductions in Cholesterol and Homocysteine trial, which was designed to assess the efficacy and safety of high-dose simvastatin, reported that 80 mg of simvastatin administered daily was associated with a much higher rate of myopathy, when compared with 20 mg of simvastatin administered daily (0.9% vs 0.03%). 4 In another meta-analysis of data from 5 statin trials, high-dose statin therapy was proved to be associated with an increased risk of new-onset diabetes, when compared with low-dose statin therapy. 5
Ezetimibe is a cholesterol absorption inhibitor that potently prevents the absorption of dietary and biliary cholesterol. When coadministrated with low-dose statin, ezetimibe provides similar LDL-C reduction as high-dose statin, 6,7 which makes it an alternative for intensive lipid-lowering treatment. It is believed that statins have pleiotropic effects apart from lipid lowering capability, and ezetimibe monotherapy in patients with hypercholesterolemia was also proved to ameliorate oxidative stress, insulin resistance, atherosclerotic and inflammatory markers, as well as atherogenic profiles. 8,9 However, the possible pleiotropic potential of ezetimibe has recently been questioned, because ezetimibe failed to demonstrate any significant changes in the primary end points of the Ezetimibe and Simvastatin in Hypercholesterolemia Enhances Atherosclerosis Regression (ENHANCE) and Simvastatin and Ezetimibe in Aortic Stenosis (SEAS) studies. 10,11 The latest Study of Heart and Renal Protection trail proved that reduction in LDL-C with 20 mg of simvastatin plus 10 mg of ezetimibe administered daily safely resulted in reduced incidence of major atherosclerotic events in a wide range of patients with advanced chronic kidney disease. 12 So far, there has been no randomized controlled trial to directly compare the effect of high-dose statin with low-dose statin plus ezetimibe on the clinical outcomes.
As endothelial dysfunction is considered to be an early step in atherogenesis and a key player in plaque progression and rupture, 13 detection of endothelial function impairment that predates the presence of clinically important plaque burden may help to identify a subgroup of patients at higher risk of future development of CV events. 14 Meanwhile, improving the endothelial function is one of the major pleiotropic effects of statin, which may provide a CV benefit beyond that expected from LDL-C lowering alone. 15 Thus, in this systemic review and meta-analysis, we aimed to determine the effect of 2 different lipid-lowering regimes (high-dose statin and low-dose statin plus ezetimibe) on endothelial function, which is assessed by flow-mediated dilation (FMD), a noninvasive measure of endothelial function of the brachial artery.
Methods
We designed a protocol that detailed the objective of our analysis, criteria for study inclusion/exclusion, assessment of study quality, primary outcome, and statistical methods in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. 16 Methods for the analysis were prespecified.
Data Sources and Searches
We conducted a search of MEDLINE (1950 to September 2011) and EMBASE (1966 to September 2011) in PubMed and EMBASE.com to identify all trials published in English, involving comparison between high-dose statin and low-dose statin plus ezetimibe (Appendix A). In addition, we performed a manual search of the literature using the references of the original manuscripts, review, and meta-analysis. Finally, a search of the Cochrane database and ClinicalTrial.org for ongoing trials or systemic reviews was conducted.
Study Selection
Two reviewers (Y.Y. and X.Z.) independently determined the study eligibility. Disagreement was resolved by consensus. Eligibility criteria of study characteristics included (1) randomized controlled trials, (2) comparing high-dose statin (defined as a daily dose of 40-80 mg) and low-dose statin (defined as a daily dose of 10-20 mg) plus ezetimibe, (3) assessment of endothelial function using FMD, and (4) reporting postintervention FMD or change from baseline FMD. Eligibility criteria of report characteristics included English literature and completely published studies (no inclusion of unpublished material and abstracts).
Data Extraction
Two authors (Y.Y. and X.Z.) extracted the following data from the included studies independently. Disagreements were resolved by discussion between the 2 reviewing authors. Information was extracted from each included trial on (1) study design (parallel study or crossover study) and sample size calculation; (2) characteristics of the participant (including age, gender, body mass index, hypertension, diabetes, and smoker); (3) type of intervention (including type, dose, and duration of statin and ezetimibe therapy); and (4) clinical outcome (pre-/postintervention or change from the baseline values of FMD, LDL-C, and C-reactive protein [CRP]) and reproducibility of FMD measurement. Both postintervention FMD and change from baseline FMD were extracted from each study, depending on what these studies have reported.
Risk of Bias Assessment
Two authors (Y.Y. and X.Z.) independently assessed the internal validity of the eligible studies according to the Cochrane Collaboration risk of bias tool. Disagreements were resolved in discussion with Shuyang Zhang until consensus was obtained. Risk of bias was described and judged in 6 specific domains: (1) sequence generation; (2) allocation concealment; (3) blinding of participants, personnel, and outcome assessors; (4) incomplete outcome data; (5) selective outcome reporting; and (6) other sources of bias (carryover effect for crossover studies). The domains of sequence generation, allocation concealment, and selective outcome reporting were reported by a single entry for each study. For incomplete outcome data, 2 entries were used because assessments generally had to be made separately for different outcomes (FMD and LDL-C). The judgments were based on the published study report that was included based on a combination of study reports, protocols, and published comments on the study. The judgments involved the answers “yes” (indicating a low risk of bias), “no” (indicating a high risk of bias), and “unclear” (if risk of bias is unknown, or if an entry is not relevant to the study).
Data Synthesis and Analysis
The primary outcome was postintervention or change from the baseline value of FMD and the secondary outcome was postintervention or change from the baseline value of LDL-C and CRP. As advised in the
Weighted mean differences were calculated using Review Manager 5.1, utilizing a random effects model (Inverse Variance). The
Using STATA 11.0, publication bias was accessed by the Begg's funnel plot and Egger weighted regression statistic with a value of
Results
Study Identification
A total of 199 studies were retrieved from the initial search, out of which 27 studies were reviewed in detail. Six trials investigating a total of 213 patients were included in the meta-analysis (Figure 1 and Table 1). Of these, 4 were parallel trials and the remaining were crossover studies. The studies were conducted in Japan, 18 Brazil, 19 Germany, 20 China (Taiwan), 21 Netherland, 22 and Sweden. 23
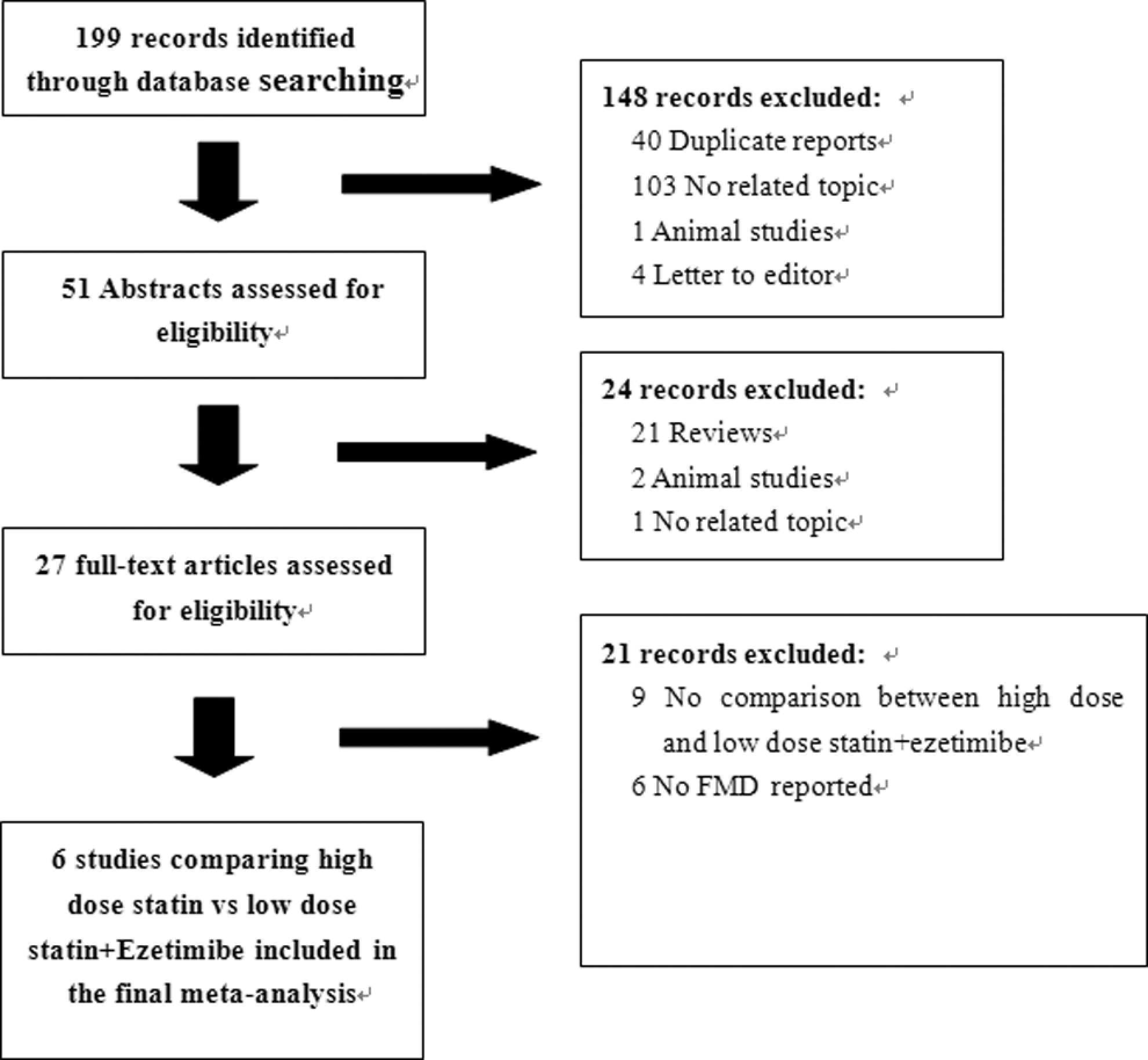
Flowchart of study selection.
Characteristics of the Included Studiesa
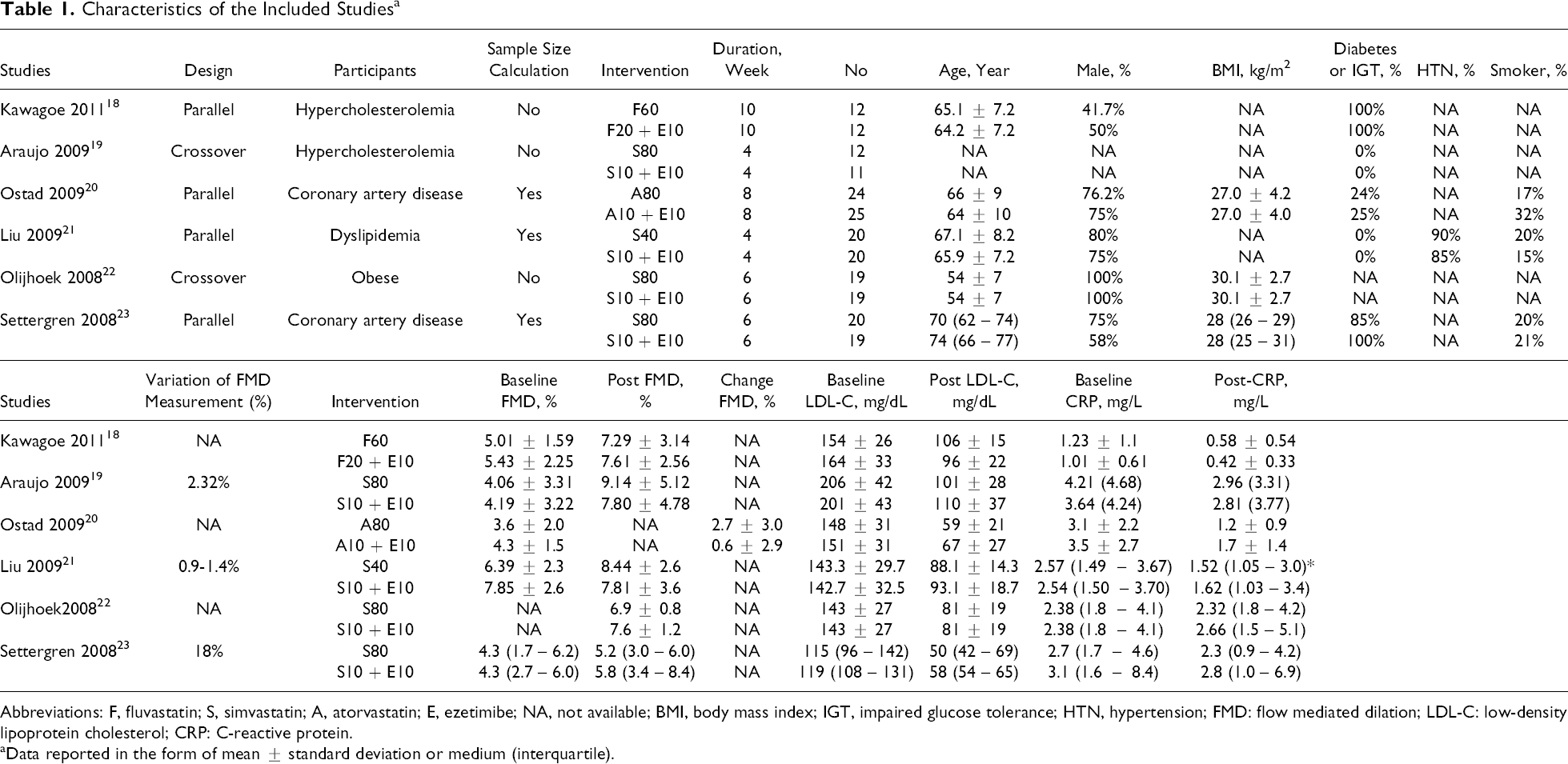

Abbreviations: F, fluvastatin; S, simvastatin; A, atorvastatin; E, ezetimibe; NA, not available; BMI, body mass index; IGT, impaired glucose tolerance; HTN, hypertension; FMD: flow mediated dilation; LDL-C: low-density lipoprotein cholesterol; CRP: C-reactive protein.
aData reported in the form of mean ± standard deviation or medium (interquartile).
Study Characteristics
A total of 107 patients treated with high-dose statin and 106 patients treated with low-dose statin plus ezetimibe were included in the analysis. In individual studies, the number of each group ranged from 11 to 25. The reported mean age of the participants ranged from 54 to 74 years. Each trial enrolled mostly men. The characteristics of the participants, statin therapy, and clinical outcome of the included trials are summarized in Table 1.
Risk of Bias
All trials were published in peer-reviewed journals. In all the trials included in this review, the methods of sequence generation and allocation concealment were not described in the study reports or study protocols. Lack of information for blinding is another problem with regard to the included studies, which made most of the studies at high risk of performance and detection bias. Two studies were at high risk of attrition bias because of missing baseline data, 22 high proportion of missing data, 19 missing data imbalance in number, and different reasons for missing data. 20 Only one of the studies had published study protocol available and had low risk of reporting bias. 20 No carryover effect was detected for 2 crossover studies. The risk of bias is summarized in Figure 2.
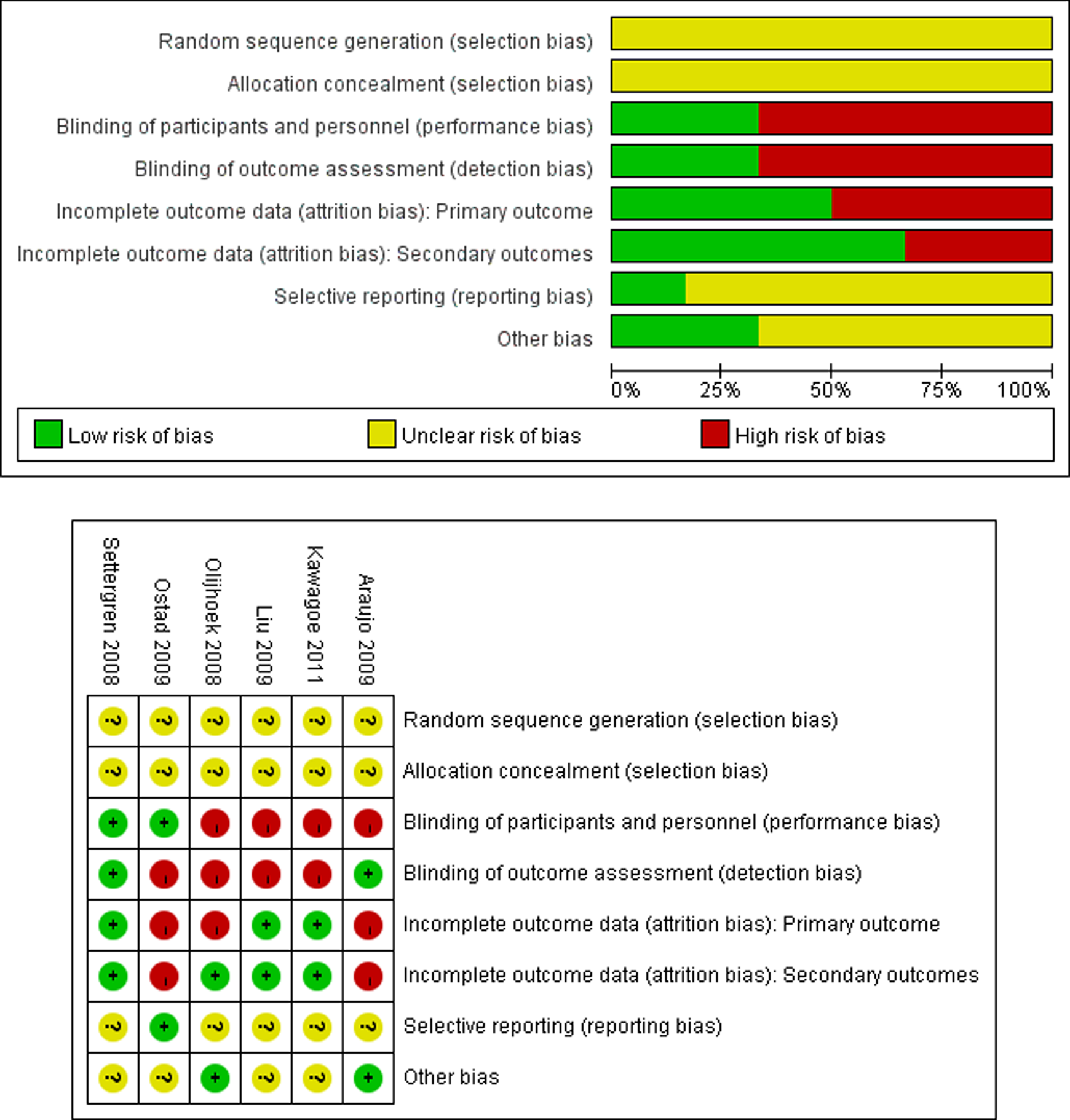
Risk of bias summary.
Quantitative Data Analysis
The pooled weighted mean difference of FMD between the 2 aggressive lipid-lowering regimes was 0.22% (95% confidence interval [CI]: −0.85%-1.29%,
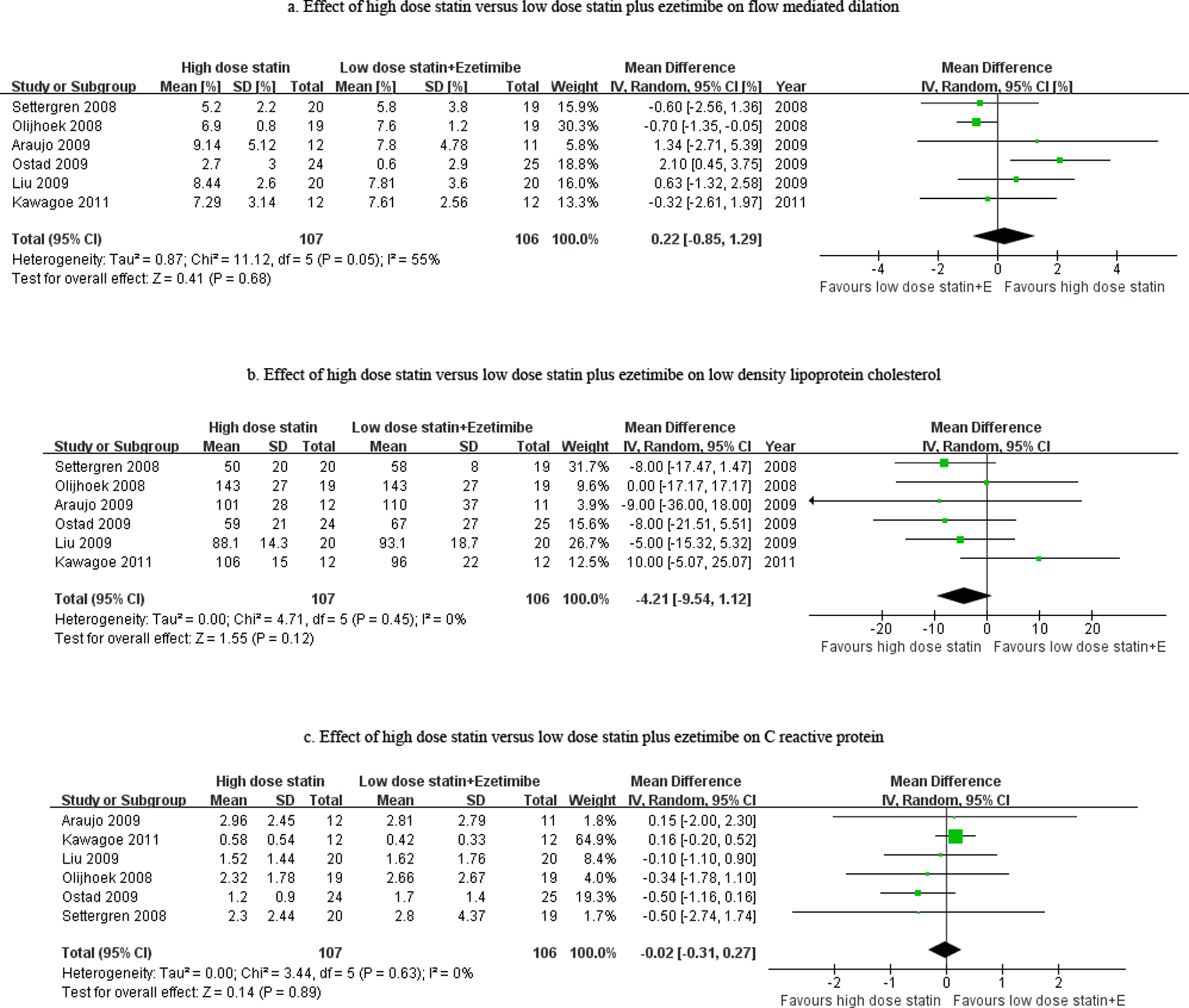
Effect of high-dose statin versus low-dose statin plus ezetimibe on flow-mediated dilation (FMD), low-density lipoprotein cholesterol (LDL-C), and C-reactive protein (CRP).
Assessment of publication bias using the Begg funnel plot (Figure 4,

Begg’s Funnel plot. SMD indicates standard mean difference.
Discussion
The main finding of our meta-analysis is that aggressive lipid-lowering treatment using the 2 different regimes, namely, high-dose statin and low-dose statin in combination with ezetimibe, did not differ with regard to their effect on endothelial function.
Studies on patients with established coronary artery disease, hypertension, or congestive heart failure have clearly demonstrated that endothelial dysfunction has fundamental prognostic implications. 24 –26 As one of the CV risk factors, hypercholesterolemia has been proved to be associated with endothelial dysfunction through several different methods. 27 –29 Cholesterol lowering with statin significantly improved endothelial dysfunction, when compared with placebo, and restoration of endothelial function was found to occur before significant reduction in serum cholesterol levels, suggesting that there may be additional effects on the endothelial function beyond that of cholesterol reduction. 30,31 Also, abrupt withdrawal of statin treatment could acutely and completely abrogate its beneficial effects on endothelial function. 32 However, this beneficial effect has been observed with other therapies besides statins. Leung et al proved that cholesterol reduction using cholesterol-reducing diet and cholestyramine also improves endothelial function in patients with hypercholesterolemia. 33 Even a single session of LDL apheresis with the reduction in total LDL and oxidized LDL could restore endothelial function. 34 This suggests that lipid lowering per se rather than the “pleiotropic effect” of statin may be more important for the improvement of endothelial function. The result of our meta-analysis demonstrated a similar LDL-lowering effect of high-dose statin and low-dose statin plus ezetimibe, which may be one of the reasons for the lack of significant difference in the improvement of endothelial function between the 2 aggressive lipid-lowering regimes.
The study by Fichtlscherer et al suggests that 40 mg of atorvastatin administered daily is more effective in improving the forearm blood flow (FBF) utilizing venous occlusion plethysmography than 10 mg of ezetimibe administered daily, thus concluding the differential effects of ezetimibe and statin on endothelial function. 35 However, more aggressive LDL-C reduction could also be observed in the atorvastatin group (49% vs 21%). Furthermore, when used as an add-on therapy in addition to chronic statin therapy, ezetimibe with 40 mg of atorvastatin administered daily was noted to provide similar LDL-C reduction and improvement in endothelial function (using SNP-induced FBF method). 35 Meanwhile, in patients with rheumatoid arthritis, both 20 mg of simvastatin and 10 mg of ezetimibe significantly reduced the total cholesterol and LDL-C. Concomitantly, Disease Activity Score, aortic pulse wave velocity, and FMD were significantly improved by both the drugs. 36 Two studies comparing rosuvastain with ezetimibe in different study populations showed varied conclusions with respect to the improvement in endothelial dysfunction.37,38 Thus, with regard to the improvement in endothelial dysfunction, lipid lowering appears to be more important than the potential pleiotropic effects of statin.
Anti-inflammatory effect, especially reduction in CRP level, has been considered as another pleiotropic effect of statin. 39 Recently, a systematic review and meta-regression analysis of 23 randomized controlled trials carried out with a variety of statin and nonstatin drugs showed that a strong correlation between the change in LDL-C and CRP and statin therapies had no significant effect on the CRP after adjusting for the change in LDL-C. 40 Our meta-analysis confirmed that the reduction in CRP did not differ between the 2 intensive regimes, showing similar reduction in LDL-C. This indicates that most of the anti-inflammatory effect of lipid-lowering therapies is related to the magnitude of change in LDL-C, and the potential non-LDL-C effects of statin on inflammation are much lower in magnitude.
Study by Berneis et al demonstrated that treatment with ezetimibe alone in healthy men was associated with the development of an atherogenic LDL subfraction profile by significantly increasing LDL4 subfraction. 41 However, this result was not consistent with studies in patients with dyslipidemia. Study by Geiss et al showed that ezetimibe decreases cholesterol in nearly all LDL subfractions including LDL4 subfraction. 42 A multicenter, randomized controlled trial further confirmed that simvastatin combined with ezetimibe was more effective than ezetimibe and simvastatin monotherapy in reducing atherogenic lipoprotein subfractions in 1397 patients with primary hypercholesterolemia. 43 Thus, ezetimibe improved not only the “quantity” but also the “quality” of LDL.
However, our study has several limitations. First, all the included studies have small sample size and lack high quality, which might make the final pooled result at risk of bias. Second, we did not have enough studies to perform a meta-regression to further determine the relationship between FMD and LDL-C. Third, studies using other methods to assess endothelial function were excluded from the meta-analysis, and the participants, type/dose of statin, and duration of therapy were not consistent across the included studies. Finally, we were unable to obtain the original data or further information regarding study design of the included studies, even though we tried to contact some of the corresponding authors before performing the meta-analysis.
High-dose statin is usually required to achieve further reduction in LDL-C, which can be problematic because the side effects of statin tend to be dose related. Combining a low-dose statin with ezetimibe could achieve the same degree of lipid-lowering effect and, therefore, might be an attractive alternative, considering the beneficial effects on endothelial function and proinflammatory markers demonstrated in the present meta-analysis. In future, large randomized controlled trial should be carried out to compare the efficacy of low-dose statin plus ezetimibe with high-dose statin on clinical outcomes.
Footnotes
Appendix A
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
