Abstract
Keywords
Background
An abdominal aortic aneurysm (AAA) is a localized dilatation of abdominal aorta (⩾ 30 mm), and a common disease, especially in older generations. 1 The diameter of the AAA correlates with increased risk of rupture, and an AAA larger than 55 mm is indicated for invasive surgical repairs such as endovascular aneurysm repair or open aneurysm repair because expected benefits overcome the natural risk of rupture and resultant death. 2 On the other hand, an undersized AAA (30–54 mm in diameter) does not yield a risk of rupture high enough to exceed the procedure-related mortality, and therefore is observed until the size reaches 55 mm. 2 Owing to continuous chronic inflammation and intraluminal arterial pressures, AAAs eventually increase in diameter.
Noninvasive pharmacotherapy for undersized AAAs (30–54 mm in diameter) is highly desired to suppress the need for surgical repairs, ruptures, and consequent death. Tireless efforts are being taken to dissect the mechanisms of AAA growth and to propose possible candidates for noninvasive pharmacotherapy for AAA in countless preclinical studies, some of which have led to clinical randomized controlled trials (RCTs). Among these candidates, antibiotics (ABx; which include doxycycline, roxithromycin, and azithromycin) and renin-angiotensin system inhibitors (RAS-I) are promising. However, even those candidate drugs with substantial supporting evidence for possible inhibition of AAA growth gave disappointing results in RCTs. These lessons highlight the difficulty of translating the success of preclinical studies to clinical usage, partly because of the complex and multifactorial nature of human AAA.
We are curious about the discrepancy between preclinical trials and human RCTs. For example, doxycycline, one of the most investigated drugs, showed repeated negative results in well-designed RCTs. Since each undersized AAA has different dynamics in its pathophysiology, we assumed there may be a good target subgroup for pharmacotherapy in undersized AAA. Therefore, we are inspired to elucidate the common factor shared in the design of human RCTs that has ‘concealed’ possible effectiveness. Herein we intended to find the subgroup for potential benefits by examining the baseline characteristics in those RCTs as variables to explain the discrepancy between preclinical trials and human RCTs.
Methods
We conducted the meta-analysis according to the PRISMA statement. 3 We forwent the approval of the Institutional Review Board due to the nature of systematic review and meta-analysis with use of already published data. This study was not registered with PROSPERO.
Search strategy and eligibility
All RCTs of pharmacotherapy in patients with undersized AAA were identified through comprehensive database search (MEDLINE and EMBASE) and additional manual search through March 15, 2021. Search words of ‘abdominal aortic aneurysm’ AND (expan* OR grow* OR dilat* OR diameter*) AND randomized were used (search strategy available in the online Appendix). RCTs with propranolol and ticagrelor were excluded; the former was proven to be harmful; the latter involves only one study. Two independent authors (TS and HT) reviewed the searched results.
Data collection and synthesis
Baseline characteristics, AAA growth from the baseline, and aneurysm-related events (diameters exceeding 55 mm, ruptures, or referral to surgery) were extracted from each study. We also extracted data of intention-to-treat analysis. The mean and SD of AAA growth rates were transformed into mm/year (mean difference [MD] < 0 favors pharmacotherapy, > 0 favors placebo) with 95% CIs. Aneurysm-related events were expressed as hazard ratios (HRs), where adjusted estimates were preferentially used when available. In one study including three arms (placebo, RAS-I, and calcium channel blocker), we only used the data of placebo and RAS-I groups. Study-specific estimates were combined using inverse variance-weighted averages of MDs and logarithmic HRs in both fixed- and random-effects (DerSimonian and Laird method) models. 4
Statistical analysis
First, we conducted meta-analysis for each drug group (ABx or RAS-I) to evaluate the MD between the drug and placebo groups. Second, multivariable random-effects restricted maximum likelihood meta-regression was performed with factors among baseline characteristics of RCTs: that is, mean of baseline diameter and drug group (ABx and RAS-I); follow-up period (month) and drug group; and duration of treatment (week) and drug group. Subgroup analyses were conducted to confirm the results of meta-regression analysis. Fixed-effect models were analyzed to confirm reproducibility of the results. An I2 index > 50% was considered for evaluating significant heterogeneity. 5 We imputed missing SDs of growth rates according to the Cochrane Handbook for Systematic Reviews of Interventions. 6 The risk of bias was also evaluated using a Cochrane Collaboration tool. 7 Funnel plot asymmetry for publication bias was examined with Egger’s linear regression tests. 8 All analyses were performed with Review Manager (RevMan) Version 5.4 software, ProMeta 3 software, and Comprehensive Meta-Analysis version 3 (Biostat, Englewood, NJ, USA). The primary endpoint was MD in aortic diameters, and the secondary endpoint was aneurysm-related events.
Results
Search strategy
Our strategy for the literature search initially yielded 480 articles (see the online Supplemental Appendix). After removal of 98 duplicate papers, 255 articles were excluded either because they were review articles or irrelevant for the current study. A further 74 papers were excluded because of other interventions, description of the trial design, they were imaging studies or used other drugs. Finally, eight RCTs were identified,9–16 as is outlined in online Supplemental Figure 1. Two studies by Vammen et al. 14 and Høgh et al. 11 recruited patients from the same Viborg County study for screening AAAs, but they are considered as separate studies because they applied different treatment regimens, as was the case in Cochrane database systematic review and meta-analysis. 17 The overall risk of bias was low (online Supplemental Figure 2).
Baseline characteristics
The current meta-analysis includes a total of 1325 patients with undersized AAAs (Table 1). The drugs investigated were doxycycline in three studies, roxithromycin in two, azithromycin in one, telmisartan in one, and perindopril in one study. Doxycycline, roxithromycin, and azithromycin were combined into the ABx group because they share the similar features of inhibiting protein synthesis in ribosome, whereas telmisartan and perindopril were in the RAS-I group. 18 The duration of pharmacotherapy ranged from 4 weeks to 2 years, with the follow-up period from 18 to 63 months (median 24 months). The inclusion criteria for baseline diameters varied slightly: four studies started at the minimum diameter of 30 mm and four RCTs at 35 mm; the maximum diameter was either 45 mm (one RCT), 50 mm (five RCTs), or 55 mm (two RCTs). As a result, mean of baseline diameters varied from 33.1 mm to 43.1 mm. The baseline diameters were median in three studies and mean in five studies. Those studies with a mean baseline diameter < 40 mm set minimum inclusion criteria at 30 mm. Ultrasound was used for aortic diameter measurement at follow-up in seven studies. As is shown in Table 2, the mean age was 68–73 years (median 71 years), with 47–100% male (median 62%). There was no specific difference in inclusion criteria between women and men. The median proportion of comorbidities or prescriptions was as follows: hypertension 90.0%, diabetes mellitus 13%, coronary artery disease 41%, current smoker 35%, β-blocker 47%, angiotensin-converting enzyme inhibitor 32%, angiotensin II receptor blocker 14%, antiplatelet 46%, and statins 65%.
Designs of included randomized controlled trials.
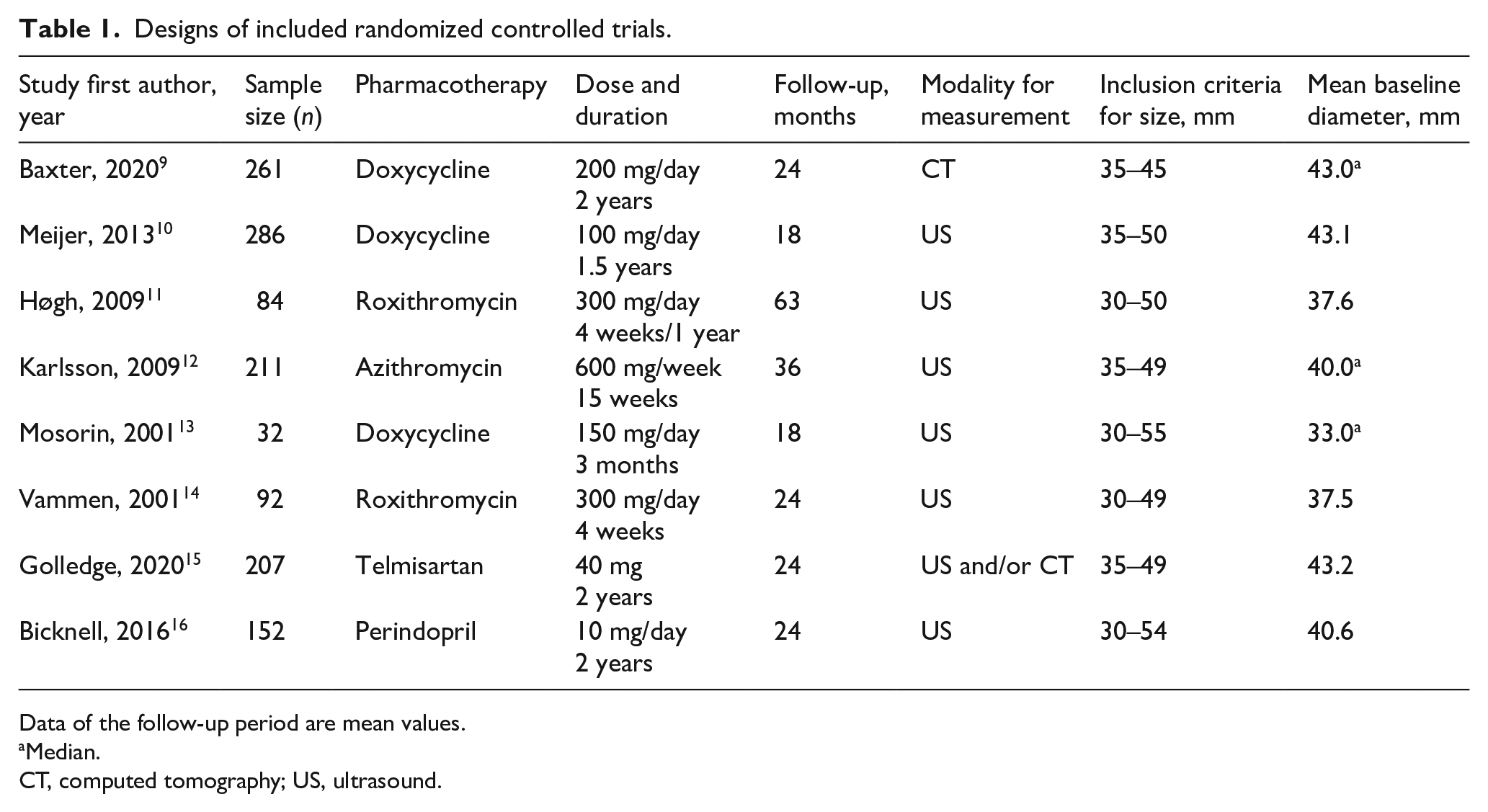
Data of the follow-up period are mean values.
Median.
CT, computed tomography; US, ultrasound.
Baseline characteristics and concomitant medications of patients included in randomized controlled trials.
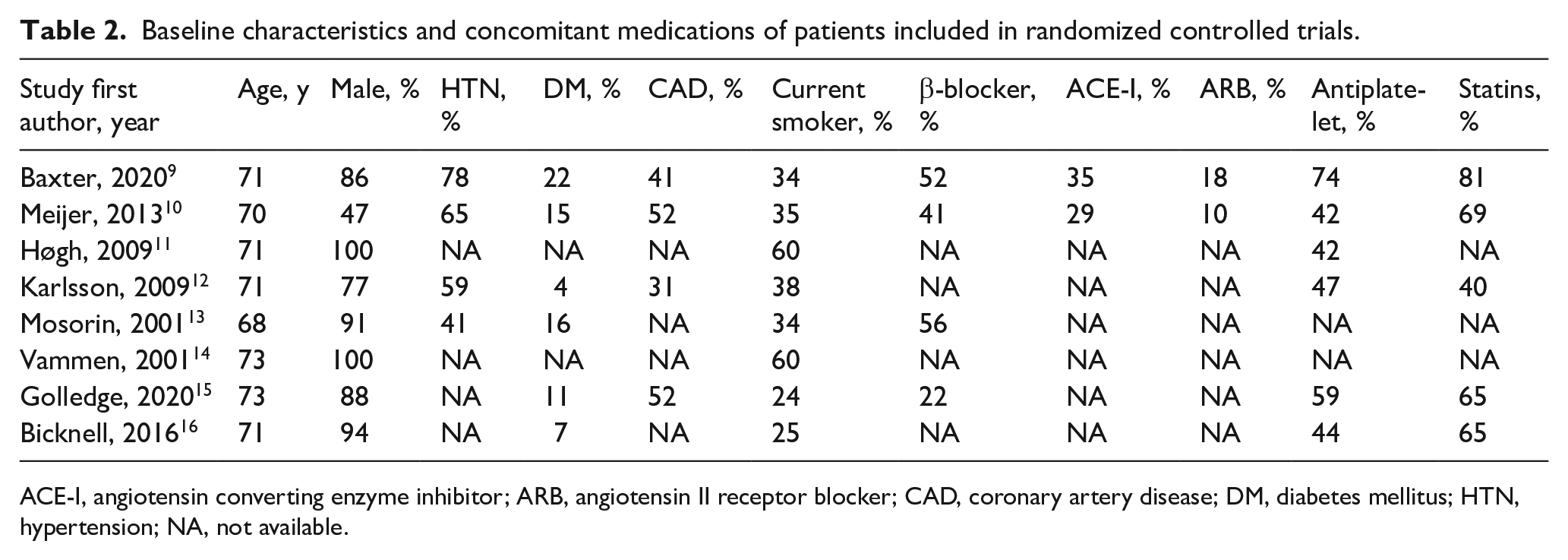
ACE-I, angiotensin converting enzyme inhibitor; ARB, angiotensin II receptor blocker; CAD, coronary artery disease; DM, diabetes mellitus; HTN, hypertension; NA, not available.
Mean difference between pharmacotherapy and placebo
First, the pooled estimates of MD in aortic diameter of each drug group (ABx or RAS-I) revealed no significant differences between the pharmacotherapy and placebo groups (ABx: MD −0.22 mm/year [95% CI −0.69, 0.25], I2 = 71%, p = 0.36; RAS-I: MD −0.02 mm/year [95% CI −0.38, 0.35], I2 = 0%, p = 0.93; Table 3, online Supplemental Figure 3). Second, the multivariable random-effects restricted maximum likelihood meta-regression was conducted for mean of baseline diameter and drug group (ABx and RAS-I), follow-up period (month) and drug group, and duration of treatment (week) and drug group. This demonstrated a statistically significant linear relationship between baseline diameter and MD in diameter changes (coefficient 0.15 [95% CI 0.0011, 0.30], p = 0.049; Figure 1A) but not for the follow-up period (coefficient −0.020 [95% CI −0.062, 0.023], p = 0.28) and duration of treatment (coefficient 0.0097 [95% CI −0.0032, 0.023], p = 0.11). The statistical significance of the baseline diameter in relation to MD in diameter changes also remained in a fixed-effect model (coefficient 0.14 [95% CI 0.047, 0.23], p = 0.003; Figure 1B). No association was identified between year of publication and MD in diameter changes. Therefore, we focused on baseline diameter for further analysis. In reference to the linear regression, these were divided into subgroups based on the baseline diameters. Subgroup analysis clearly depicted that the group with baseline diameters < 40 mm is related to significant therapeutic effect in ABx (MD −1.03 mm/year [95% CI −1.64, −0.42], I2 = 0%, p = 0.001; Figure 2A), whereas the groups with baseline diameters ⩾ 40 mm failed to show the effectiveness of pharmacotherapy, be it either ABx (MD 0.18 mm/year [95% CI −0.18, 0.53], I2 = 53%, p = 0.33; Figure 2B) or RAS-I (MD −0.02 mm/year [95% CI −0.38, 0.35], I2 = 0%, p = 0.93; Figure 2C). The reproducibility of subgroup analysis was confirmed in fixed-effect models (Table 3).
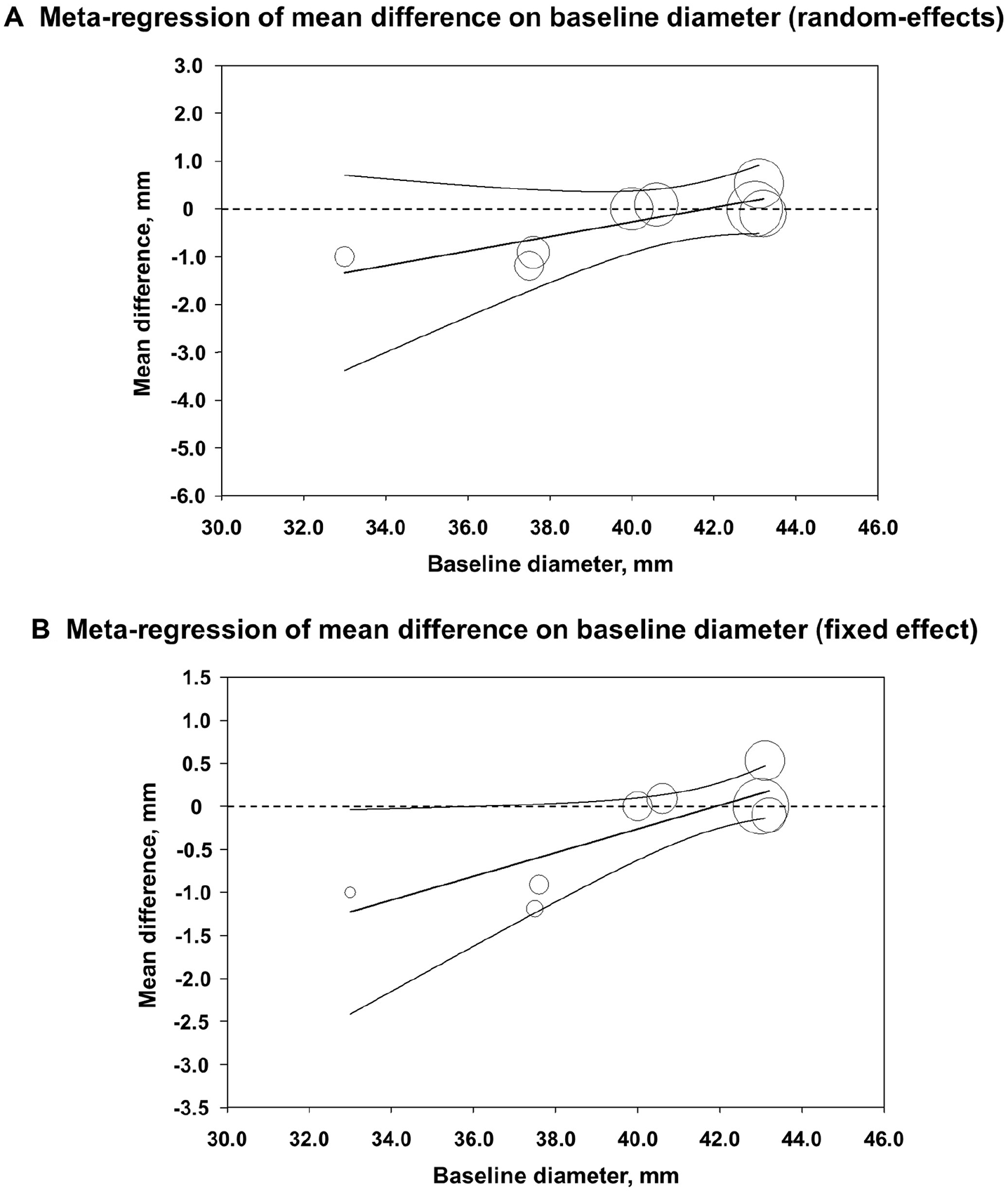
Multivariable random-effects restricted maximum likelihood meta-regression. Linear regressions of mean difference in diameter changes on mean of baseline diameter and drug group. (
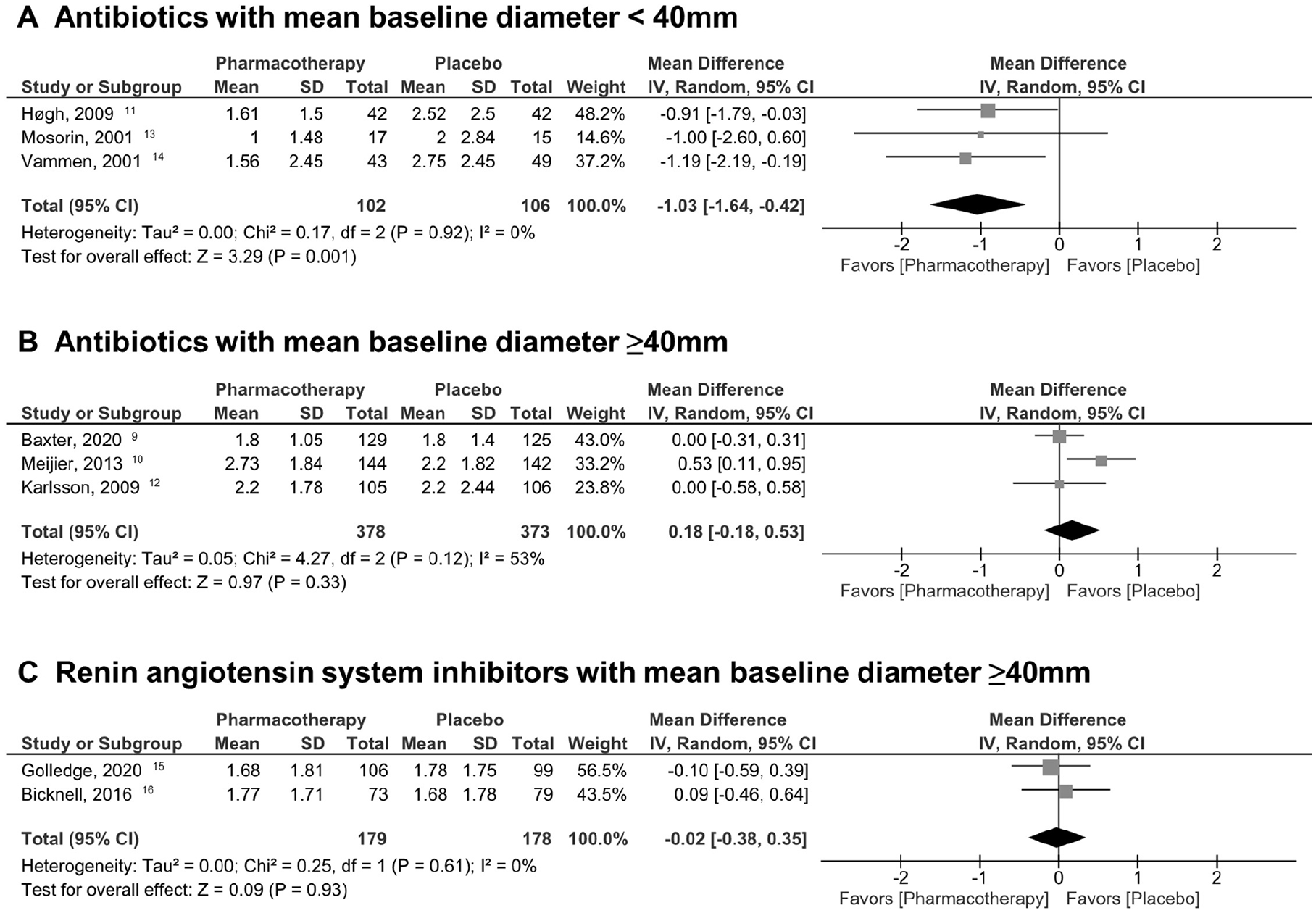
Estimation of mean difference in aortic diameter changes based on mean of baseline diameters (random-effects model).
Comparison of random-effects model and fixed-effect model for each analysis.
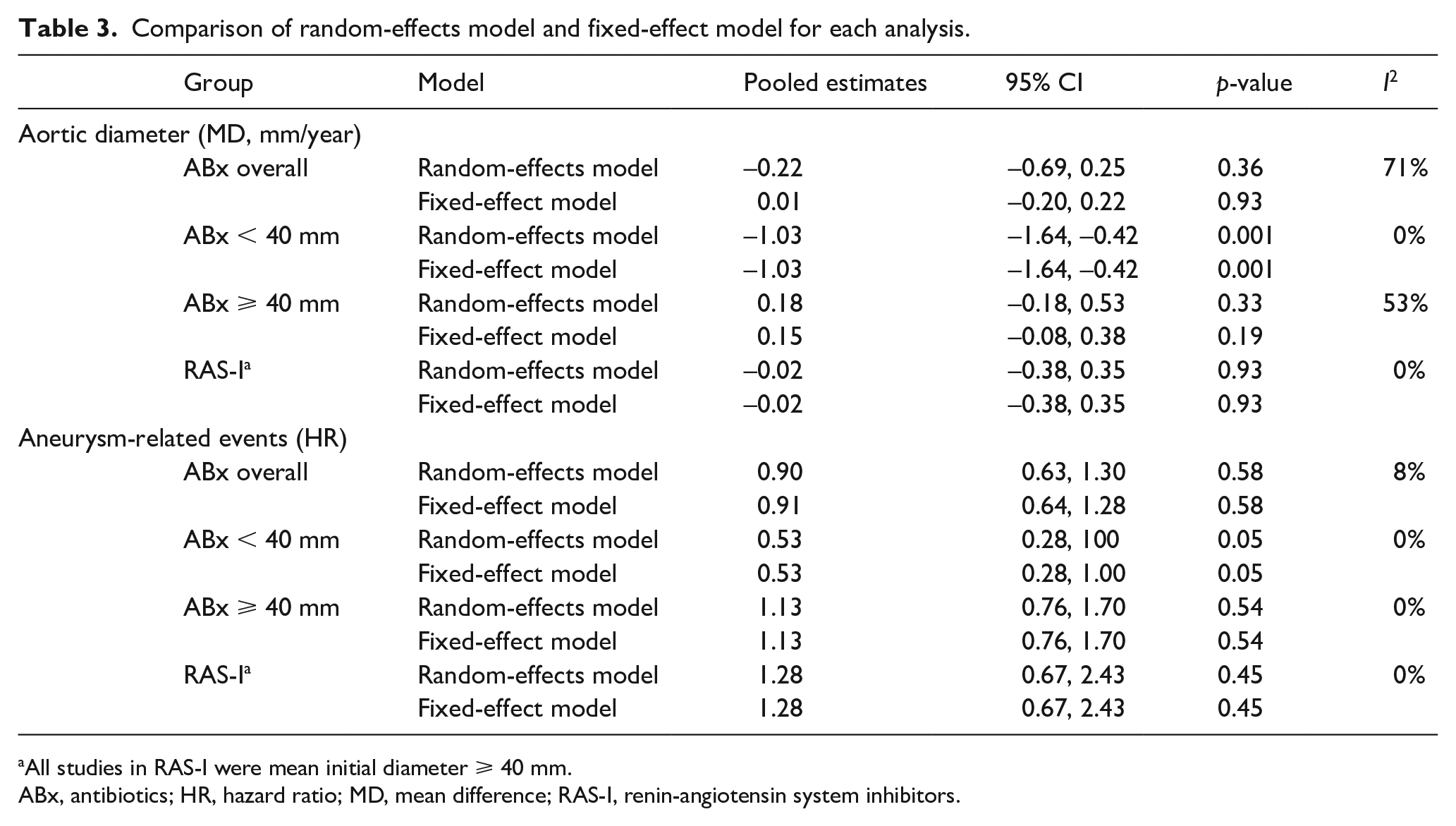
All studies in RAS-I were mean initial diameter ⩾ 40 mm.
ABx, antibiotics; HR, hazard ratio; MD, mean difference; RAS-I, renin-angiotensin system inhibitors.
Aneurysm-related event between pharmacotherapy and placebo
Similar distinct outcomes were revealed in aneurysm-related events. Overall, pharmacotherapy did not contribute to suppress aneurysm-related events (ABx: HR 0.90 [95% CI 0.63, 1.30], I2 = 8%, p = 0.58; RAS-I: HR 1.28 [95% CI 0.67, 2.43], I2 = 0%, p = 0.45; Table 3, online Supplemental Figure 4). However, subgroup analysis of ABx with mean baseline diameters < 40 mm were at borderline significance for reduction of aneurysm-related events (HR 0.53 [95% CI 0.28, 1.00], I2 = 0%, p = 0.05; Figure 3A). Subgroups with baseline diameters ⩾ 40 mm did not reduce aneurysm-related events significantly (ABx ⩾ 40 mm: HR 1.13 [95% CI 0.76, 1.70], I2 = 0%, p = 0.54; RAS-I ⩾ 40 mm: HR 1.28 [95% CI 0.67, 2.43], I2 = 0%, p = 0.45; Figures 3B, 3C). These findings were also reassured in fixed-effect models (Table 3).
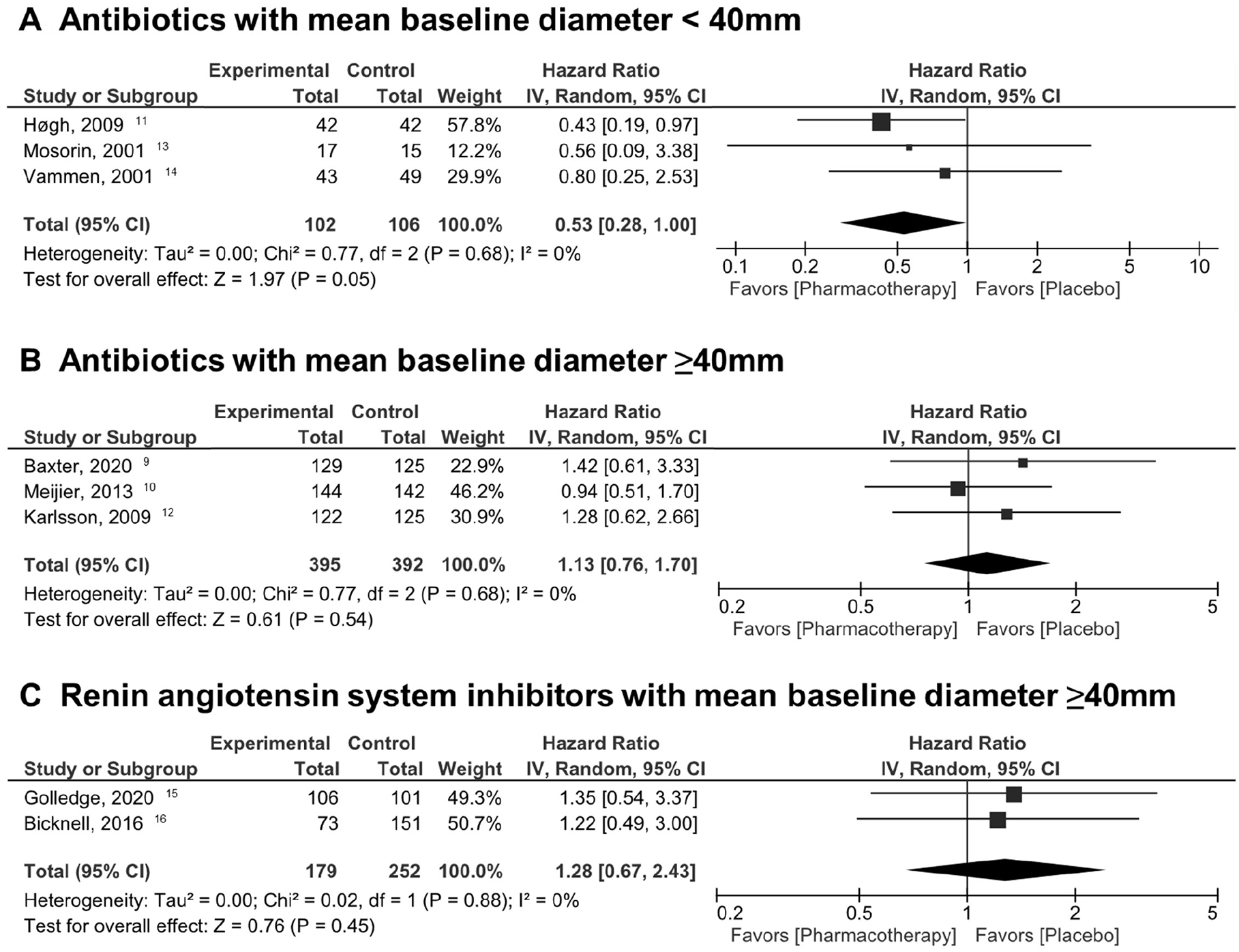
Estimation of hazard ratios of aneurysm-related events (diameters exceeding 55 mm, ruptures, or referral to surgery) based on mean of baseline diameters (random-effects model).
Publication bias
A funnel plot is shown in online Supplemental Figure 5. Although Egger’s test was p = 0.13, the possibility of publication bias is not completely denied due to the small number of original articles. 19
Discussion
The novel finding in the current meta-analysis was that effectiveness of pharmacotherapy for undersized AAAs (30–54 mm) differs depending on mean baseline diameter of RCTs. This is statistically significant for MD in aortic diameters and at borderline significance for aneurysm-related events between pharmacotherapy and placebo. Follow-up period and duration of treatment did not have any impact on the outcomes. These results suggest the need for reconsidering optimal candidates among undersized AAAs in the design of RCTs of pharmacotherapy.
The repeated ‘negative’ results of RCTs for undersized AAA were discouraging because doxycycline and RAS-I were presumably among the most promising drugs in preclinical trials. In fact, the effectiveness of pharmacotherapy in the overall population was confirmed as not significant in the current meta-analysis, where a group of doxycycline, roxithromycin, and azithromycin were merged for their similar pharmacological characteristics on ribosome and have historically been investigated in relation to Chlamydia pneumoniae infection. 18 Even within the negative data, we assumed there must be some factors in the design of RCTs which blinded us from hidden positive results. In this context, we intended to discover certain subgroups of patients with undersized AAAs who could possibly benefit from pharmacotherapy. And we finally found that the baseline diameters matter. We can interpret the results of restricted maximum likelihood meta-regression as follows. For each millimeter increase in baseline diameter, the effect of pharmacotherapy (MD in aortic diameter) is estimated to be reduced by 0.14 mm/year (fixed-effect model) or 0.15 mm/year (random-effects model) compared to placebo treatment. Therefore, earlier stages of undersized AAAs are preferable for pharmacotherapy than more advanced stages.
It is theoretically legitimate that pharmacological effect differs depending on the initial AAA size. It is generally accepted that AAA never regresses. In the earlier phase of AAA, the extracellular matrix is still preserved and the protective effects on the aortic wall can be expected. Meanwhile, in the later phase of AAA, the integrity of elastic lamellae is compromised, smooth muscle cell density is decreased, and the aneurysms are susceptible to higher pressures (Laplace’s law), recruiting more inflammatory cells.1,20,21 Supporting evidence of many drugs in preclinical trials is usually from interventions initiated at the early stage or right after inducing AAAs.20,21 Moreover, a meta-analysis on the natural course of AAA growth supports different rate of expansion depending on the initial size of AAAs (30–34 mm, 1.81 mm/year; 35–39 mm, 2.66 mm/year; 40–44 mm, 3.86 mm/year; 45–49 mm, 4.96 mm/year), indicating distinct dynamics of undersized AAAs at various diameters. 22 Admitting that the diameter of an AAA is not a single determinant for aneurysm growth rate, it is likely a strong predictor. Taking the theory and current data together, it is suggested that the earlier phase of undersized AAAs is more suitable for noninvasive pharmacological protection against the natural course of AAA enlargement.
As is shown in the current meta-analysis, it is probable that all undersized AAAs are not good candidates for pharmacotherapy; however, finding the optimal group among undersized AAA is not straightforward. Even though some papers describe smaller undersized AAAs (30–40 mm) or even ectatic aortas (25–30 mm) can grow in size, most small AAAs are stable without reaching 55 mm or experiencing rupture.23,24 More importantly, their causes of fatal outcomes are not necessarily aneurysm-related, but rather cardiovascular events or cancer.24,25 It is also noted that it takes 5 years or even longer to see the natural growth of ectatic or smaller, undersized AAAs, posing a question for the optimal follow-up term of future RCTs. On the other hand, even the study with inclusion criteria of 35–45 mm resulted in a mean baseline diameter of 43.0 mm. 9 Bearing these observations in mind, we would like to positively present the future direction for clinical trials to testify the promising noninvasive therapy for undersized AAAs. It is advisable to include earlier stages of undersized AAAs to see the therapeutic effects by pharmacotherapy.
Study limitations
Although we undertook statistical validation with random-effects and fixed-effect models, we must note that the small number of RCTs considered for the meta-regression is a limitation. The baseline diameters from five studies were mean values, which are not good measures of central tendency in some circumstances. Another limitation includes possible aggregation bias, since the baseline diameters are aggregated data at the study level. We could not obtain individual patient data to perform the Bayesian meta-regression method. 26 Yet, we believe this could summarize the best available evidence to date. Hopefully, the lessons learned from the former RCTs will further enhance the drive to realize noninvasive therapy for undersized AAAs and to improve the management of AAA as a whole.
Conclusion
The current meta-analysis of placebo-controlled RCTs of noninvasive pharmacotherapy for undersized AAAs portrayed that there is a linear relationship between mean baseline diameter of each RCT and effectiveness of pharmacotherapy. Future RCTs should preferentially include patients with smaller undersized AAAs, the candidates who potentially benefit from pharmacotherapy.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211061603 – Supplemental material for Smaller size is more suitable for pharmacotherapy among undersized abdominal aortic aneurysm: A systematic review and meta-analysis
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211061603 for Smaller size is more suitable for pharmacotherapy among undersized abdominal aortic aneurysm: A systematic review and meta-analysis by Takuro Shirasu, Hisato Takagi, Jun Yasuhara, Toshiki Kuno, K Craig Kent and W Darrin Clouse in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
