Abstract

Keywords
The most frequent location for aneurysmal degeneration of the aorta is the infrarenal segment, accounting for 90% of cases. The remaining 10% are distributed in incidence through the ascending aorta (60%), the aortic arch (10%), and the thoracic and thoraco-abdominal segments (40%).1,2 The epidemiology and associated risk factors are different among these: thoracic aortic aneurysms (TAA) are more frequent in females and females tend to be diagnosed at an older age than males. 3
Reports regarding the risk of thoracic aneurysm development after an abdominal aortic aneurysm (AAA) surgery are scarce, with non-standardized definitions of the TAA diameter and variable figures ranging from 2.2% to 27.0%.4–8 This has a growing importance because most imaging protocols after AAA repair do not specifically assess the thoracic aorta, and an early diagnosis is essential for preventive management. The aim of this study was to quantify the incidence of TAA degeneration detected in imaging controls after an elective AAA treatment and to identify potential associated risk factors for TAA development.
All patients who underwent an elective AAA treatment (open or endovascular) during a 10-year period (2003–2012) at a single tertiary-care institution were prospectively enrolled into a database. Demographics, comorbidities, preoperative aneurysm diameter, type of aneurysm, and repair technique were recorded. Also, postoperative outcomes, mortality, and morbidity over time were recorded according to reporting standards. 9 All patients underwent a complete thoracic-abdominal contrast-enhanced computed tomography angiogram (CTA) prior to the AAA repair. All preoperative CTA scans were reviewed by a vascular specialist using either OsiriX (Pixmeo; Geneva, Switzerland) or HorosTM software (Purview; Annapolis, MD, USA). All measured diameters were obtained from a centerline reconstruction of the aortic anatomy. The Institutional Review Board dismissed the need for specific informed consent given the retrospective nature of the study.
Follow-up on the endovascular aortic repair (EVAR) group was performed by means of a multiphase CTA at 1 month and 1 year after the procedure. In the absence of endoleaks, duplex ultrasound (DUS) was performed yearly thereafter. In the open surgery group (OS), DUS was performed yearly. After 5 years of follow-up, a thoraco-abdominal CTA was performed in all patients (EVAR and open), following the recently published recommendations in Europe for management of AAA. 10
The main outcome was defined as an enlargement of the ascending aortic, aortic arch, or descending thoracic diameter of 45 mm or more in any of the follow-up CTA scans that were either included in the protocol or performed to rule out other pathologies. A secondary objective was to identify comorbidities or other risk factors potentially associated with TAA development.
Continuous variables were described using mean and SD, and categorical variables were described using frequencies. For the first, normality was assessed using the Kolmogorov-Smirnov test, using median and IQR when the distribution was not assumable to a normal Gaussian curve. For the latter, Pearson’s chi-squared test, Fisher’s exact test, Student’s t-test, and the Mann–Whitney U-test were used to assess univariate associations when appropriate. A p-value under 0.05 was considered statistically significant. SPSS v.20.0 software (IBM Corp., Armonk, NY, USA) was used for all calculations.
A total of 616 patients were included (mean age 71.9 ± 8 years, 96.1% males). Fifteen patients (2.4%) had a prior history of thoracic aortic pathology and were excluded from any further analysis. Demographics, comorbidities, and surgical outcomes are depicted in Table 1.
Demographics, comorbidities, and postoperative outcomes in patients with and without a TAA development over time.
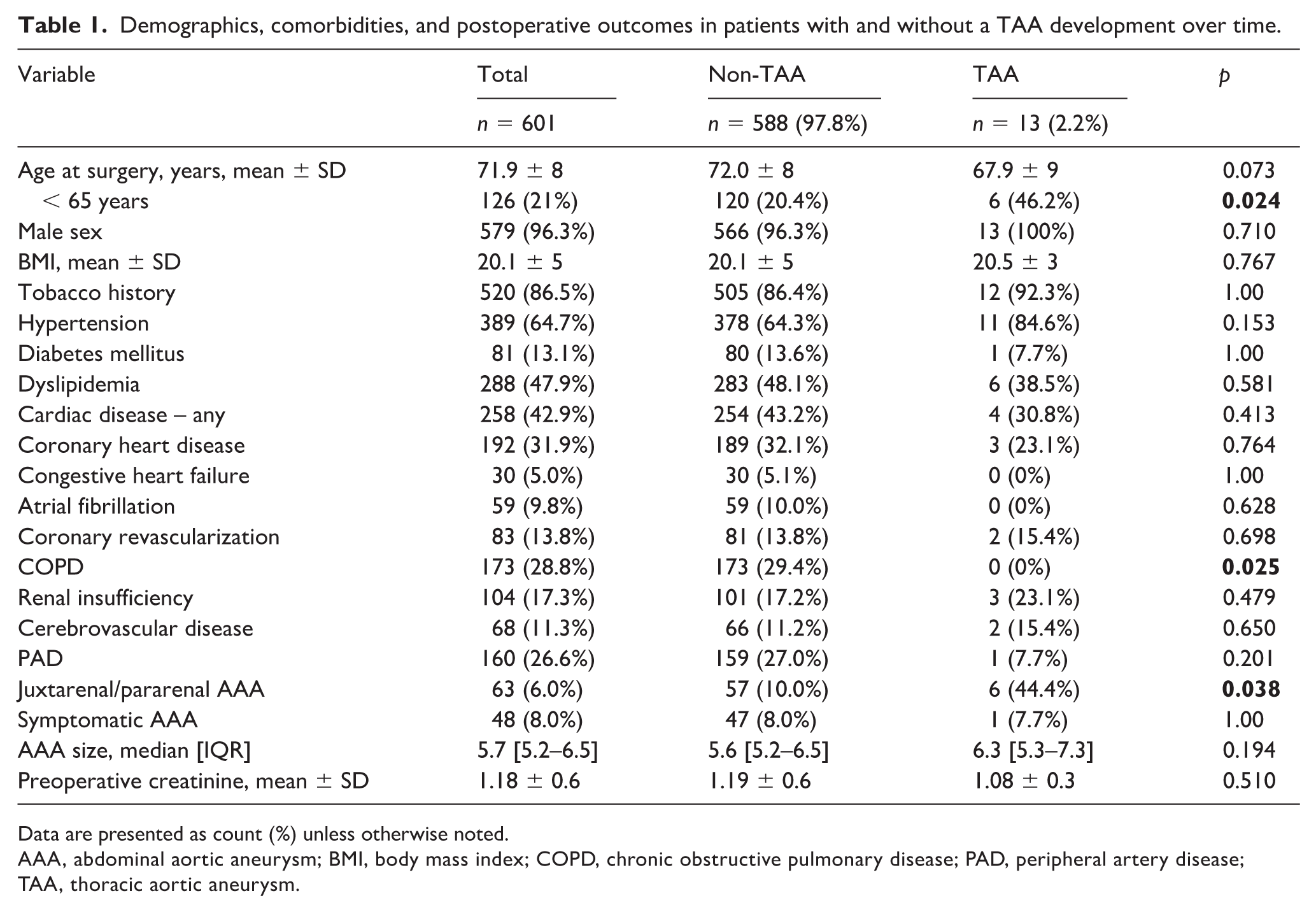
Data are presented as count (%) unless otherwise noted.
AAA, abdominal aortic aneurysm; BMI, body mass index; COPD, chronic obstructive pulmonary disease; PAD, peripheral artery disease; TAA, thoracic aortic aneurysm.
The overall mean follow-up was 82.5 ± 47 months (range 42.3–120.0 months). Thirteen patients (2.2%) developed a TAA over time: eight affected the descending thoracic aorta, three were aortic arch aneurysms, and two were ascending aortic aneurysms. The median timespan until TAA diagnosis was 59.0 months (range 40.0–111.5 months).
An increased risk of TAA after AAA repair was detected for juxtarenal or pararenal AAAs (44.4% vs 10.0%; p = 0.038) and patients under 65 years old (46.2% vs 20.4%; p = 0.024). Chronic obstructive pulmonary disease (COPD) showed a negative statistical association (0% vs 29.4%; p = 0.025) for TAA development. No other associations reached statistical significance.
Aneurysmal degeneration of the thoracic aorta after AAA repair is not uncommon. Our incidence of TAA over time (2.2% at the mean follow-up of 5 years) seems comparable to several previous descriptions, particularly with a meta-analysis by Schlösser and colleagues in 2008 that communicates similar figures (2.2%). 8 This is also concordant with the biggest series, published in the 1980s. 7 However, Chaer and colleagues investigated TAA incidence after diagnosis (not treatment) of AAA and reported a much higher incidence: 10.8% for synchronous and 12.6% for metachronous TAA. 4 Chaer and colleagues, as well as Kalman et al., 5 used a ⩾ 3 cm threshold for defining and an aneurysm in the entire aorta. Conversely, Robinson and colleagues did not mention the diameter of the TAA, although it was the cause of death. We selected a higher diameter threshold for defining aneurysmal degeneration, and included patients undergoing an open surgical repair. 6
Risk factors capable of predicting TAA development are not uniform between studies. Diabetes and tobacco use history, which have shown a protective role, 4 could not be confirmed in our series. However, our series did show a significantly lower incidence of TAA in patients with COPD. This association has not been previously described and might be explained by a lower life expectancy in this group of patients, which shortens the time in which a TAA can develop and be detected. However, our limited sample size prevented an eventual multivariate analysis, and this finding may be influenced by confounders. The paper by Plate et al. also stratified the risk of developing future aneurysms, finding a relationship with preoperative hypertension (p < 0.001) that could not be replicated in our study. 7 On the other hand, connective tissue disorders could be associated with both AAA and TAA development, as well as with aneurysms in other locations, dissections, or penetrating aortic ulcers. Approximately 25% of patients with thoracic aortic disease could have a genetic mutation involved 11 ; however, this was not assessed in our study.
Our paper also suggests that patients with juxtarenal or pararenal AAA have an increased risk of developing TAA (44.4% vs 10.0%; p = 0.038) (Table 1). This is also concordant with the results by Chaer et al., where AAA location (infrarenal AAA) was significantly associated with no TAA development over time. 4
Our study has some additional limitations that deserve to be highlighted. Being retrospective, no information regarding medical treatments associated both pre and post AAA repair could be included and analyzed. Despite all aortic measurements being performed by vascular specialists, no repeated measures were obtained, and subsequently no interobserver agreement analysis was included. Moreover, the small number of detected events (TAA development) could damper some statistical associations and prevent a sufficiently powered multivariate analysis over potential risk factors.
In conclusion, further studies with a larger sample size are needed to confirm our incidence figures. A thoraco-abdominal CTA scan every 5 years after AAA repair should be considered to rule out TAA, as recommended in the most recent guidelines from the European Society for Vascular Surgery. 10
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
