Abstract
Introduction:
Abdominal aortic aneurysm (AAA) is a relevant clinical problem due to the risk of rupture of progressively dilated infrarenal aorta. It is characterized by degradation of elastic fibers, extracellular matrix, and inflammation of the arterial wall. Though neutrophil infiltration is a known feature of AAA, markers of neutrophil activation are scarcely analyzed; hence, the main objective of this study.
Methods:
Plasma levels of main neutrophil activation markers were quantified in patients with AAA and a double control group (CTL) formed by healthy volunteers (HV) and patients with severe atherosclerosis submitted for carotid endarterectomy (CE). Calprotectin, a cytoplasmic neutrophil protein, was quantified, by Western blot, in arterial tissue samples from patients with AAA and organ donors. Colocalization of calprotectin and neutrophil elastase was assessed by immunofluorescence.
Results:
Plasma calprotectin and IL-6 were both elevated in patients with AAA compared with CTL (p ⩽ 0.0001) and a strong correlation was found between both molecules (p < 0.001). This difference was maintained when comparing with HV and CE for calprotectin but only with HV for IL-6. Calprotectin was also elevated in arterial tissue samples from patients with AAA compared with organ donors (p < 0.0001), and colocalized with neutrophils in the arterial wall.
Conclusions:
Circulating calprotectin could be a specific AAA marker and a potential therapeutical target. Calprotectin is related to inflammation and neutrophil activation in arterial wall and independent of other atherosclerotic events.
Introduction
Abdominal aortic aneurysm (AAA) is a complex disorder characterized by permanent dilatation of the aortic diameter. The mechanisms underlying AAA formation and development are not fully understood, but vascular smooth muscle cell imbalance, extracellular matrix degradation, inflammation, and immune response are known features of the disease, as is the presence of intraluminal thrombus in most cases. 1 Inflammatory cell infiltration in AAA has been profusely studied. Although macrophages predominate and have been most extensively investigated, 2 neutrophils are not only present but also actively involved in AAA. 3 Main functions of neutrophils include degranulation, phagocytosis, NETosis, cytokine release, extracellular vesicles release, and oxidative burst. 4 While performing these activities, neutrophils can release metalloproteases, interleukins, and reactive oxygen species, among other molecules, thus contributing to extracellular matrix degradation, inflammation, and oxidative stress, all processes implicated in AAA. 5 In addition, neutrophils may promote increased neutrophil infiltration or even release anti-inflammatory cytokines, thus complicating their role in AAA. Consequently, several markers of neutrophil activation have been extensively analyzed, whereas others remain poorly understood.
To evaluate the hypothesis that some of these molecules are selectively expressed and released from AAA tissues upon neutrophil activation, we have quantified their plasma levels to further explore their potential role as AAA biomarkers. With this purpose, markers of degranulation, NETosis, and cytokine release were specifically analyzed in patients with AAA.
Methods
Plasma and tissue samples
Plasma samples were obtained from 76 subjects: 38 patients with AAA who underwent open surgery or endovascular repair (aortic diameter ⩾ 5.5 cm), and 38 men aged over 55 years without evidence of AAA (19 healthy volunteers [HV] and 19 patients with severe atherosclerosis who underwent carotid endarterectomy [CE]) as a double control group (CTL). The presence of AAA was ruled out by ultrasound scanning or by an additional image technique previously performed for other medical reasons and documented in their clinical record.
Blood was collected in tubes with K2EDTA as an anticoagulant. Tubes were centrifuged at 1811 × g for 30 min at 4°C within 3 h after collection. Plasma was recovered, dispensed in aliquots, and stored at −80°C until used.
An independent set of abdominal aortic tissue samples were obtained from patients undergoing AAA open repair (n = 26) and organ donors (ODs) as controls (n = 11). When surgically feasible, a fragment of inferior mesenteric artery (IMA), nonaffected by aneurysm, was also obtained from patients with AAA (n = 6) as an internal control of healthy tissue. Samples were washed with phosphate-buffered saline pH 7.4 (PBS), immediately snap-frozen in liquid nitrogen, and stored at La Fe Biobank (La Fe University and Polytechnic Hospital) until used.
Cardiovascular risk factors were obtained by reviewing the medical records of patients with AAA and CE and interviewing HVs. Clinical parameters were obtained from the last blood test prior to the intervention or visit.
The present study was performed according to the Declaration of Helsinki and was approved by the ethical committee of the Medical Research Institute Hospital La Fe. Samples and data from subjects included in this study were managed and provided by Biobank La Fe after approval from the scientific and ethical committees. All participants agreed to donate samples to La Fe Biobank and gave written informed consent.
Quantification of plasma neutrophil activation and inflammatory biomarkers
Markers of neutrophil activation were measured in plasma samples as previously described.6,7 Calprotectin, a neutrophil cytoplasmic protein, was quantified with the Human Calprotectin ELISA Kit (Hycult Biotech, The Netherlands). Myeloperoxidase (MPO) and neutrophil elastase (NE) are neutrophil granule proteins realized upon degranulation and neutrophil extracellular traps (NETs) formation. These proteins were measured with the Human MPO ELISA Kit (Abnova, Taiwan) and the Human PMN Elastase ELISA Kit (Abcam, UK), respectively, according with the manufacturer’s indications.
DNA–histone complexes and cell-free DNA (cfDNA) were measured as markers of neutrophil nuclear content using the Cell Death Detection ELISAPLUS Kit (Roche, Germany) and the Quant-iT PicoGreen dsDNA Kit (Life Technologies, CA, USA), respectively.
Circulating interleukin-6 (IL-6) as an inflammation biomarker was quantified in plasma samples using a commercially available ELISA kit: IL-6 ELISA Kit (Diaclone, France). All the experiments were performed following the manufacturer’s instructions.
Protein quantification in tissue samples by Western blot
Protein content was extracted from frozen tissue specimens using the mirVana PARIS Kit (Invitrogen, Thermo Fisher Scientific, MA, USA). Briefly, a cross-section of tissue sample was homogenized with the TissueLyser LT (Qiagen, Germany) (homogenization conditions: 2 beads, 50 mHz, 5 min) in 625 µL cold cell disruption buffer with 7 µL Protease Inhibitor Cocktail (Calbiochem, Merk, Germany). After sonication (3 pulses of 5 s), proteins were distributed in aliquots, snap-frozen in liquid N2, and stored at −80°C until use.
The protein concentration was quantified with the Pierce BCA Protein Assay Kit (Thermo Fisher Scientific) following the manufacturer’s instructions. Next, 20 µg of protein were loaded in a 4–12% SDS-polyacrylamide gel electrophoresis (PAGE) under reducing conditions and transferred to a polyvinylidene difluoride (PVDF) membrane (20 V, 7 min) using the iBlot Gel Transfer System (Life Technologies). Membranes were blocked with 5% nonfat milk in Tris-Buffer solution containing 0.1% Tween-20 (TBS-T) for 1 h at room temperature, and then incubated overnight at 4°C with specific antibodies in the blocking buffer. Finally, membranes were incubated for 1 h with horseradish peroxidase (HRP)-conjugated secondary antibodies (dilution 1:3000) and bands were detected by chemiluminescence using enhanced chemiluminescence (ECL) (GE HealthCare, UK) in an Amersham Imager 600 (GE HealthCare). Primary antibodies were Calprotectin Mouse Monoclonal Antibody (MAC387) (Invitrogen, Thermo Fisher Scientific) at a 1:1000 dilution in TBS-T and GA-PDH rabbit monoclonal antibody (Cell Signaling technology, The Netherlands) at a 1:2000 dilution in TBS-T, as the reference control. Secondary antibodies were ECL-labeled Anti-Mouse IgG, HRP-linked F(ab′)2 fragment from sheep (GE HealthCare), and Goat Anti-Rabbit IgG (H + L)-HRP Conjugate (Bio-Rad, CA, USA).
Calprotectin localization in tissue samples
Frozen tissues were embedded in optimal cutting temperature compound and sectioned in 8–10 µm fragments using a cryostat. The general tissue structure was assessed by staining with Masson’s Trichrome Stain Kit (Polysciences, PA, USA) following the manufacturer’s indications for frozen tissues. Images were acquired with an optical microscope (Leica DMD108).
Calprotectin and NE were detected by immunofluorescence as follows. Tissue sections were fixed with 4% paraformaldehyde in PBS during 20 min at 4°C and then blocked with 3% bovine serum albumin (BSA) and 0.2% triton in PBS for 5 min followed by 1 h incubation with PBS-3% BSA at room temperature. Sections were incubated overnight at 4°C with the primary antibody and after washing three times with PBS-3% BSA they were incubated for 1 h at room te-mperature with the secondary antibody in PBS-3% BSA. Samples were washed two times for 10 min with PBS and incubated for 3 min with an Autofluorescence Quenching Kit (Vector TrueVIEW; Vector Laboratories, CA, USA). After washing for 5 min with PBS, the samples were incubated for 10 min with Hoescht 33342 (Invitrogen, Thermo Fisher Scientific) at 1:1000 in PBS and finally mounted with Vectashield Vibrance Antifade Mounting Medium (Vector Laboratories). Primary antibodies were Calprotectin Mouse Monoclonal Antibody (MAC387) (Invitrogen, Thermo Fisher Scientific) diluted 1:200 and Rabbit Polyclonal to NE (ab21595; Abcam) diluted 1:200. Secondary antibodies were Goat Anti-Mouse (H+L) cross-adsorbed secondary antibody, Alexa Fluor 594 and Goat Anti-Rabbit highly cross-adsorbed secondary antibody, Alexa Fluor plus 488 (Invitrogen, Thermo Fisher Scientific) diluted at 1:200. Fluorescence images were acquired with a confocal microscope (Leica TCS-SP5-AOBS).
Statistical analysis
Continuous variables were summarized as median and IQR and categorical variables were displayed as count and percentage. The Shapiro–Wilk test was performed to assess if data fitted to a normal distribution. Differences in the continuous variables between two groups were compared with the Mann–Whitney test. Categorical variables were compared with the chi-squared method. Correlation between continuous variables was analyzed by the Spearman test. The predictive ability of each biomarker was evaluated by estimating the area under the curve (AUC) for the receiver operator characteristics (ROC). The predictive ability of a combination of variables was evaluated with a multivariable logistic regression model. The analysis was performed using GraphPad Prism v.8.0.1 and v.10 (San Diego, CA, USA) and p < 0.05 was considered statistically significant.
Results
Clinical characteristics of the study subjects
The clinical characteristics and common medications of subjects included in the study are shown in Tables 1 and 2. The main differences were found in age since patients with AAA were older than both CE and HV. Interestingly, the neutrophil count was higher in AAA than in the overall CTL group (p = 0.039), but the lymphocyte count was similar between groups (Table 1).
Clinical characteristics of the subjects with plasma samples included in the study.
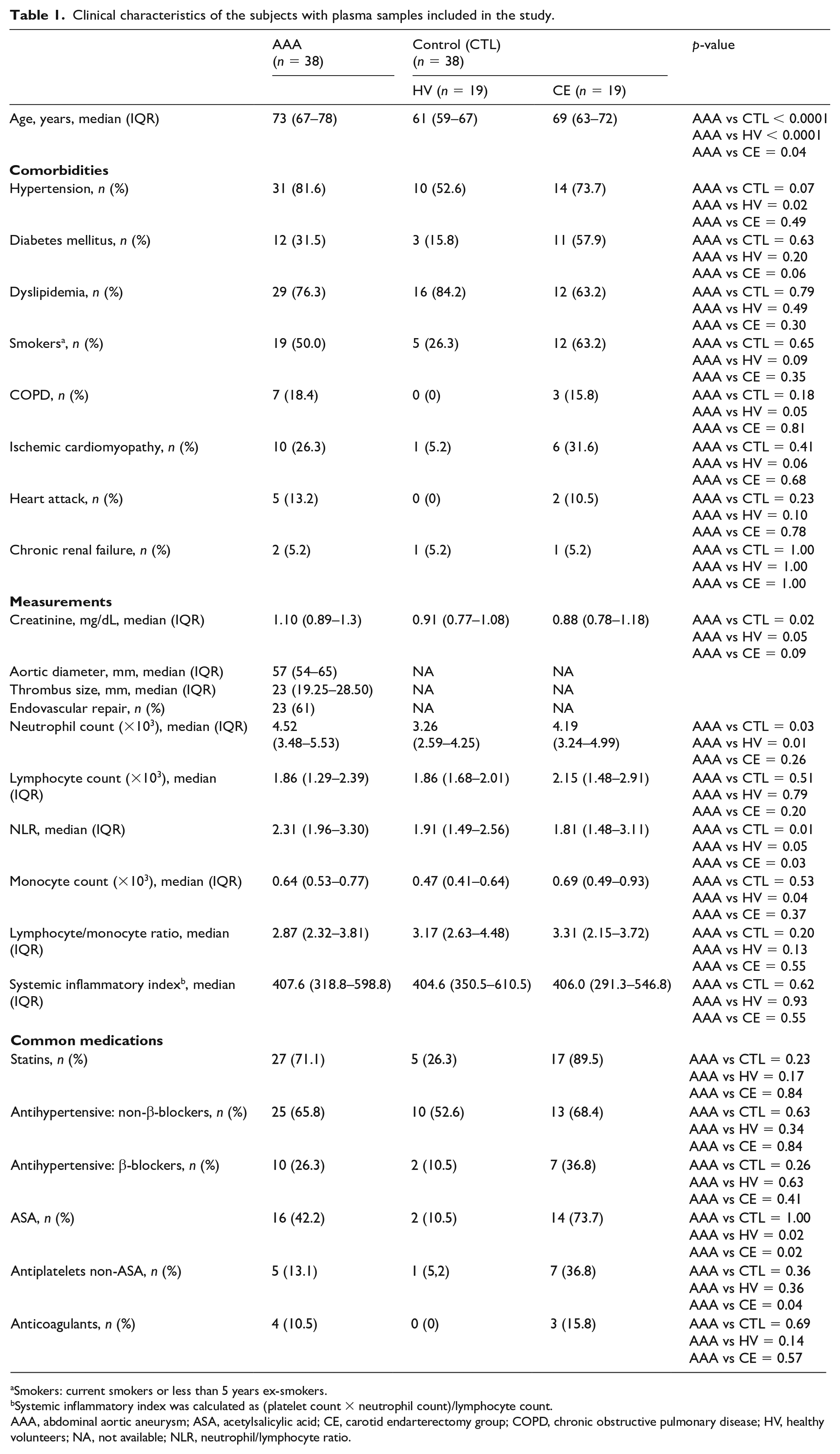
Smokers: current smokers or less than 5 years ex-smokers.
Systemic inflammatory index was calculated as (platelet count × neutrophil count)/lymphocyte count.
AAA, abdominal aortic aneurysm; ASA, acetylsalicylic acid; CE, carotid endarterectomy group; COPD, chronic obstructive pulmonary disease; HV, healthy volunteers; NA, not available; NLR, neutrophil/lymphocyte ratio.
Clinical characteristics of subjects with tissue samples included in the study.
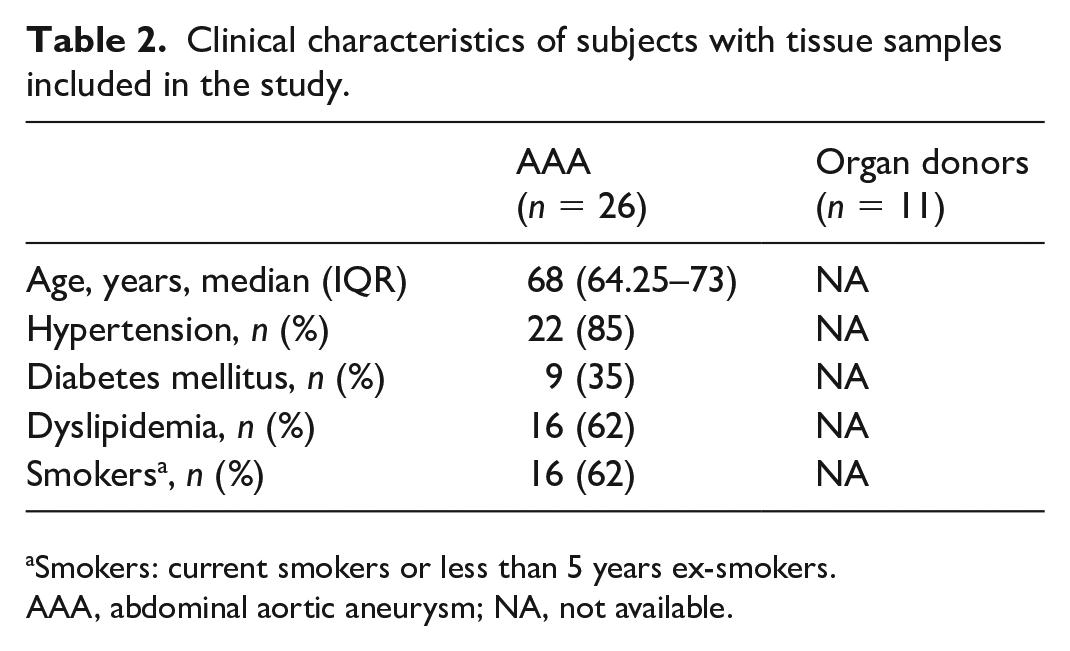
Smokers: current smokers or less than 5 years ex-smokers.
AAA, abdominal aortic aneurysm; NA, not available.
Neutrophil activation markers in plasma samples
Regarding neutrophil activation markers, calprotectin was higher in AAA than in CTL (median, [IQR]: 1073, [707.3–1451] vs 664.5, [551.8–884] ng/mL) (p = 0.0001).
Also, IL-6, an inflammatory cytokine released by several cell types, including neutrophils, showed significantly increased levels in patients with AAA compared with the CTL group (3.90, [2.00–5.46] vs 0.98, [0.28–0.98] pg/mL) (p < 0.0001).
To assess if neutrophil markers were able to discriminate AAA from other vascular disorders, the CTL group was divided into HV subjects and patients with CE. Remarkably, calprotectin levels were higher in patients with AAA than in both HV (700, [568–862] ng/µL) (p = 0.0016) and patients with CE (603, [465–914] ng/µL) (p = 0.0026), yet no differences arose in calprotectin levels between patients with CE and HV (Figure 1).
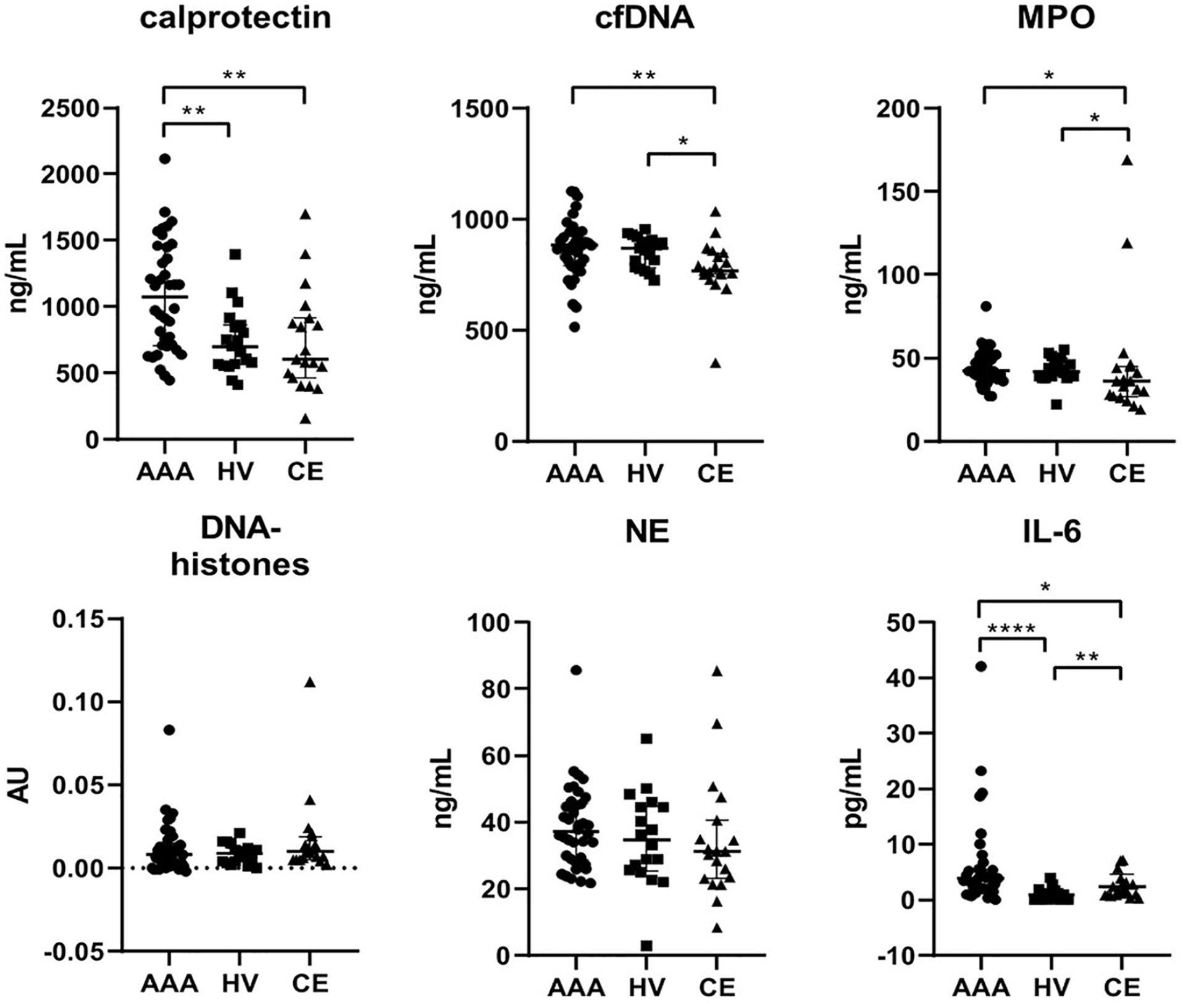
Plasma levels of neutrophil activation markers in patients with abdominal aortic aneurysm (AAA), healthy volunteers (HV), and patients with carotid endarterectomy (CE).
Interestingly, IL-6 was also higher in patients with AAA than in both HV (0.578, [0.03–1.229], p < 0.0001) and patients with CE (1.714, [0.834–3.119], p = 0.0128). IL-6 was also increased in patients with CE compared with HV (p = 0.0021), thus indicating that it could be an unspecific inflammatory marker of vascular disorders (Figure 1).
Additionally, cfDNA and MPO plasma levels were lower in patients with CE than in both patients with AAA (p = 0.0073, p = 0.0191, respectively) and HV (p = 0.0154 and p = 0.0363, respectively) (Figure 1). Thus, both cfDNA and MPO plasma levels could be useful as severe atherosclerosis biomarkers.
The ability of plasma calprotectin and IL-6 to predict AAA was subsequently evaluated. The ROC curve for plasma calprotectin rendered an AUC of 0.75 (95% CI [0.64–0.86], p = 0.0002) when AAA and CTL were compared. According to the Youden index, an optimal cut-off point for plasma calprotectin to distinguish between patients with AAA and the CTL group was established at 613.5 ng/mL, rendering a sensitivity and specificity of 0.92 and 0.47, respectively. The predictive ability of calprotectin for AAA remained similar when comparing AAA with HV (AUC 0.75 [0.63–0.88], p = 0.002). In that case, the best cut-off point was 1132 ng/mL, with a sensitivity and specificity of 0.5 and 0.95, respectively. Similarly, if patients with AAA were compared only with patients with CE, we found an AUC of 0.74 [0.60–0.88], p = 0.0031, and a cut-off of 610 ng/mL, with a sensitivity and specificity of 0.92 and 0.52, respectively (Figure 2). The ROC curve for plasma IL-6 rendered an AUC of 0.79 ([0.69–0.90], p < 0.0001) when AAA was compared with CTL. The optimal cut-off was estimated as 1.944 pg/mL, with a sensitivity and specificity of 0.79 and 0.70, respectively. This predictive ability improved when AAA was compared with HV (AUC 0.89 [0.80–0.98], p < 0.0001) with a cut-off of 1.944 pg/mL, a sensitivity of 0.79 and a specificity of 0.89. However, when comparing patients with AAA with those with CE, the predictive ability of IL-6 slightly decreased (AUC 0.70 [0.56–0.85], p = 0.0135), with a cut-off of 3.12 pg/mL, a sensitivity of 0.58 and a specificity of 0.79 (Figure 2). In addition, a slight improvement in the predictive ability to distinguish AAA from CTL, HV, and CE was observed when both variables were combined in a multivariable regression logistic analysis (AUC 0.81 [0.71–0.91], p < 0.0001), (AUC 0.90 [0.81–0.98], p < 0.0001), and (AUC 0.76 [0.62–0.89], p = 0.0017), respectively. If cfDNA was included as an additional variable, improvement was only observed when patients with AAA were compared with HV (online Supplemental Figure 1).
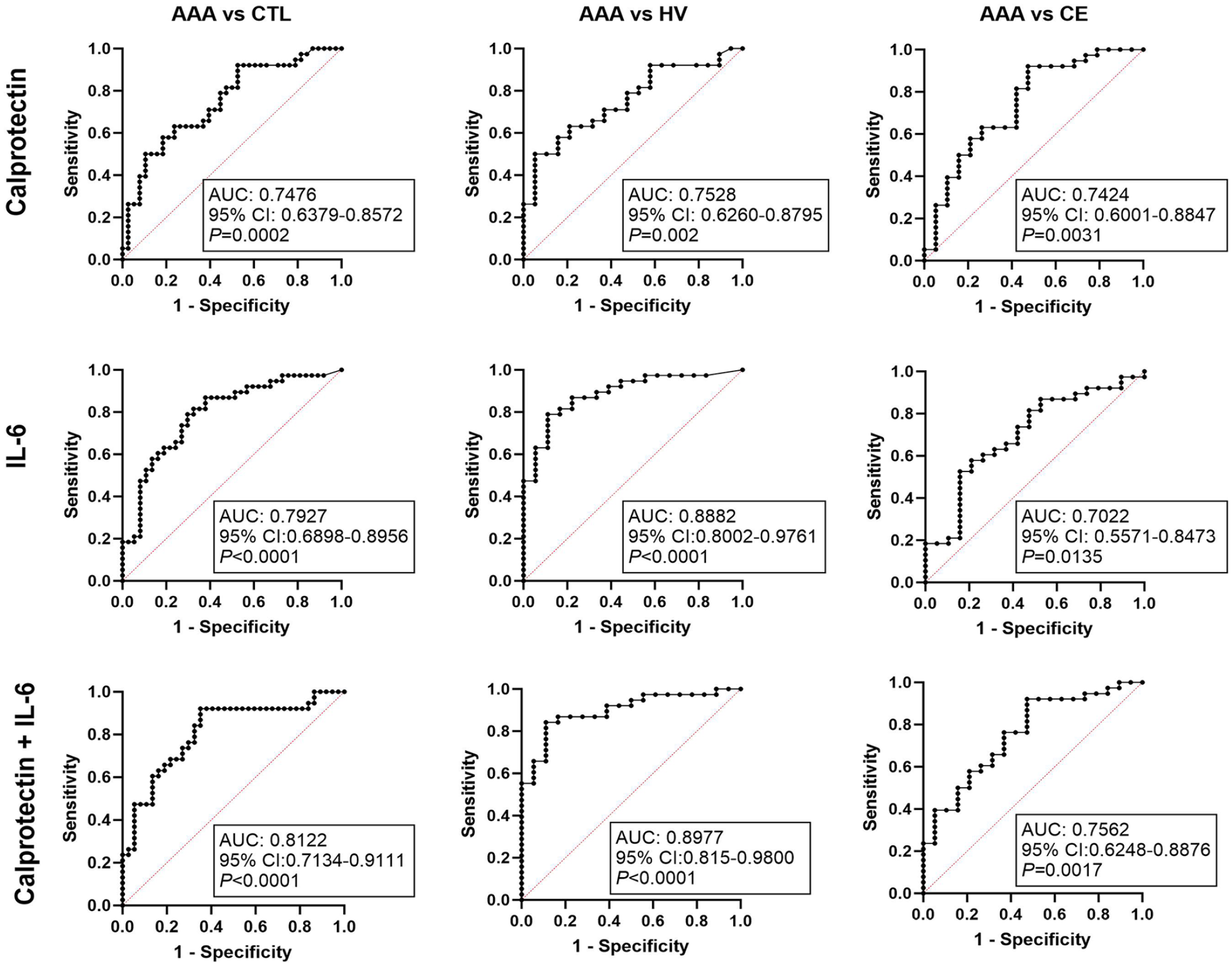
ROC curves of calprotectin, IL-6, and the logistic regression model including both calprotectin and IL-6 combined.
Finally, the correlations between neutrophil markers, clinical parameters, aortic diameter, and thrombus size were assessed (online supplemental Table 1). Interestingly, calprotectin significantly correlated with cfDNA, MPO, NE, and also with neutrophil count. Moreover, calprotectin and IL-6, the two biomarkers with the best predictive ability of AAA, also showed a positive correlation (0.455, p < 0.001). Remarkably, no correlation was found between aortic diameter or thrombus size with the other parameters analyzed. Since patients with AAA were older than both HV and CE, we also evaluated if age correlated with any neutrophil biomarker and other analytical parameters in our set of samples. We have found a correlation of age with IL-6, lymphocyte count, neutrophil/lymphocyte ratio, creatinine, and lymphocyte/monocyte ratio. However, age did not correlate with other parameters including calprotectin, NE, or cfDNA (online supplemental Table 1).
Local dysregulation of calprotectin
Next, we investigated the local expression of calprotectin in an independent set of tissue samples. Calprotectin levels were increased 360% in arterial specimens from patients with AAA compared with those from ODs (relative expression: 1.896 [1.052–3.109] vs 0.525 [0.262–0.617], p < 0.0001), and increased 466% in patients with AAA compared with the IMA samples (0.407 [0.286–0.637], p = 0.0003). Importantly, the expression of calprotectin was similar in OD tissue than in control IMA samples considered as an internal control of healthy tissue (Figure 3; online supplemental Figure 2).
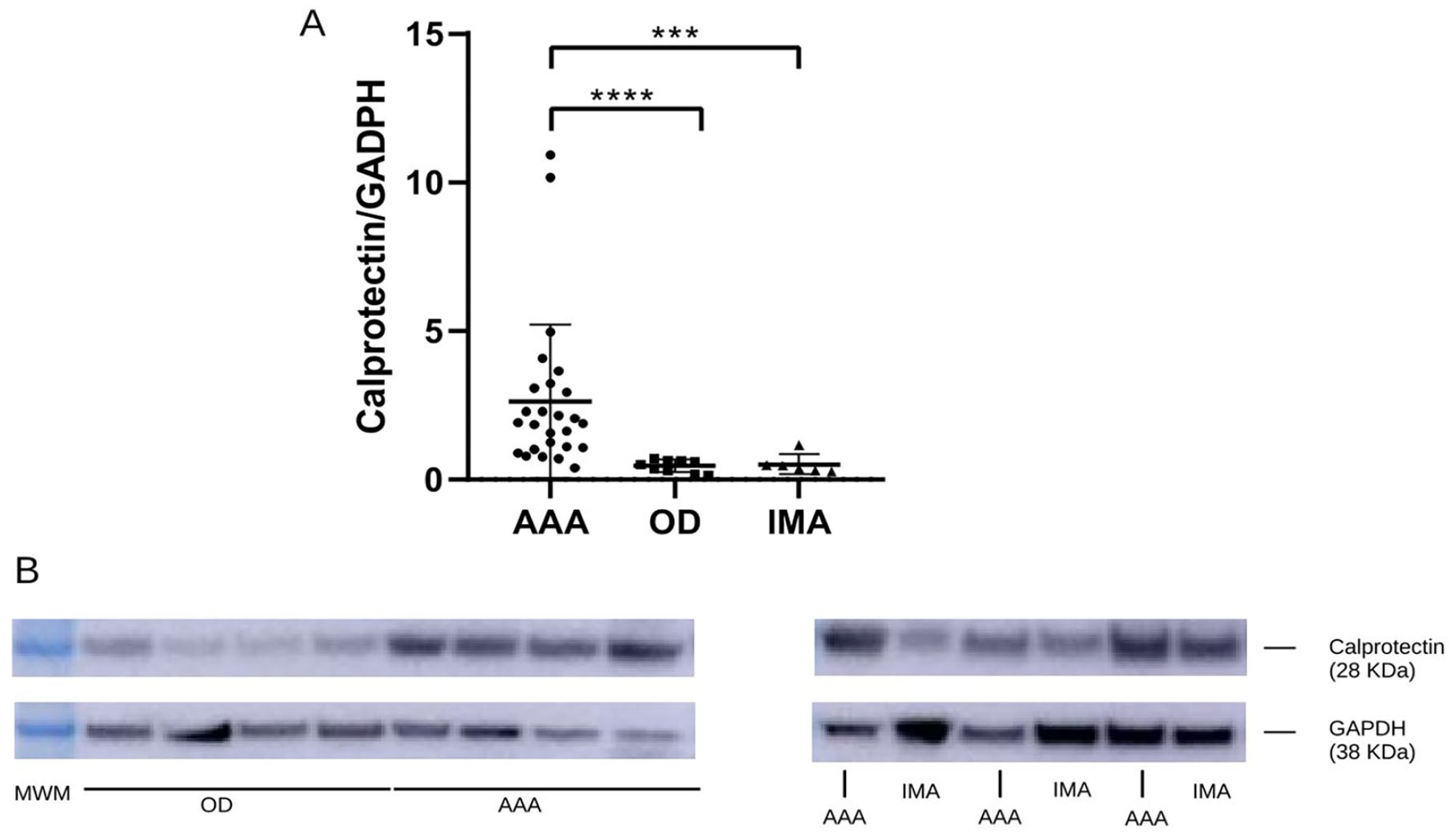
Calprotectin levels in tissue samples of AAA and IMA from patients, and aortic tissue from ODs.
Finally, we analyzed the potential role of neutrophils as a source of calprotectin in AAA affected tissue. Masson’s trichrome staining showed a typical organized structure in OD tissue specimens, and collagen disruption together with cell infiltration was evidenced in AAA tissue samples (data not shown). The release of calprotectin by infiltrating neutrophils in AAA tissue could be demonstrated by immunofluorescent colocalization of calprotectin and NE, a specific neutrophil marker (Figure 4; online supplemental Figure 3).
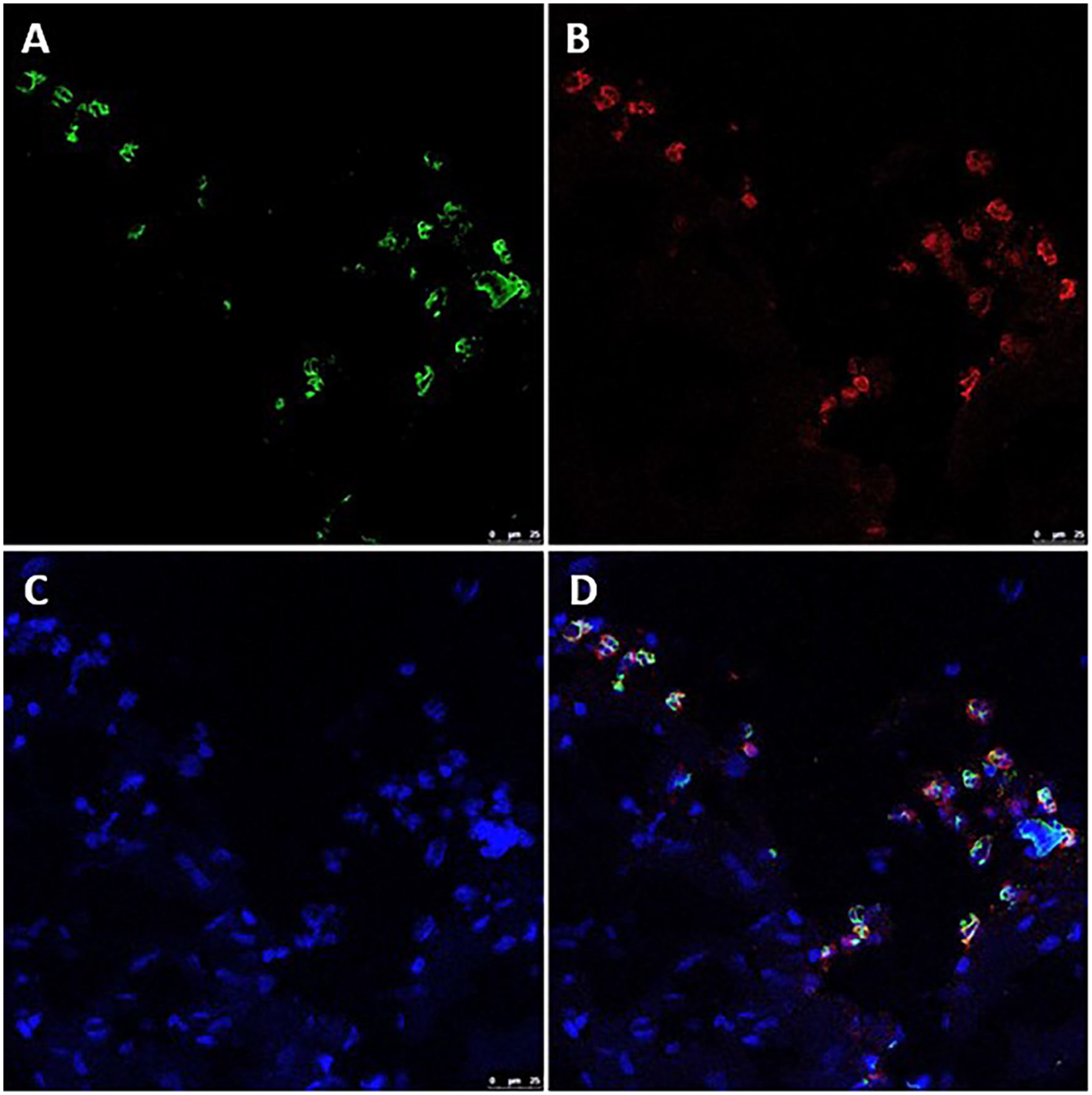
Immunofluorescent colocalization of calprotectin and neutrophil elastase in tissue samples from a patient with abdominal aortic aneurysm.
Discussion
Neutrophil infiltration has been previously described in AAA lesions, but the mechanisms by which they may contribute to AAA formation and development and their potential as a source of biomarkers remains elusive. The main objective of our study was to quantify several neutrophil activation markers associated with degranulation, NET formation and inflammation (calprotectin, MPO, DNA–histone complexes, cfDNA, NE, and IL-6) to determine their potential role as AAA biomarkers.
We observed an increase in plasma calprotectin levels in patients with AAA compared to the overall CTL group, including both HV and CE. Calprotectin is a cytoplasmic heterodimer composed of S100A8/S100A9 subunits and is mainly expressed by neutrophils. It is known to be increased in the circulation in several inflammatory disorders, such as rheumatic diseases 8 and inflammatory bowel disease. 9 In relation to cardiovascular disease, calprotectin has previously been proposed as a cardiovascular risk marker in some population studies. Healy et al. 10 proposed calprotectin levels as predictors of future myocardial infarction, stroke, and cardiovascular death in healthy women. Altwegg et al. 11 also suggested calprotectin as an early marker for the detection of acute coronary syndromes. Even more, the role of calprotectin as a predictor of thrombotic events in cancer patients has previously been suggested by our group.12,13
Notably, in a pilot study in animal models, Moris et al. 14 observed an increase in circulating calprotectin in rats with elastase-induced AAA. Furthermore, calprotectin levels were higher during AAA progression than during AAA development, suggesting that it may be a biomarker of the ongoing process. This finding strengthens our results as we only included patients with relatively large AAAs in our study.
To our knowledge, few groups have investigated the potential role of calprotectin as an AAA biomarker in humans. For example, Hauzer et al. observed increased levels of calprotectin in patients with AAA compared to controls, and interestingly, these levels were reduced after surgery. 15 They also found differences in the levels of soluble receptor for advanced glycation end products (RAGE) between the two groups, 16 but were unable to establish a correlation between both markers (calprotectin or RAGE) and aortic diameter, probably due to limited sample size. 17 Liu et al. 18 also showed increased levels of calprotectin in AAA serum samples. Our results corroborate those of Hauzer et al. and Liu et al., and all together appear to highlight the role of calprotectin as a feasible AAA biomarker. Both Hauzer et al. and Liu et al. included only healthy volunteers as a control group. In contrast, our control group comprised healthy volunteers and carotid endarterectomy patients with severe atherosclerosis, who were analyzed separately to determine whether calprotectin would be able to differentiate AAA from other vascular diseases. Interestingly, calprotectin levels were higher in patients with AAA than in the HV and CE groups, whereas HV and CE showed similar calprotectin levels, highlighting its specificity as an AAA biomarker in cardiovascular context.
Regarding other neutrophil markers, no differences in cfDNA levels were found in patients with AAA compared to CTL. However, when the CTL groups were analyzed separately, a decrease in cfDNA was found in patients with CE, emphasizing the relevance of the clinical characteristics of the control group. Delbosc et al. 19 observed an increase in plasma cfDNA levels in patients with AAA compared to healthy controls, which even correlated with AAA diameter. As no information is available on the clinical characteristics of the control group recruited by Delbosc et al., differences between the clinical groups in both studies may account for these discrepancies. 19
Similarly, we found that MPO levels appeared to be lower in patients with CE, although no differences in plasma MPO levels between the AAA and CTL groups were detected. Our results differ from other groups who found increased MPO levels in AAA. Recently, Brandau et al. 20 reported elevated plasma MPO levels in patients with AAA, not only compared to healthy controls, but also to patients with peripheral artery disease. Of note, their inclusion criteria were subjects with an aortic diameter > 3 cm (i.e., a small aneurysm at baseline with no surgical indication). In contrast, the participants in our study were surgically treated patients with a larger aortic diameter (⩾ 5.5 cm), so the expression of some neutrophil biomarkers might be exacerbated in the early stages of AAA, as previously suggested for some NET biomarkers, 21 but might stabilize in the advanced stages. However, the effect of MPO in larger AAAs appear to be controversial. Whereas Siennicka et al. 22 observed a decrease in plasma MPO levels in patients with AAA, Zagrapan et al. 23 reported an increase in this biomarker. These discrepancies between groups highlight the importance of further research into neutrophil biomarkers in AAA, as different AAA stages, inclusion criteria, and patient characteristics like comorbidities or medication may affect the levels of some of these molecules.
Inflammation of the aortic wall is one of the main hallmarks of AAA development, and the pivotal role of several cytokines in AAA has been studied previously. 24 IL-6 is a well-known marker of inflammation that has been associated with AAA. Consistent with former studies, we observed increased levels of IL-6 in the plasma of patients with AAA. Calprotectin has been suggested to increase IL-6 production in various cell types.25,26 Interestingly, we observed a positive correlation between IL-6 and calprotectin levels, supporting that both proteins may be involved in common pathways.
In agreement with other groups, 20 an increase in neutrophil count was observed in patients with AAA. Additionally, a correlation between neutrophil count and both IL-6 and calprotectin also supports the potential role of neutrophils in the inflammatory context of AAA.
Calprotectin and IL-6 are both inflammatory markers associated with various diseases. However, in the specific context of AAA, IL-6 has been extensively studied and reviewed,24,27,28 whereas calprotectin has been hardly analyzed. In our study, both calprotectin and IL-6 can discriminate between subjects with and without AAA with a high and comparable sensitivity and specificity, although calprotectin seems to be slightly better at discriminating AAA from other serious vascular diseases. Moreover, IL-6 levels seem to vary with age, probably due to systemic inflammation or inflammatory aging, 29 but there seems to be no consensus on the effect of age on circulating calprotectin level. In fact, we have observed a positive correlation between age and IL-6; however, no correlation was detected between age and calprotectin in our set of samples, reinforcing previous studies.30,31 Taking these results together, calprotectin seems to be a more reliable low-invasive biomarker for AAA than IL-6.
Interestingly, although differences between AAA and CTL were only found for inflammatory molecules (calprotectin and IL-6), most neutrophil markers were correlated among them.
Finally, we verified the local increase of calprotectin in the arterial wall of patients with AAA, not only compared with ODs but also with healthy tissue from patients with AAA, confirming that circulating calprotectin levels are probably due to arterial inflammation. Furthermore, plasma calprotectin and NE levels correlated and both markers colocalized in AAA tissue samples, which would support neutrophils as the main source of calprotectin in patients with AAA. It is worth noting that, although other inflammatory cells like monocytes could express calprotectin, it has been previously demonstrated that it is mainly released by neutrophils. 32 Consequently, in recent years, calprotectin has been assumed to be a marker of neutrophil-driven inflammation in several diseases,33–36 and thus it seems reasonable to propose that calprotectin could be involved in neutrophil activation and inflammation in the context of AAA.
Taken together, our findings suggest that neutrophil activation is a feature of AAA, and the release of cytokines and other inflammatory markers may be the most relevant neutrophil function in an advanced state of the disease.
Study limitations and strengths
The main limitation of our study is the rather limited sample size of each clinical group studied, consistent with a pilot study. In addition, as open surgery is being progressively replaced by endovascular repair whenever possible, the collection of AAA and paired healthy IMA, as well as paired plasma-tissue samples, becomes more complex. In addition, all patients studied had large aneurysms (maximum diameter ⩾ 5.5 cm), fulfilling the indication of surgical repair, and thus in an advanced state of the disease. Complementary analysis of patients with small aneurysms would be useful to better understand the potential role of calprotectin as a progression biomarker.
Otherwise, a strength of our study is that our control group includes healthy controls as well as patients with severe atherosclerosis, guaranteeing that our results are not biased by common features of vascular diseases.
Finally, our results are preliminary and need to be validated in larger cohorts and multicenter studies as the first step for their clinical application. Additionally, longitudinal studies are required for evaluating the ability of calprotectin to predict AAA progression. Emerging potential AAA biomarkers, such as calprotectin, combined with classical risk factors and current imaging techniques, may help to develop new clinical scores for better stratifying individual risk of rupture, thus contributing to a more personalized medicine.
Conclusions
Plasma calprotectin is increased in patients with AAA compared with both patients undergoing carotid endarterectomy and healthy individuals. Moreover, AAA tissue has an increased expression of neutrophil-derived calprotectin. Accordingly, the increase in plasma calprotectin levels could be related to damage in arterial tissue. Altogether, our results suggest that calprotectin could become a new reliable biomarker for AAA in the context of cardiovascular diseases.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241231494 – Supplemental material for Calprotectin as a new inflammatory marker of abdominal aortic aneurysm: A pilot study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241231494 for Calprotectin as a new inflammatory marker of abdominal aortic aneurysm: A pilot study by Emma Plana, Julia Oto, Raquel Herranz, Pilar Medina, Fernando Cana and Manuel Miralles in Vascular Medicine
Footnotes
Acknowledgements
La Fe Biobank (La Fe University and Polytechnic Hospital) is acknowledged for the storage and management of tissue samples (PT17/0015/0043). The Microscopy Unit (La Fe University and Polytechnic Hospital) is acknowledged for its support and collaboration.
Declaration of competing interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Instituto de Salud Carlos III (ISCIII) (PI20/01171, PI20/00075, FI21/00171), co-funded by the European Union (ERDF, “A way to make Eurpe”).
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
