Abstract

In patients with symptomatic lower extremity peripheral artery disease (PAD), supervised exercise training (SET) is among first-choice therapeutic options.1,2 Walking is the common training modality. 2 However, some patients have poor SET adherence, 3 suggesting the need for alternative training modalities.
Eccentric exercise (lengthening of muscle while producing force) could be an appealing option. For the same absolute workload, eccentric exercise elicits lower cardiovascular stimulation, rate of perceived exertion, and metabolic requirement than concentric exercise (shortening of muscle while producing force).4,5 In addition, for an exercise performed at a similar metabolic demand (similar oxygen consumption), eccentric exercise resulted in greater workload (four to fivefold) compared to concentric exercise.4,5 Previous investigations demonstrated that eccentric exercise training induces positive muscular adaptations in individuals with and without chronic diseases. 6 Descending stair walking (DSW), a functional form of eccentric exercise, improved muscle strength and function compared to ascending (concentric) stair walking (ASW) in older patients with and without chronic diseases.7–9 To our knowledge, the feasibility, safety, and the acute (single session) physiological responses of DSW have never been investigated in patients with PAD.
The rationale for introducing this new exercise modality may reside in its potential positive muscular alterations, despite lower cardiovascular stimulation and less exercise-induced ischemia. First, eccentric exercise may allow patients with PAD to perform (high-load) muscular exercise with lower cardiovascular stress, which may be severe during concentric exercise. 10 This seems to be an important determinant since skeletal muscle myopathy, high prevalence of sarcopenia, and impaired muscular function have been observed in patients with PAD.11–15 Second, the lower metabolic requirement of eccentric exercise may induce less exercise-induced ischemia. This might be associated with less claudication pain during exertion. Since claudication pain has a huge impact on daily life activities and is one of the main reasons to avoid exertion, the proposal of eccentric exercise seems judicious and may also induce a greater exercise appreciation. This could also have some benefits in patients with more severe exercise-induced ischemia who are unable to perform (flat) ground walking.
This pilot study aimed to investigate the cardiovascular and muscular responses to a single session of DSW and ASW in patients with symptomatic PAD. It was hypothesized that, for the same mechanical work, DSW would elicit (1) lower cardiovascular response and rate of perceived exertion and (2) lower muscle oxygen desaturation (less exercise-induced ischemia) and less claudication pain than ASW.
Symptomatic patients with chronic PAD (Fontaine stage II) with stable uni- or bilateral lower limb claudication were included. The study was approved by the local ethics committee and was conducted according to the Declaration of Helsinki. All subjects provided written, voluntary, informed consent before participation.
The exercise sessions were performed in a five-floor building at the Lausanne University Hospital. To match the total mechanical work during DSW and ASW, the number of steps and the time required to perform the total number of steps need to be similar. To that end, a single fixed step cadence (varying from 45 to 55 steps·min−1), delivered with an acoustic metronome, was chosen for each patient, ensuring that the same parameters were used during both exercise sessions. Each patient performed a single exercise session of DSW and ASW, interspaced with 1–2 weeks. The order was randomly assigned. Before each exercise session, patients performed a 5-min warm-up at 40 W on a cycle-ergometer. Each exercise session consisted of six sets of 2 min each interspaced with 2 min of resting. During rest, patients rested in a seated position and ascended or descended to the starting point (accompanied by the investigator) with an elevator. Each exercise session was performed without using the stair handrail. The number of stairs and floors performed during each set depended on the step cadence. At 45 steps·min−1, patients performed four floors (68 stairs), including three steps at the landing. At 50 steps·min−1, patients performed four floors (68 stairs), including five steps at the landing. At 55 steps·min−1, patients performed five floors (85 stairs), including three steps at the landing. The height of each stair was 17 cm.
At rest and after each set, heart rate (HR), systolic blood pressure (SBP), rate of perceived exertion (RPE) on a Borg’s scale, and claudication pain on a visual analogue scale (VAS; 0: ‘no pain’; 10: ‘maximal pain’) were assessed. Calf oxygen saturation (StO2) of the more symptomatic leg was measured by the near-infrared spectroscopy (PortaMon, Artinis, The Netherlands). Patients were asked to rate their delayed-onset muscle soreness (DOMS) on VAS (0: ‘no soreness’; 10: ‘maximal soreness’) at 24 h, 48 h, and 72 h postexercise. Maximal voluntary isometric contraction (MVC-iso) at the ankle joint was assessed pre-, immediately post-, and 48 h postexercise. Normalized MVC-iso (NMVC-iso; pre-exercise = 100%) was calculated. Linear mixed models (condition × duration) were performed to compare the different outcomes following the exercise sessions. Statistical analyses were performed with SPSS 27 software (IBM Corporation, Armonk, NY, UK).
Nine patients with symptomatic PAD were included (age: 62.0 ± 2.4 years, body mass index: 25.4 ± 1.6 kg·m−2, ankle–brachial index in the more symptomatic limb: 0.77 ± 0.05, 66% men, 33% type II diabetes, 56% hypertension, 67% current smokers, 78% dyslipidemia, 33% prior arterial revascularization).
All the patients completed DSW and ASW. Seven patients performed at 55 steps·min−1, one patient at 50 steps·min−1, and one patient at 45 steps·min−1. The mean total number of steps during DSW and ASW was 487 ± 15.
There was a significant condition (p ⩽ 0.001), duration (p ⩽ 0.001), and condition × duration interaction effect (p ⩽ 0.001) for the HR and SBP (Figures 1A and 1B). Compared to ASW, HR and SBP were significantly lower during DSW at each repetition (p ⩽ 0.001).
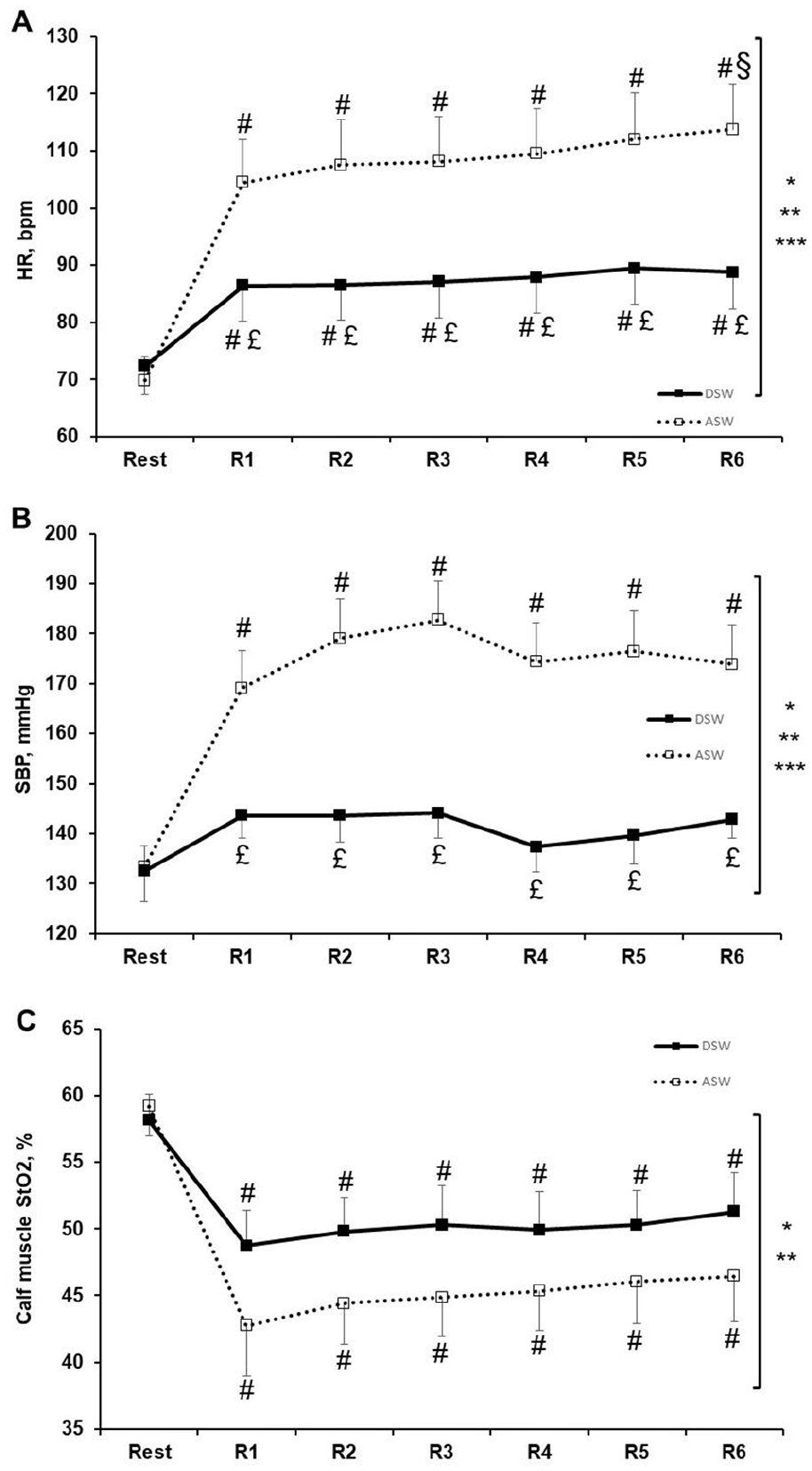
Heart rate (
There was a significant condition (p ⩽ 0.001) and duration (p ⩽ 0.001) effect, with no condition × duration interaction effect for the calf StO2 (Figure 1C), RPE, and claudication pain (online supplemental table). Compared to ASW, calf StO2 was significantly higher (p ⩽ 0.001), and RPE and claudication pain were significantly lower (p ⩽ 0.001) during DSW.
There was a significant condition (p = 0.035) and duration (p = 0.002) effect, with no condition × duration interaction effect for DOMS. Compared to ASW (24 h: 1.3 ± 0.6; 48 h: 0.9 ± 0.4; 72 h: 0.6 ± 0.4), DOMS was significantly higher following DSW (24 h: 2.7 ± 0.7; 48 h: 1.7 ± 0.7; 72 h: 0.5 ± 0.4; p = 0.035).
There was no significant condition effect, with a significant duration (p ⩽ 0.001) and condition × duration interaction effect (p = 0.002) for the NMVC-iso. Compared to ASW, NMVC-iso was significantly higher immediately postexercise (DSW: 89 ± 3%; ASW: 78 ± 3%; p = 0.002) and significantly lower 48 h postexercise (DSW: 94 ± 3%; ASW: 103 ± 2%; p = 0.030) following DSW.
When matched for mechanical work, lower muscle oxygen desaturation (less exercise-induced ischemia) was observed during DSW, suggesting a better match between oxygen delivery and consumption during eccentric exercise in patients with symptomatic PAD. This was associated with less claudication pain and cardiovascular response, as previously shown.7,9
As previously reported,4,5 DSW induced greater postexercise muscle soreness and 48 h postexercise strength loss, attesting muscle damage in the following days. This could be unfavorable for patients’ adherence. However, after a second bout of eccentric exercise, the magnitude of muscle damage is attenuated, suggesting a protective adaptation.4,5 This finding may suggest that eccentric exercise should be gradually introduced during rehabilitation in naïve individuals. 4
Some limitations exist. First, the application of this exercise protocol would be difficult for elderly patients with PAD with concomitant arthritis or for those with balance problems. Second, we evaluated the effects of a single exercise session with outcomes up to 72 h postexercise only. Further studies are needed to evaluate long-term effects of DSW. Indeed, since stimulating skeletal muscle ischemia or improving cardiovascular and endothelial function are the main postulated mechanisms usually linked to improved walking performance, 2 the potential benefits of DSW training on muscle and walking capacities remain to be determined in patients with PAD. Recent investigations showed that resistance training programs, which induce no or less ischemia during exertion and have less effect on cardiorespiratory fitness, are also effective to improve walking performance in these individuals. 16 Notably, the improved walking performance correlates with lower limb strength gain following training. 16 Overall, this indicates that muscular adaptations may (1) occur with low levels of ischemia and cardiovascular stimulation during exertion, and (2) play a pivotal role in improved physical function in patients with PAD. In this context, DSW could be an appealing training modality inducing positive muscular and functional adaptations in these individuals.
This is the first study showing that DSW is a safe and promising exercise modality in patients with symptomatic PAD, especially for those unable to perform (flat) ground walking. These results provide guidance for developing new targeted rehabilitation strategies and investigating eccentric exercise in these individuals.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211058784 – Supplemental material for Descending stair walking in patients with symptomatic lower extremity peripheral artery disease: A pilot study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211058784 for Descending stair walking in patients with symptomatic lower extremity peripheral artery disease: A pilot study by Stefano Lanzi, Paula Nussbaumer, Luca Calanca, Lucia Mazzolai and Davide Malatesta in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
