Abstract
Introduction
Apixaban is a direct oral anticoagulant (DOAC) approved for multiple indications. The need to interrupt anticoagulation in the perioperative period is common. Approaches for the discontinuation of DOACs are based upon the pharmacokinetic parameters of the drug in the context of individual patient and procedural characteristics. The US apixaban product insert specifies the last dose be held at least 24 hours before a low bleeding risk procedure and at least 48 hours prior to elective surgery or invasive procedures with a moderate or higher risk of bleeding. 1 This is based upon a population half-life of ~12 hours, only modest decreases in drug clearance with hepatic or renal dysfunction, and linearity of pharmacokinetics across clinically used doses. However, population pharmacokinetic methods have noted moderate between-patient variation based upon known parameters such as age, size, and renal function, with unexplained covariates accounting for ~30% of variation. 2 The additive effects of multiple patient characteristics may impact apixaban clearance. Treating physicians may not be aware of all clinical factors that could impact apixaban clearance. In addition, the clearance of apixaban declines after orthopedic surgery by ~25%, which returns to baseline by postoperative day 4. 3 All of these factors highlight the potential that in actual use, expected apixaban exposures may diverge from model predicted values. The effectiveness of a perioperative apixaban cessation approach in the setting of actual practice with a heterogonous patient population was studied by Douketis in the PAUSE trial. 4
We hypothesized that discontinuation of apixaban for at least 48 hours prior to surgical procedures in a naturalistic observational setting would result in a clinically insignificant residual anticoagulation as defined by an apixaban plasma concentration < 30 ng/mL.
Methods
This was a single site, prospective, observational study of patients receiving standard of care perioperative management of apixaban anticoagulation (ClinicalTrials.gov Identifier: NCT02935751). The study population was patients who were already receiving treatment with apixaban for nonvalvular atrial fibrillation or venous thromboembolism (VTE) and who required an elective major surgical or invasive procedure.
All patients had a plasma apixaban and anti-Xa level drawn at steady state at the pre-admission clinical visit and on the morning of surgery. The steady state concentration collection was taken at the pre-admission visit. The time of collection relative to last dose of apixaban was not fixed. Apixaban was stopped and restarted at the discretion of the treating physician, and there was no protocol-specified or suggested interval for cessation. Institutional guidance suggested cessation 48 hours prior to a surgical procedure. Adverse events and bleeding events were collected to 30 days postprocedure. Major bleeding was defined as fatal or in a critical organ, nonsurgical bleeding with a 2 g/dL decrease in hemoglobin or requiring transfusion of 2 units or more of blood, surgical site bleeding that required surgical intervention or resulted in hemarthrosis within 30 days of cessation of therapy. Arterial thromboembolic events, VTE and death by any cause were recorded if they occurred within 30 days of the last dose of apixaban (Figure 1).
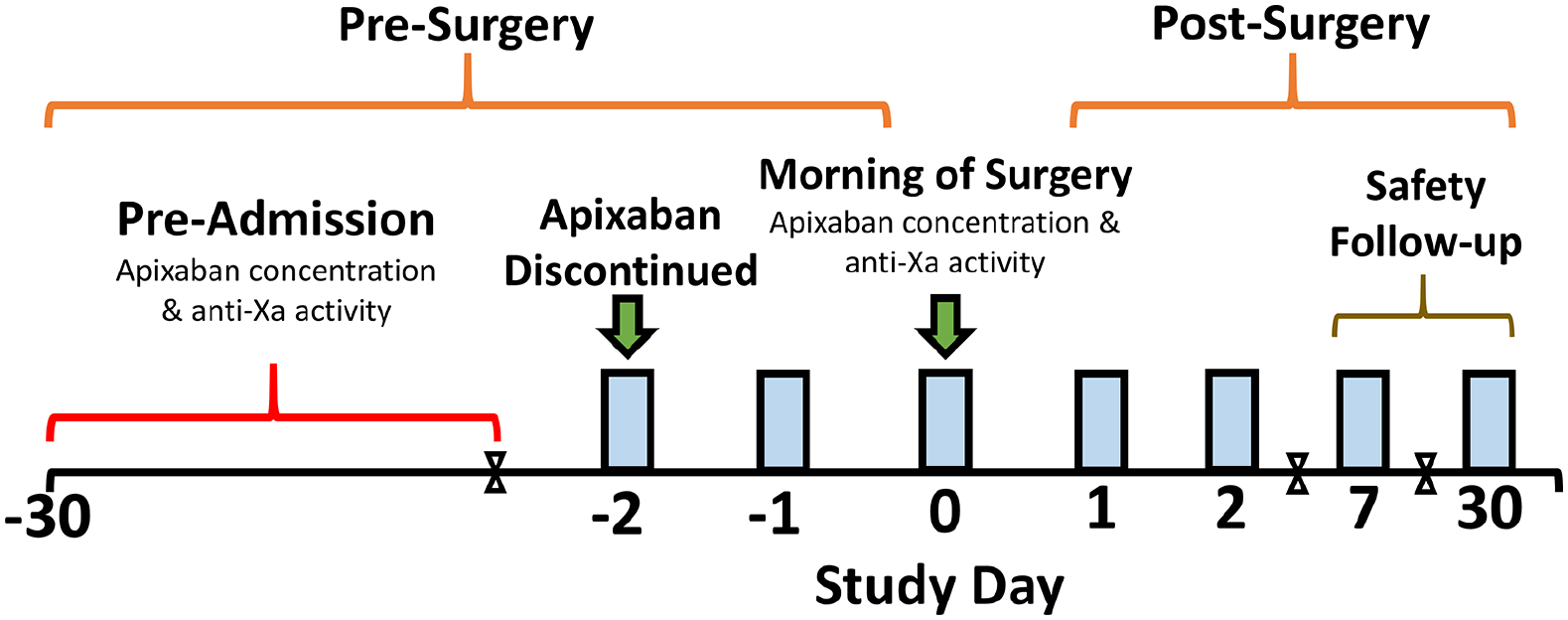
Study design. Timing of cessation of apixaban prior to surgery was at the discretion of the treating physician.
The Thomas Jefferson University Institutional Review Board approved the study, and all patients provided written informed consent.
Consecutive patients were assessed for study participation eligibility in the pre-admission testing center. Inclusion criteria included adults greater than 18 years of age; on long-term anticoagulation with apixaban (5 mg or 2.5 mg twice daily) for treatment of nonvalvular atrial fibrillation or VTE; scheduled for elective surgery or invasive procedure which required anticoagulation interruption; and able to adhere to the apixaban interruption protocol at the time of enrollment. Patients were excluded if they had more than one procedure planned within 30 days; creatinine clearance less than 30 mL/min; cognitive impairment or major psychiatric illness; previous study participation or participation in another clinical trial. Patients with a creatinine clearance < 30 mL/min were excluded at the time of this study based on our hospital’s guidelines for the use of DOACs with creatinine clearance below this value at the time the protocol was written. VTE prophylaxis is not routinely provided postoperatively pending therapeutic DOAC re-initiation. A log of doses taken by the patient for the 7 days prior to surgery was recorded. Before the procedure, patients were categorized as having a high or low bleeding risk procedure according to a prespecified classification listed in online Supplemental Table 1. Patients were followed daily during their hospitalization to assess resumption of apixaban and document hemorrhagic or thrombotic events. Patients received phone contact at days 7 and 30 postdischarge to assess bleeding, thrombotic events or hospital readmission.
Statistical methods
The primary outcome of the ADIOS study was the estimation of the proportion of patients who achieved a plasma apixaban concentration of ⩽ 30 ng/mL following at least 48 hours of discontinuation prior to surgery or invasive procedure. This threshold is based upon International Society on Thrombosis and Haemostasis recommendations for reversal, 5 expert opinion as clinically important,6–8 and represents a value below that seen after three to four half-lives based upon published pharmacokinetic parameters in patient populations. 2 The secondary outcome was the incidence of postoperative arterial or venous thromboembolic events, major bleeding and clinically significant nonmajor bleeding complications. Based on similar studies performed9,10 in which 86% of patients had a plasma level at or below 30 ng/mL at least 48 hours after discontinuation, we hypothesized that the proportion in the population was likely to be between 80% and 95% for apixaban. A total of 130 patients were chosen as having a reasonably narrow Clopper–Pearson 11 exact 95% CI at the lowest anticipated observed proportion (CI width of 0.15 if proportion is 0.80) and high confidence in the location of the population proportion if nearly all or all patients reached the threshold of < 30 ng/mL (CI widths of 0.08 and 0.03 for proportions of 0.95 and 1.00, respectively) (online Supplemental Table 2). All study variables were summarized by means and SD, or, if substantially skewed, by medians with the first and third quartiles using R version 4.0.2. Plots were generated using the R package ggplot2 (R Foundation for Statistical Computing, Vienna, Austria). Pearson’s correlation coefficients were used to estimate correlation between the apixaban concentration and anti-Xa apixaban activity. Two-sided significance was set at α = 0.05.
Analytic procedure
Plasma concentrations of apixaban were determined by liquid chromatography-tandem mass spectrometry using an AB Sciex API 3200MD, Framingham, MA, USA. 12 Commercial calibrators (Hyphen BioMed, Rue d’Eragny, 95000 Neuville-Sur-Oise, France) were used, along with rivaroxaban-d4 (Santa Cruz Biotechnology, Santa Cruz, CA, USA) as an internal standard. The calibration curve was linear over the range 0–600 ng/mL. Between-run precisions were 8.4% (at 30 ng/mL) and 4.7% (at 200 ng/mL). The lower limit of quantitation (LLOQ) was 4 ng/mL. No analytical interferences or ion suppression effects have been observed in precedent LC-MS/MS assays. 13 Anti-Xa was performed on ACL-TOP500 using Biophen Heparin LRT and Biophen Apixaban calibrator, both from Aniara Diagnostica (West Chester, OH, USA). Two level controls (~72 and ~275 ng/mL; Biophen Apixaban control) were performed on each run.
Results
Study population
The enrollment period for the ADIOS trial lasted 18 months, from December 2016 through July 2018. Of the 130 patients enrolled in the trial, 111 paired blood samples were available for the primary outcome of change in plasma apixaban concentration (Figure 2). There were 19 patients who were excluded due to missing variables, including missing apixaban concentrations at pre-admission testing, day of surgery, or both. The primary reason for a missed blood draw was last minute cancellation of the scheduled surgery (six patients), and five patients were missed because the operating room schedule in the electronic medical record was inaccurate and patients were taken for surgery earlier than anticipated. Two of the patients did not have a sample drawn because they had difficult vein access, and the study coordinator could not get the sample after three attempts. Patient apixaban dose was available for all 111 patients with paired blood samples. Patient demographics and disposition are listed in Table 1. The proportion of patients achieving apixaban concentrations of less than or equal to 30 ng/mL was 93.7% (Figure 3).
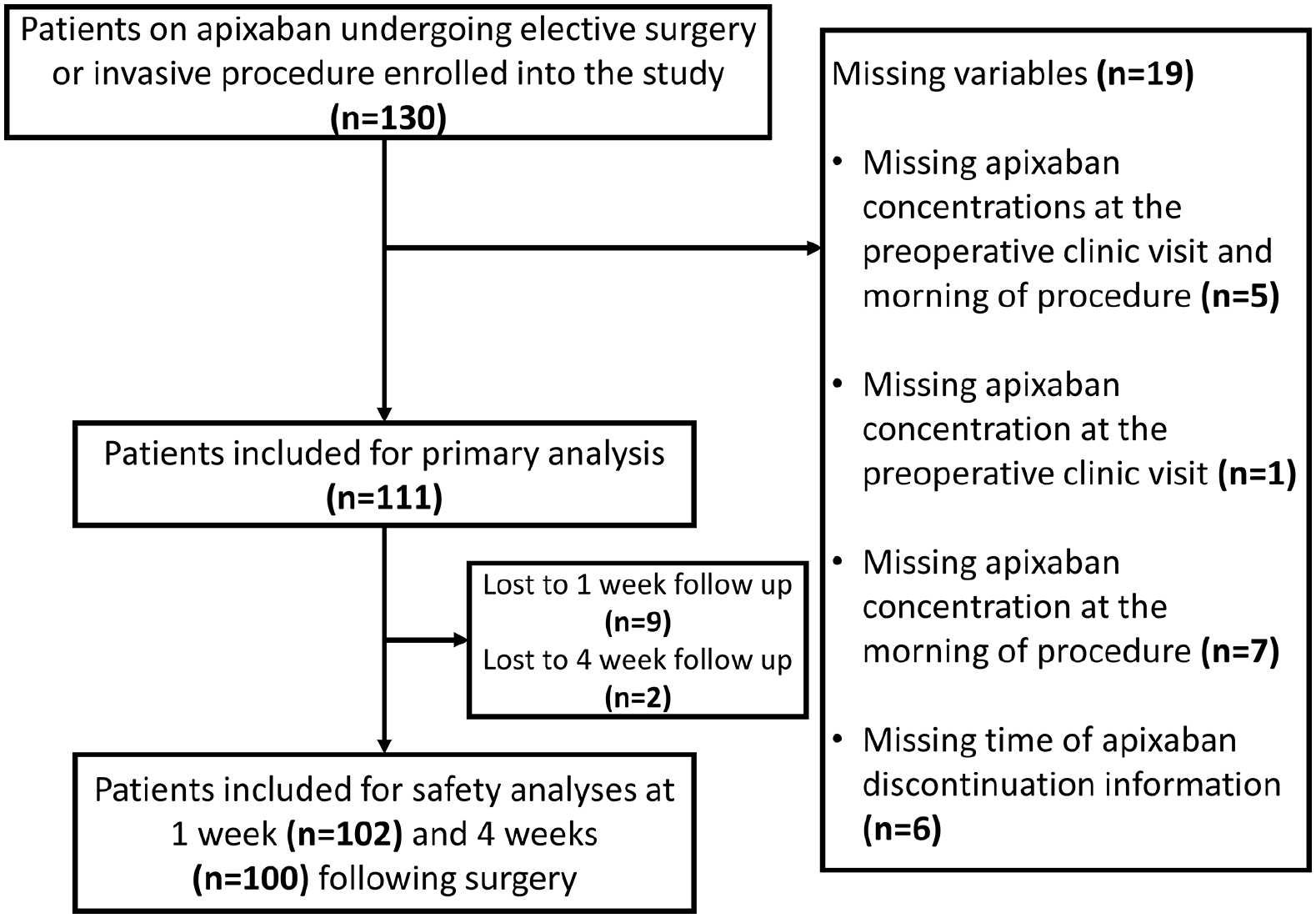
Study subject disposition.
Baseline patient characteristics.

Twelve patients were taking apixaban for multiple indications: seven for reduction of risk of stroke in nonvalvular atrial fibrillation
TIA, transient ischemic attack; VTE, venous thromboembolism.
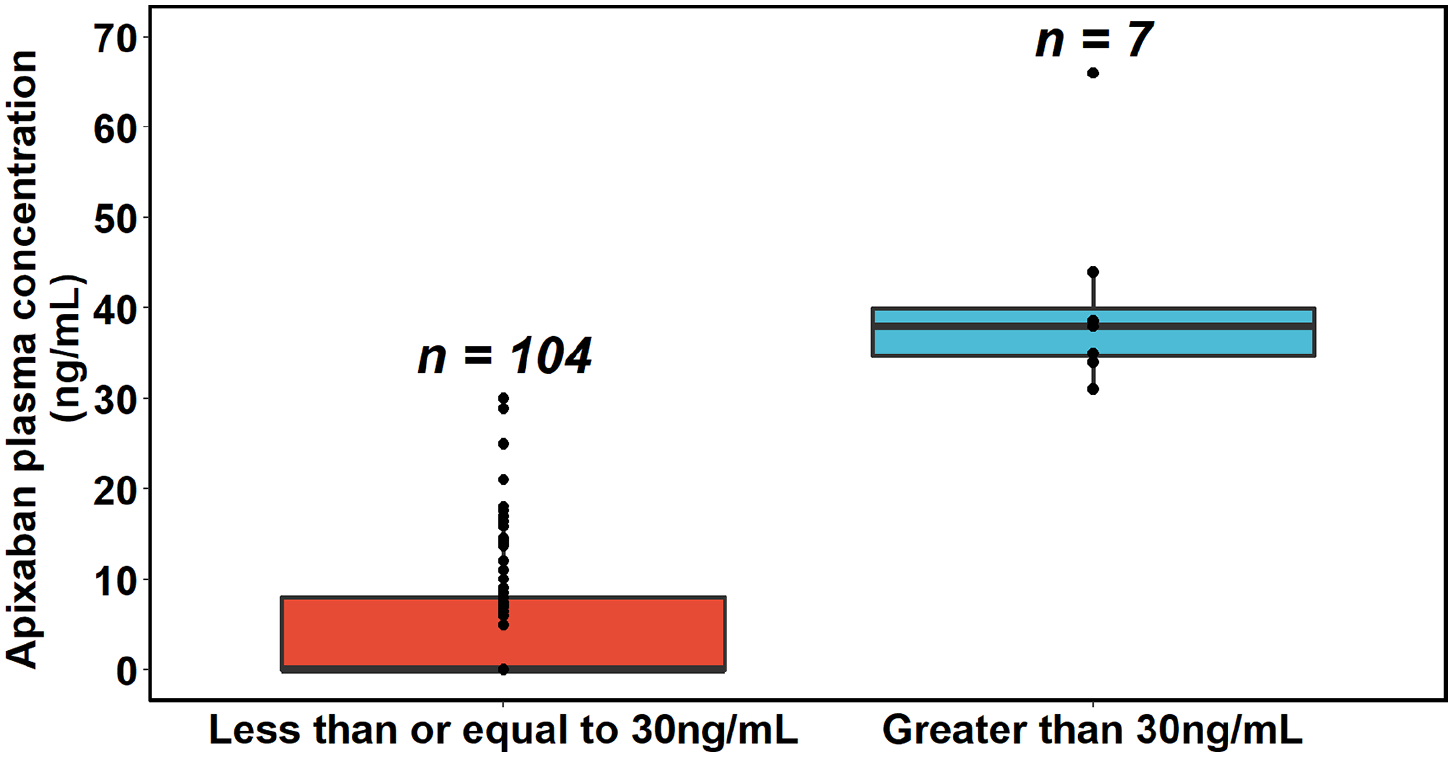
Proportion of patients grouped by apixaban concentrations > 30 ng/mL and with those achieving ⩽ 30 ng/mL, with IQRs.
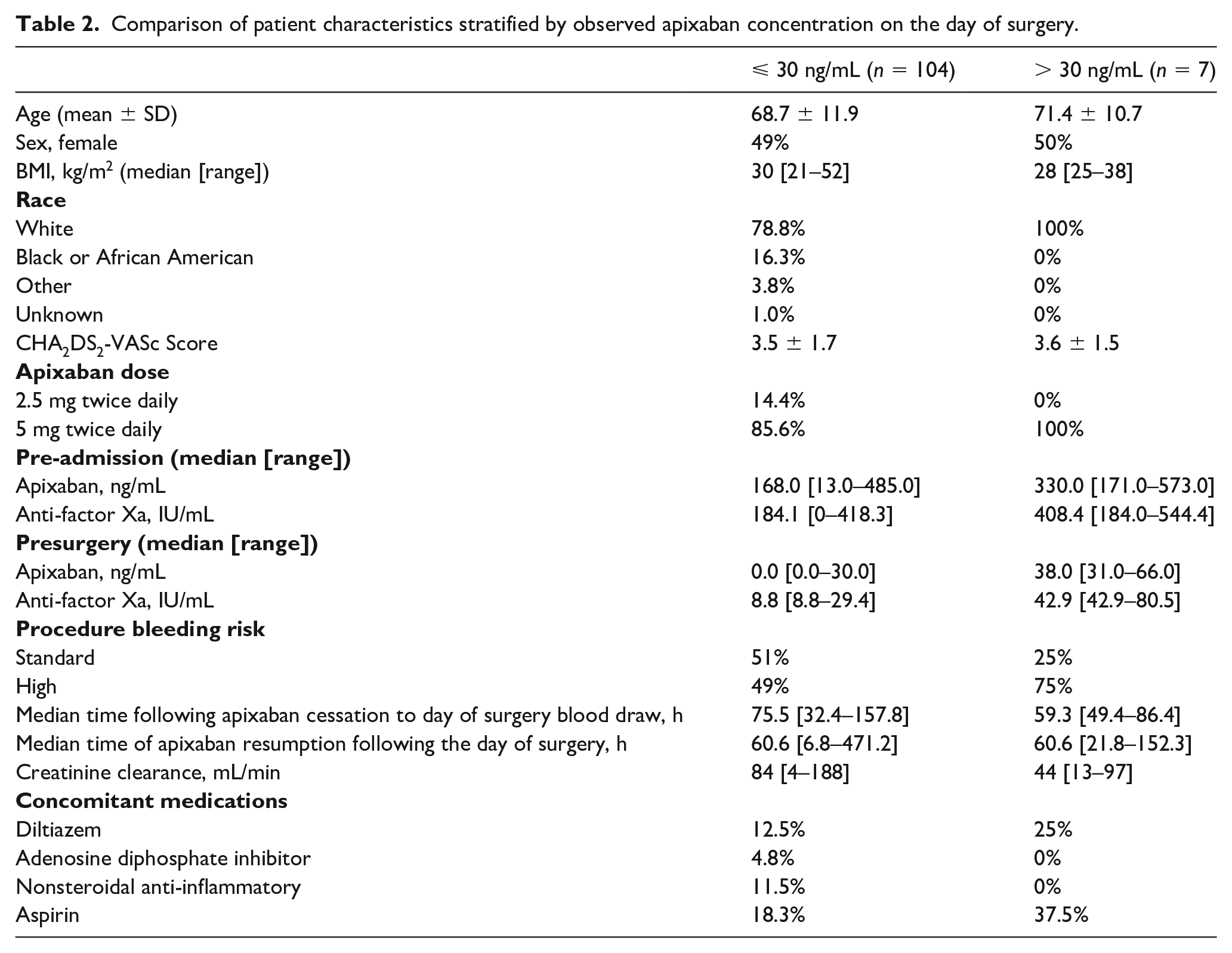
The median time between the self-reported last dose and day of surgery blood sampling was 75.5 hours (32.4–157.8 hours) for those who achieved concentrations ⩽ 30 ng/mL and 59.3 hours (49.4–86.4 hours) for those > 30 ng/mL. Figures 4 and 5 display the proportion of patients achieving target apixaban concentrations based on duration of apixaban discontinuation and dose of apixaban, respectively. All seven patients who did not achieve target plasma apixaban levels were on a maintenance dose of 5 mg twice daily. Pre-admission and preoperative concentrations of apixaban, and anti-Xa, and clinical characteristics of patients who did and did not achieve target plasma apixaban levels are presented in Table 2.
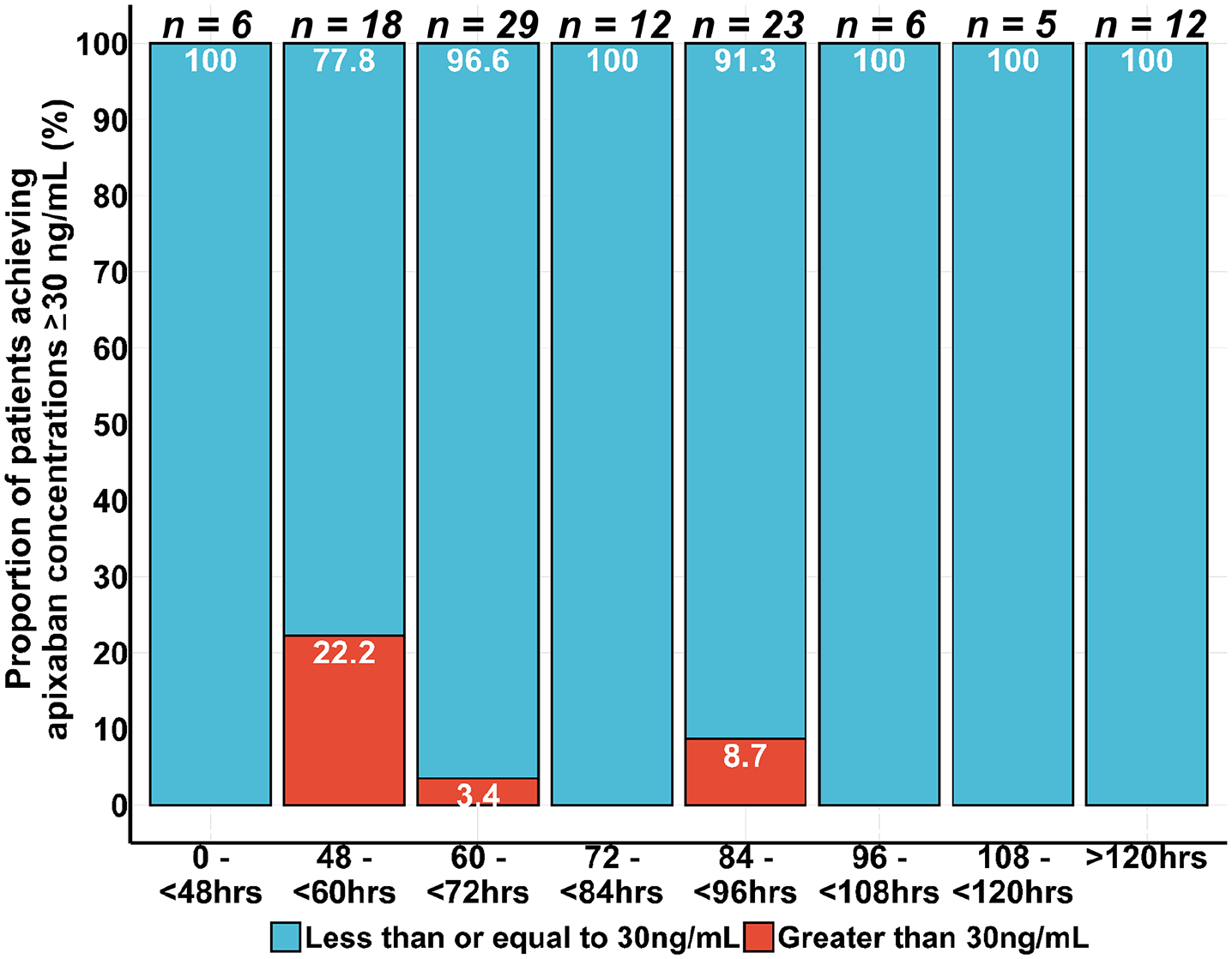
Proportion of patients who had apixaban concentrations > 30 ng/mL according to time since last dose.
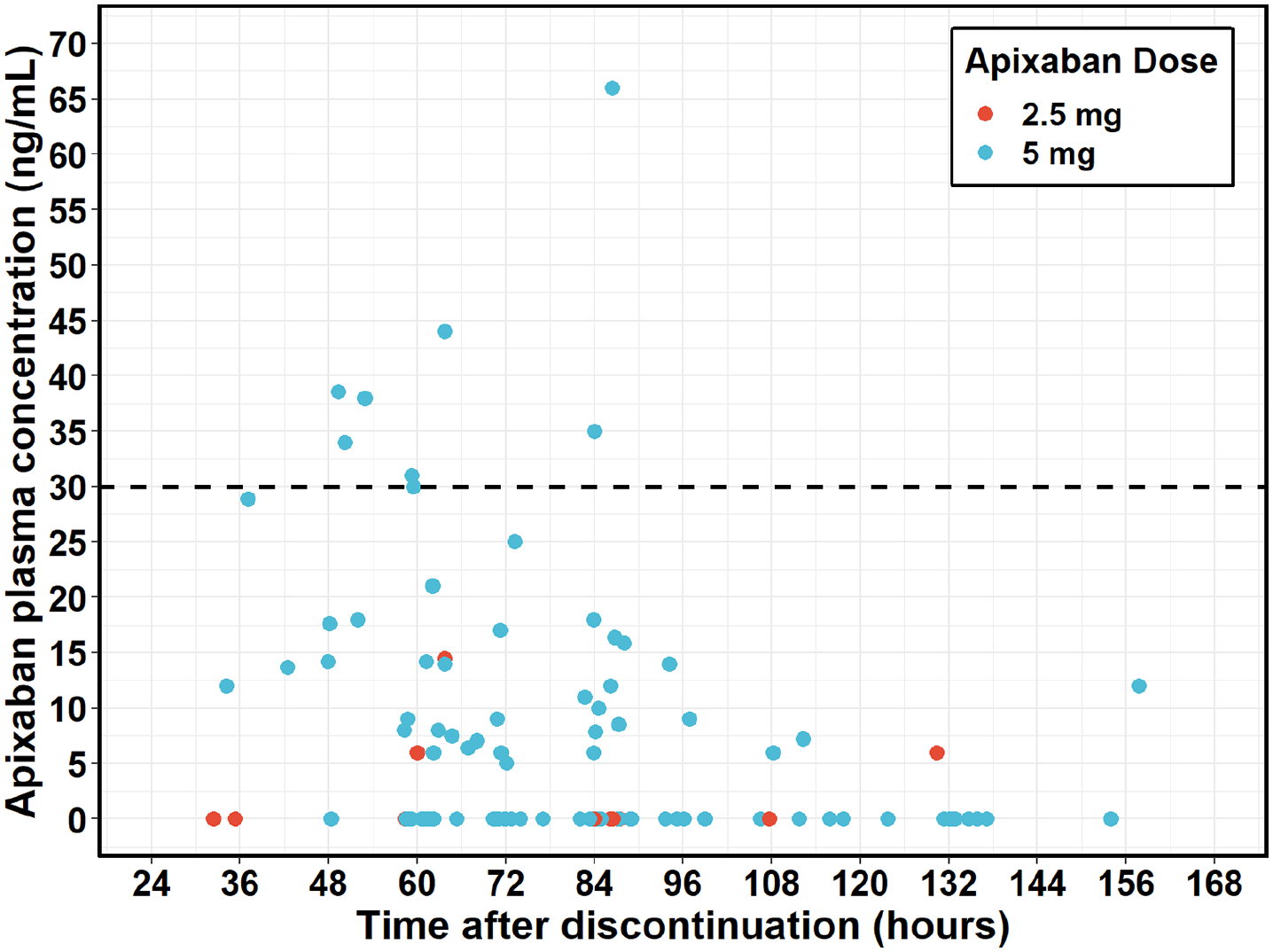
Apixaban concentration following discontinuation on the day of surgery grouped by dose (data points represent individual patient values).
Comparison of patient characteristics stratified by observed apixaban concentration on the day of surgery.
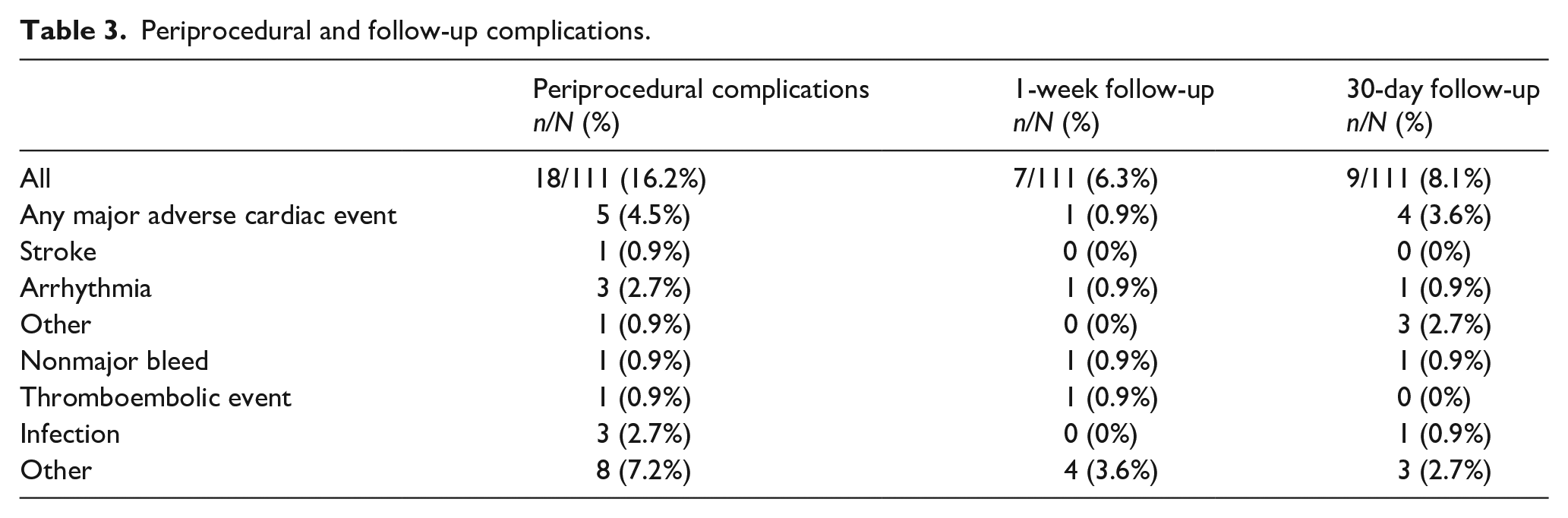
Pharmacokinetic endpoint data were available for 111 of the 130 total patient population. A total of 11 patients were lost to follow-up with nine and two patients at 1-week and 4-week follow-ups, respectively. Clinically significant nonmajor bleeding was reported in one patient at 1-week postsurgery follow-up. There were two reported venous thromboembolic events during the study and follow-up periods. Of these two, one had restart of apixaban at 50 hours and another at 158 hours postsurgery. There were 10 major adverse cardiac events, one stroke, and five arrhythmias. One patient reported both stroke and arrhythmia during the initial hospitalization. Clinical endpoint data are summarized in Table 3. A Welch two-sample t-test was used to compare the binary distribution between patients that had events to those that did not have events and revealed that there was no significant correlation between these events and mean plasma apixaban levels with a mean difference of 0.25 ng/mL (95% CI: –14.6, 15.1, p = 0.97) between both groups.
Periprocedural and follow-up complications.
Discussion
A high proportion (100%) of patients achieved target apixaban concentrations (⩽ 30 ng/mL) following at least 48 hours of discontinuation prior to surgery, and a greater proportion of patients (> 90%) achieved target apixaban levels after discontinuation for > 60 hours. Patients who did not achieve target plasma apixaban levels tended to be older and have poorer creatinine clearance, with a mean (± SD) age of 71.4 (± 10.7) years and median (range) creatinine clearance of 44 (13–97) mL/min. The median concentration observed for these patients at the pre-admission blood sampling was 330 (171–573) ng/mL. As home dosing the morning of collection at steady state will have varied, these concentrations are considered random and do not necessarily represent maximal (Cmax) or average steady state concentration for any one individual. For comparison, predicted Cmax for patients receiving 5 mg BID for stroke prevention in nonvalvular atrial fibrillation is 171 ng/mL (95% CI: 91, 321). 2 Our results suggest that the recommended 48-hour discontinuation period is adequate for most individuals to achieve adequate resolution of anticoagulation. The effect of these patient-specific factors was almost entirely absent at 60 hours, suggesting that a discontinuation period longer than 3 days will not reduce anticoagulation-associated bleeding events in the perioperative period. All seven of the patients who did not achieve the target plasma apixaban level of ⩽ 30 ng/mL were on a daily dose of 5 mg twice daily, though none were taking concomitant inhibitors of CYP450 enzymes or p-glycoprotein. Dose is a primary driver of systemic exposure. Dose, in addition to patient characteristics, concomitant medications, and perceived bleeding risk will play a role in a physician’s assessment of individualizing the appropriate discontinuation time.
Anti-Xa levels were highly correlated with plasma apixaban, especially at higher prediscontinuation concentrations. Validity of the analytic method was demonstrated by significant correlation (p < 0.001) between anti-Xa activity and plasma apixaban concentration measured on the visit prior to discontinuation and on the day of surgery (online Supplemental Figure 1). This is consistent with a known linearity between concentration and anti-Xa over a range of therapeutic doses. 2 Further external validation of our methods was indicated by an apparent positive trend between apixaban concentration and age, and a negative trend between apixaban concentration, weight, and creatinine clearance measured at steady state (first blood draw) (online Supplemental Figures 2 and 3).
No significant safety issues were observed related to the discontinuation of apixaban. Of the seven patients who had a day of surgery apixaban concentration of > 30 ng/mL, only two experienced perioperative adverse events, neither of which were bleeding. Of the remaining 104 patients, the rates of major adverse cardiac events, arterial thrombosis, recurrent VTE, and death were low, most of which were attributed to underlying disease rather than discontinuation of apixaban. The primary endpoint of this observational study was pharmacokinetic and the study was not powered to make inferences about the efficacy and safety of the apixaban discontinuation approach recommended in the product insert. Although the numbers were too small to perform a robust statistical analysis, the small rates of cardiac and thromboembolic events suggest the safety of the prescribed discontinuation period were reassuring.
Study limitations and strengths
One of the major limitations of this trial is that it was an observational study assessing provider-directed discontinuation rather than a standardized protocol, which led to a wide range of presurgery discontinuation windows. Another limitation was that the exact time of last dose for both visits was patient-reported and subject to recall bias and the variability associated with patient self-reporting in a naturalistic setting. This lack of exact dose time and limited number of samples prevented the use of population pharmacokinetic approaches that could have otherwise been used to identify covariates associated with pharmacokinetic variation. Lastly, the reported cut-off of 30 ng/mL was chosen as a reasonable surrogate of a safe level for an invasive procedure. This has not been defined in large studies with clinical endpoints. Despite the varied patient population, only one patient had an apixaban concentration > 50 ng/mL. For comparison, 50 ng/mL is the model predicted minimum apixaban concentration for patients prescribed apixaban 2.5 mg BID for the prevention of venothromboembolic events in hip or knee replacement surgery (95% CI: 23, 109). 3 While in the PAUSE trial with a DOAC 48 hours prior to surgery discontinuation time, 90.5% of the apixaban group achieved a drug plasma level of < 50 ng/mL. The major bleeding incidence was 2.96% (95% CI: 0–4.68%) in the apixaban group undergoing high bleeding risk surgery. 4 In patients with serious bleeding, only a drug concentration > 50 ng/mL is likely sufficiently high to warrant antidote administration, whereas in those requiring an urgent intervention associated with a high risk of bleeding, antidote administration is advocated if the drug concentration exceeds 30 ng/mL.5,8 In the current study, even those with > 30 ng/mL of apixaban had an order of magnitude reduction in both apixaban and anti-Xa activity on the morning of surgery compared to steady state concentration. The number of patients prescribed 2.5 mg was low. This is not an impact for those prescribed this for prophylaxis, as presurgery concentrations will be expected to be lower than those prescribed 5 mg. Although formal pharmacokinetic modeling was not done on such a small set of patients, even those taking the labeled dose reduction due to age ⩾ 80 years, body weight ⩽ 60 kg, or serum creatinine ⩾ 1.5 mg would be expected to have similar or lower apixaban pharmacokinetic exposure than those prescribed 5 mg. In totality, the findings of this study suggest no role for anti-Xa assays in the routine management of cessation of apixaban for elective surgical procedures.
A strength of this study is the generalizability of the results to patients assessed in clinical practice, as a high proportion of screened patients were enrolled (83%). Another strength is the clinical applicability of the apixaban regimen management we assessed, as most patients adhered to a physician-directed perioperative apixaban therapy interruption (95%) and resumption (93%) management protocol.
Conclusion
A large proportion of patients (93.7%; 95% CI: 88–97%) achieved plasma apixaban concentrations of ⩽ 30 ng/mL following at least 48 hours of discontinuation. A greater proportion (> 90%) of patients achieved below 30 ng/mL when apixaban was discontinued for > 60 hours. The clinical evidence here supports current pharmacokinetic discontinuation strategies based on patient factors and apixaban half-life.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211047270 – Supplemental material for Apixaban Discontinuation for Invasive Or major Surgical procedures (ADIOS): A prospective cohort study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211047270 for Apixaban Discontinuation for Invasive Or major Surgical procedures (ADIOS): A prospective cohort study by Geno J Merli, Walter K Kraft, Luis H Eraso, Taki Galanis, Lynda J Thomson, Geoffrey O Ouma, Eugene Viscusi, Jerald Z Gong and Edwin Lam in Vascular Medicine
Footnotes
Acknowledgements
We thank the following individuals from Thomas Jefferson University Hospitals: Douglas Stickle for apixaban analytic measurement; Scott Keith for power analysis; and Suzanne Adams, Daniel Schwegler, and Melissa McCurdy for data management.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Galanis has served as a speaker for Jansson. No other authors have relevant disclosures.
Funding
This research was funded by an investigator-initiated research award from Bristol Myers Squibb and Pfizer, as part of the American Thrombosis Investigator Initiated Research Program (ARISTA-USA). Edwin Lam was supported by a National Institutes of Health institutional training grant T32GM008562-24. Study design, research conduct, analysis, and manuscript preparation were conducted by the Thomas Jefferson University investigators, who had final editorial control over the published content.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
