Abstract
Little is known about the impact of oral anticoagulation (OAC) choice on healthcare encounters during venous thromboembolism (VTE) primary treatment. Among anticoagulant-naïve patients with VTE, we tested the hypotheses that healthcare utilization would be lower among users of direct OACs (DOACs; rivaroxaban or apixaban) than among users of warfarin. MarketScan databases for years 2016 and 2017 were used; healthcare utilization was identified in the first 6 months after initial VTE diagnoses. The 23,864 patients with VTE had on average 0.2 ± 0.5 hospitalizations, spent 1.3 ± 5.2 days in the hospital, had 5.7 ± 5.1 outpatient encounters, and visited an emergency department 0.4 ± 1.1 times. As compared to warfarin, rivaroxaban and apixaban were associated with fewer hospitalizations, days hospitalized, outpatient office visits, and emergency department visits after accounting for age, sex, comorbidities, and medications. Hospitalization rates were 24% lower (incidence rate ratio (IRR): 0.76; 95% CI: 0.69, 0.83) with rivaroxaban and 22% lower (IRR: 0.78; 95% CI: 0.71, 0.87) with apixaban, as compared to warfarin (IRR: 1.00 (reference)). Healthcare utilization was similar between apixaban and rivaroxaban users. Patients with VTE prescribed rivaroxaban and apixaban had lower healthcare utilization than those prescribed warfarin, while there was no difference when comparing apixaban to rivaroxaban. These findings complement existing literature supporting the use of DOACs over warfarin.
Introduction
Venous thromboembolism (VTE), which consists of both deep vein thrombosis (DVT) and pulmonary embolism (PE), has a lifetime risk of one in 12, 1 and affects approximately one million Americans annually. 2 Patients with VTE are at risk of recurrent events (for unprovoked VTE, about 10% after 1 year and 30% after 5 years)3 –5 and bleeding due to treatment with anticoagulants. VTE survivors may also experience complications such as post-thrombotic syndrome (present in nearly 50% of patients with DVT),6,7 venous ulcer, chronic thromboembolic pulmonary hypertension, 2 diminished quality of life,8 –10 anxiety, and depression.10,11 All of these factors may lead to patients with VTE having greater healthcare utilization.
Oral anticoagulation (OAC) is the mainstay for primary VTE treatment, which spans 3–6 months following the initial event. 12 Direct oral anticoagulants (DOACs; i.e. apixaban, rivaroxaban, dabigatran, edoxaban), together with warfarin, are standards of care for VTE treatment in current guidelines. 12 Post-VTE healthcare utilization patterns may differ according to the OAC prescribed. As compared to warfarin, the DOACs have generally been associated with similar risk of VTE recurrence but lower bleeding rates in Phase III randomized controlled trials13 –18 and observational investigations of real-world databases.19,20 Warfarin has a narrow therapeutic index and requires monitoring and thus may require more frequent outpatient encounters. However, the required monitoring for warfarin may improve compliance and reduce adverse events. 21 Among the DOACs, some are once daily after an initial 1–3 week acute treatment period (rivaroxaban, edoxaban) while others are twice daily (apixaban and dabigatran), and differential compliance with once versus twice daily medications may affect healthcare utilization. 22
Relatively little is known about the effect of OAC therapy choice on healthcare utilization in the context of VTE primary treatment. Using a large administrative dataset, we evaluated whether, in the first 6 months post-VTE diagnosis, healthcare utilization differed according to whether the patient was prescribed apixaban, rivaroxaban or warfarin for primary treatment of their VTE event. Dabigatran and edoxaban were not assessed as they are not frequently prescribed for the primary treatment of VTE in the United States. 23 Additionally, cancer-related VTE events were excluded since (a) healthcare utilization would be greater in this population due to cancer treatment and (b) low-molecular weight heparin is recommended for treating cancer-associated VTE.24,25 We hypothesized that the number of hospitalizations, days hospitalized, emergency department visits, and outpatient office visits would be higher among warfarin users than among users of rivaroxaban and apixaban.
Methods
IBM MarketScan® Commercial Claims and Encounters and Medicare Supplemental and Coordination of Benefits databases for calendar years 2016 and 2017 were used in the present analysis. The databases contain individual-level, de-identified, HIPPA-compliant, healthcare claims information from US employers, health plans, hospitals, and Medicare programs. 26 Individual-level identifiers allow for linkage of data across enrollment records and inpatient, outpatient, ancillary, and drug claims. As this is a commercial insurance database, individuals with no insurance are not included and individuals working at small companies are underrepresented. The University of Minnesota Institutional Review Board deemed this research exempt from review. Because these are de-identified administrative data, informed consent was not obtained or required.
Identification of VTE cases
The present analysis includes individuals aged 18–99 with incident VTE, at least one prescription for an OAC within the 31 days before or after their first VTE claim, and ⩾ 3 months of continuous enrollment prior to their first OAC prescription. As in previous work, 19 we defined VTE as having at least one inpatient claim for VTE or two outpatient claims for VTE, which were 7–185 days apart, in any position, based on International Classification of Diseases (ICD) codes (listed in online Supplemental Table 1). The positive predictive value (PPV) of this definition was 91% in a recent validation study that employed a definition similar to that used in this analysis, which was inclusive of both inpatient and outpatient encounters and additionally required treatment. 27
Participant characteristics according to oral anticoagulant prescribed for VTE primary treatment: MarketScan 2016–2017.
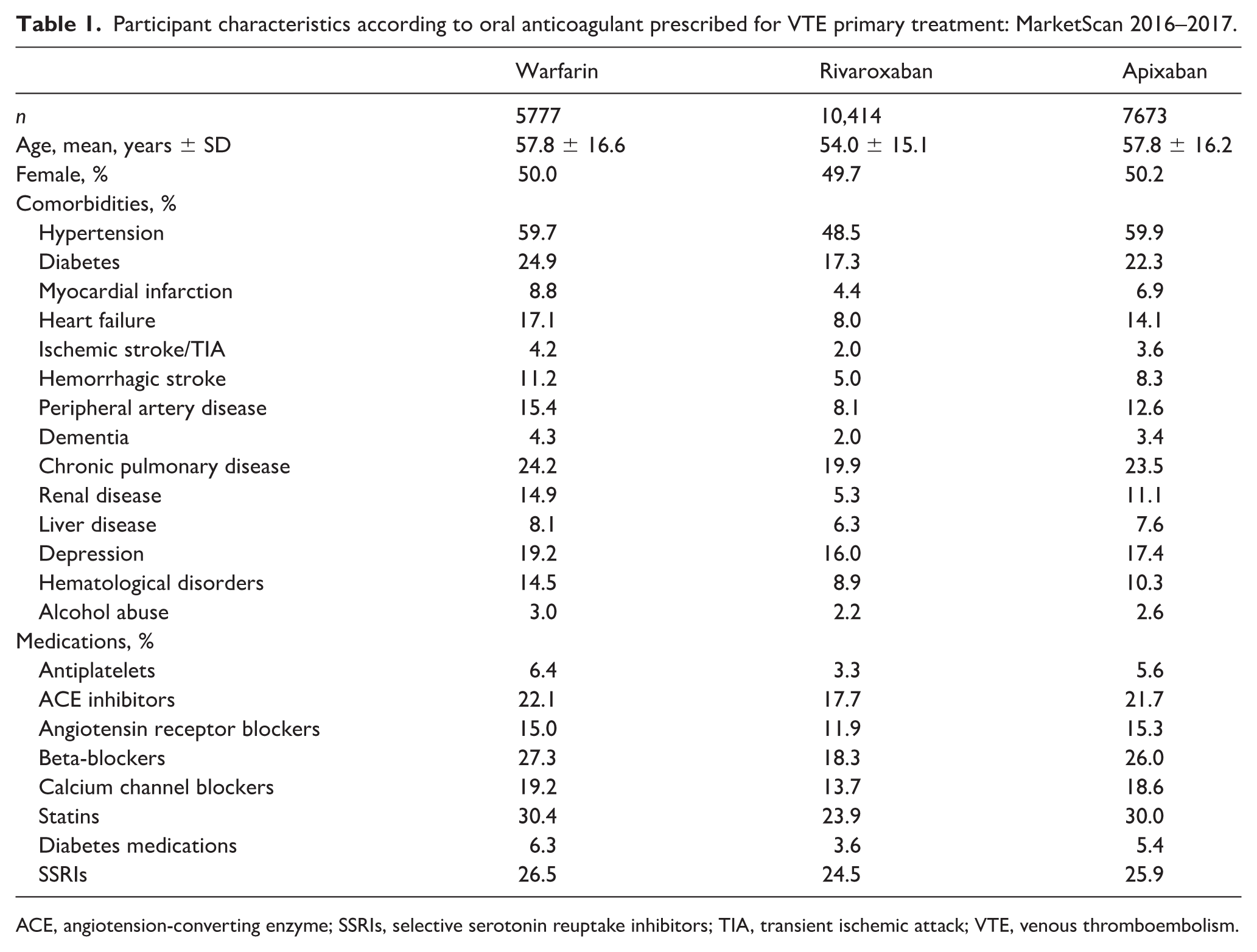
ACE, angiotension-converting enzyme; SSRIs, selective serotonin reuptake inhibitors; TIA, transient ischemic attack; VTE, venous thromboembolism.
For the present analysis we restricted our analytic sample to participants whose initial VTE occurred between January 1, 2016 and December 31, 2017, despite availability of data from earlier years. The rationale for this restriction was to (a) minimize bias related to patients with VTE prescribed soon after the DOAC approvals being different (likely healthier) than their counterparts prescribed warfarin, (b) provide contemporary estimates of healthcare utilization, and (c) allow for greater consistency since only ICD-10 codes were used with this approach. The initial sample included 88,686 patients with VTE aged 18–99 years. The analytic sample was 68,092 once limited to individuals ever prescribed an OAC in the study period; 46,507 after requiring the first OAC prescription be ±31 days of the VTE ICD code date; 32,938 after requiring ⩾ 3 months of continuous enrollment before the first OAC prescription; and 23,864 after excluding individuals who used low-molecular weight heparin as their sole anticoagulant prescription or who had evidence of cancer, as specific recommendations for treatment exist for this population. 12
Anticoagulant use
For the present analysis of anticoagulant-naïve patients with VTE, we considered only the first OAC prescribed. We identified prescriptions for apixaban, rivaroxaban, and warfarin using outpatient pharmaceutical claims data, which includes information on the National Drug Code, the prescription fill date, and the number of days supplied. Validation studies for apixaban and rivaroxaban claims have not yet been conducted. However, the validity of warfarin claims in administrative databases is excellent (sensitivity: 94%, PPV: 99%). 28
Healthcare utilization
Inpatient, outpatient, and emergency department utilization during the VTE primary treatment period (i.e. between the discharge date for the initial VTE and disenrollment or 6 months post the index date (whichever came first)) were identified using the MarketScan inpatient and outpatient databases. Inpatient claims were used to determine the number of hospitalizations and days hospitalized. Emergency department and outpatient office visits were derived from the outpatient claims and distinguished using information on place of service. In secondary analyses, we also evaluated the number of bleeding-related hospitalizations and days hospitalized. Incident hospitalized bleeding was defined according to the Cunningham algorithm, 29 as we have done previously.19,30
Assessment of pre-specified covariates
Information prior to the OAC initiation date (minimum 90 days) from all data sources in MarketScan (i.e. demographic data, inpatient, outpatient, and pharmacy claims) was used to derive pre-specified covariates. We identified 14 pre-specified comorbidities using validated algorithms29,31 applied to the inpatient and outpatient data, and eight different medication categories based on pharmacy prescription fills. These pre-specified covariates are listed in Table 1, and the codes are in online Supplemental Table 2.
IRRs for healthcare utilization according to oral anticoagulant prescribed for VTE primary treatment: MarketScan 2016–2017.
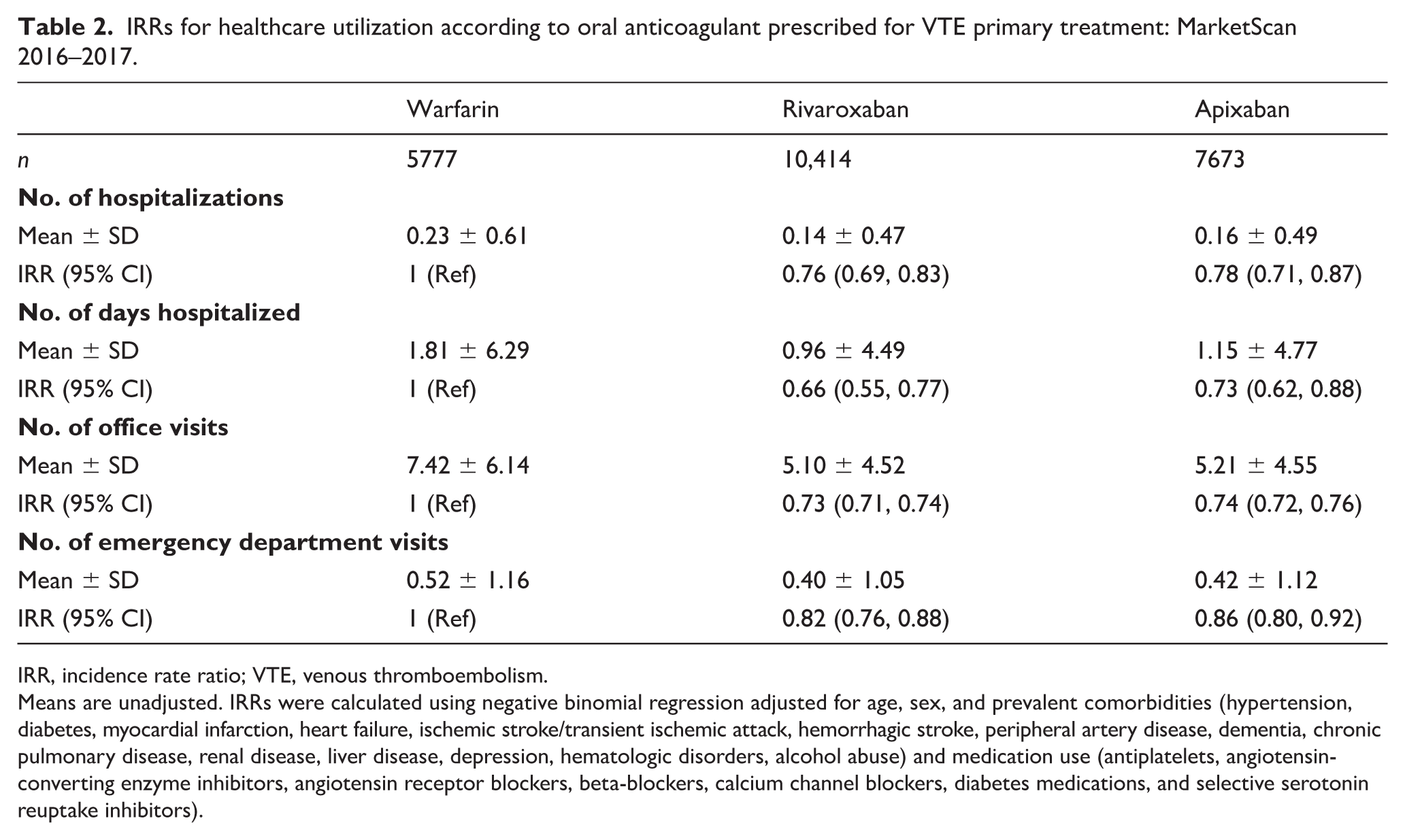
IRR, incidence rate ratio; VTE, venous thromboembolism.
Means are unadjusted. IRRs were calculated using negative binomial regression adjusted for age, sex, and prevalent comorbidities (hypertension, diabetes, myocardial infarction, heart failure, ischemic stroke/transient ischemic attack, hemorrhagic stroke, peripheral artery disease, dementia, chronic pulmonary disease, renal disease, liver disease, depression, hematologic disorders, alcohol abuse) and medication use (antiplatelets, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, beta-blockers, calcium channel blockers, diabetes medications, and selective serotonin reuptake inhibitors).
Statistical analysis
Descriptive characteristics are presented as means ± SD and percentages. All outcomes were count data and, as such, negative binomial regression was used (i.e. SAS GENMOD procedure with a negative binomial distribution and a log link) and an offset of log follow-up time was applied to account for not all participants having 185 days of follow-up. Incidence rate ratios were calculated. We adjusted for age, sex, and the comorbidities and medications listed in Table 1. Multiplicative interactions were evaluated between OAC use and sex and age (< 65 vs ⩾ 65 years). Results stratified by sex and age are also reported. In a sensitivity analysis, we restricted the sample to individuals with no evidence of kidney disease, since individuals with severe kidney disease may have additional healthcare utilization due to dialysis. We also conducted analyses to correspond to a 3-month primary treatment period, by ending follow-up 90 days after the OAC initiation date.
Additional secondary analyses were also conducted. First, using methods similar to those above, we evaluated hospitalized bleeding as an outcome (both number of hospitalizations and days hospitalized). Second, in order to evaluate confounding in a different manner, in head-to-head OAC comparisons we matched each new user to one user of the comparator OAC by age (± 1 year), sex, and drug initiation date (± 90 days) using an automated greedy matching algorithm. 32 After matching, for each OAC comparison, a propensity score for exposure to the OAC was created via logistic regression, using comorbidities and medications determined a priori (Table 1). This propensity score was then included in the model, together with age and sex. All statistical analyses were performed with SAS software, version 9.3 (SAS Institute Inc., Cary, NC, USA).
Results
Among the 23,864 patients with incident VTE included in the present analysis, 50.6% were female and the average (± SD) age at the time of VTE was 55.7 ± 16.1 years. In the 6 months following discharge from their incident VTE, 3355 individuals (14.1%) were hospitalized at least once. Allowing for multiple hospitalizations per individual, there were 4509 hospitalizations in total, the average number (± SD) of hospitalizations was 0.18 ± 0.53, and the average length of hospitalizations was 1.28 ± 5.18 days. When restricted to individuals who were hospitalized, the average number of hospitalizations and days hospitalized were 1.34 ± 0.77 and 9.72 ± 11.01, respectively. During the 6 months of follow-up, there were a total of 145,086 outpatient office visits and 11,160 emergency department visits. On average, they had 5.71 ± 5.08 outpatient encounters, and visited an emergency department 0.44 ± 1.11 times.
The initially prescribed OAC was warfarin for 24.2% of patients with VTE, rivaroxaban for 43.6%, and apixaban for 32.2%. As shown in Table 1, patients with VTE initially prescribed rivaroxaban were younger (54.0 ± 15.1 years) than those prescribed warfarin (57.8 ± 16.6 years) or apixaban (57.8 ± 16.2 years). They also tended to be taking fewer medications and have fewer comorbidities.
Table 2 presents crude mean healthcare utilization and incidence rate ratios (IRR) adjusted for age, sex, and the comorbidities listed in Table 1, with warfarin as the reference OAC. Results with rivaroxaban as the reference are shown in online Supplemental Table 3. Patients who filled claims for rivaroxaban and apixaban had fewer hospitalizations, days hospitalized, outpatient office visits, and emergency department visits relative to patients who filled claims for warfarin. For example, the mean number of hospitalizations was 24% lower (IRR: 0.76; 95% CI: 0.69, 0.83) for patients prescribed rivaroxaban and 22% lower (IRR: 0.78; 95% CI: 0.71, 0.87) for patients prescribed apixaban, as compared to those prescribed warfarin (IRR: 1.00 (reference)). When comparing apixaban to rivaroxaban, there were no differences in the number of hospitalizations, days hospitalized, or emergency department visits. For office visits, the IRR comparing apixaban to rivaroxaban was 1.02 (95% CI: 1.00, 1.04). Results were similar when we restricted to individuals with no evidence of kidney disease (data not shown) and when we restricted follow-up to the first 3 months after the initial OAC prescription (online Supplemental Table 4).
IRRs for healthcare utilization according to oral anticoagulant prescribed for VTE primary treatment by sex and age category: MarketScan 2016–2017.
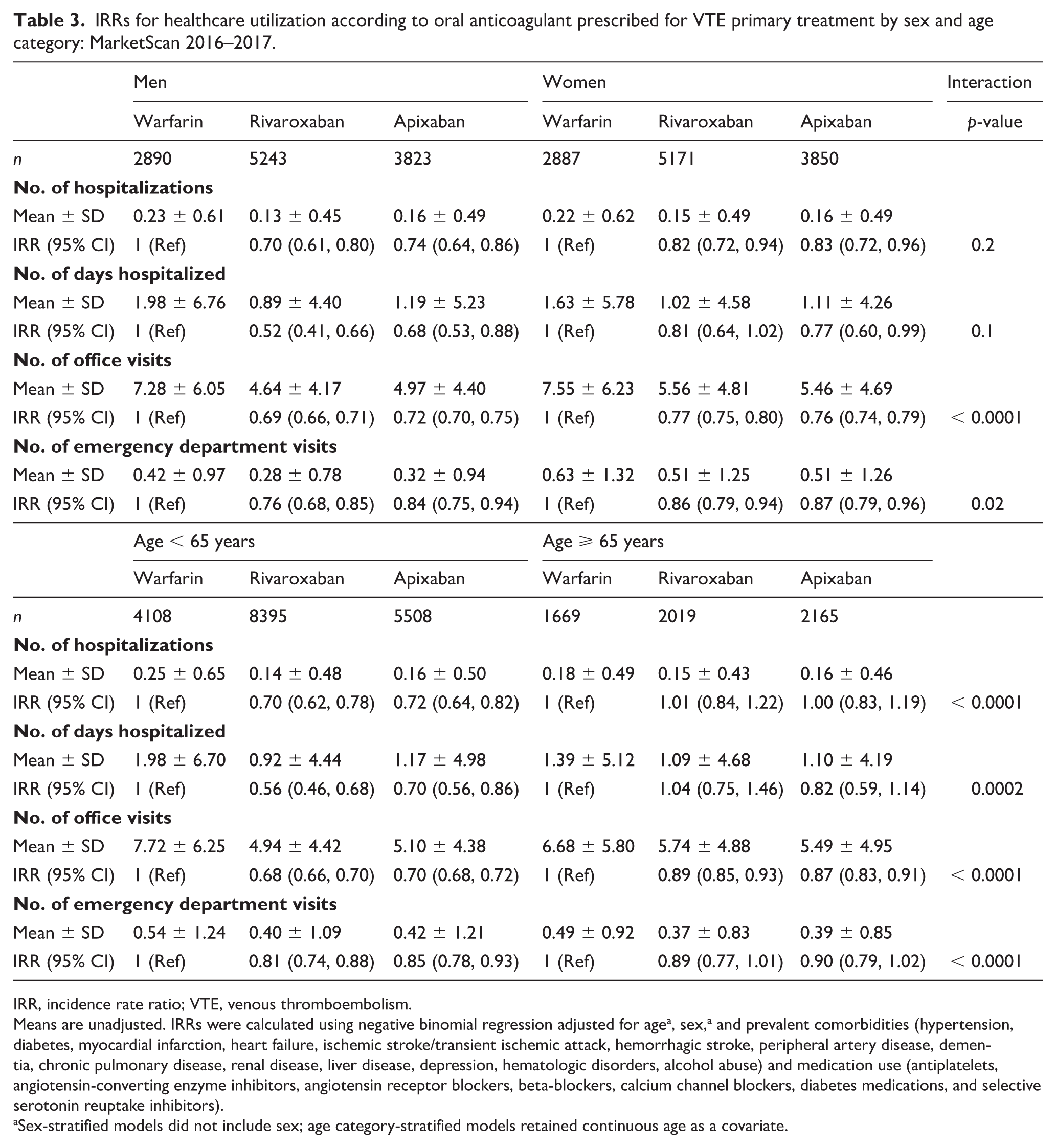
IRR, incidence rate ratio; VTE, venous thromboembolism.
Means are unadjusted. IRRs were calculated using negative binomial regression adjusted for agea, sex,a and prevalent comorbidities (hypertension, diabetes, myocardial infarction, heart failure, ischemic stroke/transient ischemic attack, hemorrhagic stroke, peripheral artery disease, dementia, chronic pulmonary disease, renal disease, liver disease, depression, hematologic disorders, alcohol abuse) and medication use (antiplatelets, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, beta-blockers, calcium channel blockers, diabetes medications, and selective serotonin reuptake inhibitors).
Sex-stratified models did not include sex; age category-stratified models retained continuous age as a covariate.
Propensity score-matched a IRRs for healthcare encounters according to oral anticoagulant prescribed for VTE primary treatment: MarketScan 2016–2017.
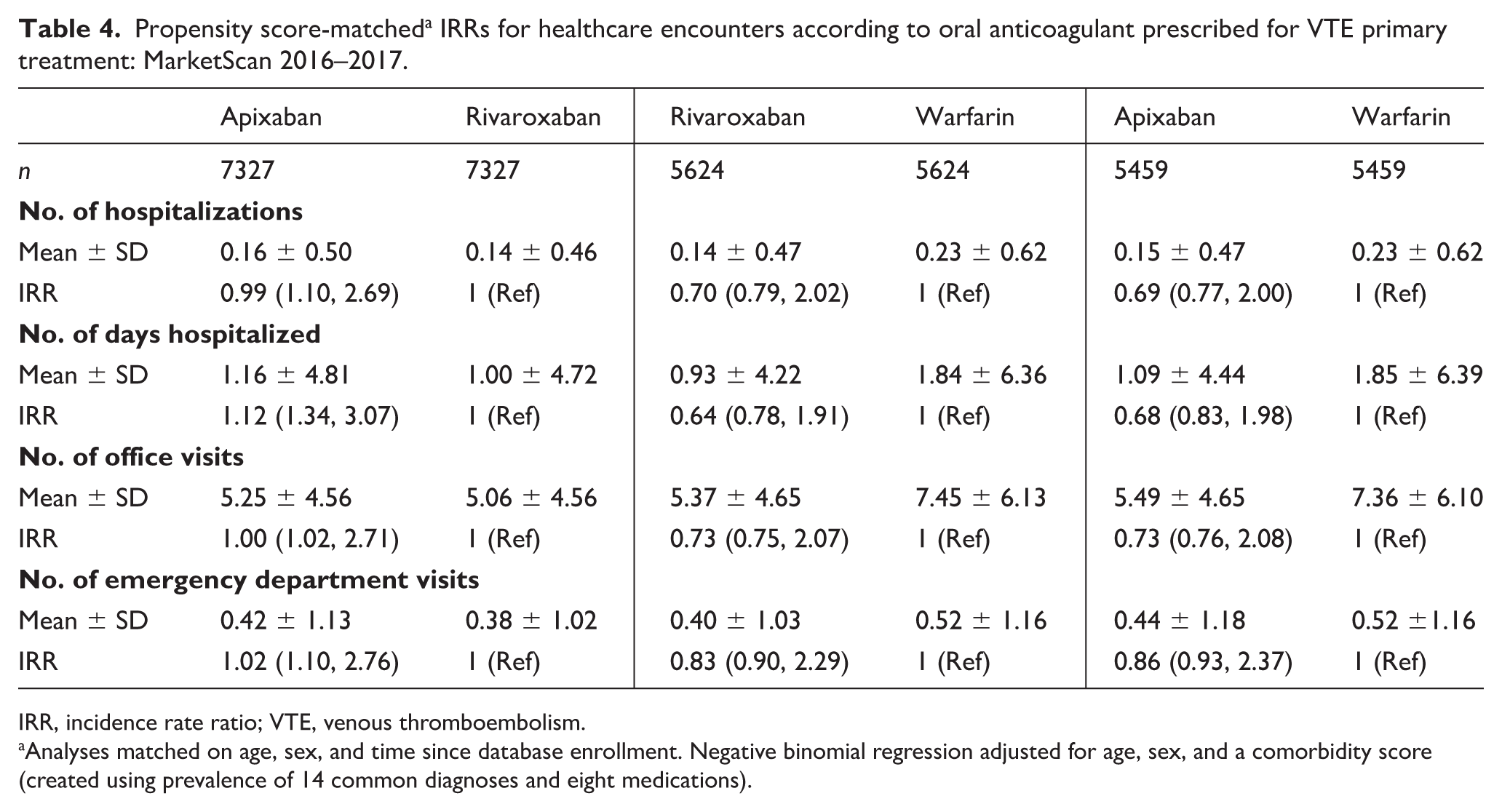
IRR, incidence rate ratio; VTE, venous thromboembolism.
Analyses matched on age, sex, and time since database enrollment. Negative binomial regression adjusted for age, sex, and a comorbidity score (created using prevalence of 14 common diagnoses and eight medications).
There were interactions by both sex and age category (Table 3). The associations between OACs and office visits and emergency department visits were more pronounced among men than women, and the associations between OACs and all healthcare utilization metrics were more pronounced among younger than older patients with VTE.
Results were similar in analyses where we matched each OAC 1:1 to a comparator OAC and then used and controlled for confounding by adjusting using a propensity score created using information on 14 common diagnoses and eight medications (Table 4).
In secondary analyses, we also evaluated outcomes including hospitalized bleeding (both number of hospitalizations and days hospitalized). Of the analytic sample, 180 were hospitalized for bleeding in the 185 days after their first OAC prescription. Among those hospitalized for bleeding, enrollees had an average of 1.10 ± 0.32 hospitalizations and spent a total of 6.21 ± 6.25 days in the hospital. The IRRs for hospitalized bleeding associated with rivaroxaban and apixaban were 0.79 (95% CI: 0.54, 1.15) and 0.57 (95% CI: 0.38, 0.87) as compared to warfarin, respectively. Likewise, as compared to warfarin users, the IRR for hospitalized bleeding days was lower among users of apixaban (0.73; 95% CI: 0.36, 1.49) and rivaroxaban (0.58; 95% CI: 0.30, 1.14).
Discussion
In this US population of 23,864 insured individuals who were treated with warfarin, apixaban or rivaroxaban for non-cancer-related VTE in 2016–2017, patients initially prescribed rivaroxaban or apixaban had fewer hospitalizations, days hospitalized, outpatient office visits, and emergency department visits relative to their counterparts prescribed warfarin over the 6 months after diagnosis. Likewise, they also had fewer bleeding-related hospitalizations and days hospitalized. The frequency of healthcare utilization was similar among patients prescribed rivaroxaban and apixaban. Healthcare utilization and the frequency of healthcare encounters are outcomes of high relevance to patients, and findings from this analysis can assist patients and providers when deciding between warfarin, apixaban and rivaroxaban.
The fact that office visits were more frequent among patients prescribed warfarin is consistent with expectation, since international normalized ratio (INR) monitoring is a vital aspect of warfarin therapy. As such, primary care providers may be more likely to see patients on warfarin than DOACs – which do not require monitoring. Both randomized clinical trials and observational comparative effectiveness studies have reported more bleeding events with warfarin as compared to the DOACs;13 –20 this may also provide some explanation for the higher number of hospitalizations, days hospitalized, and emergency room visits among warfarin users. In secondary analyses, where the outcome was bleeding-related hospitalizations, results followed a similar pattern to the primary analysis, with apixaban and rivaroxaban being associated with less hospitalized bleeding-related healthcare utilization, albeit not always statistically significantly. Very little research exists evaluating healthcare utilization among patients with VTE according to OAC therapy. In an analysis of the observational industry-funded XALIA study, which recruited from 19 European countries, Canada and Israel, hospital readmission was similar between the rivaroxaban group (2%) and the standard anticoagulation (i.e. vitamin K antagonists) group (2%). 33 Though the authors did not report results of statistical testing, in a propensity score-matched set they did demonstrate that rivaroxaban versus standard anticoagulation was associated with fewer outpatient hospital consultations, and visits with general practitioners, specialists, and nurses.
Interactions were noted, whereby the association between DOAC versus warfarin usage and healthcare utilization was weaker in patients with VTE who were older and female. Women34 –37 and older individuals37,38 have greater healthcare utilization than their male and younger counterparts. The simplest explanation for the weaker associations in women and older individuals is that these individuals have greater healthcare utilization for reasons independent of their VTE events and treatment, which may mute differences in healthcare utilization specifically related to the OAC prescribed. An alternate explanation is that incidence of adverse events – both major and minor – differ according to demographic subgroups due to sex- and aging-related differences in pharmacokinetics and pharmacodynamics (e.g. differences in volume mass or distribution, drug clearance, receptor numbers, binding, or signal transduction post-receptor binding).39 –44
Strengths and limitations
Strengths of this analysis include the relatively large samples of participants with a broad spectrum of clinical characteristics (such as may be seen in routine clinical practice), the contemporary evaluation of healthcare utilization, and the head-to-head comparison of the three OACs most commonly prescribed for VTE primary treatment. 23 Uncontrolled confounding is the most relevant potential limitation of this analysis. Our results were robust to both a traditional analysis, and a propensity score-adjusted matched analysis. Misclassification is another concern, as it is with all analyses of administrative data. However, the algorithms for VTE and warfarin prescription fills all have high PPVs. The PPV for DOAC prescription fills has not yet been quantified. An additional limitation is that with the exception of bleeding-related hospitalization, we did not evaluate the impetus for healthcare utilization. Outpatient bleeding-related healthcare utilization was not evaluated due to poor sensitivity of such definitions. We did not report healthcare costs as these vary by state, hospital, and country and relatively small differences in costs can have a profound impact on findings. We also did not try to balance the drug cost as retail costs are not representative of insurance plan costs or costs to patients. Instead, focusing on patient-centered outcomes (visits to clinics, emergency rooms, or hospitalizations) allows broader translation of our results across healthcare systems, jurisdictions, and even countries.
Conclusions
In conclusion, rivaroxaban and apixaban were associated with lower post-event healthcare utilization than warfarin in this analysis of patients who received warfarin, rivaroxaban or apixaban for VTE primary treatment in 2016–2017. There was no difference in indices of healthcare utilization based on whether the patient used rivaroxaban or apixaban. The frequency of healthcare encounters, such as emergency room visits, outpatient visits, and hospitalizations, are meaningful outcomes for patients with VTE. Further research is required to determine the relative weights of these adverse events from a patient perspective, but herein we report the absolute numbers and relative differences of several healthcare utilization indices by whether patients with VTE were initially prescribed warfarin, rivaroxaban or apixaban. This information may assist in decision making when patients and their providers are deciding between these OAC options.
Supplemental Material
Supplemental_Tables – Supplemental material for Impact of oral anticoagulation choice on healthcare utilization for the primary treatment of venous thromboembolism
Supplemental material, Supplemental_Tables for Impact of oral anticoagulation choice on healthcare utilization for the primary treatment of venous thromboembolism by Pamela L Lutsey, Richard F MacLehose, J’Neka S Claxton, Rob F Walker, Terrence J Adam, Alvaro Alonso and Neil A Zakai in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Institutes of Health National Heart, Lung, and Blood Institute grant numbers R01-HL131579 and R01-HL122200.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
