Abstract
Direct oral anticoagulants (DOACs), particularly direct factor Xa inhibitors, have been associated with prolongation of the prothrombin time and the international normalized ratio (INR). Although DOACs do not require monitoring, elevations in the INR have been reported in in vitro and observational studies. The literature surrounding the extent of elevation and the clinical significance is limited. The objective of this study was to quantify the degree of INR elevation in hospitalized patients receiving apixaban. This was a single-center, retrospective, observational analysis of adult patients who received at least 1 dose of apixaban during their hospital admission and had at least 1 INR sample collected prior to and following administration. The major end point of this study was to characterize the effect of apixaban on the INR by determining the percentage of patients with an INR higher than our laboratory defined normal (defined as INR > 1.1). Minor end point outcomes included the incidence of an INR increase >0.3 from baseline INR and additional patient-specific factors that may influence INR elevation. Seventy-nine patients were included in the analysis. On day 1 of therapy, the median (interquartile range, IQR) INR was 1.4 (1.3:1.6) with 84.5% of patients having an elevated INR. The median (IQR) INR increased to 1.5 (1.4:1.6) and 1.7 (1.5:1.9) on day 4 and day 7, respectively. Of patients whose INR increased by more than 0.3, the median (IQR) change in INR from baseline was 0.5 (0.4:0.6). Apixaban is associated with a notable increase in INR in hospitalized patients, although it is not clear the clinical impact of the increase. Although literature does not support monitoring INR as a marker of apixaban activity, it is important for clinicians to understand the association apixaban has on the INR to avoid inappropriate interpretation of routine coagulation assays.
Keywords
Introduction
Direct oral anticoagulants (DOACs) may be preferred over warfarin by both prescribers and patients alike for anticoagulant therapy. This is due to the favorable outcomes in addition to minimal monitoring of DOACs when compared to warfarin. 1 –3 While warfarin’s kinetics and impact on coagulation assays are well established within the literature, there is still a gap in knowledge surrounding DOACs, despite its increased use, particularly when assessing coagulation laboratory assessments. 4,5
The international normalized ratio (INR) is routinely used in patients receiving warfarin to assess the level of anticoagulation. 2,3 Routine monitoring is not required with DOACs, despite their association with prolongation of the prothrombin time and elevation in the INR. 5 The literature surrounding the impact on coagulation assays is very limited and generally used only as a quantitative measure with DOACs. There is a paucity of data describing the interpretation of these values with regard to safety and efficacy. 6 –9 The objective of this study was to determine the effect and degree of INR elevation associated with apixaban in inpatients.
Methods
A single-center, retrospective, observational analysis was performed at Brigham and Women’s Hospital, a 793-bed, acute, tertiary care academic medical center in Boston, Massachusetts. All patients >18 years of age admitted between January 1, 2016, and March 31, 2016, who received a dose of apixaban during their hospital admission were eligible for inclusion in the study. Patients were excluded from the study if they received warfarin, any direct thrombin inhibitor, or other factor Xa inhibitors 72 hours prior to or following apixaban administration. Patients were also excluded if at least 1 INR was not reported prior to or following a dose of apixaban. This study was approved by the institutional review board of Brigham and Women’s Hospital.
A hospital reporting system was used to identify all adult patients who were ordered and administered apixaban during the defined period. Data collected included patient demographics, pertinent past medical history, and baseline laboratory results. The use and total daily dose of apixaban prior to admission and for up to 7 days during the inpatient stay were collected, along with the use of inhibitors and inducers of the cytochrome P450 (CYP) 3A4 enzyme and p-glycoprotein (PGP). Cytochrome P450 3A4 and PGP inhibitors and inducers were determined by the Food and Drug Association Drug Development and Drug Interactions list. 10 Patient’s baseline INR were also collected (defined as INR upon hospital admission), INR the day prior to administration of the first apixaban dose, and any INR obtained day 1 through 7, if available. The prothrombin time (PT)/INR was determined using the Stago Star Evolution (Diagnostica Stago), which detects clotting of citrated plasma electromechanically after addition of a rabbit brain thromboplastin-STA Neoplastin. 11 Serum creatinine and blood urea nitrogen were collected starting on day 1 of the first apixaban dose and daily throughout and up to day 7. Liver function tests were collected on day 1 of apixaban administration and on hospital days 5 to 7 if available.
The major outcome of this analysis was to determine the percentage of patients with an INR elevated greater than our laboratory-defined normal (defined as INR >1.1) at any point during therapy. Minor end points included the incidence of INR elevation ≥0.3. Risk factors for INR elevation including drug–drug interactions, acute liver dysfunction (defined as alanine aminotransferase and aspartate aminotransferase 3 times the upper limit of normal), and acute kidney injury (as defined by the Acute Kidney Injury Network criteria) were also collected and analyzed.
Results
During the study period, 109 adult patients who were ordered apixaban of which 79 (72.5%) patients were included in the analysis. Fifteen patients were excluded because an INR was never collected following the initiation of apixaban, 14 patients were excluded due to medications administered known to impact the INR, and 1 patient was excluded as they met both exclusion criteria. Baseline demographics are presented in Table 1. Only 1 patient had a history of hepatic disease, while 16 patients had chronic kidney disease. The majority of patients were on apixaban prior to admission with the most common indication being atrial fibrillation. The median (interquartile range, IQR) baseline INR was 1.2 (1.2:1.4) and median (IQR) INR on the day prior to apixaban initiation was 1.3 (1.2:1.4).
Baseline Demographics.

Abbreviations: ALT, alanine transaminase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; CKD, chronic kidney disease; ICU, intensive care unit; INR, international normalized ratio; IQR, interquartile range; PTA, prior to admission; SCr, serum creatinine; TDD, total daily dose; VTE, venous thromboembolism.
aData presented as median (IQR).
bReduced dose = 2.5 mg twice daily.
On day 1 of therapy, the median (IQR) INR was 1.4 (1.3:1.6), with 84.5% of patients having an INR greater than 1.1 (Table 2). The median (IQR) INR increased to 1.5 (1.4:1.6) and 1.7 (1.5:1.9) on day 4 and day 7, respectively (Figure 1). Throughout the 7-day collection period, the INR was found to be elevated in greater than 80% of patients in 6 of the 7 days.
Percentage of INR Greater Than Normal.a

Abbreviations: INR, international normalized ratio; IQR, interquartile range. aDefined as >1.1.
bData presented as median (IQR).
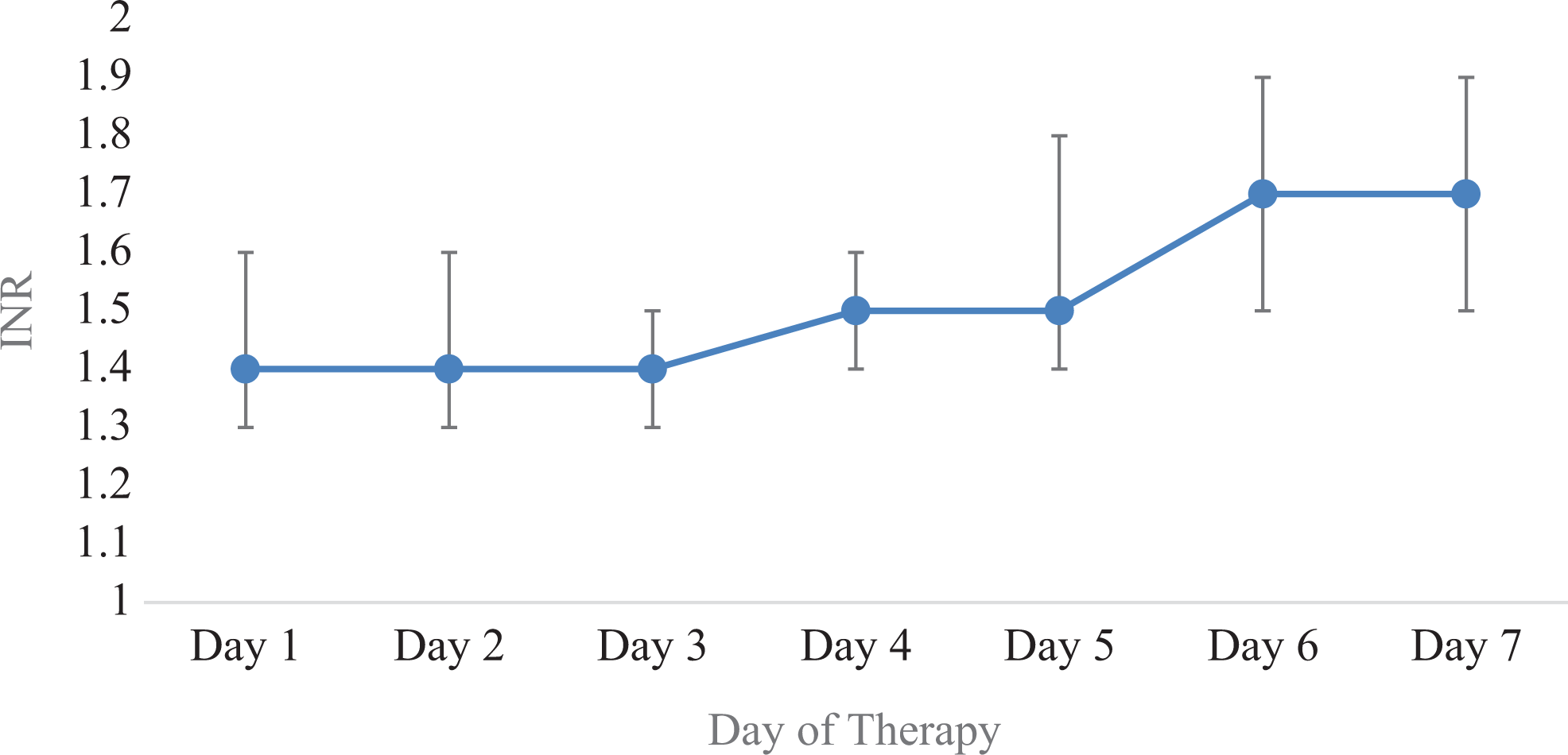
Daily median international normalized ratio (INR).
Twenty-five (13.2%) of the INRs collected were elevated greater than 0.3 from baseline, whereas 24 (11.9%) INRs were elevated greater than 0.3 when compared to the INR collected the day prior to initiation, with a median (IQR) delta of 0.5 (0.4:0.6) and 0.6 (0.5:0.7), respectively (Table 3). Of the patients who were found to have a change in the INR greater than 0.3, 39 (49.2%) had a risk factor for an elevated INR (Table 4).
Incidence in Change of INR > 0.3.
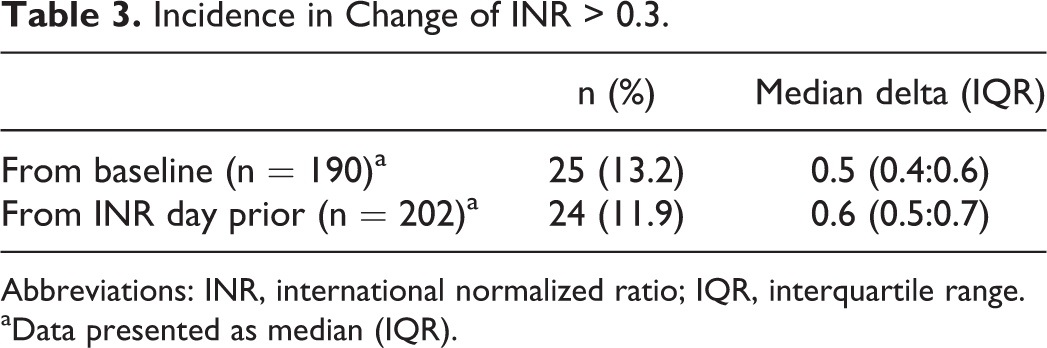
Abbreviations: INR, international normalized ratio; IQR, interquartile range.
aData presented as median (IQR).
Percentage of Patients With Any Risk Factors for INR Elevation.
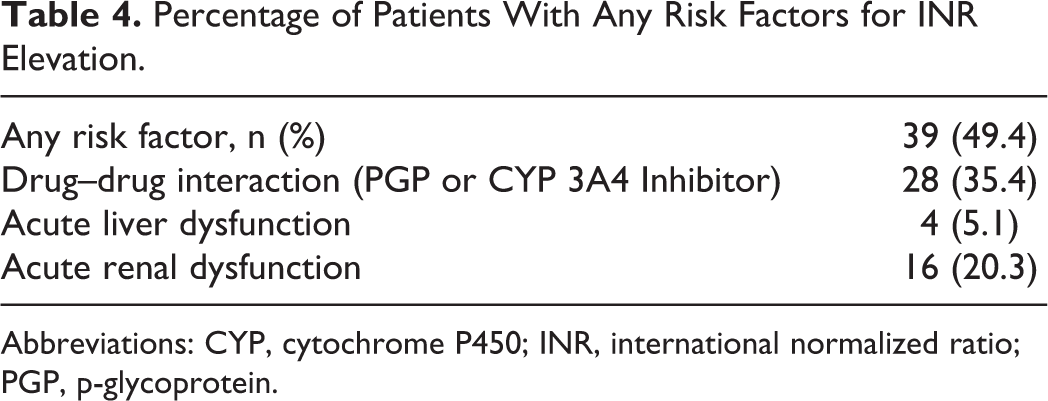
Abbreviations: CYP, cytochrome P450; INR, international normalized ratio; PGP, p-glycoprotein.
Discussion
In vitro and limited observational analyses have previously demonstrated a low to modest impact on the INR due to apixaban in medically ill patients. 4 –8 Frost and colleagues analyzed the effect of rivaroxaban and apixaban dose on coagulation assays in healthy patients. Their study found a modest increase in the INR from apixaban, which was found to be impacted by overall drug concentration. 9 Due to the specific binding to factor Xa by apixaban, the mechanism differs from warfarin and makes the INR unreliable for clinical interpretation. The sensitivity of the PT varies considerably between each DOAC and can be effected by the reagent used as well as the plasma concentration of the drug at the time of collection. 4,6,9 Additionally, because both bound and unbound serine proteases are inhibited, the measure of the clotting time does not equate to the same level of anticoagulation as seen with warfarin. 6 However, there are limited data that help quantify the extent to which apixaban impacts the INR, which if interpreted incorrectly could negatively impact patient care.
This analysis adds meaningful context to quantify the effect apixaban will have on the INR. As DOACs are becoming more popular and convenient therapy options for patients, it is important for providers to understand the impact these medications have on routine coagulation laboratory results. Despite the INR not being reliable method to monitor the level of anticoagulation with DOACs, the PT and INR were still routinely found to be collected at our institution. This may be due to “standard” laboratory monitoring along with unfamiliarity with appropriate monitoring for DOACs by providers.
This analysis found elevated median INR values above laboratory normal value in most patients following the first dose of therapy and up to 80% of patients by day 7 of therapy, with a median (IQR) INR of 1.7 (1.5:1.9). The extent of INR elevation may be impacted by multiple factors, such as interacting medications and existing comorbid conditions; however, 13% of collected INRs had a delta greater than 0.3, with a median (IQR) change of 0.6 (0.5:0.7). This degree of elevation can be significant if interpreted incorrectly, leading to inappropriate therapy or reversal of anticoagulation. One patient in our analysis received phytonadione due and elevated INR.
Due to the metabolism and elimination of apixaban, this study focused on identifying patients with acute liver and renal function, along with potential drug interactions, that could impact serum concentrations of apixaban. 12 As previous data identified that drug concentration plays an important role in apixaban’s effect on the INR, these few but not all-inclusive risk factors are important to take into consideration. As seen in Table 4, roughly 50% of the study population had at least 1 risk factor that may impact the overall serum concentration of apixaban. Due to our small patient population, overall risk factors were not analyzed individually in regard to degree of INR elevations; however, future analyses may be warranted to further investigate the overall impact.
Our findings have limitations that must be considered when extrapolating the results to the general population. This is a retrospective single-center analysis, so it is difficult to consider and account for all factors that may be impacting INR fluctuations during hospitalization. This analysis also did not capture the time of INR collection in regard to dose administration, which may impact the effect on the INR as drug concentration is a known factor affecting INR. Additionally, more than half of the patients in this analysis were on apixaban prior to hospital admission, and as such the initiation impact of apixaban on INR may be underreported.
Conclusion
Apixaban is associated with a notable increase in INR in hospitalized patients. The clinical impact of the increase in INR is not well understood. Members of the health-care team should be aware of the potential for increased INR in patients on apixaban to avoid inappropriate therapy.
Footnotes
Authors’ Note
Kovacevic, Lupi, Wong, Gilmore, and Malloy do not report any affiliation with or financial interest in a commercial organization that poses a conflict of interest with this article. The protocol was reviewed and approved by the Brigham and Women’s Hospital Institutional Review Board prior to the start of this study.
Author Contribution
Kovacevic, M contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; and gave final approval. Lupi, K, Gilmore, J, and Malloy, R contributed to conception and design; contributed to analysis and interpretation; critically revised manuscript; and gave final approval. Wong, A contributed to conception and design; contributed to acquisition; critically revised manuscript; and gave final approval. All authors agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
