Abstract
Catheter-directed thrombolysis (CDT) has been utilized as an adjunct to anticoagulant therapy in selected patients with deep vein thrombosis (DVT) for approximately 30 years. CDT used to be limited to patients with DVT causing acute limb threat and those exhibiting failure of initial anticoagulation, but has expanded over time. Randomized trials evaluating the first-line use of CDT for proximal DVT have demonstrated that CDT does not produce a major reduction in the occurrence of post-thrombotic syndrome (PTS) and that it is poorly suited for elderly patients and those with limited thrombus extent or major risk factors for bleeding. However, CDT does offer selected patients with acute iliofemoral DVT improvement in reducing early DVT symptoms, in achieving reduction in PTS severity, and in producing an improvement in health-related quality of life (QOL). Clinical practice guidelines from medical and surgical societies are now largely aligned with the randomized trial results. This review offers the reader an update on the results of recently completed clinical trials, and additional guidance on appropriate selection of patients with DVT for catheter-directed thrombolytic therapy.
Keywords
Introduction
Deep vein thrombosis (DVT) causes substantial patient harm and has an annual incidence of approximately 1:1000 persons in the United States. 1 For many years, anticoagulation has been the mainstay of therapy for acute DVT since it reduces the occurrence of fatal pulmonary embolism (PE), symptomatic nonfatal PE, and recurrent venous thromboembolism (VTE). 2 Despite the use of anticoagulant therapy, many patients suffer long-term after-effects of DVT, specifically venous valve damage and post-thrombotic syndrome (PTS). This observation has prompted the evaluation and use of fibrinolytic drugs to actively dissolve thrombus in order to preserve long-term venous function.3,4
In recent years, the DVT treatment landscape has evolved, with widespread adoption of direct-acting anticoagulant drugs (DOACs), increased awareness of PTS and post-PE syndrome, a focus on cost-effectiveness, patient–provider collaborative decision-making, and increased integration of catheter-based therapies. Several pivotal randomized trials and prospective registries now enable greater insight into the associated risk-benefit ratio. This article will present the results of recent clinical trials, updated clinical practice guidelines, and evolving clinical practice, and thereby provide guidance on the use of thrombolytic therapy in the contemporary management of DVT.
Relevant trends in DVT treatment
For this review, the authors supplemented their existing article repositories with a limited PubMed search aimed at identifying recent randomized trials and clinical practice guidelines.
There is greater appreciation among clinicians that preventing recurrent VTE should not be the sole objective of DVT therapy. Among patients with symptomatic proximal DVT, nearly half will develop PTS and approximately 5% will develop venous ulceration.3,5,6 PTS is a leading predictor of patient health-related quality of life (QOL) 2 years after a DVT episode, and the severity of PTS correlates closely with the degree of QOL impairment.3,7 In addition, some patients with extensive thrombus experience severe leg pain and swelling that limits their activity for weeks to months after DVT onset.
With the advent of DOACs, the acceptability and feasibility of long-term anticoagulation have increased. Four DOACs have been approved for the treatment of VTE: rivaroxaban, apixaban, dabigatran, and edoxaban. These agents offer comparable efficacy and improved safety compared with warfarin and have largely reset expectations among many providers and patients for what constitutes a quality patient-centered experience with long-term anticoagulant therapy. 2 The FDA has approved targeted reversal agents for DOACs. 8 Some studies suggest that DOACs may prevent PTS better than warfarin. If proven definitively, this could reduce enthusiasm for more aggressive strategies such as thrombolytic therapy. 9
More clinicians are now aware that catheter-directed thrombolysis (CDT) offers a potential for rapid thrombus dissolution with smaller doses of fibrinolytic drug than are needed for systemic thrombolysis. New endovascular tools such as intravascular ultrasound have improved venous diagnosis, and dedicated venous stents have the potential to offer more precise and long-lasting treatment.10–12
Finally, there has been increasing teamwork among medical and endovascular providers, resulting in completion of important clinical trials and greater collaboration around important multidisciplinary initiatives to improve patient care, awareness, and education in VTE. Over time, these interactions have improved the level of refinement in developing clinical practice guidelines that reflect the clinical experiences of diverse specialties and offer the promise of moving forward with greater collaboration.
New insights into the clinical effectiveness of CDT and related therapies
CDT has integrated catheter-mounted devices that deliver additional energy (mechanical, ultrasound) into venous thrombus to enhance thrombus removal and reduce the need for larger doses of fibrinolytic drugs. Together with the incorporation of venous angioplasty and stent placement into CDT procedures, these changes have reduced treatment times.13,14
Several randomized controlled trials provide clinicians with guidance in identifying appropriate patients for CDT (Table 1). The first trial was the CAVENT Trial, a multicenter, open-label, randomized controlled trial in Norway that assigned (1:1 ratio) 209 patients with acute proximal DVT extending above the mid-thigh to receive, or not receive, traditional infusion CDT. CDT reduced the occurrence of PTS at 2 years (41% CDT vs 56% No-CDT, p = 0.047) and at 5 years (42% CDT vs 70% No-CDT, p < 0.01) follow-up.5,15 However, most PTS cases were mild, and no QOL benefit was identified beyond 6 months. 16 The U.S. ATTRACT Trial, funded by the National Institutes of Health (NIH), randomly assigned 692 anticoagulated patients with acute proximal DVT extending above the popliteal vein to pharmacomechanical CDT (PCDT; infusion CDT with use of mechanical thrombectomy devices), or No-PCDT. In this study, PCDT exerted no effect on 2-year PTS occurrence (47% PCDT vs 48% No-PCDT, p = 0.56). 6 Subgroup analysis found that patients ⩾ 65 years old were more likely to develop PTS after use of PCDT, compared with younger patients (p = 0.04). Finally, the Dutch CAVA Trial randomized 184 patients with acute iliofemoral DVT (defined as involving the common femoral vein or iliac vein) to receive, or not receive, ultrasound-assisted CDT (UA-CDT), and found no effect upon 1-year PTS (29% UA-CDT vs 35% No-UA-CDT, p = 0.42) or QOL. 17 The above trials were restricted to patients with lower extremity DVT. There are still no rigorous randomized trials evaluating CDT for upper extremity DVT.
Drug dosing and clinical outcomes in multicenter randomized catheter-directed thrombolysis trials.
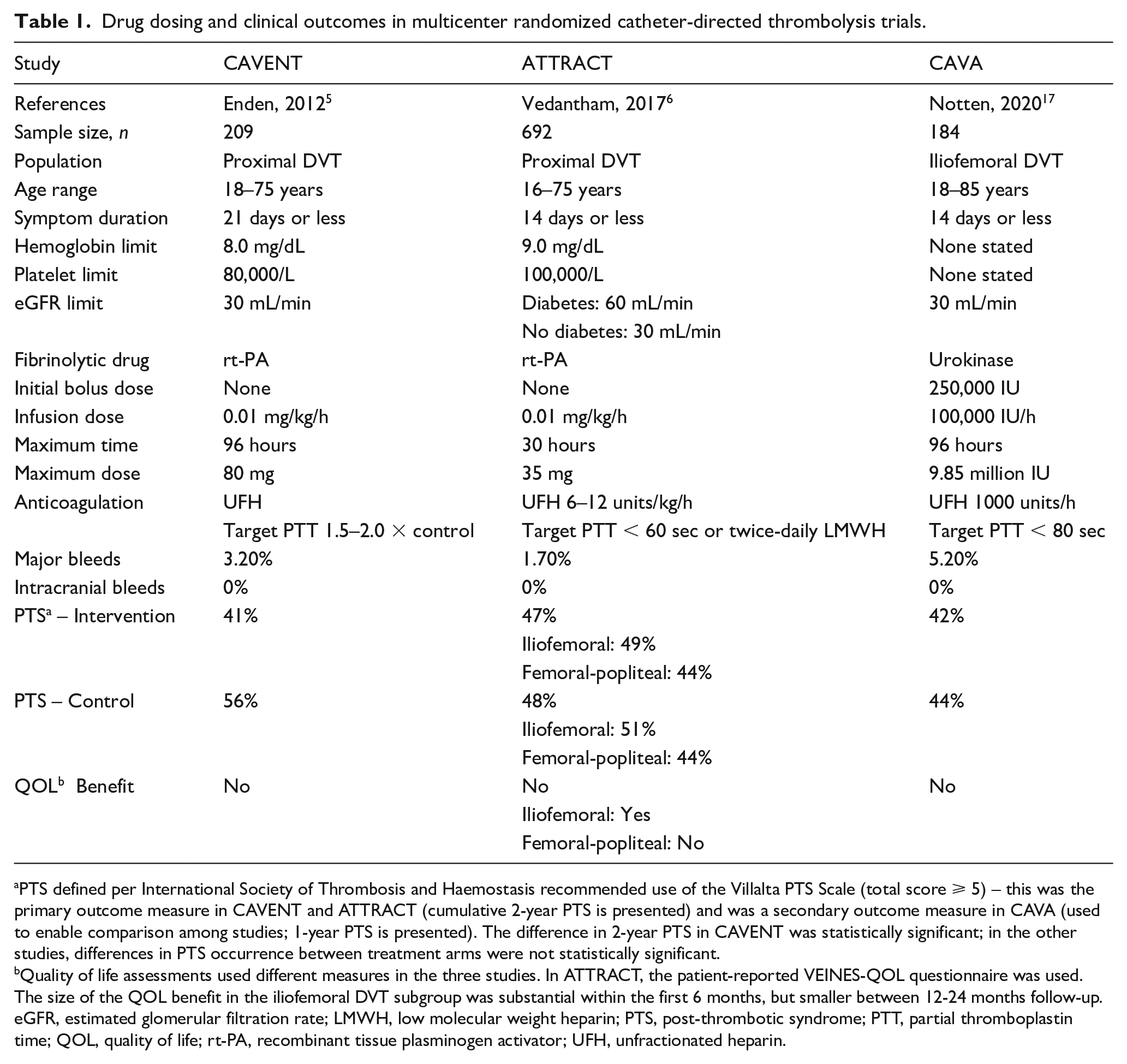
PTS defined per International Society of Thrombosis and Haemostasis recommended use of the Villalta PTS Scale (total score ⩾ 5) – this was the primary outcome measure in CAVENT and ATTRACT (cumulative 2-year PTS is presented) and was a secondary outcome measure in CAVA (used to enable comparison among studies; 1-year PTS is presented). The difference in 2-year PTS in CAVENT was statistically significant; in the other studies, differences in PTS occurrence between treatment arms were not statistically significant.
Quality of life assessments used different measures in the three studies. In ATTRACT, the patient-reported VEINES-QOL questionnaire was used. The size of the QOL benefit in the iliofemoral DVT subgroup was substantial within the first 6 months, but smaller between 12-24 months follow-up.
eGFR, estimated glomerular filtration rate; LMWH, low molecular weight heparin; PTS, post-thrombotic syndrome; PTT, partial thromboplastin time; QOL, quality of life; rt-PA, recombinant tissue plasminogen activator; UFH, unfractionated heparin.
Additional analyses of the ATTRACT Trial data have yielded important insights. First, although the occurrence of PTS was not reduced at any time point beyond 6 months, the severity of PTS over 2 years was significantly reduced in the PCDT recipients (p < 0.01). 6 Second, PCDT led to greater relief of leg pain and swelling within the first month. Third, these benefits in reducing PTS severity and in improving early symptom relief were primarily confined to patients presenting with acute iliofemoral DVT, with no benefits seen in patients with DVT limited to the femoral-popliteal veins18–20 (Table 1). In the iliofemoral DVT subgroup, the effects of PCDT on early symptom relief were associated with sizable early benefits in venous disease-specific QOL within the first 6 months (10 points on the validated VEINES-QOL scale, p < 0.01). The beneficial effects of PCDT on 2-year PTS severity were associated with a late venous disease-specific QOL benefit of smaller size (6 VEINES-QOL scale points, p = 0.01). 20
The time dependence of thrombus removal, flow restoration, and vein wall injury have prompted questions about whether the clinical effects of CDT/PCDT may vary by thrombus age. In one post hoc analysis of ATTRACT, PCDT may have been more effective in patients who entered the study at intermediate time points (symptom duration 4–8 days), compared with those randomized earlier or later. 21 However, this analysis was limited by its post hoc nature and the iterative data mining used to select the cut-points. The trial’s original prespecified subgroup analyses did not find patients with 0–7 days of symptoms to fare differently in PTS occurrence or QOL change from those with 7–14 days of symptoms.6,20 Hence, the above findings require additional confirmation before application to clinical practice.
The above studies were all of small to medium size with limited ability to make reliable observations on long-term disease mechanisms. Follow-up imaging in CAVENT and ATTRACT demonstrated that (a) CDT/PCDT was highly effective in removing thrombus initially (80% reduction in thrombus volume); (b) CDT/PCDT recipients were more likely to have open veins with less thrombus on late imaging than nonlysed patients; and (c) maintenance of open veins correlated with reduced PTS.22,23 However, even in patients treated with CDT/PCDT, a large proportion of treated veins had residual thrombus during follow-up. In ATTRACT, 40% of venous segments were noncompressible on ultrasound at 1-month and 1-year follow-up in the PCDT-treated group; similar observations were made in CAVENT. Hence, while symptomatic recurrent DVT did not differ statistically between treatment groups, it is possible that subclinical recurrent thrombosis may have contributed to the weak long-term effects of CDT/PCDT.
Catheter-based devices enable venous thrombus removal with use of limited or no thrombolytic drug (Figure 1). These options may be particularly useful for patients at high bleeding risk. The AngioJet Rheolytic Thrombectomy System (Boston Scientific Corporation, Marlborough, MA, USA), which uses high velocity saline jets to fragment and aspirate thrombus, is effective in removing thrombus after an initial fibrinolytic drug infusion and can sometimes remove limited amounts of thrombus without a fibrinolytic drug. In the ATTRACT Trial, use of the AngioJet for drug delivery and thrombus aspiration led to 80% thrombus clearance and (along with adjunctive balloon angioplasty and/or stent placement) flow restoration in the iliofemoral (100%) and femoral-popliteal (95%) venous segments. 10 The feasibility of removing venous thrombus has also been reported for the Indigo (Penumbra, Inc., Alameda, CA, USA), ClotTriever (Inari Medical, Inc., Irvine, CA, USA), and Cleaner (Argon Medical, Frisco, TX, USA) devices; however, no published prospective studies have documented their efficacy for DVT treatment. A prospective registry found the AngioVac System (Angiodynamics, Inc., Queensbury, NY, USA) to frequently enable aspiration of bulky thrombi in the inferior vena cava (IVC) or right heart through a large sheath after creation of a venous–venous bypass circuit. 24
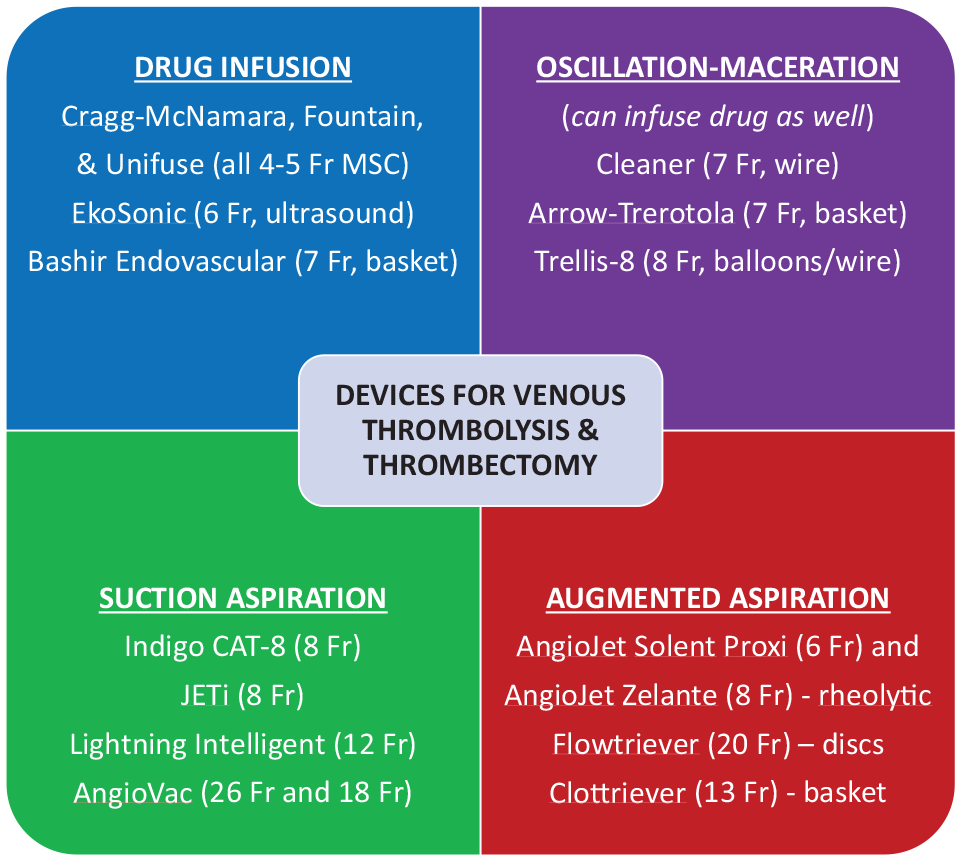
Mechanical thrombectomy devices that have been used for treatment of DVT. Little quality information is available on the effectiveness of these devices; only the AngioJet Solent Proxi and EkoSonic catheters have had device-specific outcomes reported from randomized trials.10,25,26 The Trellis-8 catheter is no longer available in the United States but is included since it was used in a randomized trial. 10 The AngioVac device requires a perfusionist to establish a venous–venous bypass circuit.
Endovascular thrombolysis methods have not been compared head-to-head in large, well-designed studies. In one pilot randomized trial, 48 patients with acute iliofemoral DVT received CDT using an ultrasound drug delivery catheter (EkoWave; Boston Scientific Corp.). This study did not find differences in thrombus removal, PTS, or safety between patients randomized to have the ultrasound energy utilized versus not utilized.25,26
The cost-effectiveness of endovascular intervention for acute DVT has been evaluated in two randomized studies (Table 2). In the CAVENT Trial, the use of additional CDT for acute proximal DVT was estimated to cost an additional US$20,429 per quality-adjusted life-year (QALY) gained, suggesting that it may be a cost-effective approach. 27 Cost-effectiveness results from the ATTRACT Trial were less favorable. The use of additional PCDT as a routine strategy for acute proximal DVT was estimated to cost US$222,041 per QALY gained, constituting low-value care by current US cost-effectiveness thresholds.28,29 Subgroup analysis in ATTRACT estimated the incremental cost-effectiveness of PCDT to be US$137,526 per QALY gained for acute iliofemoral DVT, suggesting PCDT to be of intermediate value for that subpopulation. For patients with isolated femoral-popliteal DVT, PCDT was associated with higher costs and lower projected QALYs than standard medical therapy, rendering standard medical therapy an economically dominant treatment strategy. The different cost-effectiveness estimates between the above studies are likely to stem from the different health systems in which the trials were carried out (Norway vs US), the greater use of mechanical thrombectomy devices and stents in ATTRACT, and the differences in the efficacy of CDT/PCDT in the two trials (sustained reduction in PTS with CDT in CAVENT yielded higher QALY gains than in ATTRACT, where the benefit was limited to the first 6 months).
Cost-effectiveness of endovascular thrombolysis in randomized trials.
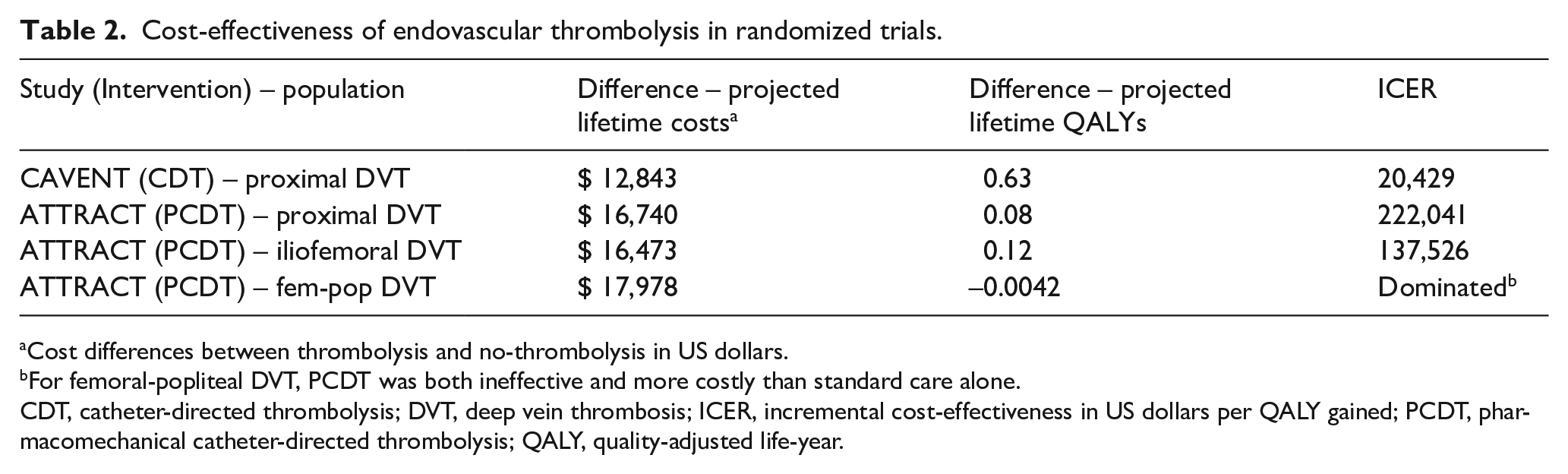
Cost differences between thrombolysis and no-thrombolysis in US dollars.
For femoral-popliteal DVT, PCDT was both ineffective and more costly than standard care alone.
CDT, catheter-directed thrombolysis; DVT, deep vein thrombosis; ICER, incremental cost-effectiveness in US dollars per QALY gained; PCDT, pharmacomechanical catheter-directed thrombolysis; QALY, quality-adjusted life-year.
New insights into the safety of CDT and related therapies
Safety considerations are paramount in considering the appropriate use of CDT and related endovascular procedures. In particular, it is important to pro-actively consider and make active efforts to mitigate the risk of bleeding related to use of fibrinolytic drugs and venous catheter placement. A fundamental rationale for the replacement of systemic thrombolysis with CDT procedures for DVT has been the potential for fewer major bleeding complications due to the reduced fibrinolytic drug doses used with CDT (e.g., mean dose of recombinant tissue plasminogen activator [rt-PA] in ATTRACT was 20 mg).
Good practices endorsed by societal practice guidelines30,31 include: (a) careful pretreatment planning and patient selection. Patients with active/recent bleeding, recent surgery or trauma, severe liver disease, known bleeding diatheses, abnormalities of coagulation or of platelet number or function, history of stroke, and lesions in critical locations (e.g., central nervous system – patients with malignancies known to metastasize to the central nervous system should undergo brain imaging to exclude metastatic lesions) should be excluded from receiving DVT thrombolysis; (b) pretreatment planning – the time since last anticoagulant therapy should be known and any long-acting anticoagulants should be allowed to become subtherapeutic before thrombolysis is initiated. For patients receiving unfractionated heparin, partial thromboplastin time (PTT) values should not be supra-therapeutic during thrombolysis. Hematocrit and renal function should be known; (c) careful procedural technique – venous access should be obtained with ultrasound guidance. When recombinant t-PA (rt-PA) is used, weight-based dosing at 0.01 mg/kg/h (not to exceed 1.0 mg/h) is recommended; (d) close monitoring – during thrombolytic infusions, patients should be placed at bedrest with immobility of the catheter-bearing extremity, frequent contact with nursing staff, and blood draws for hematocrit, platelet count, and PTT at least every 12 hours. There must be excellent communication between physician and nurse, and the physician should be immediately informed of important findings that may represent signs of current or impending bleeding, such as marked peri-catheter oozing, minor sentinel bleeds (e.g., epistaxis), elevated PTT levels, or new symptoms (e.g., headache, neurological symptoms). Arterial punctures and intramuscular injections should be avoided if possible. Thrombolytic progress should be assessed by venography at least every 24 hours to enable timely cessation of the thrombolytic infusion.
Randomized trials with stringent patient selection criteria will often have lower rates of bleeding than real-world registry populations. Rates of major bleeding in the intervention versus control arms of the three trials above were, respectively, 3.2% versus 0% (CAVENT), 1.7% versus 0.3%, p = 0.049 (ATTRACT), and 5.6% versus 0% (CAVA, which used a higher age cut-off for inclusion), with no fatal or intracranial bleeds5,6,15 (Table 1). These findings were in general accord with the 2.8% major bleeding rate observed in a pooled analysis (n = 1531) of observational studies published from 2004 to 2014, suggesting that modern CDT can be delivered with reasonable safety using stringent patient selection criteria, meticulous procedural technique, and quality peri-procedure monitoring. 30 A large observational study using the National Inpatient Sample administrative database estimated the rates of intracranial bleeding to be approximately 0.9% in patients with DVT who received thrombolytic therapy, versus 0.3% in patients who did not receive thrombolysis (p = 0.03), which is consistent with a previous registry.15,32
In the ATTRACT Trial, PCDT increased major bleeding (1.7% PCDT vs 0.3% No-PCDT, p = 0.049). 6 Of the six major bleeds in the PCDT arm, none was fatal or intracranial; two each involved the gastrointestinal tract, the venous access site, and the retroperitoneum (the latter necessitating catheter embolization). The risk of major bleeding with PCDT was higher in patients ⩾ 65 years of age (p = 0.0007) compared with younger patients. Fifteen PCDT recipients in ATTRACT had a major or minor bleed within 10 days after randomization. Higher systolic blood pressure, worse renal function, and age ⩾ 65 years were associated with an increased risk of any (major or minor) bleeding. However, multivariable logistic regression found age ⩾ 65 years to be the only independent predictor of the risk of any (major or minor) bleeding.
In addition to preventing bleeding, additional measures to ensure patient safety are to: (a) prehydrate patients with preexisting renal insufficiency; (b) premedicate patients with contrast allergies; (c) monitor vital signs and oxygen saturation; (d) use a meticulous sterile technique; (e) ensure adequate anticoagulation before, during, and after the endovascular procedure to prevent PE; and (f) avoid the routine use of IVC filters before infusion CDT. The risk of symptomatic PE with infusion CDT is approximately 1%; in contrast, long-term risks of filters may include device migration, embolization, fracture, and recurrent DVT. 33 Placement of a retrievable filter may occasionally be reasonable for patients with extensive thrombus who have poor underlying cardiopulmonary status; if placed, filters should be removed as soon as clinically appropriate after PCDT; and (g) operators should be aware of device-specific risks – for example, the AngioJet (Boston Scientific Corp.) is known to cause bradycardia infrequently and met-hemoglobinuria commonly. Aspiration devices (e.g., Indigo, ClotTriever) can remove large amounts of blood very quickly if not closely monitored. Devices that require large sheaths (e.g., AngioVac) may increase the risk for access site complications. Device-specific rates of recurrent DVT have not been prospectively documented to date.
Treatment recommendations and converging clinical practice guidelines
In June 2019, the National Institute of Health and Care Excellence (NICE) in the United Kingdom issued guidance acknowledging that PCDT can be used for patients with acute iliofemoral DVT with special arrangements for informed consent, local governance, and quality improvement, but that it remains investigational for femoral-popliteal DVT. 34 The 2020 American Society of Hematology VTE Treatment Guidelines state that thrombolysis is reasonable to consider for patients with limb-threatening DVT (phlegmasia cerulea dolens) and for selected younger patients at low risk for bleeding with symptomatic DVT involving the iliac and common femoral veins, but that its use should be rare for femoral-popliteal DVT. 2 In 2021, the European Society of Vascular Surgery issued guidelines that recommend consideration of early thrombus removal strategies for selected patients with acute iliofemoral DVT but not for less extensive DVT. 35 These and previous guidelines also suggest use of CDT and first rib resection for selected, highly symptomatic patients with axillosubclavian DVT who are at low risk for bleeding when there is demonstrable thoracic outlet compression. Overall, the newfound concordance of guidelines from medical and surgical societies is a welcome advance.
It is important to distinguish among (a) the rare patients with DVT causing acute limb-threatening circulatory compromise such as phlegmasia cerulea dolens (urgent CDT/PCDT is recommended unless bleeding risk is prohibitive); and (b) first-line versus salvage uses of CDT/PCDT. We suggest first-line use of CDT/PCDT in young (< 65 years), symptomatic patients with acute iliofemoral DVT, low bleeding risk, and high preexisting functional status. Because the predominant benefits of CDT relate to symptom relief over the first 6 months, it may be reasonable to first try anticoagulation alone for 5–7 days in patients with iliofemoral DVT whose symptoms do not limit activity, to monitor them closely, and to utilize CDT/PCDT if symptoms worsen. In contrast, patients with DVT limited to the femoral, popliteal, and/or tibial veins should rarely be lysed. In informed consent discussions, the risks of major or intracranial bleeding, the reasonable likelihood of seeing short-term benefits in symptom relief, and the more modest possibility of experiencing a reduction in long-term PTS severity should be mentioned.
Future directions
The demonstration of apparent differences in treatment outcome among anatomical DVT subgroups should prompt efforts to identify high-risk patients (e.g., iliofemoral DVT) for closer monitoring and investigation to determine if they would benefit from augmented antithrombotic, compressive, anti-inflammatory, or endovascular treatment strategies. More study is needed to determine the optimal concomitant antithrombotic therapy to utilize during and after CDT and to understand the full range of factors that influence the development of PTS over time.
Patients with cancer merit specialized consideration since they are at higher risk of bleeding and re-thrombosis even with anticoagulant therapy alone, and these risks may be compounded by new therapies such as angiogenesis inhibitors (promote bleeding), novel biologics (promote thrombosis), and other agents. Since such patients have been largely excluded from randomized trials of endovascular interventions to date, additional studies targeted to the cancer population should examine differences in venous biology, the impact of chronic venous symptoms upon quality of life, and the effects of medical and endovascular interventions.
In 2020, the coronavirus pandemic demonstrated a potential relationship between viral infection and thrombosis that is still being characterized. 36 These clinical observations directed new attention to the biology of thrombosis; in particular, in situ venous thrombosis. New insights gained from the study of the novel coronavirus will be relevant to our overall understanding of the genesis of PTS and the impact of catheter-directed therapies.
Conclusions
Recently completed randomized trials have yielded important insights into the short-term and long-term effects of thrombolytic therapy in patients with DVT (Table 3). Key points to remember regarding this topic are summarized in Table 4. At present, the use of CDT should focus on patients with acute iliofemoral DVT, severe symptoms, low bleeding risk, and good functional status, with patient–provider collaborative discussions placing more emphasis on the likelihood of achieving short-term symptom relief and QOL improvement rather than long-term benefits. Careful patient selection is important to optimize safety. In particular, elderly patients are at increased risk for bleeding with thrombolysis. Fortunately, treatment guidelines from medical and surgical societies have become more closely aligned, boding well for future efforts to develop additional thrombolytic therapy strategies that optimize patient outcomes.
Important recent publications.
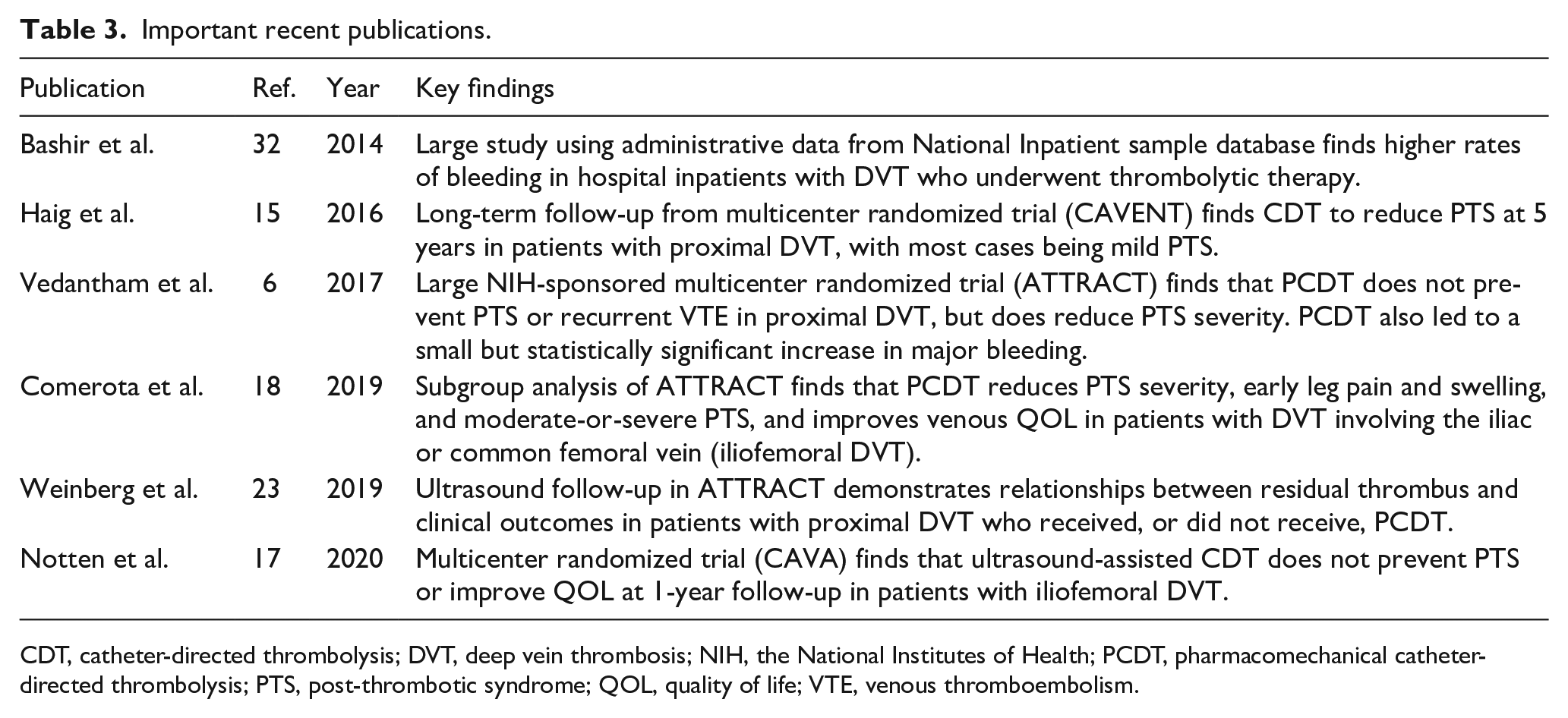
CDT, catheter-directed thrombolysis; DVT, deep vein thrombosis; NIH, the National Institutes of Health; PCDT, pharmacomechanical catheter-directed thrombolysis; PTS, post-thrombotic syndrome; QOL, quality of life; VTE, venous thromboembolism.
Key points to remember.

CDT, catheter-directed thrombolysis; DVT, deep vein thrombosis; PE, pulmonary embolism; PTS, post-thrombotic syndrome; VTE, venous thromboembolism.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Samuel Z Goldhaber – research grant support from Bristol Myers Squibb, Boston Scientific Corporation, Janssen; personal fees from Bayer, Agile. Elizabeth A Magnuson – research grant support from Abbott Vascular, Cardiovascular Systems, Inc., Corvia Medical, Edwards Lifesciences, Svelte Medical Systems. Khaja M Chinnakondepalli – nothing to disclose. David J Cohen – research grant support and consulting fees from Abbott Vascular, Boston Scientific Corporation, Medtronic, Phillips. Suresh Vedantham – research grant support (in kind support only) from Medi USA.
Funding
Dr. Vedantham’s work is supported by grants from the National Heart Lung and Blood Institute (U01-HL088476, UH3-HL138325) and National Center for Advancing Translational Sciences (UL1-TR002345).
