Abstract
Venous thromboembolism (VTE) is a common vascular condition. New medications are available to prevent hospital-associated VTE. Strategies are being studied to increase appropriate diagnostic testing utilization. Management of deep vein thrombosis (DVT) and pulmonary embolism (PE) has evolved with the advent of new anticoagulant options and catheter-directed intervention. In light of this, providers are commonly challenged with the decision regarding inpatient versus outpatient management. Which patients require long-term (> 3 months) anticoagulation is challenging and multiple clinical prediction models may be used to help determine the risk–benefit ratio in each patient. The management of VTE is an ongoing area of research and is rapidly evolving.
Keywords
Continuing medical education (CME) credits and maintenance of certification (MOC) points are available for reading this article. This opportunity is made possible through a joint partnership with University of Virginia School of Medicine (UVASOM). For instructions, please visit the Society for Vascular Medicine’s website at
The University of Virginia School of Medicine (UVASOM) is accredited by the ACCME to provide continuing medical education for physicians. UVASOM designates this journal-based CME activity for a maximum of one AMA PRA Category 1 Credit™. Participants should claim only the credit commensurate with the extent of their participation in the activity. UVASOM, as an accredited provider, awards 1 hour of participation (consistent with the one AMA PRA Category 1 Credit™) to participants who successfully complete this educational activity. Successful completion of this CME activity, which includes participation in the evaluation component, enables the participant to earn up to one MOC II point in the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) program. It is the CME provider’s responsibility to submit participant completion information to ACCME for the purpose of granting ABIM MOC II credit. UVASOM maintains a record of participation for 6 years.
The faculty, staff and planning committee of the University of Virginia Office of Continuing Medical Education have no financial affiliations to disclose. The CME planning committee disclosed the following: Heather Gornik is supported by CVR Global and receives intellectual property rights from Summit Doppler Systems, Inc. and intellectual property rights and stock/ownership from FlexLife Health. Aditya Sharma is supported by National Institute of Health Sciences, AstraZeneca, and Biomet Biologics. Valerie Clark has no financial affiliations to disclose. The authors have disclosed the following: Maya Serhal is supported by DAIICHI Sanyko. Geoff Barnes is supported by BMS/Pfizer, Janssen, Portola, Blue Cross Blue Shield of Michigan, and the National Heart, Lung, and Blood Institute (K01HL145367).
Participants are expected to download and read the article, then complete the CME evaluation and MOC post-test questions with an 80% score or higher to receive credit. The post-test is available online from
Introduction
Venous thromboembolism (VTE), comprised of deep vein thrombosis (DVT) and/or pulmonary embolism (PE), is a common vascular condition. With the introduction of new treatment strategies and policy efforts focused on reducing the development of VTE, much has changed in the approach towards prevention and treatment of VTE over the past decade. In this review, we aim to update clinicians on the approaches to VTE prevention, diagnosis, and treatment with a focus on recently published studies.
The annual incidence of VTE is estimated at more than 300,000–600,000 in the United States and more than 700,000 across six European countries. 1 While the incidence of VTE grows exponentially as people age, PE accounts for an increasing proportion of all VTE events among these older adults. Overall lifetime estimates of VTE in the United States are 8.1% (95% CI, 7.1% to 8.7%). 1 These estimates are higher among African Americans (11.5%) and obese individuals (10.9%). Recent data have suggested that the overall incidence of VTE has not changed significantly since the early 2000s despite efforts to improve VTE prevention in the hospital setting. 2
Approximately 20% of all VTE events are classified as ‘provoked’ because of recent immobilization, trauma, surgery, or hospitalization. 1 An additional 30% of VTE events are associated with cancer, while the remaining 50% are considered ‘unprovoked’. Additional independent risk factors for the development of VTE include increasing age, confinement in a hospital or nursing home, active cancer, presence of a central venous catheter or transvenous pacemaker, and a recent pregnancy.
Prevention of venous thromboembolism (VTE)
Patients hospitalized for acute medical illness are often at risk for VTE. While the overall risk of VTE has been estimated at 3.3%, the majority of these events occurred in the first 30 days, but most often after hospital discharge. 3 This is particularly true in North America and Western Europe where hospital length of stay is declining. Current use of in-hospital VTE prophylaxis centers on either unfractionated heparin or low-molecular-weight heparin (LMWH) therapy. 4 However, appropriate use of pharmacologic prophylaxis ranges between 50% and 90% at different hospitals. 5
A number of risk assessment models exist for hospitalized patients. These include the Caprini and Rogers risk scores, most commonly used in surgical patients, and the Padua, IMPROVE, Kucher, and Intermountain scores for hospitalized medical patients.6–10 Implementation of these tools to identify appropriate patients for VTE prophylaxis is an important and ongoing area of research. In general, multi-component strategies are needed, including integration into electronic medical record and computerized physician order systems, robust education, and multidisciplinary leadership teams.
Prevention in the non-surgical hospitalized patient
While most clinicians focus on the prevention of hospital-associated VTE with in-hospital use of anticoagulants, recent evidence has suggested that the risk of hospital-associated VTE persists beyond the hospitalized period. In fact, one nationwide study from the United States demonstrated that more than one-half of all hospital-associated VTE events occurred after hospital discharge. 3
Prior studies have examined the potential role of extending VTE prophylaxis beyond the initial hospital period for medically ill patients. In the Extended Clinical Prophylaxis in Acutely Ill Medical Patients (EXCLAIM) trial, extended-duration enoxaparin reduced the incidence of VTE compared to placebo (2.5% vs 4.0%, absolute difference 95% CI, −0.5% to −2.5%) but with an increase in major bleeding risk (0.8% vs 0.3%, absolute difference 95% CI, 0.1% to 0.9%). 11 In the Apixaban Dosing to Optimize Protection from Thrombosis (ADOPT) trial, low-dose apixaban (2.5 mg twice a day for 30 days) was compared to enoxaparin (40 mg daily for 6–14 days) and demonstrated no significant reduction in VTE events (2.7% vs 3.1%, relative risk (RR) 0.87, 95% CI, 0.62 to 1.23) and an increased risk of major bleeding (0.5% vs 0.2%, RR 2.58, 95% CI, 1.02 to 7.24). 12 In the Multicenter, Randomized, Parallel Group Efficacy and Safety Study for the Prevention of Venous Thromboembolism in Hospitalized Acutely Ill Medical Patients Comparing Rivaroxaban with Enoxaparin (MAGELLAN), low-dose rivaroxaban (10 mg daily for 31–39 days) was compared to enoxaparin (40 mg daily for 6–14 days). 13 In this study, rivaroxaban was associated with a similar risk of VTE (2.7% vs 2.7%, RR 0.97, 95% CI, 0.71 to 1.31) but more bleeding (2.8% vs 1.2%, RR 2.3, 95% CI, 1.63 to 3.17) than enoxaparin. Overall, these trials are felt to have been unsuccessful either due to inadequate selection of high-risk patients or medication doses and durations that do not optimally balance VTE prevention with bleeding risk.
Most recently, the newest direct oral anticoagulant (DOAC), betrixaban, was tested in the Acute Medically Ill VTE Prevention with Extended Duration Betrixaban (APEX) trial. 14 In this trial, patients aged 40 years or older who had been hospitalized for an acute medical illness were randomized to receive either betrixaban 80 mg daily for 35–42 days or enoxaparin 40 mg daily for 5–14 days. In an attempt to enrich the study for a population at higher risk for VTE, they developed a three-cohort analysis plan based largely on findings from the ADOPT and MAGELLAN trials.12,13 The first cohort included patients with an elevated D-dimer level at the time of admission. The second cohort included those patients with an elevated D-dimer or who were aged ⩾ 75 years. The final cohort included all eligible patients. As shown in Table 1, the primary composite outcome (asymptomatic proximal DVT and symptomatic VTE) occurred numerically less often in the betrixaban arm than the enoxaparin arm from cohort 1. However, this did not reach the pre-specified threshold for statistical significance. There was a statistically significant reduction in VTE from cohort 2 and the overall trial population; however, these were considered exploratory. Major bleeding risk was similar in both randomized groups. However, major and clinically relevant non-major bleeding was more common in the betrixaban group compared to the enoxaparin group in all three analysis cohorts. Secondary analysis from the APEX trial has demonstrated significant reductions in VTE risk among patients with a prior history of VTE (hazard ratio (HR) 0.54, 95% CI, 0.36 to 0.81) and a reduction in ischemic stroke (0.48% among betrixaban vs 0.91% among enoxaparin, RR 0.53, 95% CI, 0.30 to 0.94).15,16
Safety and efficacy outcomes of extended DOACs or standard enoxaparin for VTE prophylaxis in hospitalized medical patients.
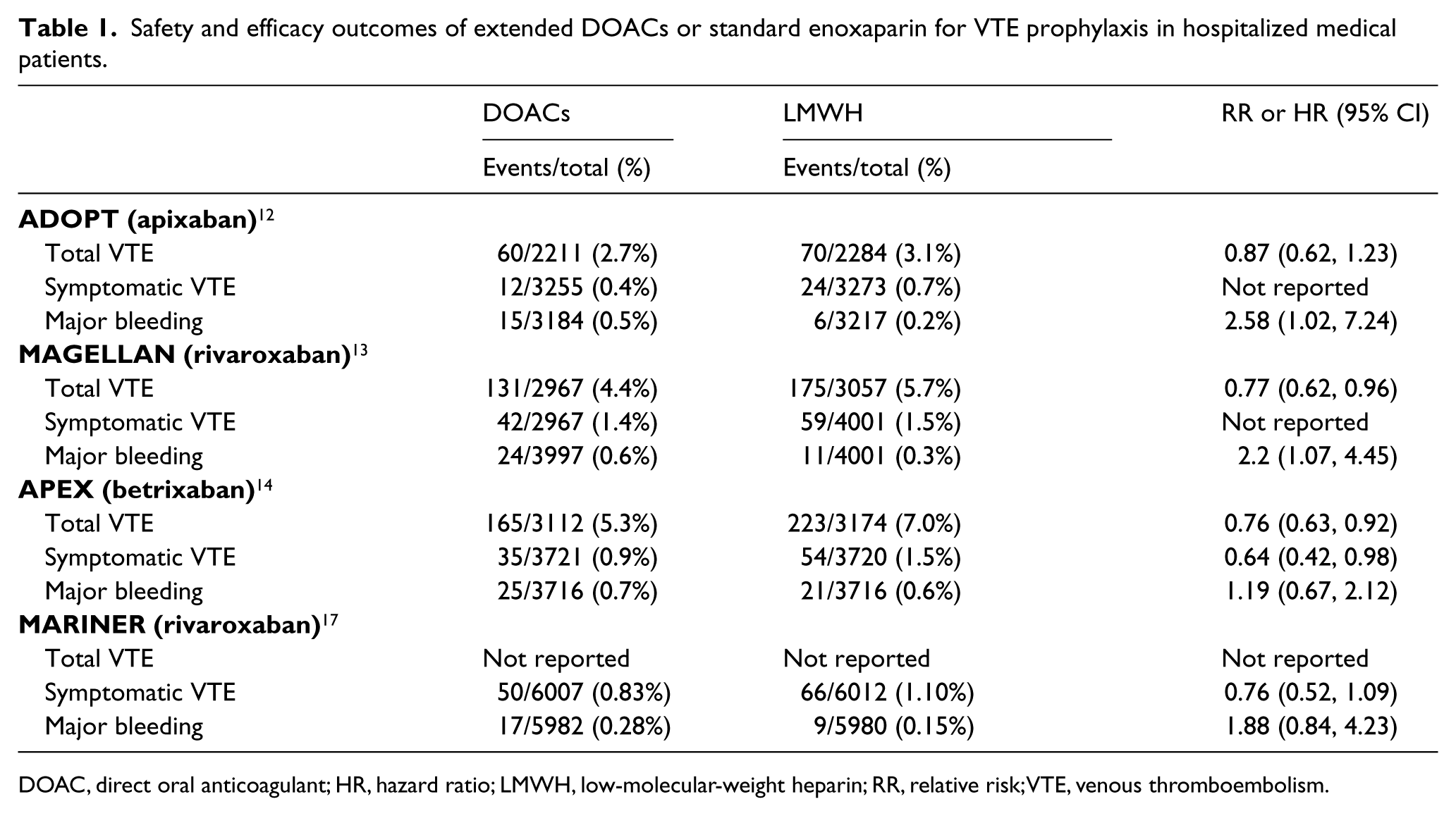
DOAC, direct oral anticoagulant; HR, hazard ratio; LMWH, low-molecular-weight heparin; RR, relative risk; VTE, venous thromboembolism.
Based on an independent analysis of this study’s dataset, the US Food and Drug Administration (FDA) has approved the use of betrixaban for extended VTE prophylaxis in hospitalized acutely ill medical patients. However, betrixaban currently has not been approved by the European Medicines Agency. It is unclear exactly why betrixaban was able to show favorable risk-to-benefit outcomes while enoxaparin, rivaroxaban, and apixaban were not able to demonstrate this favorable benefit. However, some have speculated it may be due to the higher-risk patient population, liberal use of D-dimer testing, longer half-life, and less renal clearance compared to the other agents.
The recently published Rivaroxaban for Thromboprophylaxis after Hospitalization for Medical Illness (MARINER) study randomized hospitalized medical patients at high risk for VTE to receive either rivaroxaban 10 mg daily or placebo for 45 days at the time of hospital discharge (Table 1). 17 Of the 12,019 patients included in the intention-to-treat analysis, the composite of symptomatic VTE or VTE-related death occurred in 50/6007 (0.83%) patients in the rivaroxaban group compared to 66/6012 (1.10%) patients in the placebo group (HR 0.76, 95% CI, 0.52 to 1.09). Symptomatic, non-fatal VTE occurred less often in the rivaroxaban-treated group (0.18% vs 0.42%, HR 0.44, 95% CI, 0.22 to 0.89). Major bleeding occurred in 17/5982 (0.28%) of the rivaroxaban-treated patients compared to 9/5980 (0.15%) of the placebo-treated patients (HR 1.88, 95% CI, 0.84 to 4.23).
Prevention in the surgical patient
For patients undergoing hip and knee surgery, VTE is a well-recognized complication. Guidelines typically recommend anticoagulant prophylaxis for 10–35 days. 18 Recently, use of DOACs has become common given their convenience and efficacy compared to enoxaparin and warfarin. Given aspirin’s low-cost, well-established, side-effect profile, and prior evidence for effective thromboprophylaxis, a multicenter randomized trial was recently conducted in Canada to compare the use of low-dose aspirin (81 mg daily) to rivaroxaban (10 mg) in postoperative orthopedic surgery patients. 19 All patients were given rivaroxaban 10 mg daily for the first 5 days postoperatively and then were randomly assigned to either continue rivaroxaban or switch to aspirin. Therapy was continued for an additional 9 days following total knee arthroplasty and for an additional 30 days following total hip arthroplasty. The primary effectiveness outcome (symptomatic VTE at 90 days) occurred in 0.64% of aspirin-treated patients and 0.70% of rivaroxaban-treated patients (p < 0.001 for non-inferiority, p = 0.84 for superiority). Major bleeding occurred in 0.47% of aspirin-treated patients and 0.29% of rivaroxaban-treated patients (risk difference 0.18%, 95% CI, –0.65% to 0.29%).
Owing to the high risk of VTE, patients undergoing abdominal or pelvic surgery for cancer are recommended to receive extended VTE prophylaxis (4 weeks) with LMWH as long as the bleeding risk is not prohibitive. A recent meta-analysis confirmed that the use of extended VTE prophylaxis was associated with reduced incidence of VTE without an increased risk of major bleeding or a change in 3-month mortality. 20
Clinical diagnosis of VTE
Classic symptoms of DVT include erythema, warmth, discomfort, or asymmetric limb swelling. However, these are neither sensitive nor specific on their own, as a variety of other conditions (including cellulitis) can lead to similar symptoms. To assist with the diagnosis, a variety of clinical prediction rules have been developed to quantify the ‘pretest probability’ of a DVT. These include the Hamilton score, the QThrombosis algorithm, the Amsterdam Maastricht Utrecht Study on thromboEmbolism (AMUSE) score, and the Wells score. Of these, the Wells score is most commonly known and used. A score is calculated based on the results of the clinical variables, and the pre-test probability for DVT is determined. Often, these risk prediction models (especially the Wells score) are combined with a moderately or highly sensitive D-dimer test to improve their sensitivity.21–26 After calculating the pre-test probability, the clinician should then determine if further imaging is needed.
Diagnosis of PE in the emergency department can be challenging, given that the most prominent symptoms (chest discomfort and shortness of breath) can overlap with other common and serious conditions (e.g. acute coronary syndrome). Again, multiple clinical prediction models may be used to help determine pre-test probability of PE. The Wells score incorporates assessment of multiple factors including clinical gestalt, comorbidities, and signs and symptoms at the time of presentation (Table 2). 27 The Pulmonary Embolism Rule-out Criteria (PERC) involves assessing eight clinical items to identify patients at very low pre-test probability for PE (Table 2). In the multicenter randomized clinical trial (PROPER), patients with a low gestalt clinical probability of PE were assessed. 28 Using crossover cluster randomization at the center level, patients found to have no items on the PERC score were ‘ruled out’ for acute PE. Use of the PERC score was associated with fewer computed tomography (CT) scans (13% vs 23%, p < 0.001) and a 36 minute (95% CI, 4 to 68 minutes) shorter emergency department stay, with only one patient subsequently being diagnosed with a PE from the PERC score group compared to the control group.
PE clinical prediction rules.

Score > 6 points places patient at high probability for PE; score of > 2 and < 6 points places patient at moderate probability for PE; score of < 2 points places patient at low risk for PE.
Presence of any PERC element suggests further evaluation is needed.
Surgery or trauma within 4 weeks requiring general anesthesia.
Oral contraceptives, hormone replacement, or estrogen-containing hormone use in either men or women.
DVT, deep vein thrombosis; PE, pulmonary embolism.
D-dimer has long been used to assist in the diagnosis of VTE. More recently, the role of age-adjusted D-dimer has been explored in patients with VTE. The plasma concentrations of D-dimer in healthy patients increases with age. 29 A D-dimer value > 500 µg/L was found in 16%, 29%, and 50% of patients between 50 and 60 years of age, 60 and 70 years of age, and ⩾ 70 years of age, respectively. Multiple authors have suggested using an age-adjusted cut-off value for D-dimer (not 500 µg/L for everyone).30–33 In the ADJUST-PE study, a retrospective review was performed and a progressive D-dimer cutoff adjusted to age was validated in a sample of 1712 patients. Authors suggest that the optimal age-adjusted cutoff was defined as the patient’s age multiplied by 10 in patients aged ⩾ 50 years. Notably, in patients with a D-dimer level between 500 µg/L and the age-adjusted cutoff, only 0.3% (95% CI, 0.1% to 1.7%) were subsequently diagnosed with VTE (non-fatal PE). Use of the age-adjusted D-dimer increased the proportion of patients aged 75+ years where PE could be excluded from 6.4% to 29.7% without any additional false-negative findings. 34 D-dimer must be interpreted in the clinical context of the patient.
Management of acute DVT: the ‘initial’, ‘long-term’, and ‘extended’ phases
After the diagnosis of VTE has been made, clinicians must decide whether or not anticoagulation is indicated. In select patients, clinical surveillance may be appropriate. For example, in patients with isolated subsegmental PE with no associated proximal DVT and who are at low risk for VTE recurrence, there is a Class 2C recommendation for clinical surveillance over anticoagulation. 35 Patients with isolated distal DVT, without severe symptoms or risk factors for propagation (including markedly elevated D-dimer, extensive thrombosis, thrombosis close to the proximal veins, no reversible provoking factor, active cancer, inpatient hospitalization, and history of VTE), serial imaging with duplex ultrasonography is recommended (Class 2C recommendation).
If a decision is made to initiate treatment with anticoagulation, then the treatment of VTE can be divided into three phases: initial, long-term, and extended phases of anticoagulation. The ‘initial’ phase of anticoagulation encompasses the first 5–10 days of treatment and historically represents the time patients are on parenteral anticoagulation. The ‘long-term’ phase represents the first 3–6 months of anticoagulation. The ‘initial’ and ‘long-term’ phases of anticoagulation are critical in preventing propagation of thrombosis and embolization. The ‘extended’ phase extends beyond the initial 3–6 months. 36 Clinical decision-making regarding the duration of anticoagulation and risk–benefit ratio of anticoagulation is critical and an area of recent research. Key factors that influence risk–benefit analysis include the presence of any provoking factors, age, sex, obesity, and signs of the post-thrombotic syndrome.
Anticoagulants (Figure 1) can broadly be divided into parenteral anticoagulants (heparins, fondaparinux, and direct thrombin inhibitors), vitamin K antagonists (warfarin), and DOACs (includes Xa inhibitors and direct thrombin inhibitors). Though DOACs do not require heparin bridging (unlike warfarin), a lead in with a parenteral anticoagulant for 5 days is recommended in patients being treated with dabigatran and edoxaban.
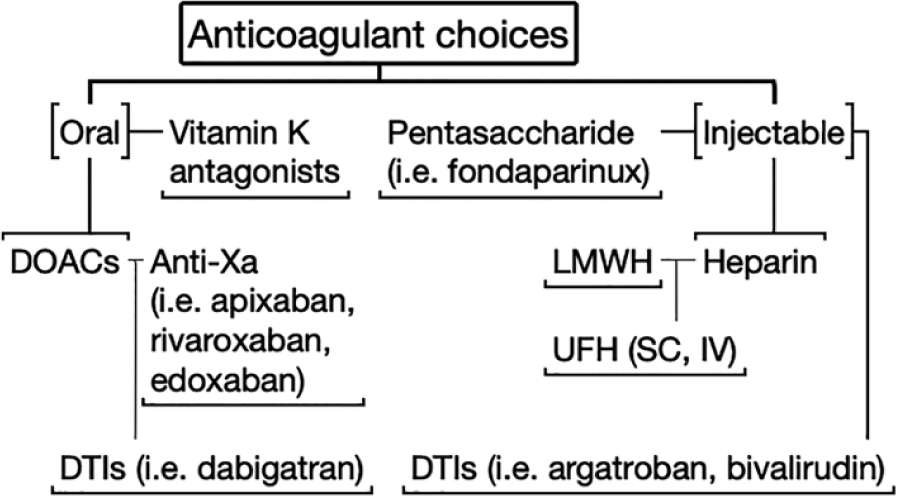
A summary of anticoagulant choices for acute VTE.
Inpatient versus outpatient treatment of VTE
Given multiple oral anticoagulant options, clinicians commonly face a challenging decision regarding whether to treat patients with VTE as inpatients or outpatients. The American College of Chest Physicians (ACCP) recommend home treatment whenever feasible (Grade 1B). 35 Broadly speaking, two major considerations include safety and practicality. Factors that should be considered include the availability and cost of the medication, the ability of patients to follow directions, and the ability of patients to have close follow-up.
When managing PE, multiple clinical prediction tools have been developed that help risk stratify patients to guide physicians regarding inpatient versus outpatient management, including the Pulmonary Embolism Severity Index (PESI) score. Aujesky and colleagues demonstrate no statistical difference in outcomes in patients with low-risk PE, with a PESI score of I or II, supporting outpatient management of these patients. 37 However, no externally validated clinical prediction tool is available to predict the safety of outpatient DVT management.
In the past decade, the introduction of DOACs and oral only therapy has made outpatient treatment of acute VTE more streamlined and acceptable. In particular, use of either rivaroxaban or apixaban (both now available with starter packs) is guideline-recommended for initial therapy and will avoid the need for hospitalization to administer parenteral heparin. 35
Multiple studies have compared outpatient and inpatient management of VTE. However, it is important to note that patients in these studies were not randomized to in-home versus outpatient treatment, and there were high exclusion rates. Preliminarily, it appears that at-home treatment of DVT may be associated with lower rates of major bleeding. 38 Characteristics that may favor inpatient management include recent surgery (within 7 days), other active medical issues that require inpatient evaluation, cardiopulmonary instability, severe symptomatic venous obstruction, severe thrombocytopenia, medical non-compliance, geographical or telephone inaccessibility, poor hepatic function, and unstable renal function. 39 Patients undergoing outpatient treatment must be cautiously selected and, ideally, have short-term follow-up with providers. Most patients with acute DVT, especially those with isolated distal DVT, can usually be treated as an outpatient. 40
Patients who have symptomatic venous obstruction may benefit from hospitalization. This can serve to initiate anticoagulation, allow for leg elevation and compression, and monitor for signs of progressive disease or impending phlegmasia. Additionally, patients may be considered for advanced therapeutic options for the treatment of extensive venous thrombosis – namely, pharmacomechanical catheter-directed thrombolysis (CDT). 41
Catheter-directed therapies for DVT
Two large randomized trials have been undertaken to determine the efficacy of catheter-directed therapy and reducing the risk of post-thrombotic syndrome. In the first study, the Catheter-directed Venous Thrombolysis in acute iliofemoral vein thrombosis (CaVenT) researchers enrolled a total of 209 participants who were randomized to CDT and conventional treatment with anticoagulation alone. 41 The absolute risk reduction in post-thrombotic syndrome among patients who underwent CDT was 14.4% (9% CI, 0.2% to 27.9%) at 24 months. There was a total of 20 bleeding complications related to CDT, including three major and five clinically relevant non-major bleeding events. 41
Subsequently, the Acute Venous Thrombosis: Thrombus Removal With Adjunctive Catheter-Directed Thrombolysis (ATTRACT) trial enrolled a total of 691 patients with acute proximal DVT (iliofemoral or femoropopliteal) to receive anticoagulation alone or anticoagulation plus pharmacomechanical thrombolysis. Post-thrombotic syndrome was common and not statistically different in both groups after 24 months (47% in the intervention group vs 48% in the control group). Notably, there appeared to be a trend towards lower risk of post-thrombotic syndrome in patients with iliofemoral DVT after CDT compared to anticoagulation alone. 42 The DUTCH CAVA-trial is ongoing and results are eagerly awaited (NCT00970619).
In light of these two large randomized trials, anticoagulation and compression remain first-line therapy for the vast majority of patients with acute DVT. In the 2012 ACCP guidelines, use of graduated compression stockings for 2 years after diagnosis of acute, symptomatic DVT to prevent the occurrence of post-thrombotic syndrome was recommended. 38 Subsequently, in 2014, the Compression Stockings to Prevent Post-Thrombotic Syndrome (SOX) trial was published, casting doubt on the notion that compression stockings reduce the risk of post-thrombotic syndrome. 43 However, poor adherence to treatment in the setting of intention-to-treat analysis may account for the negative results of the study. In the most recent iteration of the ACCP guidelines in 2016, graduated compression stockings are no longer recommended for prevention of post-thrombotic syndrome. 35 Still, they can be highly effective as a treatment for patient discomfort and swelling in both the acute and chronic phases. Management of acute DVT is summarized in Figure 2.
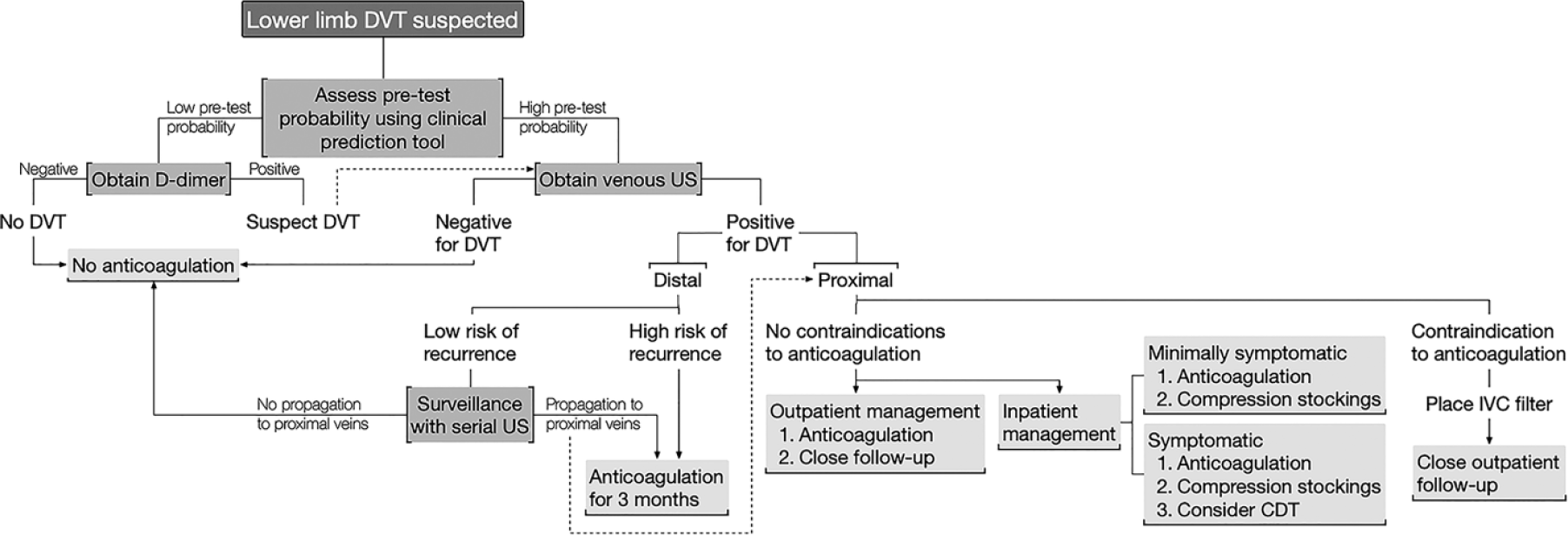
An algorithm to guide the initial evaluation of patients with suspected lower limb DVT.
Special populations
Patients with cancer are among the highest risk patients to develop VTE, representing nearly one in five of all VTE cases. 44 Certain cancer subtypes are more strongly associated with VTE risk, including gastric, pancreatic, brain, gynecologic, lung, renal, bladder, bone and hematologic malignancies. Patients with cancer-associated VTE are also at the highest risk of VTE recurrence. 45
Based largely on two randomized trials, guidelines have consistently recommended LMWH over warfarin or DOACs for the treatment of cancer-associated VTE.35,39,46,47 The recently published Hokusai VTE Cancer study compared dalteparin (a LMWH) to edoxaban for prevention of VTE recurrence in cancer-associated VTE patients. 48 Importantly, all patients received 5 days of LMWH prior to randomization, and treatment was continued for 6–12 months. In this study, patients receiving edoxaban had a similar risk of the composite primary endpoint (recurrent VTE or major bleeding) as patients receiving LMWH (12.8% vs 13.5%, HR 0.97, 95% CI, 0.70 to 1.36; p = 0.006 for non-inferiority). Notably, both major bleeding (HR 1.77, 95% CI, 1.03 to 3.04) and the composite of major or clinically relevant non-major bleeding (CRNM) (HR 1.40, 95% CI, 1.03 to 1.89) were higher in the edoxaban group. This was particularly true for patients with gastrointestinal cancer. Concurrently, a pilot study comparing rivaroxaban to LMWH in patients with cancer-associated VTE demonstrated favorable outcomes for DOAC therapy (HR 0.43, 95% CI, 0.19 to 0.99). 49 Based on the results of these two studies and other observational data, clinicians continue to gain comfort with DOAC therapy in a select, but growing number of patients with cancer-associated VTE. 50
Anticoagulant selection in patients with VTE and a known thrombophilia remains uncertain. Some data from the large phase 3 randomized trials of DOACs versus warfarin suggest that use of DOACs may be safe in many thrombophilic patients. 51 However, a recently published study of patients with ‘triple positive’ anti-phospholipid antibody syndrome demonstrated an excess of VTE events among the rivaroxaban-treated patients compared to the warfarin-treated patients. 52 Additional studies are ongoing in patients with anti-phospholipid antibody syndrome (NCT03684564 and NCT02295475).
Additionally, the efficacy and safety of DOAC use in patients with extremes of age or weight are not known; the major studies available assessing DOAC use had an average age of enrollment in the 50s and the majority of patients enrolled had a weight ranging from 60 kg to 100 kg.53–57 As noted in a recent guidance statement, caution should be used in young and elderly patients, and patients with extremes of weight or body mass index. 58
Superficial vein thrombosis is also a risk factor for future VTE. Current guidelines recommend treating these patients with a 45-day course of fondaparinux or LMWH. 35 In the recently published Rivaroxaban versus Fondaparinux in the treatment of Superficial Vein Thrombosis (SURPRISE) study, patients with symptomatic superficial-vein thrombosis were randomized to receive rivaroxaban 10 mg daily or fondaparinux 2.5 mg daily for 45 days. 59 In this study of 472 patients, the primary efficacy outcome (a composite of symptomatic VTE, progression or recurrence of superficial-vein thrombosis, and all-cause mortality) occurred in 3% of the rivaroxaban group and 2% of the fondaparinux group (HR 1.9, 95% CI, 0.6 to 6.4; p = 0.0025 for non-inferiority). There were no major bleeding events in either group.
Long-term and extended phase management of VTE
Determining how long to continue anticoagulation is one of the most critical decisions regarding DVT management. 60 This is often guided by the location of the thrombus (isolated distal DVT vs proximal DVT or PE), the presence of any provoking factors, and if this is a first or recurrent VTE. In patients with isolated distal DVT, recurrent VTE risk is low with a HR of 0.49 (95% CI, 0.34 to 0.71). 61 Therefore, patients with isolated distal DVT can often receive a limited duration of anticoagulation or no anticoagulation at all. Patients with a first VTE can be categorized as having had a strong provoking risk factor (e.g. recent surgery with general anesthesia, estrogen-containing oral contraceptive use), a weakly provoking risk factor (e.g. airplane travel) or no provoking risk factor (‘unprovoked’). If strongly provoking risk factors are present and reversible, 3–6 months of anticoagulation is recommended. Patients with ‘unprovoked’ DVT or DVT provoked by a persistent risk factor may benefit from a longer course, but this has to be weighed against the cost and risk of bleeding associated with extended courses of anticoagulation. Patients with VTE provoked by minor transient or persistent risk factors may benefit from extended courses of anticoagulation (including low-dose DOAC therapy) if their risk of VTE recurrence outweighs their risk of bleeding. 62
The 2016 ACCP guidelines recommend treatment with anticoagulation for at least 3 months over treatment for a shorter duration (Grade 1B) in patients with unprovoked DVT of the leg. 35 Recommendation is made to assess the risk–benefit ratio of extended therapy after patients have completed 3 months of anticoagulation. Mention is made to consider bleeding risk, sex of the patient, and D-dimer level.
Predicting VTE recurrence risk
Unfortunately, to date, a tool to formally assess bleeding risk in patients with history of VTE is not available. However, clinical prediction tools have been developed to help guide therapy in patients with ‘unprovoked’ DVT, based largely on recurrence risk. Perhaps the most commonly used scoring systems are the DASH score, the Vienna Prediction Model, and the ‘Men continue and HERDOO2’ score.63–65 These studies prospectively enrolled patients with a first unprovoked VTE event and assessed risk of recurrence. The ages of patients in the DASH study were slightly older. Additionally, the Vienna Prediction Model researchers obtained extended follow-up data.
To develop the DASH score, Tosetto and colleagues performed a meta-analysis that included 1818 patients with unprovoked VTE who had completed 3 months of anticoagulation. 63 Cox regression analysis was performed and four factors were found to be associated with increased risk of recurrent VTE: elevated D-dimer (after stopping anticoagulation), Age < 50 years, male Sex, and Hormone use at VTE onset. Patients with a DASH score of ⩽ 1 had an annual incidence of recurrent VTE of 3.1% (95% CI, 2.3 to 3.9), suggesting that these patients could discontinue anticoagulation. On the other hand, those with a score > 1 had a recurrence risk of > 5% annually (ranging from 6.4% to 12.3%), which would favor long-term anticoagulation therapy.
Developers of the ‘Men continue and HERDOO2’ model found that men were at high risk of recurrent VTE, with a 13.7% annual risk of recurrence in men who discontinued anticoagulation. 64 The risk of recurrent VTE in women was determined by allotting points for the following features: hyperpigmentation, edema, redness, D-dimer > 250 µg/L, obesity (BMI ⩾ 30), and older age (> 65 years). Women who had two or more of these risk factors were found to have a high risk for recurrent VTE, with an annual risk of 14.1%, in comparison to women with 0–1 risk factors who had an annual recurrence risk of 1.6%. This was recently validated externally in 44 secondary and tertiary centers in seven countries. 66
The Vienna Prediction Model was developed using a Cox proportional hazards ratio; a prognostic tool was developed that incorporated sex, site of the VTE event, and D-dimer. 65 The Vienna Prediction Model was later updated to allow prediction of recurrent VTE events at 3 weeks after anticoagulation and at 3, 9, 15, and 24 months. 67 Both of these models were externally validated.68,69
The Vienna Prediction Model and the DASH score both suggest a low risk of recurrent VTE in select male patients; however, in each of these derivation studies, men were still at increased risk of VTE recurrence compared to women.
However, despite these clinical prediction rules, a subset of patients remains in whom there is ‘clinical equipoise’ regarding maintenance of anticoagulation. More recently, there has been research regarding use of low-dose DOACs to reduce the risk of recurrent VTE. These low-dose DOACS are presumably associated with a lower risk of bleeding than full-dose DOACs. The AMPLIFY-EXT study and the EINSTEIN CHOICE trial showed evidence of a reduction of recurrent VTE in patients taking apixaban 2.5 mg twice a day and rivaroxaban 10 mg daily, once patients had completed 3–6 months of anticoagulation.62,70 For many patients at moderate risk of recurrence, this low-dose option presents a favorable balance between preventing VTE recurrence and avoiding bleeding risk associated with anticoagulant therapy. Two trials studied the role of low-dose aspirin for extended VTE prophylaxis and demonstrated benefit. 71 However, aspirin is considered to be less effective than DOAC therapy, with similar bleeding risk. Therefore, its use is largely limited to select cases with other indications for aspirin (e.g. recent coronary stenting) or for patients who cannot afford DOAC therapy but warrant extended prophylaxis. 62 Further research is needed to identify when to use low-dose versus full-dose anticoagulation in the extended treatment phase. Although the trials of low-dose extended anticoagulation were conducted only in patients for whom there was clinical equipoise about the need for extended anticoagulation, their use in patients with a clear indication for extended courses of anticoagulation remains to be fully defined; low-dose anticoagulant therapy may be considered in patients with an ongoing indication for anticoagulation who may be at higher risk for bleeding.
Summary
VTE remains a significant public health burden, despite the development of new preventative and treatment strategies. Continued efforts are needed to implement this evidence into practice so as to reduce the incidence and recurrence of VTE. The strategies presented in Table 3 are strongly recommended to decrease the high degree of morbidity and dysfunction associated with the post-thrombotic and post-PE syndromes.
Key points to remember for VTE.

CDT, catheter-directed thrombolysis; DASH, D-dimer, age < 50 years, male sex, hormone use; DVT, deep vein thrombosis; HERDOO2, hyperpigmentation, edema, redness, D-dimer, obesity, and older age; PTS, post-thrombotic syndrome; VTE, venous thromboembolism.
Footnotes
Declaration of conflicting interests
The authors have disclosed the following: Maya Serhal is supported by DAIICHI Sanyko. Geoffrey Barnes is supported by BMS/Pfizer, Janssen, Portola, Blue Cross Blue Shield of Michigan, and the National Heart, Lung, and Blood Institute (K01HL145367).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
