Abstract
Deep vein thrombosis (DVT) is a common disorder affecting approximately 900,000 new patients in the United States each year. Although the mainstay of treatment of DVT patients is therapeutic anticoagulation, some patients remain significantly symptomatic and therefore require more advanced interventions such as catheter-directed thrombolysis (CDT). We describe a case series of 13 patients with acute symptomatic inferior vena cava (IVC) and iliofemoral DVT that were treated with CDT using the Bashir Endovascular Catheter (BEC). We report the first-in-human use of BEC, which is a novel pharmacomechanical thrombolysis device. All the treated patients had complete and rapid resolution of their symptoms with excellent venous outflow. Despite initial promising results, larger studies using this catheter design will be needed to assess the role of BEC-directed therapy on rates of post-thrombotic syndrome and bleeding complications.
Keywords
Introduction
Deep vein thrombosis (DVT) is a very common disorder that affects approximately 900,000 patients each year in the United States. 1 Anticoagulation, which is the mainstay of DVT treatment, has been shown to significantly reduce the incidence of acute DVT-related complications such as pulmonary embolism (PE). 2 Nevertheless, some patients with proximal DVT (iliocaval and femoropopliteal) remain significantly symptomatic despite therapeutic anticoagulation and may thus require more advanced therapies.
Catheter-directed thrombolysis (CDT) is an endovascular technique by which a lower dose of thrombolytic agent is infused locally within the thrombus to cause thrombus dissolution. This approach frequently employs adjunctive pharmacomechanical thrombectomy 3 wherein various thrombectomy devices are used to lessen the thrombus burden. The recently completed randomized controlled ATTRACT trial has shown a greater improvement in leg pain and swelling among patients with iliofemoral DVT who underwent CDT treatment compared to those treated with anticoagulation therapy alone. 4 Patients with proximal DVT appear to have an improvement in their quality of life at 1 and 6 months after CDT 5 ; this is especially true if CDT is successful in achieving more than 90% venous patency. 6 EKOS (Boston Scientific; Marlborough, MA, USA), which was the most commonly used CDT device in recent clinical trials,7,8 is a single-lumen infusion catheter which utilizes ultrasound energy to augment thrombolysis. 9 Although low-frequency ultrasound has been shown to be effective in accelerating thrombolysis in animal models, 10 subsequent analyses have not found any significant differences between ultrasound-assisted and unassisted tissue plasminogen activator (tPA) infusion among patients with iliofemoral DVT. 11
The Bashir Endovascular Catheter (BEC) (Thrombolex, Inc., New Britain, PA, USA), which has been cleared by the United States (US) Food & Drug Administration (FDA) for infusion of physician-prescribed fluids (including thrombolytics) into the peripheral vasculature, 12 is a novel device that was developed to improve the efficacy of CDT by distributing the thrombolytic agent over a wide cross-sectional area. The BEC contains an expandable infusion basket comprised of six separate mini-infusion catheters, each having eight laser-drilled infusion holes (Figure 1). Once the infusion basket of the BEC is within the thrombus, it is expanded in a spiral fashion prior to initiating tPA infusion. The intent of the mechanical thrombus maceration using the BEC basket is to increase the surface area over which tPA can interact with thrombus. In this report, we present the first-in-human experience of using the BEC for treatment of severe symptomatic inferior vena caval and iliofemoral DVT.
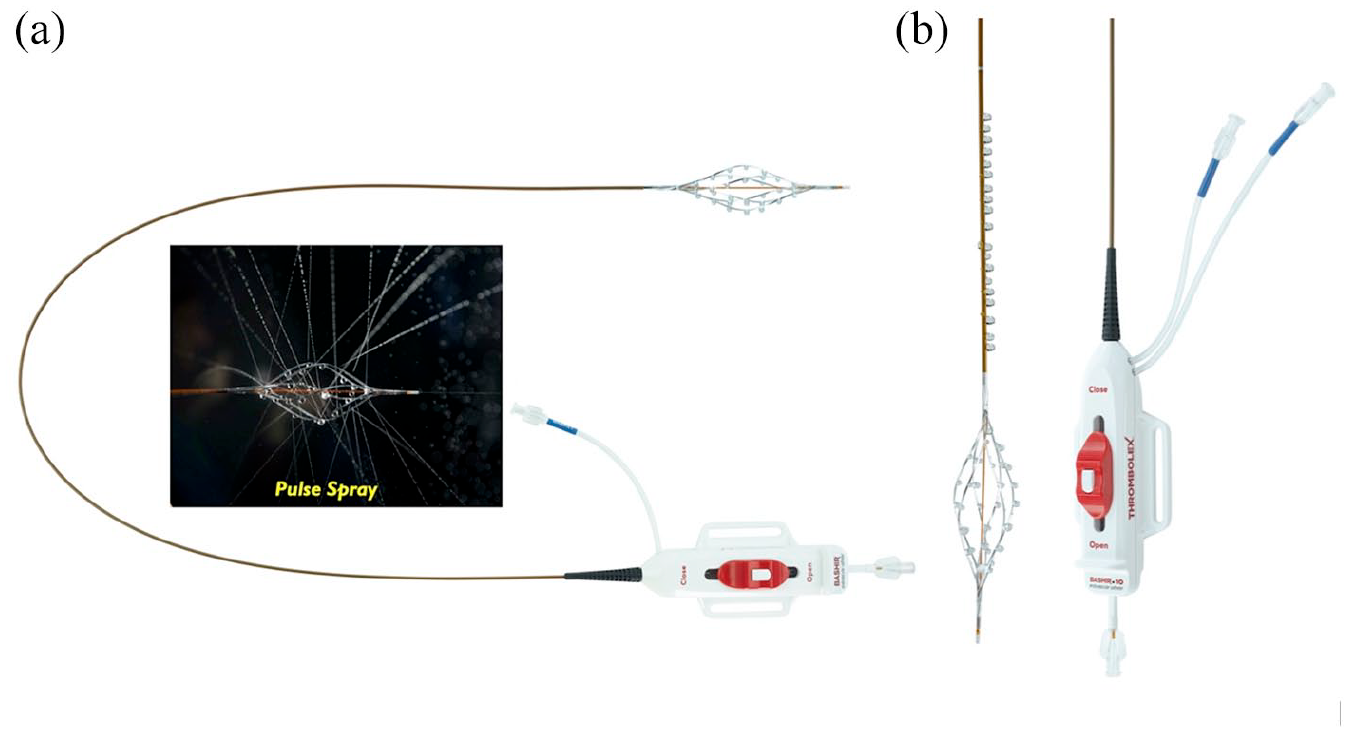
Methods
The proposal for this project underwent a thorough internal committee review by the institutional review board and was approved. Informed consent was obtained for all patients as part of the procedure only. The consent form included a statement to inform patients that the device was developed by a physician at our institution. This was explained to the patient prior to using the device. Given the retrospective nature of this case series, no additional consents were obtained other than the procedural consent.
We included 13 consecutive patients with a diagnosis of iliocaval and/or iliofemoral DVT who underwent treatment with BEC in our institution. Patients were included if they were > 18 years old, had a diagnosis of iliocaval and/or iliofemoral DVT with symptom onset of < 2 weeks’ duration. Patients were considered for treatment with BEC if they had significant persistence of symptoms (such as back pain, thigh/hip/calf pain, marked swelling, heaviness) despite a trial of therapeutic anticoagulation. None of the patients had evidence of phlegmasia.
Ultrasound-guided venous access (popliteal or common femoral vein) was obtained using a 7 cm 21-gauge micropuncture needle. After placement of an 8 Fr sheath, venography was performed to define the extent of thrombus burden. A soft-angled Glidewire supported by a 4 Fr Glidecath (Terumo, Somerset, NJ, USA) was then used to cross the thrombotic segment in all patients. Manual aspiration thrombectomy using an 8 Fr MPA guiding catheter and a 60 cc syringe was performed in select patients with extensive thrombus burden. A 0.018” Steelcore wire was then placed across the thrombotic segment and BEC (7 Fr device) was delivered to the most proximal part of the thrombosed vein. After expanding the BEC basket, a bolus of tPA (1 mg in 25 mL saline) was administered. Sequential openings of the basket and administration of tPA boluses were then performed across the entire length of the thrombosed segment in the cranial-to-caudal fashion. Finally, the BEC was re-advanced proximally, the basket was expanded, and tPA infusion initiated at 0.005 mg/kg/hr up to a maximum of 0.5 mg/hr. Additional tPA at a rate of 0.005 mg/kg/hr up to a maximum of 0.5 mg/hr was administered via the popliteal or common femoral vein sheath; therapeutic anticoagulation with unfractionated heparin was used in all patients to maintain a partial thromboplastin time (PTT) of around 50 msec.
All patients were observed overnight in the cardiac intensive care unit (CICU), after which they returned to the cath lab for repeat venography. In cases of residual thrombus, adjunctive rheolytic thrombectomy was performed using an 8 Fr Zelante AngioJet (Boston Scientific). Peripheral IVUS assessment was then performed and balloon angioplasty ± stenting performed if necessary.
Results
A total of 13 patients underwent pharmacomechanical thrombolysis using the BEC. Table 1 shows the procedural characteristics of the treated patients. The mean age of the patient population was 47.2 years. Seven patients had iliofemoral and six patients had inferior vena cava (IVC) filter-associated iliocaval DVT. The indication for intervention in all patients was persistence of severe symptoms despite therapeutic anticoagulation.
Procedural characteristics of 13 patients with iliocaval and/or iliofemoral deep vein thrombosis treated with the Bashir Endovascular Catheter.
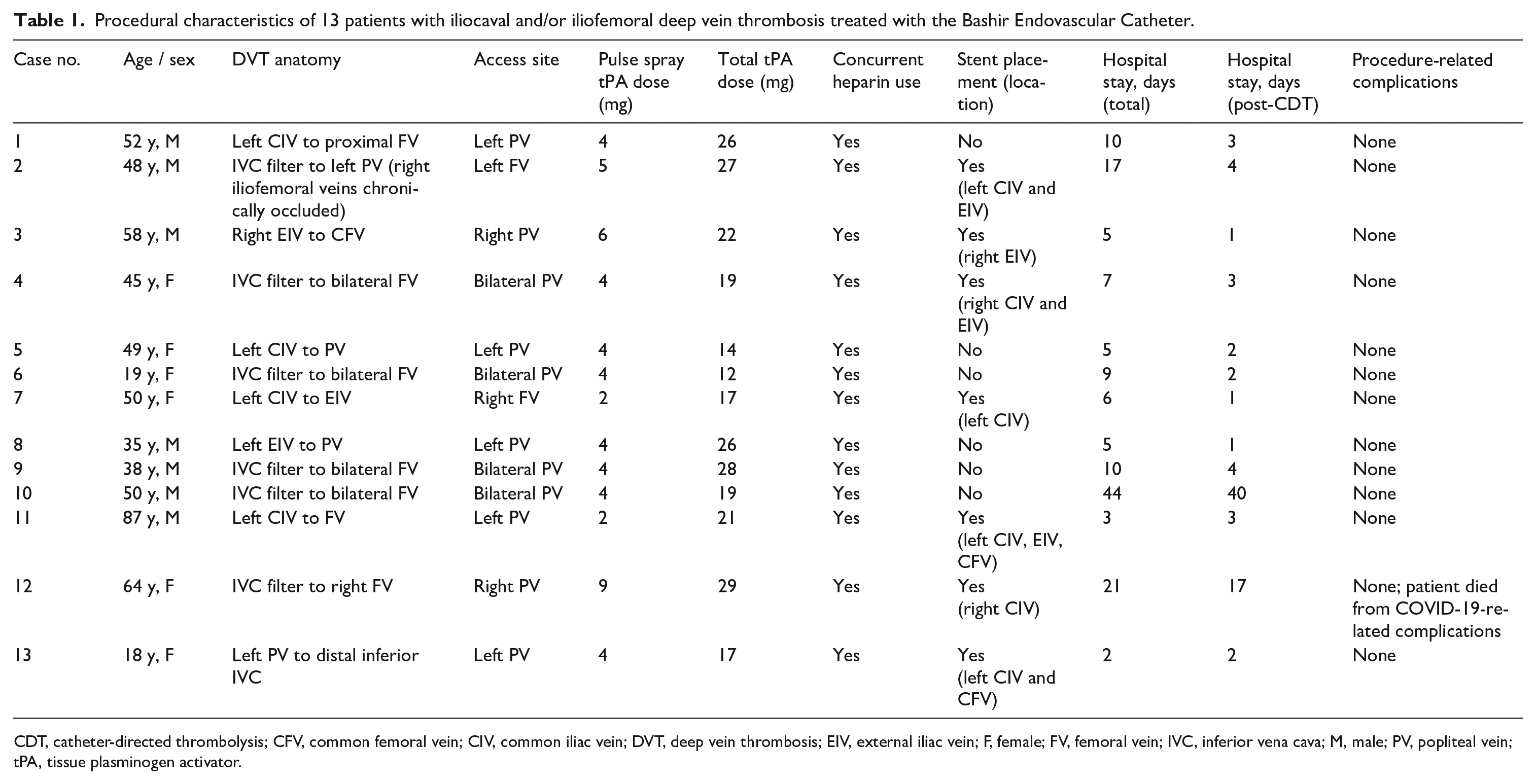
CDT, catheter-directed thrombolysis; CFV, common femoral vein; CIV, common iliac vein; DVT, deep vein thrombosis; EIV, external iliac vein; F, female; FV, femoral vein; IVC, inferior vena cava; M, male; PV, popliteal vein; tPA, tissue plasminogen activator.
The popliteal vein was accessed in 11 patients and the common femoral vein in two patients. Device placement was successful in 100% of all patients. The average pulse spray dose of tPA was 4.3 mg with a mean total tPA dose of 21.3 mg. All patients received a concomitant infusion of unfractionated heparin during thrombolysis and were monitored in the CICU for the duration of tPA infusion. There were no significant device-associated complications, procedure-related complications, or significant bleeding events in any of the patients.
Successful thrombolysis was achieved in all patients. Figures 2 and 3 show representative images of two treated patients: the first patient with iliofemoral DVT and the second patient with IVC filter-associated iliocaval DVT. Seven patients required iliac vein stenting, five of whom had evidence of May–Thurner anatomy. There was significant clinical improvement after completion of the procedure in all cases. The average total length of hospital stay was 11 days (with a range of 2–44 days); average stay after completion of thrombolysis was 6.3 days.
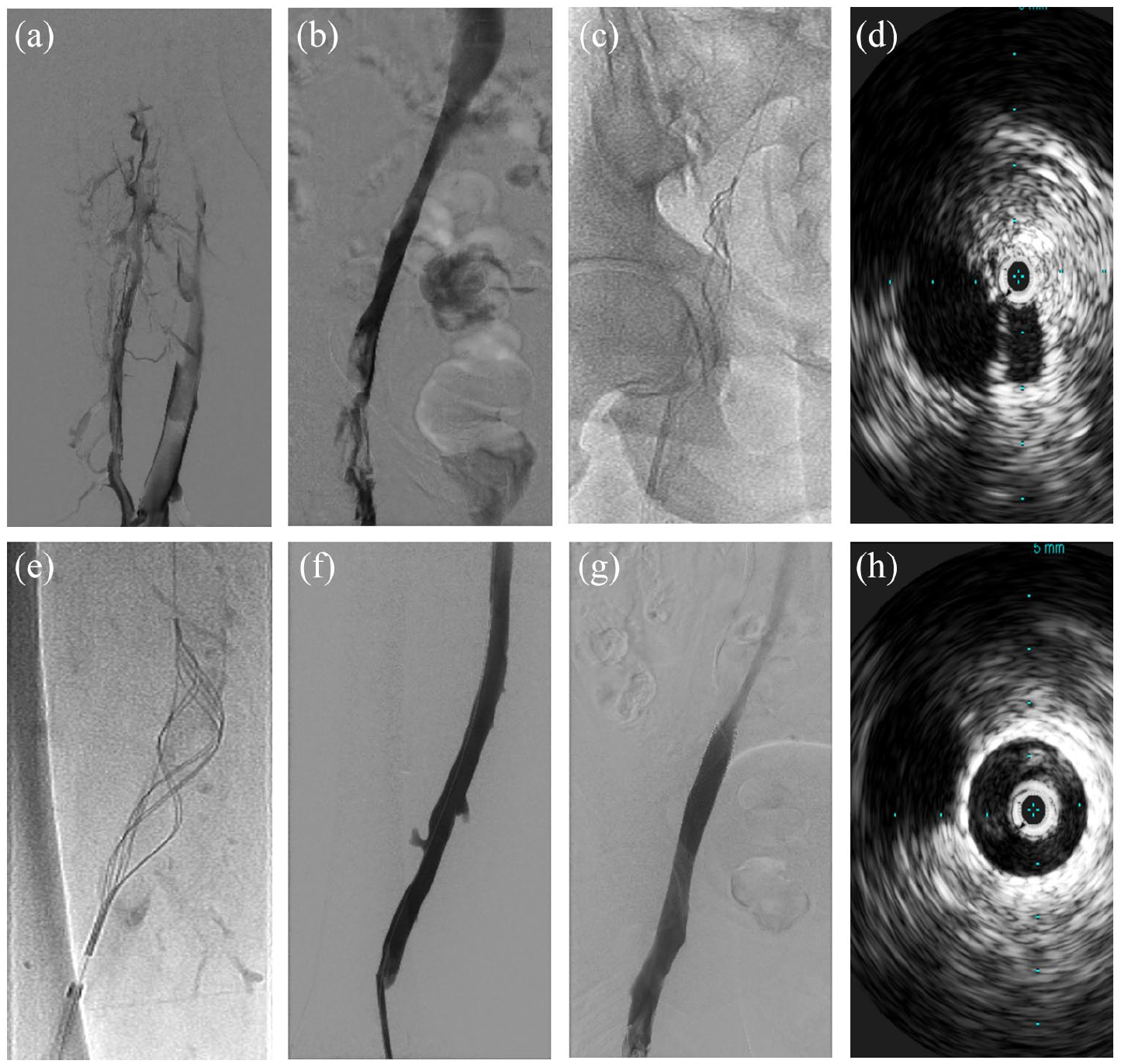
Case 2: Acute symptomatic iliofemoral thrombosis. (
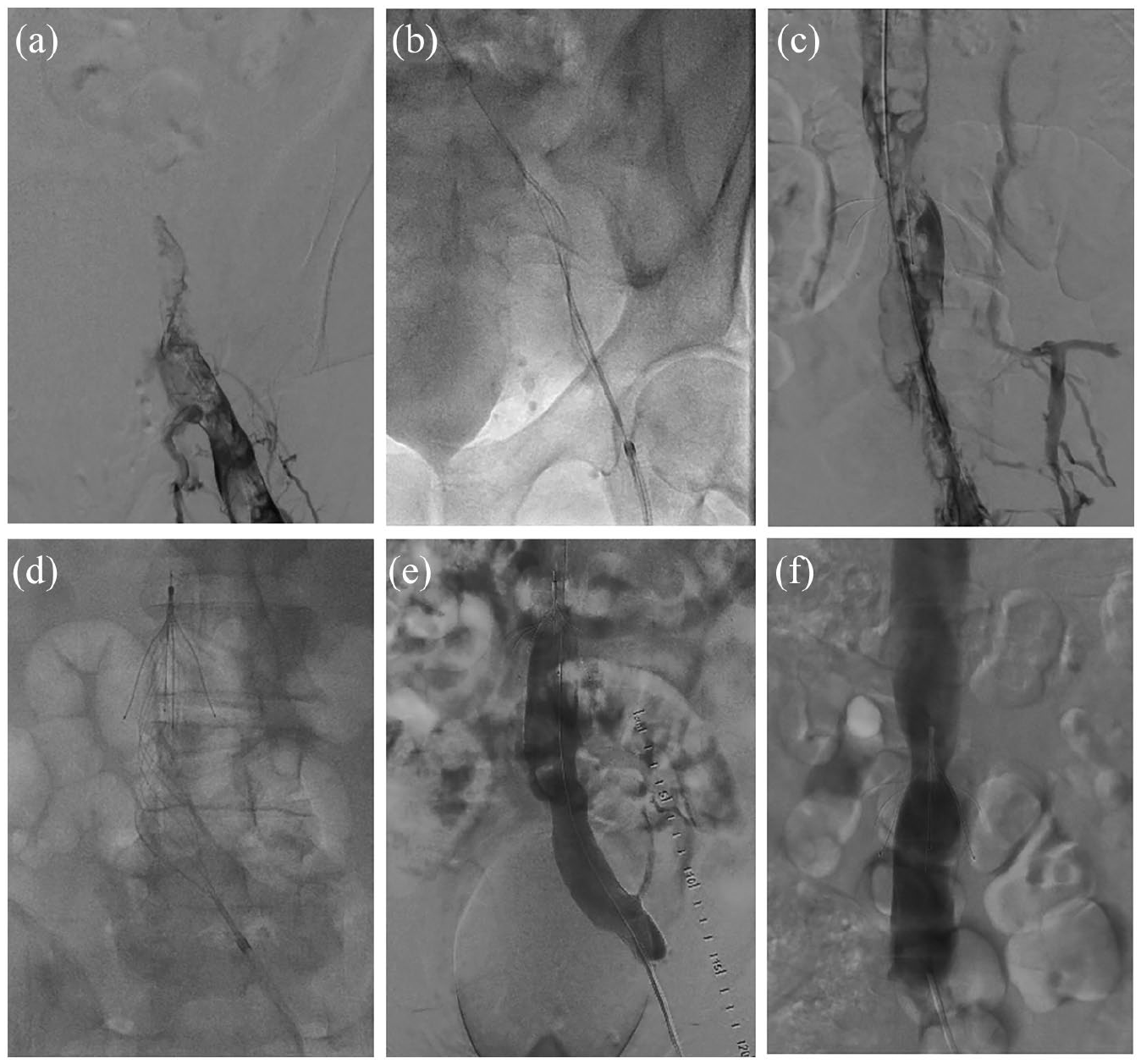
Case 3: Iliocaval DVT. (
Posthospitalization follow-up data were available in nine of the 13 treated patients. Three patients were lost to follow-up and one patient died during index hospitalization due to COVID-19-related complications; mortality was determined to be unrelated to CDT. Follow-up appointments, which were scheduled for all patients prior to hospital discharge, were timed to approximately 2–4 weeks postdischarge.
Of the nine patients who had outpatient follow-up, three patients reported significant improvement in their quality of life and had minimal residual post-thrombotic symptoms; four patients reported no significant residual post-thrombotic symptoms. Two patients (both of whom underwent their index CDT for IVC filter-associated iliocaval thrombosis) underwent reintervention for re-thrombosis. The cause for re-thrombosis in both patients was determined to be noncompliance with their respective outpatient anticoagulation regimens.
Discussion
In the present case series, we evaluated the efficacy and safety of CDT using the BEC (a novel device for pharmacomechanical thrombolysis) among 13 consecutive patients presenting with acute inferior vena caval and iliofemoral DVT. All the patients had successful CDT with prompt resolution of symptoms and without any significant bleeding complications. Despite the average total length of hospital stay being 11 days, the average time from completion of CDT to hospital discharge was 6.3 days among our patients.
Unlike the currently available devices, which are comprised of a single infusion lumen, the BEC contains an ‘infusion basket’ which is composed of six interconnected infusion catheters (each with eight laser-drilled holes for infusion of physician-prescribed fluids). Upon expansion of the basket within a thrombotic segment, the spiral-shaped basket creates multiple channels within the clot into which tPA can be introduced first as a bolus followed by a continuous infusion. The theoretical advantage of creating more than one channel inside a thrombus is the potential to restore native blood flow more rapidly in order to augment endogenous fibrinolysis, as well as create a larger surface area within the thrombus for exogenous tPA activity.13,14 When compared to traditional infusion catheters, these features may result in faster and more complete reduction of thrombus burden.
In our series, all patients received a pulse spray dose of tPA prior to initiation of continuous infusion. This is different from our approach when performing thrombolysis using a single-lumen infusion catheter, in which case tPA bolus is not administered. The idea behind this maneuver is to initiate thrombolysis as early as possible in the treatment course instead of waiting until after performing thrombectomy. Furthermore, administration of tPA bolus through the expanded BEC basket allows the drug to be distributed over a wider surface area within the thrombus. Despite the administration of a pulse spray dose, the total average tPA dose at the completion of treatment was 21.3 mg. This is similar to the average total tPA dose used in recent studies where the total dose ranged between 20 mg and 21 mg.7,11
Although the role of CDT for management of caval and iliofemoral DVT is still being defined, it is clear that some patients remain symptomatic despite therapeutic anticoagulation and thus require advanced therapeutic interventions. Importantly, the benefit of these interventions needs to be considered in the context of the increase in bleeding complications seen in patients treated with CDT. Although we did not observe any significant bleeding events in our series, the rate of major bleeding complications in the CDT arm of the recently published ATTRACT trial was 1.6%. 7 Therefore, significant attention needs to be paid to the patient’s clinical status in addition to close monitoring of coagulation parameters (such as complete blood count, fibrinogen, PTT) throughout the duration of thrombolytic infusion.
Study limitations
Our study has several limitations. Given a small overall sample size and no available comparator group, the comparative effect of BEC use on the total tPA dose used, length of hospital stay, risk of bleeding complications, or long-term risk of developing post-thrombotic syndrome could not be assessed. Although the unique catheter and basket design have the theoretical potential of improving the safety and efficacy of CDT, larger studies will be needed to assess the true impact of this technology. Long-term follow-up will also be required to determine the impact of BEC-guided CDT on the risk of post-thrombotic syndrome among patients with caval and proximal lower extremity DVT.
Conclusion
In our case series, CDT using the Bashir Endovascular Catheter for treatment of patients with caval and iliofemoral DVT resulted in significant acute symptom improvement and was successful in reducing the overall thrombus burden. The device also appears to be safe in terms of bleeding complications and risk of distal thrombus embolization. Larger studies using this new catheter design are needed to assess the role of BEC-directed CDT in reducing the rates of long-term complications such as post-thrombotic syndrome.
Footnotes
Declaration of conflicting interests
Dr Bashir is the inventor of the Bashir Endovascular Catheter and has equity interest in Thrombolex, Inc. No financial support was received from Thrombolex. The remaining authors have no conflicts of interest to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
