Abstract

Keywords
The calf muscle pump is a robust venous pump that is a major contributor to blood return from the lower extremities. Recent studies have found reduced calf pump function (CPF) to be a predictor of venous thromboembolism (VTE) and mortality.1,2 Measurement of mortality and VTE risk in prior studies used previously established clinical cut-off values to classify normal versus reduced CPF. In this study, we sought to re-evaluate quantitative plethysmography measurements (passive outflow, passive refill, and postexercise refill) to determine if these previously established thresholds of CPF could be refined when it relates to prediction of deep vein thrombosis (DVT).
To accomplish this, we used long-term outcome data from the Rochester Epidemiology Project and examined Olmsted County residents who underwent clinical venous plethysmography testing between 1998 and 2015. Patients were excluded if they had a history of VTE or if there was evidence of venous outflow obstruction on the index plethysmography study; see previously published methods. 1 CPF measurements were analyzed as refill volumes as a percent of leg passive drain refill (PDR) volume (standard) or leg outflow volume, or using the crude volume refilled after exercise (toe-lifts), regardless of testing methodology (strain gauge or air). In addition to plethysmography data, venous continuous wave Doppler assessment was performed/supervised by Registered Vascular Technologists at each plethysmography study and the presence of and location of deep venous insufficiency was analyzed in conjunction with data on overall venous hemodynamic insufficiency. The venous plethysmography parameters of each examined leg were evaluated with the outcome of interest being imaging- or autopsy-confirmed ipsilateral DVT (proximal or distal) after the index study using Cox proportional hazard models. The Charlson Comorbidity Index (includes: myocardial infarction, congestive heart failure, peripheral vascular disease, stroke or transient ischemic attack, dementia, chronic obstructive pulmonary disease, connective tissue diseases, peptic ulcer disease, liver diseases, renal diseases, diabetes mellitus, hemiplegia, chronic kidney disease, cancers, and HIV/AIDS) as well as age, body mass index, and sex were evaluated initially in univariates analyses. The research was reviewed and approved by Institutional Review Boards from Olmsted Medical Center and Mayo Clinic in Rochester, MN.
A total of 3064 legs (1532 patients) were analyzed with a median follow-up of 11.7 years. The median age of the cohort was 63.9 years (SD 18.4) and the majority were female (69%). CPF was reduced by previously established criteria bilaterally in 38%, unilaterally in 23%, and was normal bilaterally in 39%. Venous insufficiency by plethysmography was present (mild, moderate, or severe) in 52.6% of right legs and 55% of left legs (p = 0.11). The primary outcome of ipsilateral leg DVT occurred in 1.8% of right legs and 2.8% of left legs (p = 0.053).
We determined the optimal predictive cut-offs for CPF using continuous variables for each calculation method. Each method for calculation of CPF was highly associated with development of ipsilateral leg DVT. However, the parameter that was most predictive was the standard calculation of CPF using passive refill volumes in comparison to exercise refill volumes (calf ejection fraction). The optimal predictive cut-off for ipsilateral DVT was determined to be an ejection fraction of ± 23.1%, which on univariate analysis demonstrated a hazard ratio (HR) of 2.57 (95% CI 1.61–4.13). Kaplan–Meier curves for incident DVT over the study follow up demonstrate separation of the curves between the two groups after 3–4 years (online Supplementary Figure). Increasing age (HR 1.03, 95% CI 1.02–1.05), increasing body mass index (HR 1.22, 95% CI 1.14–1.31), presence of proximal deep vein incompetence by continuous wave Doppler assessment (HR 1.67, 95% CI 1.02–2.73), and presence of severe hemodynamic incompetence by venous plethysmography (HR 2.98, 95% CI 1.50–5.93) were all statistically significant predictors for ipsilateral DVT. The Charlson Comorbidity Index was not an independent predictor in this analysis (age removed from calculated value) (p = 0.10) nor was the patient’s sex (p = 0.10) or increased pulsatility as assessed by continuous wave Doppler (p = 0.55). A multivariable Cox proportional hazard model was performed using CPF ± 23.1% including all covariates with p < 0.1 in univariate analysis. In this analysis, only reduced CPF < 23.1% (HR 1.78, 95% CI 1.61–4.13) and proximal deep vein incompetence (HR 1.73, 95% CI 1.07–2.79) were independent predictors for ipsilateral leg DVT (Table 1). Although severe overall hemodynamic insufficiency was a strong predictor in univariate analysis, it was not an independent predictor within the model. Both age and body mass index also were not statistically significant in the multivariable model.
Multivariable model results for first ipsilateral deep vein thrombosis per leg.
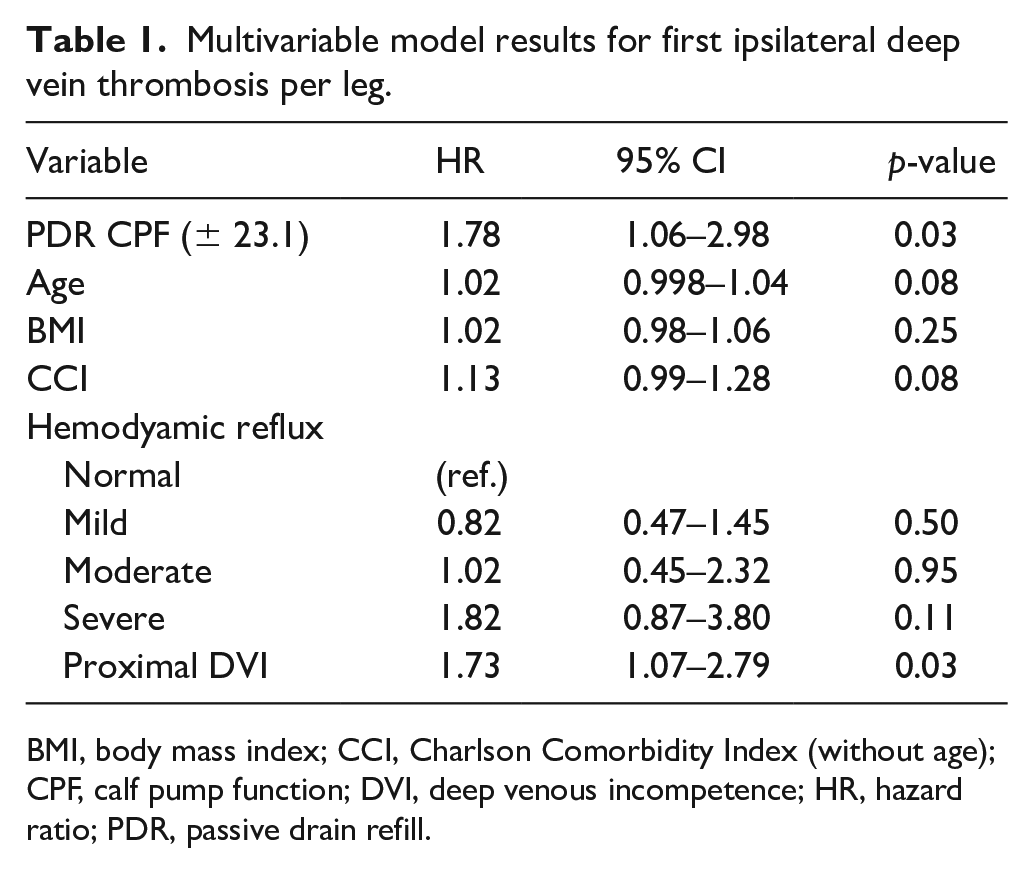
BMI, body mass index; CCI, Charlson Comorbidity Index (without age); CPF, calf pump function; DVI, deep venous incompetence; HR, hazard ratio; PDR, passive drain refill.
These results demonstrate that the previously established clinical cut-offs for CPF on standard plethysmography studies (ejection fraction of 45%) can be refined for better prediction of ipsilateral DVT with the most optimal calculation method for calf pump ejection fracture being confirmed on this analysis: [postexercise refill volume ÷ PDR volume]. In the multivariable models, physiologic testing of venous efficiency from the lower extremities using CPF and proximal deep vein incompetence by continuous wave Doppler assessment are the only statistically significant parameters for DVT prediction. The findings suggest that well-established epidemiologic VTE risk factors such as age and body mass index might be surrogate markers for abnormalities in venous physiology due to their association with CPF and deep vein incompetence.
The population studied in this analysis, on the account of referral for testing, reflect a population that is enriched for reduced CPF and other venous diseases compared to the general population. Therefore, additional prospective studies are needed to understand if the risk estimates observed in this study are similar in other populations. Unlike our previous analyses of VTE risk, we did not include pulmonary embolism as an outcome in this study due to the per leg analysis and inability to attribute the venous thromboembolic event to one leg or the other. We do not believe that this diminishes the significance of these findings, but rather highlights ongoing need for further research to better understand what contributes to embolization of lower-extremity DVT. It is not known whether CPF may fluctuate or change over time, and what factors may contribute to this, and it is not known if subsequent changes may increase or decrease DVT risk. It has been shown that CPF can be improved with specific exercises, bringing up the possibility that this may be a modifiable risk factor. 3 This study suggests that venous physiologic measurements may better predict or significantly refine current clinical VTE risk prediction tools and should be studied in a prospective manner to determine their clinically utility.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X221124996 – Supplemental material for Reduced calf pump function and proximal deep vein incompetence are predictors for ipsilateral deep vein thrombosis
Supplemental material, sj-docx-1-vmj-10.1177_1358863X221124996 for Reduced calf pump function and proximal deep vein incompetence are predictors for ipsilateral deep vein thrombosis by Damon E Houghton, Aneel Ashrani, David Liedl, Ramila A Mehta, David O Hodge, Thom Rooke, Paul Wennberg, Waldemar Wysokinski and Robert McBane in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This publication was made possible through the support of the Eduardo G. Mestre and Gillian M. Shepherd, M.D., Clinician Career Development Award Honoring John T. Shepherd, MD, and the Rochester Epidemiology Project, which is supported by the National Institute on Aging of the National Institutes of Health (Award no. R01AG034676). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
