Abstract
Keywords
Background
Postthrombotic syndrome (PTS), a consequence of untreated venous hypertension, is characterized by leg swelling, pain, heaviness, and skin ulceration. This syndrome ultimately affects 40–60% of deep vein thrombosis (DVT) patients: 7–16% of whom develop severe PTS, with the highest incidence in patients with iliofemoral obstruction.1 –3 Relieving venous outflow obstruction has been shown to decrease the incidence of severe PTS in patients with acute iliofemoral DVT.2,4,5 Over time, the acute thrombus matures into an adherent, collagen-rich structure, occasionally causing persistent outflow obstruction. 6 No medical therapies exist for these chronic luminal changes. Recanalization and stenting of chronic DVTs is effective at relieving symptoms of PTS by relieving venous hypertension. 7 The technical success of such procedures is significantly limited due to (a) an inability to cross chronic thrombi to allow for recanalization and stenting, and (b) poor stent patency rates when compared with nonthrombotic venous stenting. These suboptimal results are thought to be driven in part by a lack of specific, suitable endovascular tools. 8
To date, all thrombectomy and thrombus crossing devices have been designed for acute/subacute thrombi. 9 Owing to the complex mechanical interactions between endovascular devices and organizing thrombus, devices effective in the setting of acute thrombus do not translate for application to chronic thrombi. Depending on the mechanism of action of the device, the thrombus may experience tensile, compressive, and shear forces in a time-dependent manner. Optimizing critical design features that govern these interactions requires a fundamental understanding of the mechanical properties of chronic DVT. Owing to the complex biologic response involved in thrombus organization and maturation, 10 in vitro models of thrombus are limited by the inability to adequately reproduce DVT composition and adherence to the vein wall.
The vital need for a reliable, endovascular, unilateral, subacute, and chronic DVT preclinical model was recently highlighted by the American Heart Association’s venous thromboembolism priorities 11 and the Society of Interventional Radiology Foundation’s Research Consensus Panel on Deep Venous Disease.12,13 Herein we characterize the mechanical and histopathologic properties of subacute and chronic, proximal DVT in a novel pig model utilizing components of stasis, endothelial injury, and hypercoagulability to generate unilateral long-segment iliac thrombosis, generating histologic and imaging features consistent with chronic thrombus. We describe, for the first time in a large animal model, variations in mechanical properties of subacute compared to chronic thrombus.
Methods
Study design
Time points for acute (2 hours), subacute (7 days), and chronic (14 days) DVT were determined a priori based on previous literature in porcine DVT models. 13 Previous studies demonstrate that thrombolytic efficacy drops from 86% at 3 days to 11% at 14 days post-DVT with advanced signs of thrombus organization and abundant collagen deposition 12–15 days post-DVT.14,15 Intravascular ultrasound (IVUS; Philips Vision 0.035 catheter), duplex, and venography were performed at the time of thrombosis. In accordance with our standard quality protocols, a midpoint duplex ultrasound was performed 4 days postthrombosis to identify early recanalization or dissolution of thrombi.
Animals
This study was approved by the University of Michigan Institutional Animal Care and Use Committee. Female farm swine weighing 60–80 kg underwent induction and maintenance general anesthesia with 1.5–3.0% isoflurane and 100% oxygen. All operations were performed under sterile conditions. Swine were placed supine on an endovascular surgery table and the following anatomic landmarks identified with duplex ultrasound: bilateral common femoral veins, iliac veins, inferior vena cava (IVC) confluence, and right external jugular (EJ). In swine, the EJ is larger than the internal jugular vein (IJ). The right EJ and common femoral vein (CFV) were cannulated with a 6F and 8F vascular sheath, respectively (Figure 1A). The sheaths were positioned in bilateral iliac veins and a diagnostic venogram was performed to identify the IVC confluence. IVUS confirmed the origin of the hypogastric vein and identified any large iliac tributaries. A vascular plug (9-MVP; Medtronic, USA) was used to occlude the origin of the ipsilateral hypogastric and a high-riding profunda femoris vein if present (Figure 1B). A 12-mm Wallstent (Boston Scientific, Marlborough, MA, USA) was partially deployed on the sterile field, frayed using a 19-gauge needle to unravel the ends of the stent for 15 mm, and recaptured into the sheath on the back table. The stent was placed into the iliac vein and partially deployed (Figure 1C). It was passed central to caudal four times to excoriate the endothelium, then resheathed and withdrawn. The iliac segment was isolated with 11.5-mm diameter Berenstein occlusion balloons (Boston Scientific). Thrombin (100 units/mL, 400 units total; Sigma-Aldrich, St Louis, MO, USA) was delivered in divided doses into the isolated segment via the balloon catheters (Figure 1D). A duplex ultrasound was performed at 1 hour and 2 hours. The balloons were deflated and vascular sheaths withdrawn. The animal was awoken from anesthesia and allowed to ambulate freely. DVT duration was: 0 days (n = 1), 7 days (n = 3), and 14 days (n = 3). The single day-0 animal was performed for proof of concept of the model. The 7-day and 14-day animals represented proposed subacute and chronic thrombi, respectively. Duplex ultrasound scans were performed on day 0, day 4, and day of harvest. Additionally, on the day of harvest, a venogram was performed. Technical success was defined as persistent partially or totally occlusive iliac thrombus at the prespecified time points. At the conclusion of the study, animals underwent a laparotomy and sternotomy. The pulmonary arteries were examined for pulmonary embolism. The iliac veins and IVC were isolated and immediately processed for mechanical testing or histopathology.
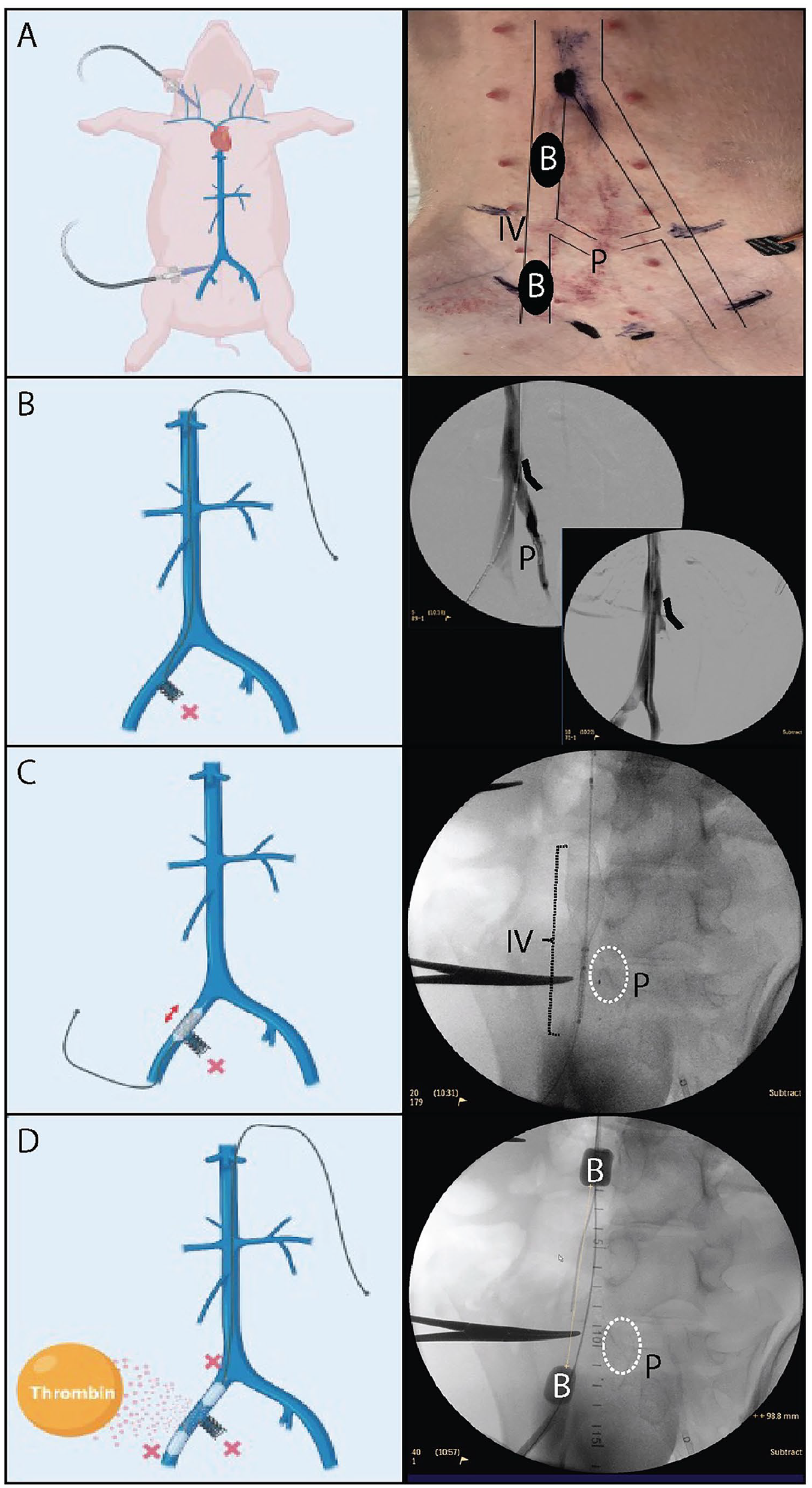
Totally endovascular unilateral iliac vein thrombosis model.
Human thrombus samples
The use of human samples was approved by the Institutional Review Board (IRB) under IRB HUM00107111. Consent was waived by the IRB, as the samples were part of routine clinical care. Human iliofemoral DVT samples were obtained from samples biopsied during open or endovenous treatment. They were fixed and processed in a similar fashion to the porcine samples, as described below.
Histopathology
En bloc iliac vein and thrombus samples were formalin fixed for 48 hours. The distal half of each thrombus was used for histologic evaluation. The specimens were paraffin-embedded, sectioned, and stained with standard hematoxylin and eosin (H&E), Masson’s trichrome, and Martius Scarlet Blue (MSB). Slides were analyzed by a cardiovascular pathologist blinded to the animal treatment and duration of thrombosis. The following nomenclature was used for characterization: fresh thrombus – intact blood elements without signs of degeneration, consistent with 0–2 days and inclusive of periprocedural thrombus; organizing thrombus – fibrin material with cellular infiltrate inclusive of leukocytes and myointimal cells; myointimal cell ingrowth – fibroblast/smooth muscle cells and collagen replacing the thrombus material in the lumen; hemosiderin deposition – presence of iron pigmentation within macrophages or extracellular matrix; diffuse intimal thickening – numerous myointimal cells with surrounding collagen and extracellular matrix material (e.g., proteoglycan accumulation) expanding the intima; may include leukocytes and/or form continuum with organizing thrombus. The vein wall and thrombus characteristics were summarized using a semiquantitative scoring system as follows: (–) denotes feature is not present, (+) denotes minor histologic feature, (++) denotes histologic feature present in majority of samples, and (+++) denotes histologic feature present throughout all samples. Thrombi were characterized and aged according to standard pathologic criteria applied to human hematoma and thrombus analysis by a cardiovascular pathologist.16 –21 Thrombus composition, including collagen and erythrocytes, was quantified using Image J software.
Mechanical testing
Thrombus stiffness, representing the resistance of the sample to deform under an applied load, was evaluated using a TA Instruments RSA-G2 solids analyzer (TA Instruments, New Castle, DE, USA) with a force resolution of 10 µN. Samples of harvested thrombi were stored in phosphate buffered saline (PBS) at 4°C prior to mechanical testing, which occurred immediately after necropsy. The thrombus was freed from the vein wall and sectioned to a length of approximately 5 mm. The sample was subjected to uniaxial compression loading for 20 seconds of 0.025 N preload force, followed by unconfined compression with a displacement rate of 0.025 mm/s. The test was terminated when the force exceeded 15 N.
22
Deformation of the sample was portrayed in an engineering stress–strain curve. Engineering stress was defined as force divided by the initial cross-sectional area and the engineering strain was defined as the current sample length minus the initial sample length divided by the initial sample length. The onset point was defined as the intersection of the lines tangential to the initial and final point on the stress–strain curve to quantify the differences in the inflection point of the curves between the two timepoints. Tangent lines were calculated using equation:
where
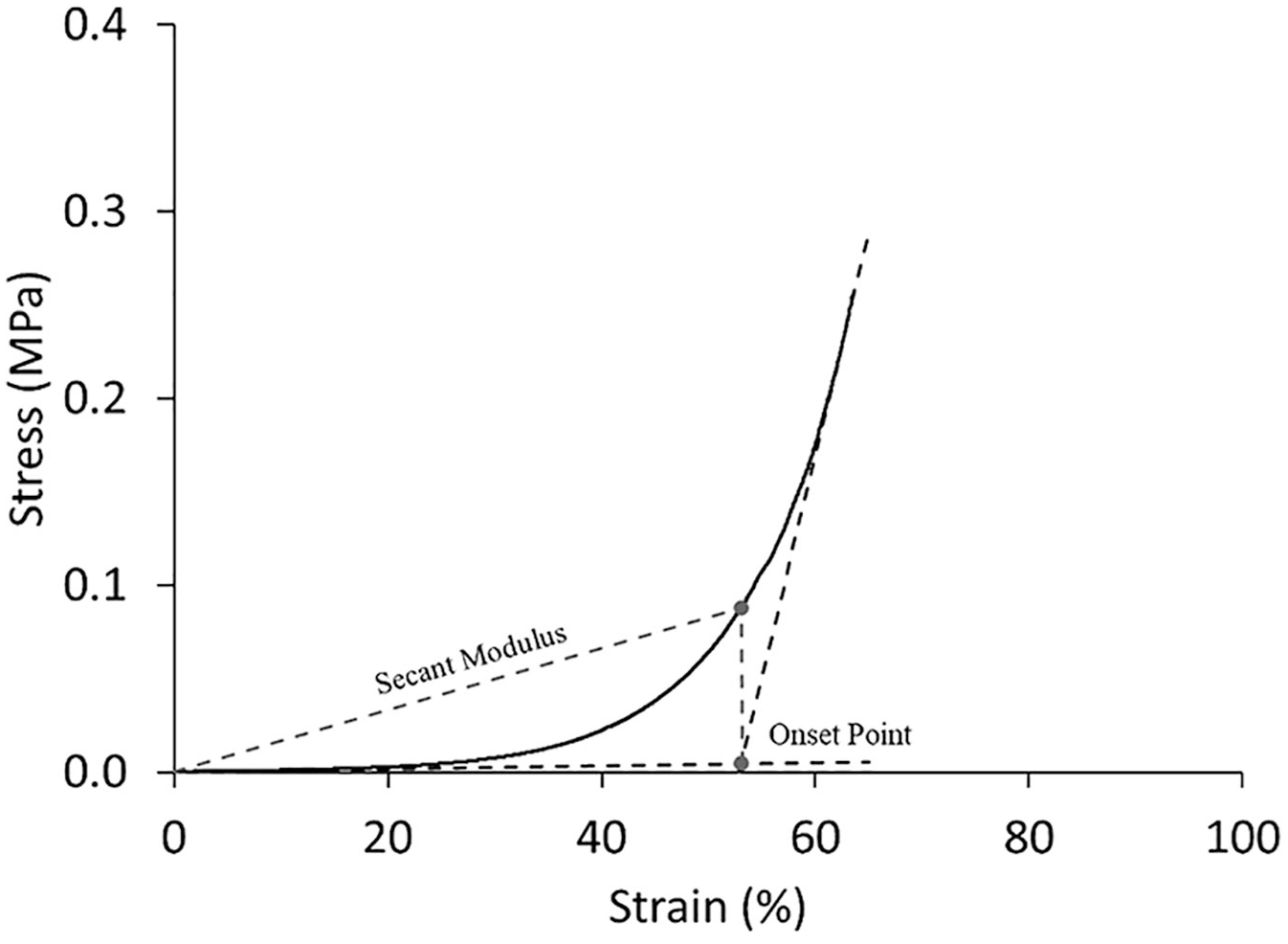
Graphic representation of the onset point and secant modulus. The onset point was defined as the intersection of the lines tangential to the initial and final point on the stress strain curve to elucidate differences in the inflection points of the curves between the two timepoints. The thrombi’s deformation resistance was quantified with secant modulus at the onset point.
Statistical analysis
Continuous variables were evaluated with a Student’s t-test, where appropriate. Outliers were determined using Grubbs’ method at α = 0.1. Statistical analysis was performed in GraphPad Prism 10.0.2 and mechanical testing data were analyzed in Hyperfit, Minitab, Matlab R2019b, Microsoft Excel, and TA Instruments TRIOS V4.4.1.41651.
Results
Model
Among severn female farm swine undergoing this novel, totally endovascular unilateral DVT model, technical success was 100% in DVT creation as confirmed via duplex ultrasound. No animals suffered main branch pulmonary embolism. The average animal weight was 72 ± 6.7 kg. Perimeter-derived external iliac vein areas measured by IVUS were 68.5 ± 10.7 mm2. The average length of iliac thrombus generated at the index procedure was 7.1 ± 1.4 cm. A total of three animals needed adjunct procedures to achieve complete venous occlusion during the index procedure, including: two requiring endovascular plugs to occlude high-riding profunda femoris veins and one requiring additional thrombin and balloon occlusion time due to nonocclusive thrombi at 2 hours.
Venographic, duplex, and gross histology characteristics demonstrated changes consistent with human DVT evolution over 14 days. At day 0, there was no evidence of venous collaterals, whereas hypertrophy of multiple pathways was evident by day 7, and robust collateralization noted by day 14 (Figure 3). Acute thrombi were consistently identified by large, noncompressible veins on duplex ultrasound. By day 14, the thrombosed veins met human diagnostic criteria for chronic DVT, including contracted and hyperechoic appearance. 24 Only day-0 and day-7 thrombi could be easily separated from the adjacent vein wall. By day 14, the thrombi had formed thick, collagen-rich material quite adherent at the vessel interface (Figure 3). One animal was maintained 21 days postthrombosis. This animal demonstrated near-complete recanalization of the thrombosed iliac vein via duplex ultrasound, venography, and IVUS (Supplemental Figures 1–3).
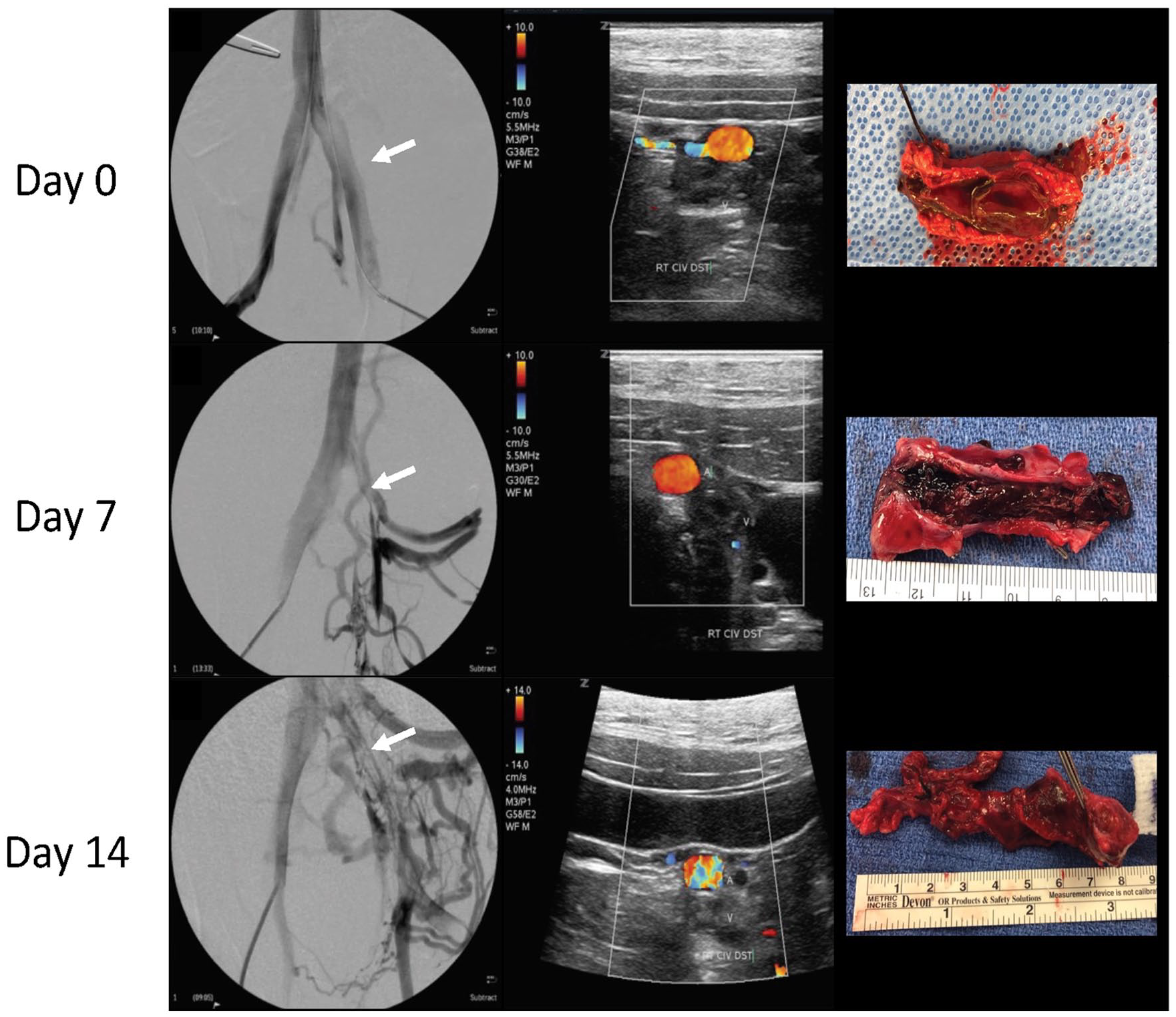
Representative venography, duplex, and gross histology images postthrombosis. Acute (day 0) DVT, subacute (day 7) DVT, and chronic (day 14) DVT: typical imaging and gross pathology findings associated with acute, subacute, and chronic thrombus (top to bottom): normal venogram; 7-day venogram with noticeable development of collateral venous pathways around obstructing thrombus; and 14-day venogram with extensive, robust collateralization. White arrows denote the occluded area. Duplex ultrasonography, demonstrating progression of acute thrombus in the dilated vein to contracted, chronic thrombus over time. Gross pathology of days 0, 7, and 14 thrombi.
Histopathology
Day 0: Compared to normal (contralateral) iliac vein (Figure 4A, B), the postthrombotic vein wall (Figure 4C, D) was found to have occasional prominent neutrophils (black arrowhead) and focal endothelial denudation. Experimental thrombus (Figure 4E
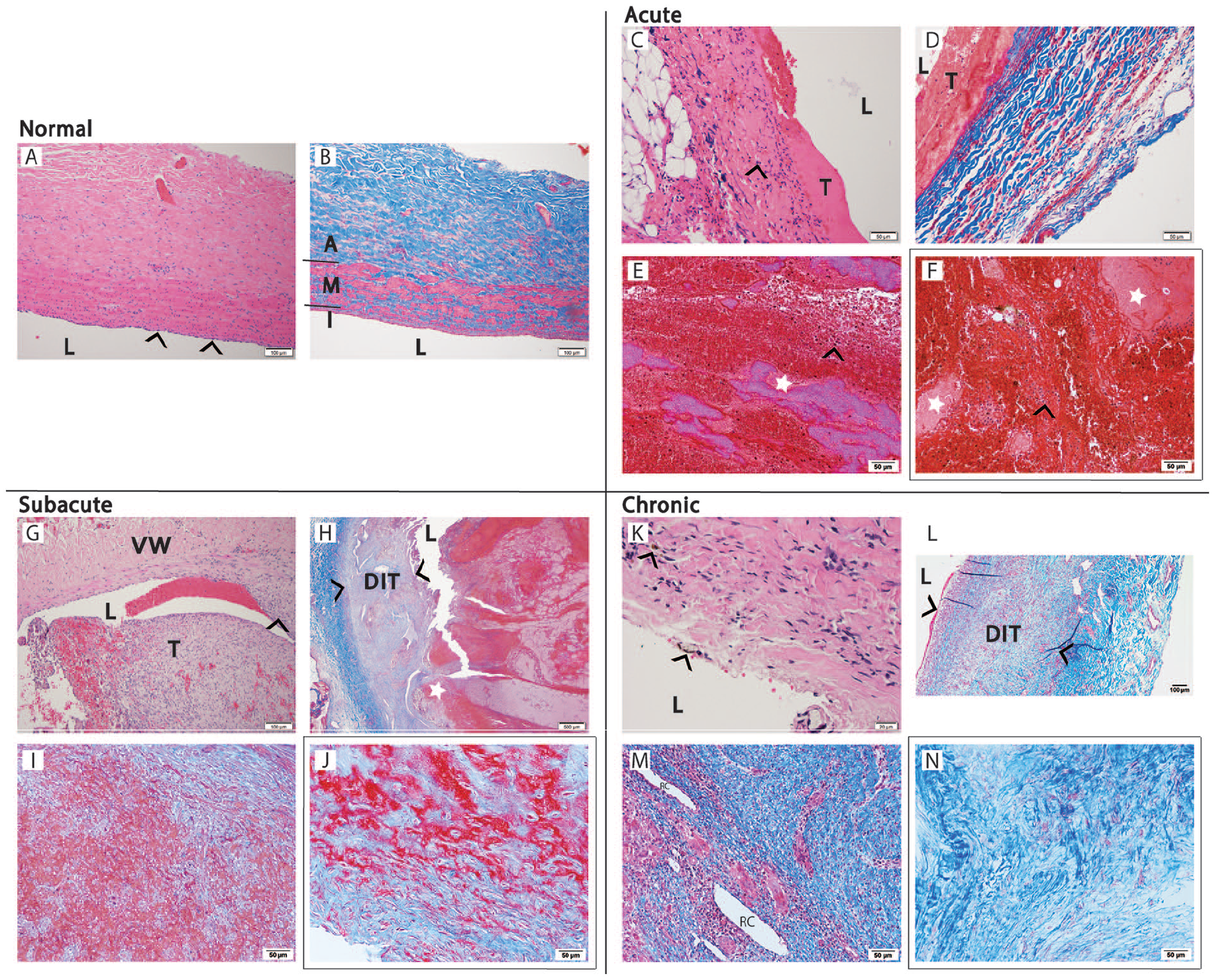
Representative human and porcine histology throughout the chronicity spectrum demonstrating vein wall and thrombus changes over time.
Day 7: Inflammatory cell infiltrate was noted at the thrombus–vein wall interface, and multinucleate giant cells were noted in focal areas of chronic inflammation (Figure 4G). Diffuse fibrosis and intimal thickening were observed in the vein wall adjacent to the thrombus in all specimens (Figure 4H). In all cases, a central thrombus appears fresher, < 2 days in age. Marked mesenchymal ingrowth and collagen deposition were noted in some cases. The organizing thrombus (Figure 4I) bears resemblance to human thrombus of approximately 2 months in age (Figure 4J).
Day 14: Focal hemosiderin deposits were noted within the vein wall–thrombus interface (Figure 4K). Diffuse intimal thickening was a feature of the vein wall regardless of the location of the thrombus (Figure 4L). Recanalization channels (Figure 4M) and foci of chronic inflammation were common within the thrombi. The dense collagen seen in the 14-day experimental thrombus was akin to human comparator thrombus of 3 years in age (Figure 4N).
Analysis of histopathology demonstrated features consistent with chronic thrombus at day 14 with myointimal cell growth, organized thrombus, and chronic inflammatory cells (Figure 4). Similarly, vein wall fibrosis and diffuse intimal thickening, characteristics of late vein wall injury, 21 were present in 14-day animals but not 7-day animals. These findings are summarized in Table 1. Based on histologic analysis of MSB-stained thrombi (Figure 5A), the collagen content was 41% in 7-day compared to 55% in 14-day thrombi (p = 0.17), and the thrombus erythrocyte percentage was 4.3% in 7-day compared to 2.2% in 14-day thrombi (p = 0.21) (Figure 5B).
Characteristics of thrombus and vein wall in representative histology slides from days 0, 7, and 14 samples.
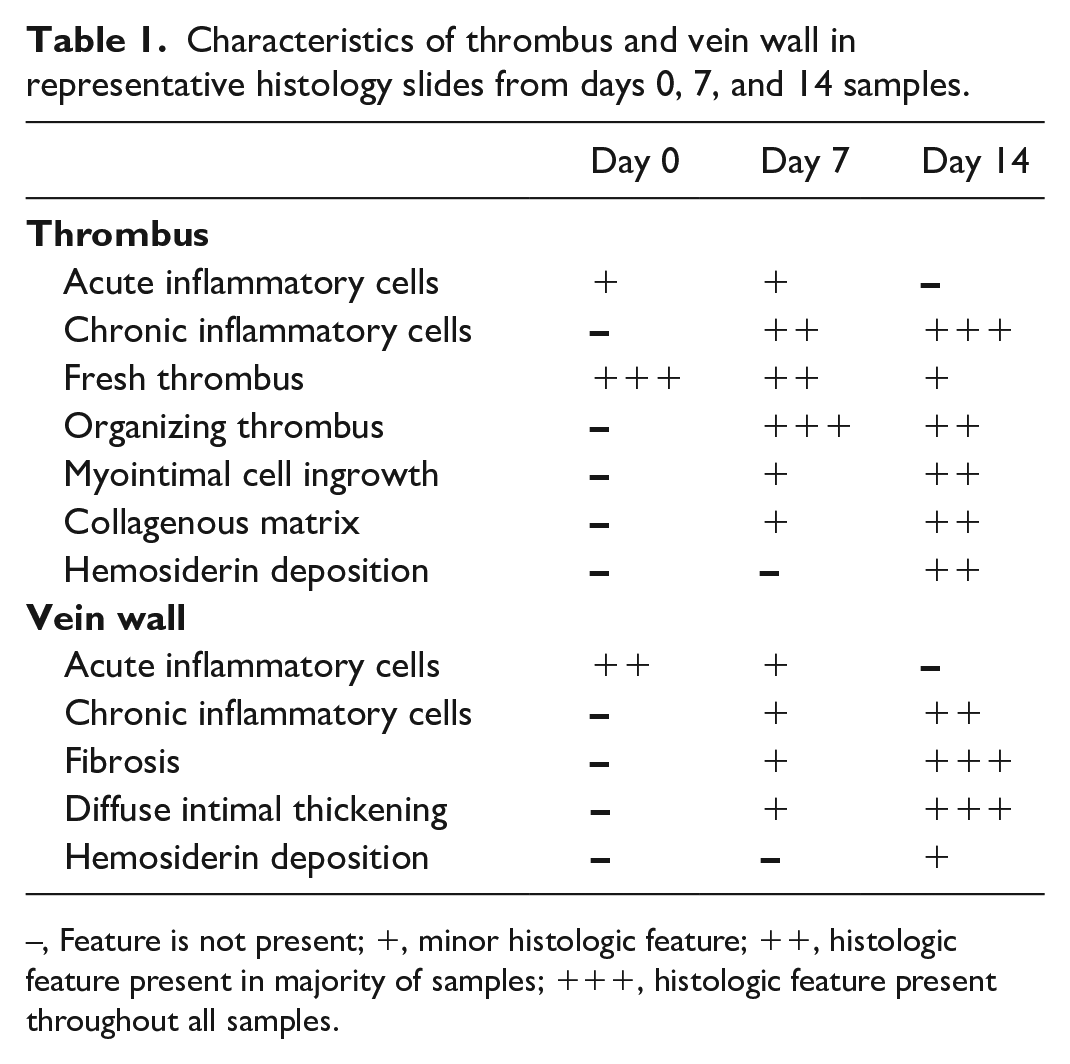
–, Feature is not present; +, minor histologic feature; ++, histologic feature present in majority of samples; +++, histologic feature present throughout all samples.
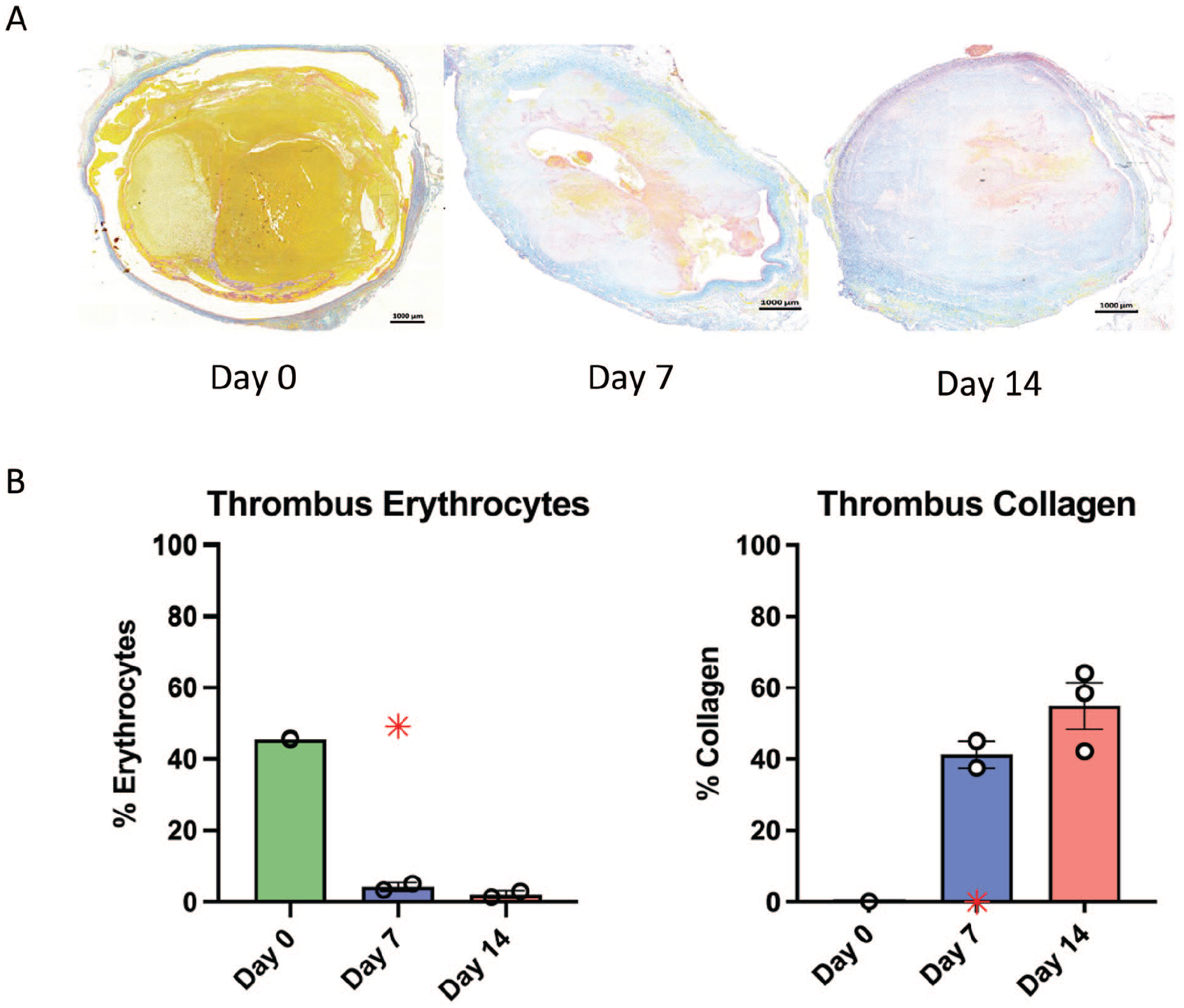
Representative histology of thrombi with quantification of thrombi erythrocytes and collagen.
Mechanical testing
During uniaxial compression loading, the onset point, which represents the amount of compression required to load the thrombus fiber network, was significantly higher in 7-day compared to 14-day thrombi, 66.6% versus 35.3%, p = 0.005 (Figure 6). The secant modulus was measured at the onset point and was 153.8 kPa in 7-day thrombi and 275.99 kPa in 14-day thrombi, p = 0.18. The Yeoh hyperelastic material model fits, defines, and predicts the mechanical response to the uniaxial compressive testing of the 7-day and 14-day thrombi. For best fit approximation, outliers from the data set were disregarded. Mean stress–strain curves were created through 80% strain for 7-day thrombi and 44% strain for 14-day thrombi based on the remaining data, due to force-based limits applied to the mechanical testing. The material constants for these fits can be found in Figure 7. The model and experimental data showed considerable agreement with R2 values of 0.987 and 0.976 for 7-day and 14-day thrombi, respectively. Yeoh hyperelastic model analysis of the mean stress–strain curves demonstrate a substantial increase in 2C10 in 14-day thrombi, which represents the shear modulus of the thrombi (1.85 kPa vs 2.85 kPa).
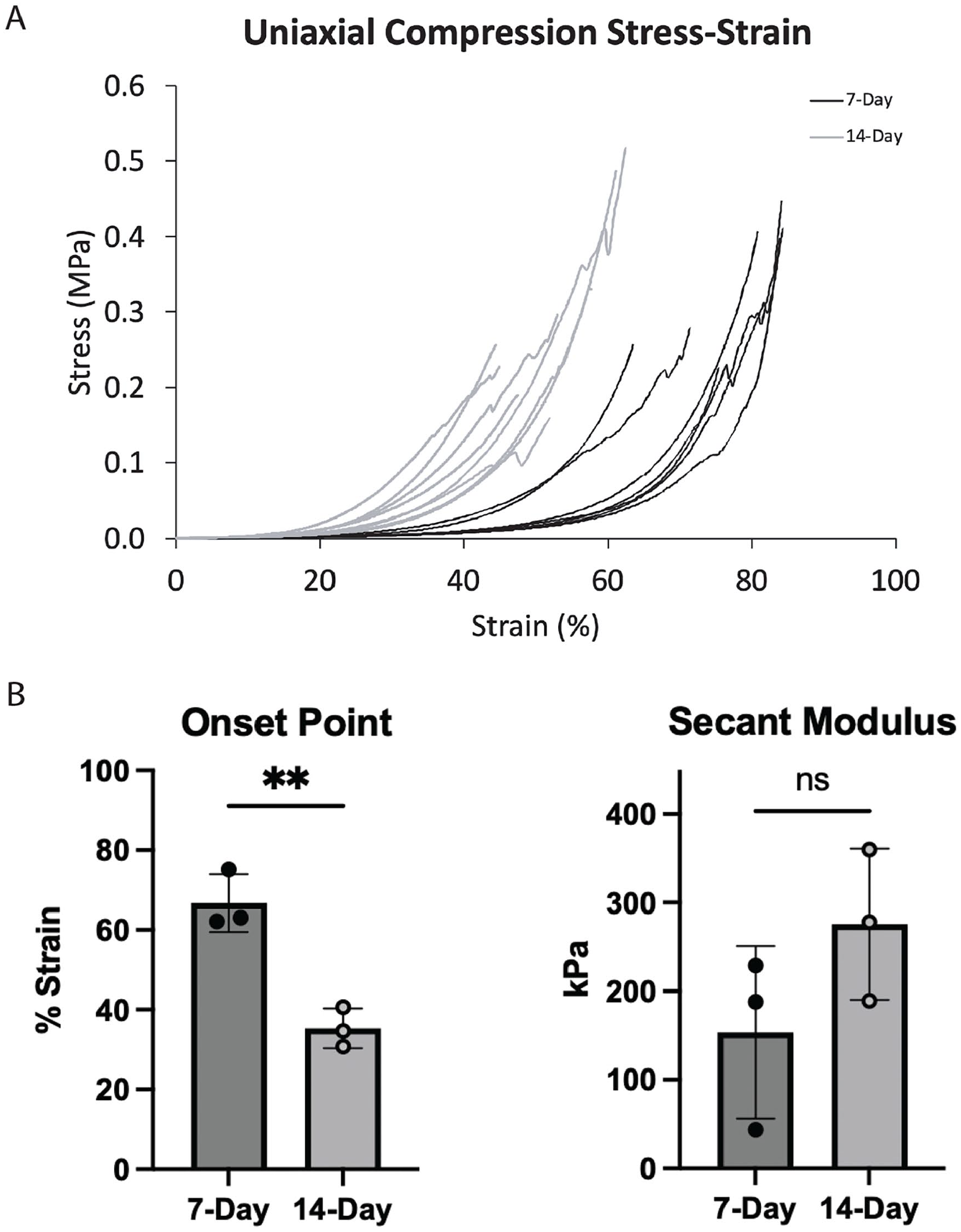
Mechanical characterization of day 7 and day 14 thrombi.
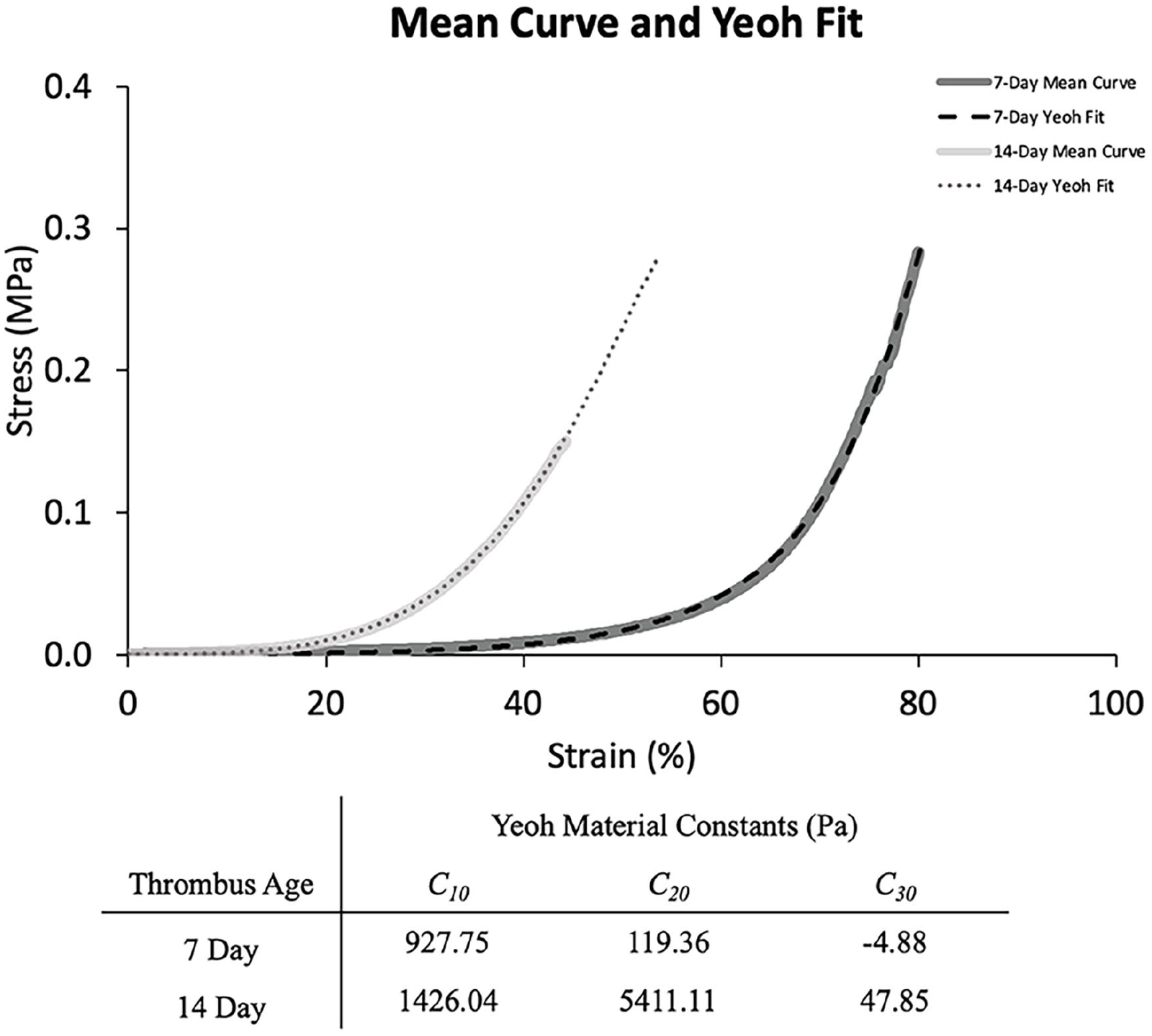
Mean stress–strain and Yeoh hyperelastic model curve fits with calculated Yeoh material constants. The increased stiffness of 14-day thrombi compared to 7-day thrombi is demonstrated by a leftward shift of the mean stress–strain curves and substantially larger C10 in the 14-day Yeoh fit.
Discussion
The results of this study demonstrate a novel porcine model of subacute and chronic, unilateral, DVT producing thrombi with ultrasound, venographic, and histopathological characteristics resembling those of human samples. 21 Current treatment paradigms indicate catheter-directed therapies in acute iliofemoral DVTs to reduce PTS severity; however, no specific, FDA-approved, chronic DVT crossing device is available.25,26 In this model, analysis of 7- and 14-day histopathology suggests the development of representative thrombi for preclinical testing of novel endovascular devices to address chronic DVT.
Current models of subacute and chronic DVT are limited by inadequate vessel diameter for testing of endovascular devices, residual foreign material, and prolonged time (⩾ 3 weeks) to develop representative chronic DVTs. 13 Through endothelial injury and temporary unilateral iliac vein balloon occlusion with thrombin injection, the proposed model produced consistent, long-segment DVT in large vessels without these limitations. At 14 days, gross and microscopic histopathology of the thrombi were consistent with chronic DVT. 27 Key features contributing to the increased complexity of catheter-based treatment of chronic DVTs – increased collagen content and vessel wall adherence – were successfully produced in this model. 28 Unilateral, large animal DVT model development has been pursued in an attempt to produce a model most representative of clinical scenarios, particularly to employ a vessel diameter appropriate for novel endovascular device testing. As recently as 2022, Schwein et al. described a totally endovenous model of cavoiliac DVT through balloon occlusion and thrombin injection. 29 This produced consistent cavoiliac thrombus, but occlusion of bilateral iliac veins eliminates the contralateral iliac vein as an internal control. Additionally, Shi et al. describe the need for external iliac vein occlusion to reduce collateralization to the thrombosed segment. 30 These models required open suture ligation, rendering them unsuitable for future endovascular testing due to complete interruption of the iliac vein. Through abrasive endothelial injury and external iliac vein endovascular plug, the model presently described incorporates the advantages of each model, while avoiding open surgery or caval thrombi. Methods employed elsewhere in preclinical vascular surgery models (i.e., Fogarty catheter, Edwards Lifesciences, Irvine, CA endothelial denudation and coil embolization of the external iliac vein) were not trialed herein but would be expected to be suitable alternatives.
In developing endovascular devices for crossing chronic DVTs, understanding and quantification of the mechanical properties imparted by increasing chronicity of thrombi are critical. Until this point, mechanical characterization of chronic DVTs has been limited to ultrasound elastography. Though noninvasive, there are limitations to ultrasound elastography. Namely, the calculation of Young’s modulus requires measurement of applied force. As the force is manually applied through the ultrasound transducer and various intervening tissues attenuate the applied force, it is not possible to accurately measure the force applied to the tissue of interest. As such, it is not possible to compute Young’s modulus or other mechanical characteristics reliant on applied force. Given the variations in measurement, some assert that measurements produced by ultrasound elastography may only be compared as binary measurements (softer or harder) when completed in the same study with the same transducer. 31
In this study, we observed that the strain required to significantly load the fiber network of the thrombus in compression was lower in 14-day thrombi when compared to 7-day thrombi. This suggests that the fiber network contained within the 14-day thrombus is denser and/or less permeable to fluid. The compressive stiffness (secant modulus) of the thrombus was 1.8 times higher in 14-day thrombi when compared to 7-day thrombi. Yeoh model fits for 7-day and 14-day thrombi are presented. These nonlinear, hyperelastic representations of the experimental data characterize the mechanical response of the material under compressive loads and can be utilized in modeling efforts to develop devices that can cross or deploy within a chronic DVT to overcome the shear and compressive resistance of the thrombus.
The thrombi produced by this model exhibit a significant increase in stiffness/mechanical properties compared with acute thrombi and thrombus analogues previously reported in the literature.32,33 The increase in compressive mechanical properties seen in this study validates the root of existing difficulties in addressing chronic DVTs with devices designed for acute DVTs. To overcome these observed mechanical changes, catheter or wire properties must be adjusted to reinforce tip design (i.e., stiffness and/or geometry) for targeting chronic DVT penetration while allowing flexibility for maneuvering the venous system. Similarly, mechanical thrombectomy catheter retrieval elements (i.e., stentrievers) must have adequate radial force to overcome the stiffness of chronic DVT. These observations may allow accurate ex vivo model design of subacute and chronic thrombi for high throughput preclinical testing of devices with varying mechanical characteristics.
Limitations
Though this study demonstrates a critical step forward on the path to endovascular devices designed specifically for chronic DVTs, there are several limiting factors. Foremost, though the thrombi and data were reproducible, the number of swine in each group was low. One 7-day thrombus was excluded from histologic analysis by a blinded vascular histopathologist. The thrombi demonstrated peripheral areas of chronicity with central acute thrombus, suggestive of perimortem thrombosis of a nonoccluded segment of vein. There are inherent limitations in validation of the model, as no parallel study exists on the mechanical characterization of human chronic DVTs. The mechanical testing performed utilized a force-based preload of the samples. However, due to the biologic variability of the samples, varying thrombi diameters create variable starting points in the strains of the samples. Regression of the stress–strain curves was performed to approximate strain-based zero points for the data prior to model analysis, which does create room for error in the data, but allows for more appropriate approximation of the mechanical properties. Despite this, the study provides mechanical properties to correlate with clinically available testing, allowing for improved diagnosis and management of chronic DVTs.
Conclusions
This study describes a newly developed, totally percutaneous model for generation of chronic thrombus in 2 weeks. The model demonstrates low morbidity and cost, and represents similar venographic and histopathologic characteristics to human correlates. Therein, the success of the model provided the opportunity for the first published mechanical characterization of chronic thrombus, quantifying increases in compressive stiffness related to histologic increases in collagen content. This clinically representative model and properties allow for targeted endovascular device development for crossing and retrieving chronic thrombi, ultimately preventing postthrombotic syndrome. Future efforts will be directed at the correlation of findings to clinically available testing such as ultrasound elastography and creation of ex vivo models to capture the mechanical properties of in vivo DVT more accurately for utilization in device design.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X241308481 – Supplemental material for Variation in compressive mechanical properties between subacute and chronic venous thrombosis in a novel unilateral iliac thrombosis model
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X241308481 for Variation in compressive mechanical properties between subacute and chronic venous thrombosis in a novel unilateral iliac thrombosis model by Nathaniel J Parchment, Christian Argenti, Sriganesh B Sharma, Oscar Moreno, David Gordon, Laura Durham, Veronica Forsythe, David M Williams, Thomas W Wakefield, Nathan Weidenhamer, Daniel Durant Myers, Minhaj S Khaja, Peter K Henke, Albert J Shih and Andrea Tara Obi in Vascular Medicine
Footnotes
Acknowledgements
The authors wish to thank Andy Poli (University of Michigan) for assistance with the technical aspects of mechanical testing and data analysis, and Angela Hawley (University of Michigan) for assistance with histologic processing. Partial data were presented at the virtual 2020 American Venous Forum. A commentary by Li and Jaffer accompanies this article. 34
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was partially funded by a scientific grant from Medtronic, Inc. This funding was used for supplies and swine. The analysis and interpretation were completed independently of Medtronic, Inc. The final manuscript was prepared by the authors and was sent to Medtronic, Inc. for review prior to submission for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

