Abstract
Introduction:
Prior data suggest associations between hearing loss, cardiovascular (CV) risk factors, and CV disease. Whether specific hearing loss patterns, including a strial pattern associated with inner ear vascular disease, are associated with systemic endothelial dysfunction and carotid intima–media thickness (IMT) remains unclear.
Methods:
We evaluated participants without prevalent CVD in the Framingham Offspring Study who underwent formal audiogram testing and brachial and carotid artery ultrasounds. Audiograms were categorized as normal or as belonging to one of four abnormal patterns: cochlear-conductive, low-sloping, sensorineural, or strial. Endothelial function as measured by brachial artery flow-mediated dilation (FMDmm and FMD%). Internal and common intima–media thicknesses (icIMT and ccIMT, respectively) were compared between audiogram patterns.
Results:
We studied 1672 participants (mean age 59 years, 57.6% women). The prevalence of each hearing pattern was as follows: 43.7% normal; 20.3% cochlear-conductive; 20.3% sensorineural; 7.7% low-sloping; and 8.0% strial. Strial pattern hearing loss was nearly twice as prevalent (p = 0.001) in those in the highest quartile of ccIMT and nearly 50% higher in those in the highest icIMT quartile (p = 0.04). There were no statistically significant differences between the prevalence of the strial pattern comparing the lowest quartiles of FMDmm and FMD% with the upper three quartiles. Age- and sex-adjusted linear regression models did not show significant associations between the vascular measures and hearing patterns.
Conclusion:
Abnormal hearing patterns were not significantly associated with impaired brachial FMD and increased carotid IMT after adjusting for age and sex effects, which may reflect age and sex-related distributional differences based on hearing loss pattern.
Keywords
Introduction
In the inner ear, the stria vascularis, which supplies the cochlear scala media and maintains the endocochlear potential, is a prime example of microcirculation with a vulnerable target end-organ. 1 The stria vascularis is fed by the radial branches of the spiral modiolar artery without any collaterals or anastomoses providing an environment with heightened sensitivity to impaired blood flow and oxygen delivery. Reduced strial blood flow results in a specific, strial pattern of hearing loss. 2 This pattern is typified by reduced hearing acuity across all frequencies with a larger adverse impact at low frequencies (250–1000 Hz) on audiometric examination relative to other common forms of hearing loss (online Supplemental Figure 1).2,3
The anatomical vulnerability of the cochlea to reduced strial blood flow suggests that identification of a strial hearing loss pattern by audiometry could be a sensitive method for early detection of individuals without prevalent atherosclerotic disease but with endothelial dysfunction or increased carotid intima-media thickness (IMT) and, therefore, at high risk for ischemic cardiovascular events.4–6 Whether specific hearing loss patterns differ in their association with the presence of systemic endothelial dysfunction or increased carotid IMT in a population without known coronary artery disease remains unclear. 4 We hypothesized that a strial pattern of hearing loss would be associated, to a greater extent than other hearing loss patterns, with impaired endothelium-dependent vasodilation and increased carotid IMT in participants in the Framingham Heart Study’s Offspring Cohort without prevalent cardiovascular disease at the time of hearing testing.
Methods
Participants
The Framingham Offspring Study enrolled 5124 participants (52% women) between 1971 and 1975 who have completed wide-ranging questionnaires and assessments every 4 to 8 years since enrollment. 7 A total of 3532 and 3539 participants participated during study visits six (1995–1998) and seven (1998–2001), respectively. During study visit six, 2329 participants completed audiograms. Of these, 2040 (88%) had a concomitant carotid artery ultrasound during the same study visit. During study examination cycle seven, brachial artery flow-mediated dilation (FMD) measurements were done on 2123 (91%) of the participants who had an audiogram during the previous study visit. An additional 451 participants were excluded who had prevalent cardiovascular disease. The Framingham Heart Study protocols were approved by the Institutional Review Board at Boston University Medical Center. All study participants gave written informed consent prior to participating in the study. This data analysis was additionally approved by the Institutional Research Board at the Medical College of Wisconsin.
Audiogram
A standard examination consisting of pure tone threshold audiometry in each ear and a word recognition test was performed at study visit six. 8 The audiometric examination environment, equipment, and measurement techniques adhered to the American National Standards Institute’s accepted methodology. The audiogram from the ear with lower pure tone average (PTA) (i.e., better hearing) was used for analysis to focus on associations between hearing loss and systemic disease that may not be reflected by using the ear with worse hearing. Audiograms were categorized using the methods described in Friedland et al. into one of five patterns depending on the pattern of hearing loss: normal, cochlear-conductive, low-sloping, sensorineural, and strial. 2
Brachial artery flow-mediated dilation (FMD) and carotid intima–media thickness (IMT) measurements
Both methodologies have been previously described. Please see the online Supplemental Methods for greater detail.9,10
Statistical methods
Summary statistics were calculated for all study variables; continuous variables were summarized by the mean and standard deviation and categorical variables were summarized by the frequency and percentage. The frequency of missing values was reported for each variable and all subsequent analyses employed an available-case approach. Primary outcomes included two surrogate markers of cardiovascular health: percent FMD (FMD%) and common carotid artery IMT (ccIMT). Secondary outcomes included baseline brachial artery diameter, absolute FMD (FMDmm), and internal carotid IMT (icIMT). Each study dependent variable was analyzed as a continuous measure. Skewed to the right positive continuous dependent variables (ccIMT and icIMT) were log-transformed prior to analyses with audiogram pattern as an independent variable.
Comparisons were made between patient characteristics and sex, audiogram category, and each binomial and continuous variable. Two-group comparisons were made using Wilcoxon rank-sum test for continuous variables, and Fisher’s exact test for categorical variables; comparisons between three or more groups were made using Kruskal–Wallis and chi-squared tests, respectively. Exact tests were used when small cell counts were observed.
Additionally, participants were divided into four equal quartiles based on the following parameters: FMD%, FMDmm, ccIMT, and icIMT. The three highest quartiles for FMD measurements and the three lowest quartiles for ccIMT and icIMT were combined into a single category and compared to the lowest FMD and highest IMT quartiles, respectively, using Wilcoxon rank-sum test for continuous variables and Fisher’s exact test for categorical variables.
Multiple regression modeling was used to make adjusted comparisons of each dependent variable by audiogram pattern; linear regression was employed for continuous variables. Adjustment variables were selected from known CV risk factors associated with the vascular measures, including age, sex, systolic blood pressure, body mass index, use of lipid-lowering medication, smoking, and heart rate. 11 Backwards variable selection procedures were used to develop the final models. The goodness-of-fit of each linear regression model was described by the coefficient of determination, R2.
All p-values were two-sided and p < 0.05 was considered statistically significant. No adjustments were made for multiple testing. Data processing was performed using SAS software, version 9.4 (SAS Institute, Cary, NC, USA), and statistical analysis was performed using R, version 3.3.4 (R Foundation for Statistical Computing, http://www.R-project.org). The corresponding author had full access to all the data in the study and takes responsibility for its integrity and the data analyses.
Results
Participant characteristics
We studied 1672 participants (mean age 59 years, 57.6% women). The audiograms were organized into one of five unique categories based on the pattern of hearing loss: normal (43.7%, n = 732), cochlear-conductive (20.3%, n = 339), sensorineural (20.3%, n = 339), low-sloping (7.7%, n = 128), and strial (8.0%, n = 134). The baseline characteristics of these participants are presented in Table 1, categorized by type of hearing loss. Individuals with strial and low-sloping hearing loss were generally older, with a higher prevalence of hypertension and lipid-lowering therapy and higher pulse pressure than those with normal hearing or other hearing loss patterns. We also looked at the characteristics of individuals who did and did not undergo audiograms (online Supplemental Table 1). Of all subjects who underwent brachial reactivity testing, those who did not undergo audiogram testing were, on average, 1 year older, and generally had a higher prevalence of diabetes, recent smoking, hypertension, and a higher average ccIMT. Of all subjects who underwent brachial reactivity testing, those who underwent both audiogram and carotid imaging compared to audiogram alone had a higher body mass index, a greater prevalence of diabetes, and were less likely to be on hormone replacement therapy (online Supplemental Table 2).
Participant characteristics stratified by audiogram pattern.
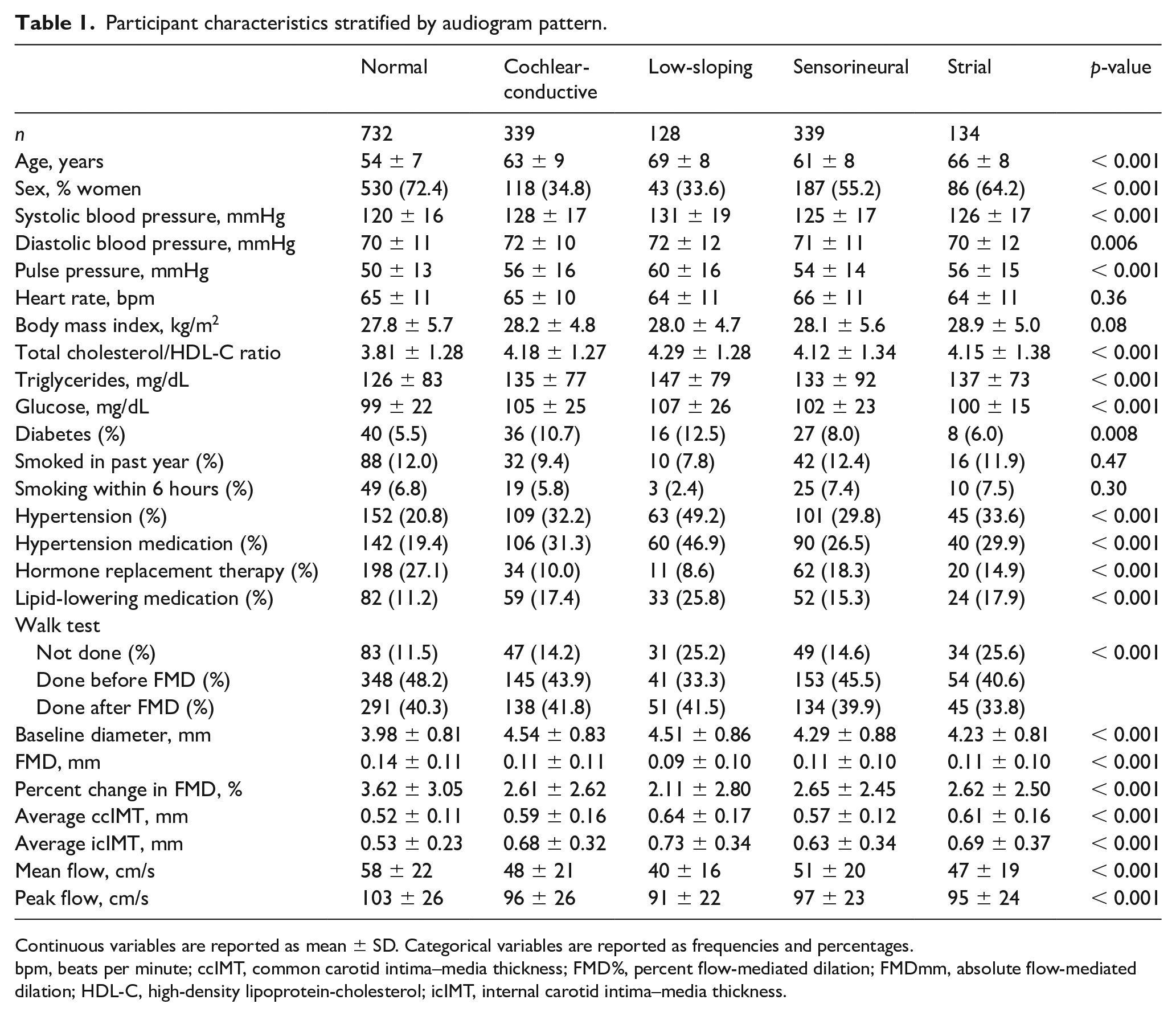
Continuous variables are reported as mean ± SD. Categorical variables are reported as frequencies and percentages.
bpm, beats per minute; ccIMT, common carotid intima–media thickness; FMD%, percent flow-mediated dilation; FMDmm, absolute flow-mediated dilation; HDL-C, high-density lipoprotein-cholesterol; icIMT, internal carotid intima–media thickness.
Participants were divided into quartiles of FMD%, ccIMT, and icIMT. Those in the lowest FMD% quartile or FMDmm (i.e., the greatest degree of impairment in FMD), were compared with the higher three quartiles (online Supplemental Tables 3 and 4) and the highest quartiles of ccIMT and icIMT were compared to their lower three quartiles (Supplemental Tables 5 and 6). In unadjusted analyses, the most impaired/pathological quartile of each measurement had a significantly lower number of participants with normal hearing (46.4 vs 36.4% for FMDmm (p = 0.001); 46.4 vs 38.4% for FMD% (p < 0.001); 50.6 vs 23.2% for ccIMT (p < 0.001); 49.4 vs 25.1% for icIMT (p < 0.001). While all patterns of hearing loss were numerically more prevalent in those in the most diseased quartiles of FMD%, ccIMT, and icIMT, strial pattern hearing loss was nearly twice as prevalent (12.1 vs 6.7%, p = 0.001) in those in the highest quartile of ccIMT and nearly 50% more prevalent in those in the highest icIMT quartile (10.7 vs 7.3%, p = 0.040). There was no statistically significant difference between the prevalence of the strial pattern in those in the lowest quartile of FMDmm and FMD% compared to the upper three quartiles (FMDmm: 8.0 vs 8.3%, p = 0.91 and FMD%: 8.1 vs 8.0%, p > 0.99).
Association of hearing patterns with endothelial function
Brachial FMD% and FMDmm were significantly lower in participants with any type of hearing loss (p < 0.001) relative to the normal hearing pattern (Table 1). Multiple linear regression demonstrates abnormal hearing pattern was associated with lower FMD% (Supplemental Table 7, model R2 = 0.04, p < 0.001 for the audiogram pattern variable) and FMDmm (Supplemental Table 8, model R2 = 0.02, p <0.001). However, these associations were not significant after adjusting for age and sex (FMD%: model R2 = 0.10, p = 0.63 for the audiogram pattern variable; FMDmm: model R2 = 0.07, p < 0.001 for overall model, p = 0.88 for audiogram data).
Following stratification of FMDmm (Figure 1A) and FMD% (Figure 1C) measurements into four age-based categories (ages 33–54, 55–60, 61–68, and 69–88 years), we observed no statistically significant differences in either of these measurements by hearing pattern within each category.
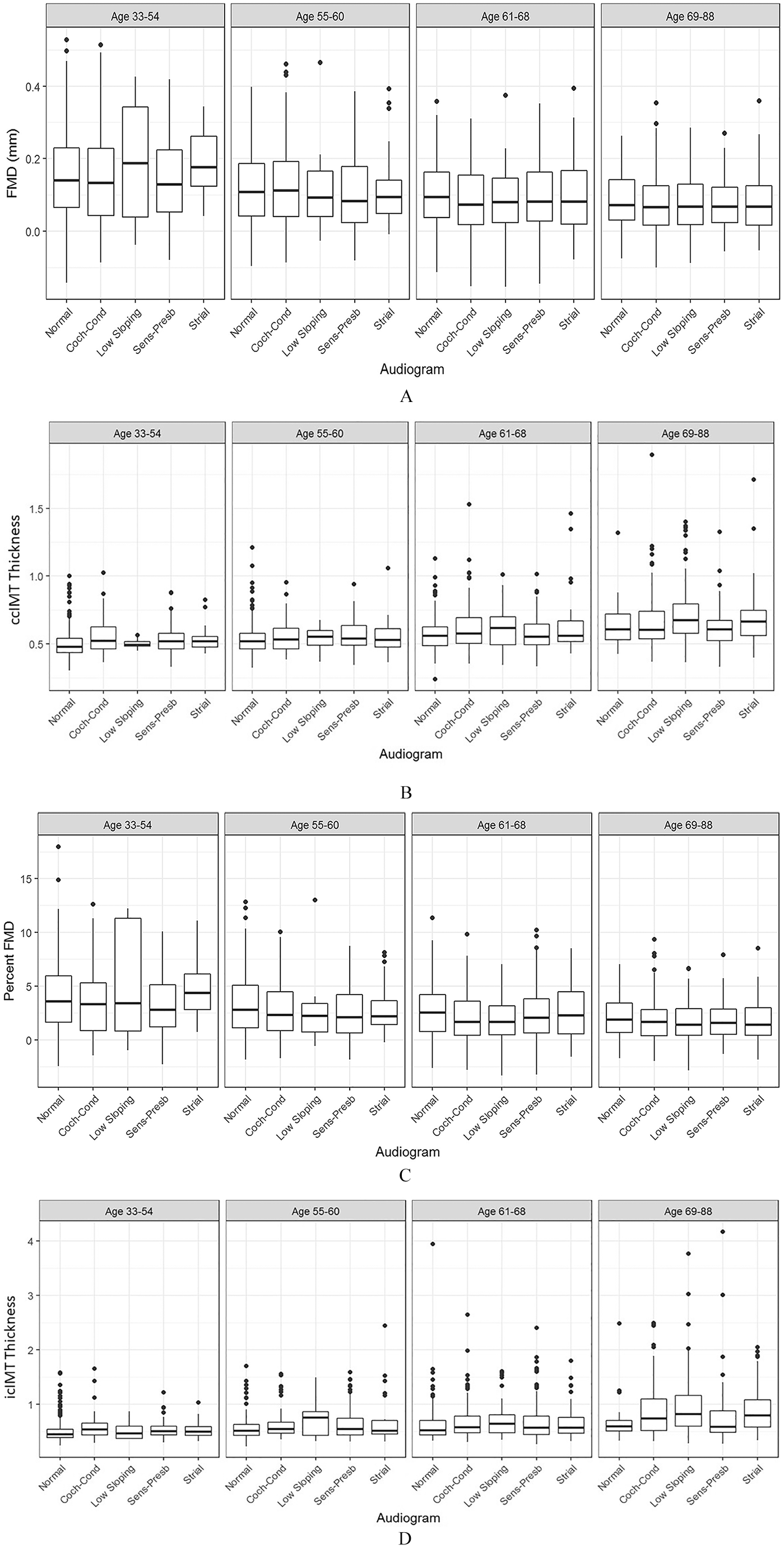
Audiometric patterns and measurements of vascular function and structure by age groups. Study participants were divided into four different age categories: (1) 33–54 years, (2) 55–60, (3) 61–68, and (4) 69 and over. Average brachial FMDmm
Hyperemic flow is a significant driver of the brachial FMD response, with greater flow associated with greater shear and generally a larger vasodilatory response. While the flow stimulus is lower in subjects with abnormal hearing patterns (Table 1), no association was found between hearing patterns and hyperemic flow velocities following age and sex adjustment, with age as the only variable significantly associated with either flow velocity measurement (data not shown).
Association of hearing patterns and carotid IMT measurements
ccIMT was significantly greater in individuals with abnormal hearing patterns compared to normal hearing patterns (Table 1). In linear regression models, while an abnormal hearing pattern was associated with higher ccIMT in unadjusted analyses (Supplemental Table 9, model R2 = 0.10, p < 0.001 for the hearing loss variable), this association was completely attenuated following age and sex adjustment (model R2 = 0.19, p = 0.13 for the hearing loss variable).
icIMT was significantly greater in individuals with abnormal hearing patterns compared to normal hearing patterns (Table 1). An abnormal hearing pattern was associated with higher icIMT in unadjusted analyses (Supplemental Table 10, model R2 = 0.09, p < 0.001). After adjusting for age and sex, icIMT was not associated with hearing loss (model R2 = 0.18, p < 0.001 overall, p = 0.37).
Kruskal–Wallis tests compared ccIMT (Figure 1B) and icIMT (Figure 1D) measurements into the same four age-based categories as FMDmm and FMD%; we found no differences in either ccIMT or icIMT by hearing pattern (p = NS).
Sex-stratified associations between hearing patterns and FMD and IMT
Additional regression analyses stratified by sex failed to find sex-specific associations between hearing patterns and impaired FMD or the thickest IMT categories. The sex-stratified analyses did not result in any age-adjusted associations between hearing patterns and vascular measurements (data not shown).
Discussion
Our analyses of these data in individuals without prevalent cardiovascular disease demonstrate that hearing loss was associated with reduced dynamic endothelium-dependent vasodilation of the brachial artery and increased carotid IMT in unadjusted analyses. However, these associations were not specific to any hearing pattern and adjustment for age and sex eliminated these associations. Further, comparisons of FMD and carotid IMT measurements for each hearing pattern within narrower age ranges failed to demonstrate any meaningful differences in these measurements between hearing patterns. Taken together, these data suggest that while hearing loss is associated with impaired systemic endothelial function and adverse structural changes of the carotid artery, these associations are most likely explained by age- and sex-related variations of these measurements. In the context of prior work, these data represent the largest and, to our knowledge, the only study to date, studying associations between hearing loss patterns and human vascular pathophysiology.
Few studies to date have looked at associations between hearing loss and brachial artery FMD. A small study of 29 patients with sudden sensorineural hearing loss demonstrated brachial FMD% was lower in individuals with this syndrome compared to control participants. 5 No differences were seen in carotid IMT compared to controls. 5 Low-density lipoprotein (LDL) cholesterol and fibrinogen apheresis as a treatment for sudden sensorineural hearing loss was associated with improved brachial FMD% in a report on six sudden sensorineural hearing loss (SSHL) patients. 12 Our data are the first, to our knowledge, to investigate and report on the associations between chronic hearing loss and hearing loss patterns and brachial artery FMD in a large, well-phenotyped population using highly standardized methods.
Two studies have examined the association between carotid IMT and hearing loss.6,13 In a cohort of 2619 participants (ages 45–81 years), carotid IMT was associated with self-reported hearing disorders following multivariable adjustment. 6 In the Beaver Dam Offspring Cohort (n = 1984), higher mean carotid IMT and the presence of plaque were associated with a 15% increase in the incidence of hearing impairment at 5 years of follow-up following multivariable adjustment, albeit more weakly than age and male sex. 13 Differences between the findings in the Framingham Offspring Cohort compared to these studies may relate to differences in the study populations regarding how hearing loss was categorized, differences in statistical power, or differences in measurement quality. Our data extend current findings by demonstrating the carotid IMT is not associated with any specific hearing loss pattern following adjustment for age and sex.
Previous epidemiological studies have investigated associations between different metrics of hearing loss, hearing loss patterns, and cardiovascular risk factors and prevalent cardiovascular disease.3,14–16 These data generally support the concept that hearing loss is associated with prevalent cardiovascular disease and traditional cardiovascular risk factors, particularly age, with some variability between studies based likely on the heterogeneity of the diagnosis of hearing loss and study populations. Our data extend these findings by demonstrating, in a population of participants without prevalent cardiovascular disease, that hearing loss is also associated with imaging-based predictors of cardiovascular disease, endothelial dysfunction, and increased carotid IMT, in unadjusted analyses that disappear following adjustment for age and sex.
Study limitations
Our study has several limitations. Strial pattern hearing loss is frequently confounded by concomitant higher frequency hearing loss. 3 Our study separated participants into their dominant audiogram pattern, which may have resulted in classifying some with mixed strial and higher frequency hearing loss into a non-strial category. This may in part account for the relatively small percentage of strial hearing patterns (n = 134, 8.0%) noted in our study population. Strial pattern hearing loss can be seen following gamma knife radiosurgery for inner ear tumors. Given the relative rarity of these tumors and their primarily unilateral appearance, it is unlikely our results are confounded by complications following gamma knife surgery. The preponderance of the study population is largely comprised of individuals of European ancestry residing in New England, which limits generalizability of our study findings to other populations. Measurements of FMD% and audiograms, while of high quality in this study, were made over 20 years ago and more contemporary technologies are available for the measurement of both metrics today. Also, given the technology at the time of data acquisition, we were unable to measure carotid plaque explicitly. Therefore, we cannot exclude independent associations between the presence of carotid plaque and hearing loss patterns. Additionally, the retrospective and cross-sectional nature of this study allows us to draw conclusions about associations but not causality. Future studies should assess the prospective validity of our findings. These limitations are balanced by the several strengths, including having a large, well-characterized cohort without prevalent cardiovascular disease with high-quality audiogram, brachial FMD, and carotid ultrasound data.
Conclusion
Although unadjusted analyses suggest associations between hearing loss and impaired endothelium-dependent vasodilation in the brachial artery as well as increased carotid IMT in individuals without prevalent cardiovascular disease, these unadjusted associations disappear following age and sex adjustment. No specific pattern of hearing loss appears to be associated with adverse vascular changes in our study population.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X211025087 – Supplemental material for Abnormal hearing patterns are not associated with endothelium-dependent vasodilation and carotid intima–media thickness: The Framingham Heart Study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X211025087 for Abnormal hearing patterns are not associated with endothelium-dependent vasodilation and carotid intima–media thickness: The Framingham Heart Study by Sudhi Tyagi, David R Friedland, Lisa Rein, Sergey S Tarima, Christopher Mueller, Emelia J Benjamin, Ramachandran S Vasan, Naomi M Hamburg and Michael E Widlansky in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Framingham Heart Study is conducted and supported by the National Heart, Lung, and Blood Institute (NHLBI) in collaboration with Boston University (Contracts No. N01-HC-25195, HHSN268201500001I, and 75N92019D00031). Dr Tyagi received support from T32GM089586. Dr Benjamin is supported by HL092577, HL141434, AG066010, and 2U54HL120163; American Heart Association 18SFRN34110082; brachial measurements were supported by 1R01 HL60040. Dr Hamburg is supported by an AHA Strategically Focused Research Network grant focused on cardiometabolic health and HL007224. Dr Vasan is supported by the Evans Medical Foundation and the Jay and Louis Coffman Endowment from the Department of Medicine, Boston University School of Medicine. Dr Widlansky is supported by HL125409, HL128240, HL144098, R38HL143561, and R61AT010680.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
