Abstract
Flow-mediated dilation (FMD) is recognized as a non-invasive endothelial function bioassay. However, FMD’s relationship with endothelial cell oxidative stress in humans is yet to be determined. Here, we sought to determine if FMD was associated with endothelial nitric oxide synthase (eNOS) and endothelial oxidative stress in humans. Twenty-seven apparently healthy young men (26.5±5.9 years) underwent brachial artery FMD testing and endothelial cell biopsy from a forearm vein. Non-normalized FMD (%) and three different brachial artery FMD normalizations were performed: (1) peak shear rate (%/SR); (2) area under the SR curve until peak dilation (%/AUC); and (3) AUC 30 seconds before peak dilation (%/AUC30). Immunofluorescence quantification was used to assess eNOS expression and nitrotyrosine (NT), a criterion marker of endothelial oxidative stress. Values for eNOS and NT expression were reported as a ratio of endothelial cell to human umbilical vein endothelial cell average pixel intensity. NT expression was significantly correlated with FMD normalized by AUC30 (r = −0.402, p<0.05). Other FMD normalizations and non-normalized FMD were not significantly correlated with NT expression (r range = −0.364 to −0.142, all p>0.05). There were no significant correlations between eNOS expression and normalized and non-normalized FMD (r range = −0.168 to −0.066, all p>0.05). In conclusion, brachial artery FMD is associated with venous endothelial cell oxidative stress. However, this association is observed only when FMD is normalized by AUC30.
Introduction
Endothelial dysfunction (ED) is the first pathophysiological step that produces atherosclerosis, which is responsible for ≈90% of all cardiovascular diseases.1–4 ED is generally defined as the decrease in nitric oxide (NO) bio-availability within the endothelium, normally attributed to an increased endothelial oxidative stress.1–3,5,6 In humans, early stages 7 and changes after interventions8–10 of ED can be assessed non-invasively via flow-mediated dilation (FMD).11–15 Briefly, FMD measures the dilatory capacity of a blood vessel, typically the brachial artery, which is exposed to a blood flow-induced stimulus (i.e. shear stress) elicited by reactive hyperemia.12,13 Although FMD is recognized as a bioassay for ED,11–15 with excellent clinical significance,16,17 few studies have shown a relationship between FMD and endothelial oxidative stress in humans. 18
FMD is based on two physiological principles: (1) blood flow-induced shear stress activates the endothelium to produce NO; and (2) NO elicits vascular dilation via relaxation of vascular smooth muscle.12,14 When there is an increased endothelial oxidative stress, endothelial NO synthase (eNOS) is uncoupled, producing a decrease in NO production and bioavailability.5,6,19 Thus, an increased endothelial oxidative stress should decrease endothelial reactivity to shear stress, which will negatively impact FMD.
Accordingly, the purpose of the present study was to determine if FMD is associated with endothelial oxidative stress and eNOS in humans. We used non-normalized and normalized FMD, as suggested by recent reports.14,15,20–25 Three different FMD normalization schemes were used: two routinely cited in the literature and one based on NO half-life and blood-flow patterns. We hypothesize that FMD would be associated with endothelial oxidative stress and eNOS expression.
Methods
Subjects
Twenty-seven young men, 19–34 years of age, with different activity background (e.g. sedentary, active recreational) were enrolled in the study. All subjects were apparently healthy with no known cardiovascular disease or major cardiovascular risk factors. Exclusion criteria included known cardiovascular disease including cardiac arrhythmias, prescription medication, ‘over-the-counter’ painkillers such as non-steroidal anti-inflammatory drugs (NSAIDs) or aspirin, or nutritional supplements containing antioxidants. The study was approved by the Institutional Review Board at the University of Florida and written informed consent was obtained from all subjects. Female subjects were not recruited due to significant variations in vascular function during the four phases of the menstrual cycle. 26
Experimental protocol
Subjects were asked to visit the laboratory twice, on two different days, with at least 48 hours between visits. The first visit was scheduled to perform some baseline measurements such as height, weight, and resting blood pressure, and a maximal aerobic capacity (VO2max; Parvomedics Inc., Sandy, UT, USA) test on a cycle ergometer (Hudson EC-400, The Hudson Fitness Inc., Dallas, TX, USA). During the second laboratory visit, venous endothelial cells were harvested from the left arm and brachial FMD was performed in the right arm. All subjects were tested at the same time of day and after 8 hours of fasting, with no caffeine intake for at least 12 hours and consumption of a low nitrate diet for 48 hours.
Venous endothelial cell harvesting
The following procedure was used for collection and preparation of venous endothelial cells and measurement of protein expression using quantitative immunofluorescence, as previously described.27–29 Two sterile J-wires (Daig Corp., Minnetonka, MN, USA) were advanced into an antecubital vein (≈4 cm beyond the tip of the catheter) and retracted through an 18-gauge catheter. The wires were then transferred to a dissociation buffer solution where 200–300 endothelial cells were recovered via a washing and centrifugation protocol. Collected cells were fixed with 3.7% formaldehyde, plated on poly-
Brachial artery flow-mediated dilation (FMD)
Following a 10-minute rest period, brachial blood pressure was measured in triplicate via an automated non-invasive device (BpTRU BPM-100; VSM MedTech Ltd, Canada). Brachial endothelial-dependent arterial vasodilation was performed using high-resolution ultrasound (HDI 3000; ATL Inc., Bothell, WA, USA) and following international guidelines.13,14 After lying quietly for 15 minutes, a 10.5 MHz linear phase array ultrasound transducer was used to image the right brachial artery longitudinally and recorded directly to a digital storage device via a super video interface (Pinnacle Systems GmbH, Avid Technology Inc., Burlington, MA, USA). Imaging was performed with the ultrasound probe fixed approximately 5 cm above the antecubital fossa.
Brachial artery diameters were determined every 5 seconds for 150 seconds from 30 seconds before cuff deflation using automated edge-detection software (Vascular Research Tools, Medical Imaging Applications LLC, Coralville, IA, USA). Peak systolic blood-flow velocity was also recorded every 5 seconds for 150 seconds from 30 seconds before cuff deflation. Brachial peak diameter was identified as the single peak diameter observed during the plateau phase after cuff deflation. 30 Brachial FMD was calculated as relative (%FMD = peak diameter/baseline diameter). In our laboratory, the coefficient of variation for peak %FMD using this method is ~8%. 31
Non-normalized FMD (%) and three different brachial artery FMD normalizations were performed (Figure 1): (1) peak shear rate (%/SR); (2) area under the SR curve (%/AUC) until peak dilation; and (3) AUC 30 seconds before peak dilation (%/AUC30). SR and AUC normalizations are often described in the literature.14,15,23,24,32,33 AUC30 normalization is based on two physiological principles: (1) NO half-life 34 and (2) turbulent blood flow that might be present immediately after the cuff release, at the beginning of reactive hyperemia. 35 Peak FMD is normally observed 45–80 seconds after cuff deflation; 14 however, NO half-life is <10 seconds. 34 According to previous studies,35,36 it is highly probable that blood flow during the first 15–30 seconds after cuff deflation is turbulent, which may inhibit endothelial function.37,38
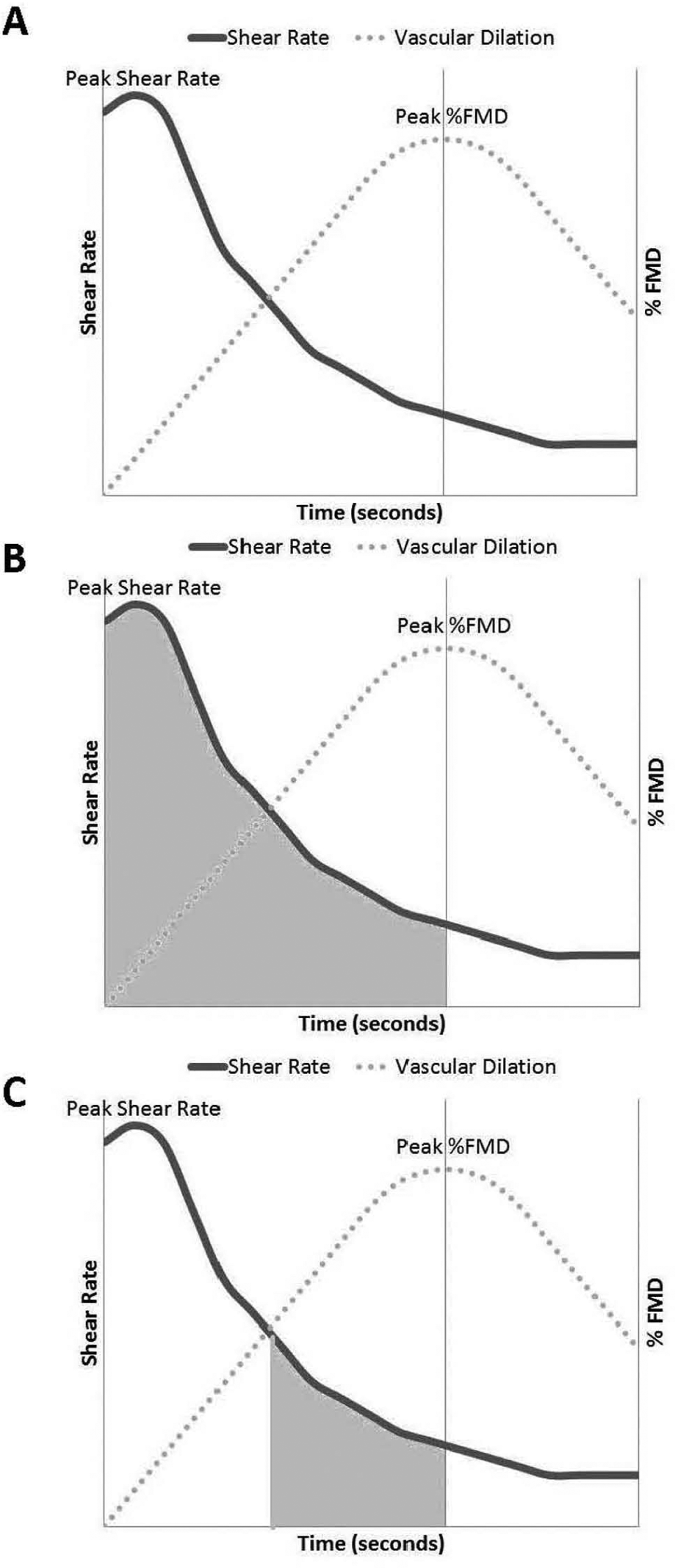
Graphic representation of all three flow-mediated dilation (FMD) normalization strategies. (A) Peak shear rate (SR); (B) area under the SR curve (AUC) until peak dilation; and (C) AUC 30 seconds before peak dilation (AUC30).
Molecular analysis
Cells were rehydrated and non-specific binding sites were blocked with 5% donkey serum. Cells were incubated with monoclonal antibodies for eNOS (from Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) and nitrotyrosine (NT) (Millipore, Billerica, MA, USA). Cells were then incubated with an Alexafluor 555 fluorescent secondary antibody (Invitrogen Corp., Carlsbad, CA, USA).
For analysis, slides were viewed with a fluorescence confocal microscope system (VT-Infinity3 2D Array Laser Confocal system; VisiTech Sunderland, UK and the Olympus BX51WI; Melville, NY, USA) and cell images were captured digitally by an ImagEM electron multiplier CCD camera (Hamamatsu, Japan). Endothelial cells were identified by staining for von Willebrand factor (Dako, Glostrop, Denmark and Jackson ImmunoResearch Labs, West Grove, PA, USA) and nuclear integrity was confirmed with DAPI (4’,6-diamidino-2-phenylindole hydrochloride) (Invitrogen Corp., Grand Island, NY, USA). Once endothelial cells with intact nuclei are identified, they were analyzed with ImageJ software (National Institutes of Health, USA). Five to 10 consecutive cells, starting from the top-left corner of each slide, were analyzed from each slide. Values for each protein were reported as a ratio of endothelial cell to human umbilical vein endothelial cell average pixel intensity. Although this analysis may have some caveats, especially when comparing cultured cells with biopsied cells, it has been validated in previous studies.27,28
Statistical analysis
Descriptive statistics, including mean, standard deviation (SD), standard error of the mean (SEM), and minimum and maximum values were obtained. Normal distribution for all dependent variables was confirmed using Shapiro–Wilkins and Smirnoff tests (at least one test p>0.05). Linear correlations between eNOS expression and raw and normalized FMDs were performed. The same analysis was performed between NT expression and raw and normalized FMDs. Data are expressed as mean ± SD unless otherwise stated. All statistical analyses were performed using SPSS (version 19.0; IBM, Chicago, IL, USA), and statistical significance was set at p<0.05.
Results
Normality analyses of the dependent variables showed that all data were normally distributed.
Table 1 shows the general characteristics of the study subjects. Age, height, weight, body max index, resting systolic and diastolic blood pressures, and VO2max are for young, healthy, non-active individuals.
Group characteristics.
Pearson’s correlation coefficients and statistical significance are shown in Table 2. There was no significant correlation between eNOS expression and FMD. FMD normalized by AUC30 and NT expression was the only significant association observed.
Pearson’s correlation coefficients and statistical significance between endothelial cellular markers and FMD.
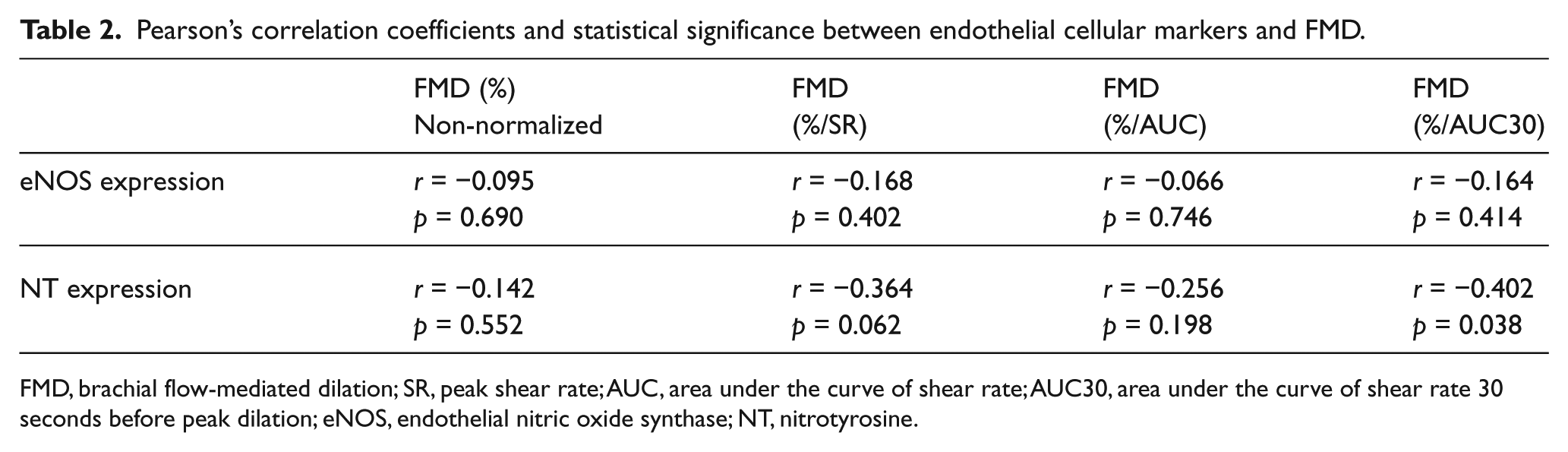
FMD, brachial flow-mediated dilation; SR, peak shear rate; AUC, area under the curve of shear rate; AUC30, area under the curve of shear rate 30 seconds before peak dilation; eNOS, endothelial nitric oxide synthase; NT, nitrotyrosine.
Figure 2 illustrates the association between NT expression and normalized FMD (i.e. %/AUC30) and representative pictures of low and high expression of NT.
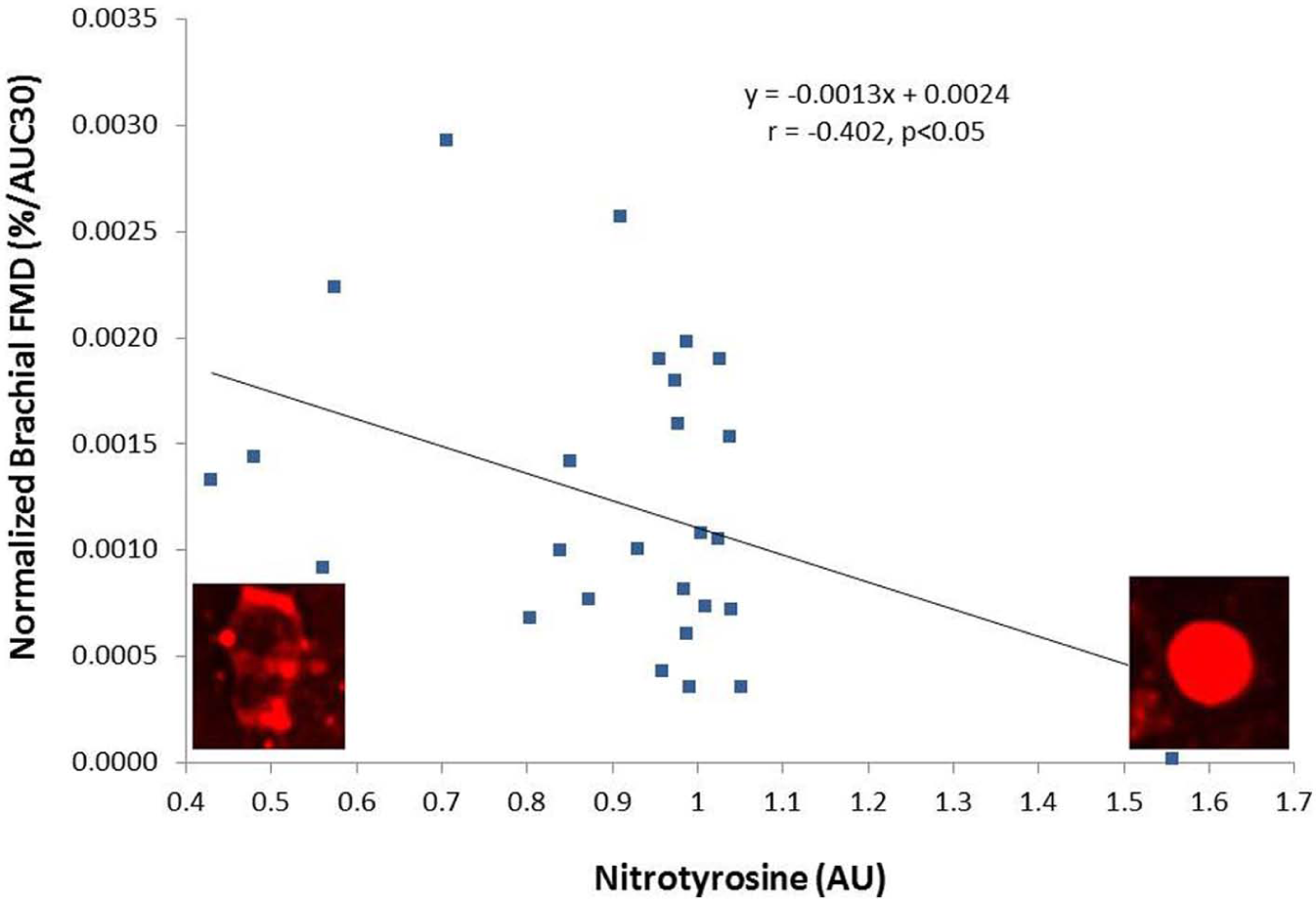
Association between nitrotyrosine, a marker of endothelial cell oxidative stress, and normalized brachial flow-mediated dilation (%FMD/AUC30).
Discussion
The principal finding of the present study was that there is an inverse relationship between brachial artery FMD and NT content in venous endothelial cells, supporting the hypothesis that FMD is associated with endothelial cell oxidative stress.
Although FMD is a bioassay for endothelial-dependent vasodilation, the relationship between endothelial oxidative stress and FMD is not well established. Several studies have reported inconsistent results between overall oxidative stress and FMD.39–41 For example, Martino et al. 41 observed that hypercholesterolemic children had attenuated brachial FMD and increased urinary isoprostanes, a marker of overall oxidative stress, when compared to normo-cholesterolemic children. In addition, the authors observed a significant inversed correlation between FMD and urinary isoprostanes (r = −0.388, p<0.005), but only in the hypercholesterolemic children and no association was observed in the normo-cholesterolemic group. 41 Tycinska et al., 39 using a 3-month crossover design to see the effects of atorvastatin in blood pressure and FMD, observed that changes in FMD were associated with changes in blood pressure. However, no relationship between FMD and serum nitric oxide, serum total antioxidant status, serum endothelin-1, and plasma peroxides measurements were observed. 39 Finally, Johnson et al. 40 studied the effects of different doses of exercise on FMD and plasma thiobarbituric acid reactive substances (TBARS), a marker for overall oxidative stress. Although the findings showed an increased TBARS on both high-intensity exercise modalities, one exercise modality showed a decreased FMD and the other exercise modality showed an increased FMD. In aggregate, results from the studies cited above are inconsistent and do not confirm the possible relationship between oxidative stress and FMD. We speculate that the incongruent outcomes in prior studies can be attributed to the fact that none of them used a specific marker for endothelial cell oxidative stress.
In contrast, Donato et al. 42 observed a significant inverse relationship between FMD and arterial endothelial cell nitrotyrosine expression (r = −0.62, p<0.01) in a subset of 11 healthy, young and 11 healthy, older subjects. Although the present study harvested venous endothelial cells, our findings are similar to Donato et al.’s study. The main difference between both studies is that Donato et al. used non-normalized FMD 42 and the significant association in the present study was observed only when FMD was normalized (%/AUC30) (Table 2). The lack of association between NT expression and non-normalized FMD in the present study, compared to Donato et al.’s study, 42 may be related to cell origin (i.e. venous vs arterial) and sample demographic differences (e.g. average age, sample size, and level of activity). Moreover, it would be interesting to see if Donato et al.’s study correlation between NT and FMD increases if FMD is normalized (e.g. %/AUC).
In general, there is a consensus about the needs for FMD normalization and the general recommendation is that normalization of FMD should be performed.14,15,20,22–25,32 Indeed, an international expert panel has recently confirmed that normalizing FMD with the shear stress stimulus is reasonable in order to keep FMD assessment clinically relevant. 43 A lingering problem, however, is that there is presently no consensus about how to normalize FMD. Pyke and Tschakovsky, experts in the field, have extensively studied different ways to normalize FMD. Peak shear rate and AUC are two recommended normalization schemes.22–25 In the present study, neither FMD normalized by peak shear stress nor FMD normalized by AUC of shear rate were associated with endothelial oxidative stress (Table 2). However, FMD normalization with AUC30 was significantly associated with endothelial oxidative stress.
Fluid biomechanics is an important factor in endothelial physiology.37,44 Thus, the blood-flow pattern produced by FMD’s reactive hyperemia should play a key role in vascular reactivity. At peak shear rate, blood flow is primarily antegrade and turbulent due to the pulsatile flow and increased flow velocity. 45 Then, blood flow decreases its velocity and flow transitions predominately to antegrade and laminar with an increased shear stress until around peak dilation, where shear stress declines due to a larger vessel diameter. 37 At peak dilation brachial blood flow starts showing a bi-phasic flow pattern, with some retrograde laminar flow, as observed during resting measurements. Based on Womersley’s approximation for pulsatile flows and an estimated hematocrit of 50%,35,36 in the present study the blood-flow pattern at peak shear rate was confirmed as primarily antegrade and turbulent (data not shown). Normalizing FMD using AUC30 before peak dilation would include only antegrade laminar blood flow, which is known to guarantee endothelial activation. In contrast, normalizing FMD using AUC at peak shear rate and peak shear rate will include some antegrade turbulent blood flow, which may inhibit endothelial function.37,38 Additionally, normalization using AUC30 better represents the half-life of NO (Figure 1C). 34
The present study was not without limitations. First, endothelial cell biopsies in the present study were performed on an arm vein versus an arm artery. However, Donato et al. 42 have shown no significant differences between venous and arterial NT expression in either young or older healthy individuals. Harvesting venous endothelial cells is attractive in that it is technically less demanding and confers significantly less patient risk and patient burden. Second, the lack of association observed in the present study with eNOS expression may be related to the fact that we did not study phosphorylated eNOS, which has been shown to be a more sensitive marker of NO production.37,46
In summary, FMD is associated with venous endothelial oxidative stress when it is normalized by an AUC of shear rate 30 seconds before peak dilation. Although shear stimulus should be the primary factor to normalize FMD, endothelial activation also depends on blood-flow patterns. Future FMD normalization should include individualized blood-flow patterns to determine when the shear stimulus is exclusively antegrade and laminar.
Footnotes
Acknowledgements
The authors would like to acknowledge Ms Elizabeth Zennah and Mrs Barbara Gómez-Munizaga for their technical help collecting and isolating endothelial cells.
Declaration of conflicting interest
The authors report no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
