Abstract
Introduction:
Exercise training improves walking capacity in patients with intermittent claudication (IC). Endothelial progenitor cells (EPCs), endothelial microparticles (EMPs), and endothelial dysfunction could play a role in this process.
Methods:
We measured EPCs and EMPs in a group of 60 patients with IC, and in a control group of 20 individuals without IC, before a treadmill test and 2, 24, and 48 hours after the test. Thirty patients with IC were randomly assigned to perform a 12-week home-based exercise training program. The EPC count, flow-mediated dilation (FMD) of the brachial artery, pain-free walking time (PFWT), and maximum walking time (MWT) were measured at the baseline and after the exercise training program.
Results:
In patients with IC, EMPs significantly increased 2 hours after the treadmill test, whereas EPCs significantly increased after 24 hours. Among the subjects assigned to complete the training program, we observed a significant increase in the number of EPCs after 12 weeks, as well as an improvement in FMD, PFWT, and MWT. A significant correlation between the variation of EPCs, FMD, and MWT was found. The increase of EPCs and FMD were independent determinants of the walking capacity improvement, without significant interaction.
Conclusion:
Our results suggest that EPCs mobilization contributes to the improvement of walking capacity in patients with IC undergoing structured physical training. A number of different, partly independent, mechanisms are involved in this process, and our results highlight the potential role of EMPs release and endothelial function improvement.
Keywords
Introduction
Intermittent claudication (IC), defined as a reproducible ischemic leg pain on walking, is the most common symptom of peripheral artery disease (PAD) of the lower limbs. Physical activity is recommended to improve walking capacity in patients with IC. 1 The efficacy of home-based physical activity in improving the walking capacity of patients with IC is well established. Although the efficacy of supervised exercise was demonstrated to be superior to home-based exercise, 2 the latter could optimize compliance of patients because of easier availability. 3
Several pathophysiological mechanisms have been proposed to explain the beneficial effect of physical exercise in patients with PAD, including an improvement in endothelial function, skeletal-muscle metabolism, blood viscosity, inflammatory status, and biomechanics of walking. 4
Endothelial dysfunction, defined as a reduced capacity of vasculature to dilate in response to an ischemic stress, has a relevant role in the pathogenesis of PAD and its cardiovascular comorbidities.5,6 It is the result of multiple factors associated with atherosclerosis, including reduced bioavailability of nitric oxide, oxidative stress, and inflammation.7,8
Endothelial progenitor cells (EPCs) are immature, bone marrow-derived cells, which contribute to angiogenesis and vascular repair. Their circulating number inversely correlates with cardiovascular risk and positively correlates with endothelial function. 9
Endothelial microparticles (EMPs) are small cellular fragments released by the endothelium in response to different stimuli. 10 Accumulating evidence has associated an increased number of circulating EMPs to endothelial dysfunction 11 and cardiovascular disease, 12 so they are considered as a biologic signal of endothelial damage. Their pathophysiological role in atherosclerotic cardiovascular disease is still a matter of debate, since different authors describe them as damage, or healing, promoting factors, as well as neutral participants in the process. 13
The effect of exercise training on EPCs and EMPs is not well documented in humans, and particularly in subjects with IC.
The aim of the present study is to investigate whether a 12-week, structured, home-based training program increases the number of circulating EPCs. Secondary endpoints of the study are correlations between the change in circulating EPCs and variations of EMPs, endothelial function, and walking capacity.
Methods
Study design
This study is composed of two phases:
Observational controlled phase involving patients with IC and matched controls without IC;
Interventional, randomized, unblinded phase involving patients with IC assigned to structured, home-based, exercise training versus behavioral advice.
Patients
From October 1, 2018 to September 30, 2019, patients with a history of IC, consecutively referred to a vascular ultrasound facility, were enrolled in the study. Definition, diagnostic work-up, and medical treatment of PAD were in accordance with the 2017 ESC/ESVS Guidelines on the Diagnosis and Treatment of Peripheral Arterial Diseases. 1 The diagnosis was confirmed by color-Doppler ultrasonography, showing a resting ankle–brachial index (ABI) < 0.9 and the presence of significant stenosis or occlusion. Significant stenosis was defined as an intra-stenosis peak systolic velocity (PSV) > 200 cm/s, a PSV ratio across the stenosis > 2, or distal demodulation at pulsed Doppler waveform analysis. 14
Subjects with no history of IC and ABI ⩾ 0.9, balanced for age, sex, smoking habit, body mass index (BMI), systolic blood pressure, and lipid profile were recruited at the outpatient clinic of the same institution and served as controls.
Exclusion criteria were critical limb ischemia (Fontaine stages III and IV), inability to perform a walking test for conditions other than claudication, chronic inflammatory conditions, history of recent (< 6 months) acute coronary syndrome, or cerebrovascular event and lower limb revascularization during the training. Patients who modified their pharmacological treatment during the trial, or in the 30 days prior to enrollment, were also excluded from the study. The flow-chart diagram of enrollment is depicted in online Supplementary Figure 1.
Both patients and controls performed a standard treadmill test according to the protocol described by Strandness, 15 using a motorized treadmill with a constant speed of 2 km/h and a fixed slope of 12°, for 5 minutes. Pain-free walking time (PFWT) was defined as the walking time before the occurrence of claudication, and maximum walking time (MWT) was defined as the time until walking arrest due to unbearable limb pain.
Blood samples were collected in the morning, after an 8-hour fasting period, and at least 48 hours after the last physical activity session (for patients enrolled in the training group). EMPs and EPCs were quantified before the treadmill test and 2, 24, and 48 hours after the test. Thirty subjects out of the IC group were randomly assigned to a 12-week ischemic exercise training program. All patients were prescribed an appropriate pharmacological treatment, as recommended by guidelines, if not contraindicated. Patients in the nontraining group were advised to perform regular aerobic activity, but did not enter a specific training program. ABI, PFWT, MWT, flow-mediated dilation (FMD), and EPCs were measured at baseline and after the 12-week intervention.
All procedures were conducted in accordance with the 1964 Helsinki Declaration and its later amendments. The study was approved by the Ethics Committee of University of Perugia (Protocol number: 2019-36). All subjects signed an informed consent form to voluntarily participate in the study. The study is registered at ClinicalTrials.gov (NCT04302571). The study was registered after completion of the recruitment of patients.
Measurements and definitions
Age, sex, and smoking habits were recorded. Major atherosclerotic cardiovascular events (MACE) were defined as a myocardial infarction, myocardial revascularization or ischemic stroke that occurred at least 6 months prior to enrollment. Diabetes mellitus was defined according to the 2018 American Diabetes Association (ADA) Guidelines. 16
Weight and height were measured, and BMI was calculated. Brachial blood pressure was measured in a quiet place after appropriate resting. Total cholesterol, triglycerides, high-density lipoprotein (HDL) cholesterol, and glucose were determined by the enzymatic-colorimetric method (Dimension Autoanalyzer; DADE Inc., Newark, NJ, USA); low-density lipoprotein (LDL) cholesterol was calculated using the Friedewald equation.
Assay of circulating endothelial progenitor cells (EPCs) and endothelial microparticles (EMPs)
Circulating EPCs and EMPs were counted as previously described.12,17 Mononuclear cells were isolated from peripheral venous blood by density centrifugation. Freshly isolated mononuclear cells were incubated with PE-conjugated antibodies against human KDR (R&D Systems, Minneapolis, MN, USA) and FITC-conjugated antibodies against human CD34 (Beckman Coulter, Inc., Fullerton, CA, USA). Mouse isotype-identical antibodies at a fixed concentration served as controls (Beckman Coulter). EPCs were defined as CD34+/KDR+ cells in the gate of viable lymphocytes, after exclusion of nonviable mononuclear cells by staining with propidium iodide (online Supplementary Figures 2 and 3).
EMPs were assayed immediately after venipuncture. Specimens were centrifuged to obtain a platelet-poor plasma, and later incubated with anti-CD31-PE (Beckman Coulter, Inc.) plus anti-CD42-FITC (Beckman Coulter). EMPs were defined as CD31+/CD42– particles with a diameter < 1.5 µm (Molecular Probes; Invitrogen, Eugene, OR, USA) (online Supplementary Figures 4 and 5). Quantitative analysis was performed on a Coulter Epics XL (Beckman Coulter).
Endothelial function assessment
Endothelial function was assessed as FMD of the brachial artery by ECG-gated ultrasonography,
18
performed by a trained physician, blinded to the allocation of subjects. The measurement was taken on the nondominant arm in a supine position, after 10 minutes rest. The brachial artery was scanned above the antecubital crease with a linear 5–12 MHz linear probe (MyLab 50; Esaote, Genoa, Italy). The diameter of the brachial artery was measured as the distance between media–adventitia interface at the anterior and posterior wall. Hyperemia was induced by inflation of a pneumatic cuff at 250 mmHg for 4 minutes on the proximal portion of the forearm. The measurement was taken before cuff inflation and 60 seconds after rapid deflation. The average of three measurements, repeated with a time lapse of 5 minutes between each other, was used for the analysis. FMD was expressed as
Exercise training
Graduate technicians, with a Physical Exercise and Sports degree, instructed subjects randomized to the training program to perform physical activity at home. According to the American Heart Association guidelines, 19 five weekly training sessions were scheduled over a 12-week period. Each session lasted 60 minutes, divided into a 45-minute aerobic workout and 15 minutes of circuit training. The aerobic workout consisted of walking on a flat floor or slight incline (maximum slope 3%): patients were advised to walk until they reached submaximal walking capacity (80% of their MWT), and rest when they experienced pain in the lower limbs; they were then advised to resume walking once the pain had improved. Resistance training consisted of calisthenic exercises focused on the trunk and lower limb muscles (bicycle, flutter kicks, squats, lunges, calf raises, wall sits, and power press) with at least three repetitions for each exercise, interspersed by 30 seconds’ recovery. The duration of repetitions increased every 3 weeks.
Statistical analysis
Data are expressed as mean ± SD or as median (IQR) for normally and non-normally distributed variables, respectively. Categorical variables are expressed as number (%). Statistical significance was accepted at a 0.05 level of probability. Based on the results of our pilot study, we expected an increase of MWT ⩾ 50 s with a SD = 48 s; for a statistical power of 0.95, we estimated a sample size ⩾ 23 subjects for each group, for a total number ⩾ 46 participants. Data were analyzed by SPSS statistical software, version 17.0 (SPSS Inc., Chicago, IL, USA). Normality of variables distribution was tested using the Kolmogorov–Smirnov nonparametric test. Missing data accounted for < 5% for any given parameter and were excluded from analyses. Comparisons between groups were performed through the two-tailed Student’s t-test or the Mann–Withney U-test for normally and non-normally distributed variables, respectively. Variations were analyzed by paired two-tailed Student’s t-test or the Wilcoxon test for normally and non-normally distributed variables, respectively. Bonferroni correction for multiple comparisons was employed. The distribution of frequencies was analyzed through Fisher’s exact test. Correlation coefficients between variables were calculated through the Pearson or Spearman correlation analysis, as appropriate. Independent predictors were identified through a multiple regression analysis model. Non-normally distributed variables were log-transformed before performing linear regression analysis.
Results
Seventy-two patients were screened for inclusion and exclusion criteria. Following the exclusion of 12 patients, 60 patients with IC, and 20 control subjects, were enrolled (online Supplementary Figure 1) in the study. Clinical and biochemical characteristics of the enrolled subjects are summarized in Table 1. No significant difference was observed between patients and controls in clinical and biochemical parameters except for the history of previous MACE, which were more prevalent in the control group. Patients had higher levels of circulating EMPs, whereas no significant difference was detected in the values of circulating EPCs.
Clinical and biochemical characteristics of the subjects.
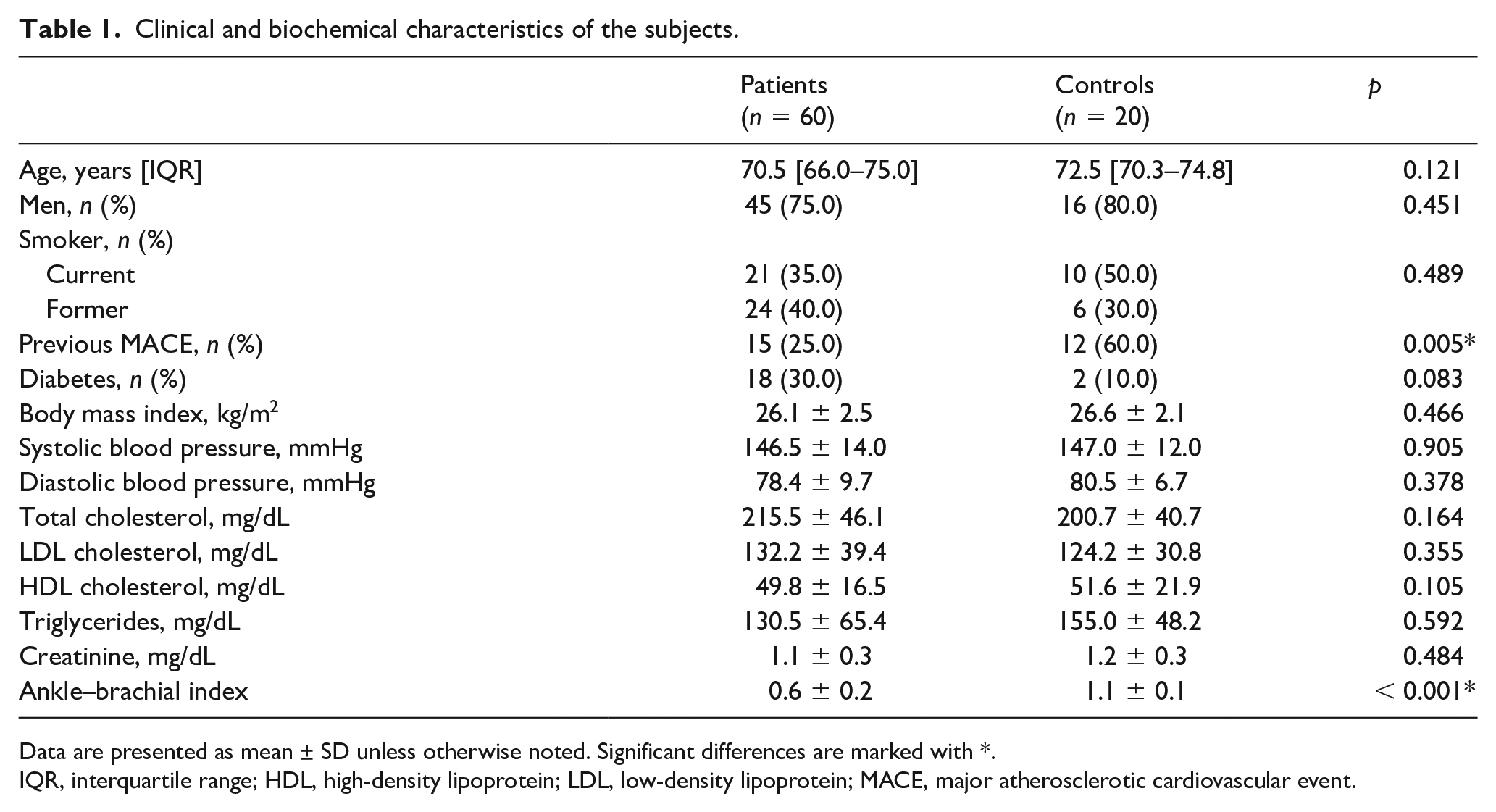
Data are presented as mean ± SD unless otherwise noted. Significant differences are marked with *.
IQR, interquartile range; HDL, high-density lipoprotein; LDL, low-density lipoprotein; MACE, major atherosclerotic cardiovascular event.
All subjects enrolled in the control group completed the treadmill test, confirming that they did not have a symptomatic PAD. Conversely, patients with IC had a median PFWT of 98.5 s (IQR 85.0–110.0 s) corresponding to a pain-free walking distance of 55.2 m (IQR 47.6–61.6 m), and a MWT of 181 s (IQR 163.5–204.5 s) corresponding to a maximum walking distance of 101.4 m (IQR 91.6–114.5 m). These values correspond to a moderate/severe impairment of walking capacity (Lériche–Fontaine stage IIb).
As shown in Figure 1, after the treadmill test, the number of both circulating EMPs and circulating EPCs significantly increased in patients, with EMPs reaching a peak after 2 hours and EPCs reaching a peak after 24 hours. Forty-eight hours after the test, the number of EPCs returned to baseline levels, while the number of EMPs was still higher than the basal values and persistently higher than the control group. Significant differences between time points were preserved after correction for multiple comparisons (overall adjusted p < 0.001 for both EPCs and EMPs).
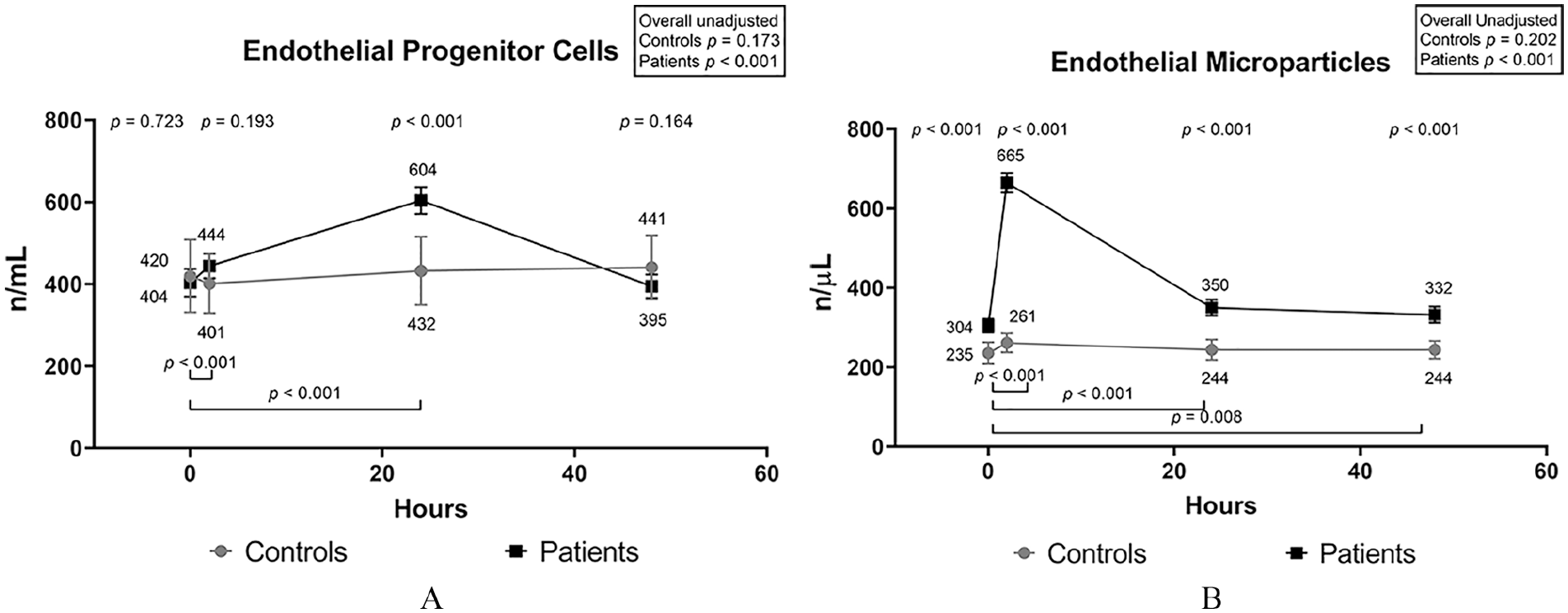
Time-plotted average values of circulating endothelial microparticles (
In the whole group of patients and controls, the change between the baseline and the peak value of EPCs was positively correlated with the change of EMPs (ρ = 0.675, p < 0.001). The correlation was lost when considering the group of patients only (ρ = 0.208, p = 0.111). The peak changes of both EPCs and EMPs were inversely correlated with PFWT (Figure 2).
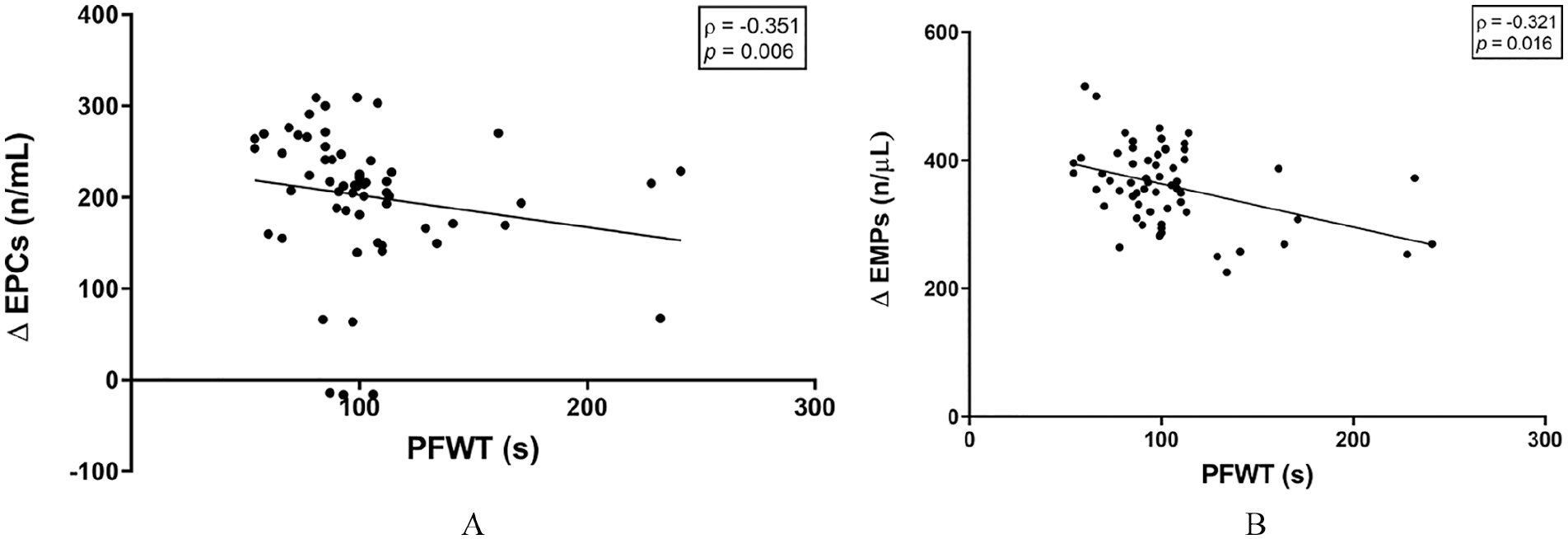
Correlation of pain-free walking time with the change in endothelial microparticles and endothelial progenitor cells from baseline to peak value, after a treadmill test: (
The exercise training was well tolerated. No subject reported injuries during the training sessions, or dropped out of the program. No significant difference was detected between patients in the training and nontraining groups, in terms of the EPCs count, FMD, and walking times at baseline (online Supplementary Table 1).
The 12-week ischemic exercise training program induced a significant increase in EPCs count, FMD, PFWT, and MWT in patients who performed exercise training (online Supplementary Table 2). The average increase in EPCs, FMD, PFWT, and MWT was significantly superior in the training group than in the nontraining group (Figure 3).
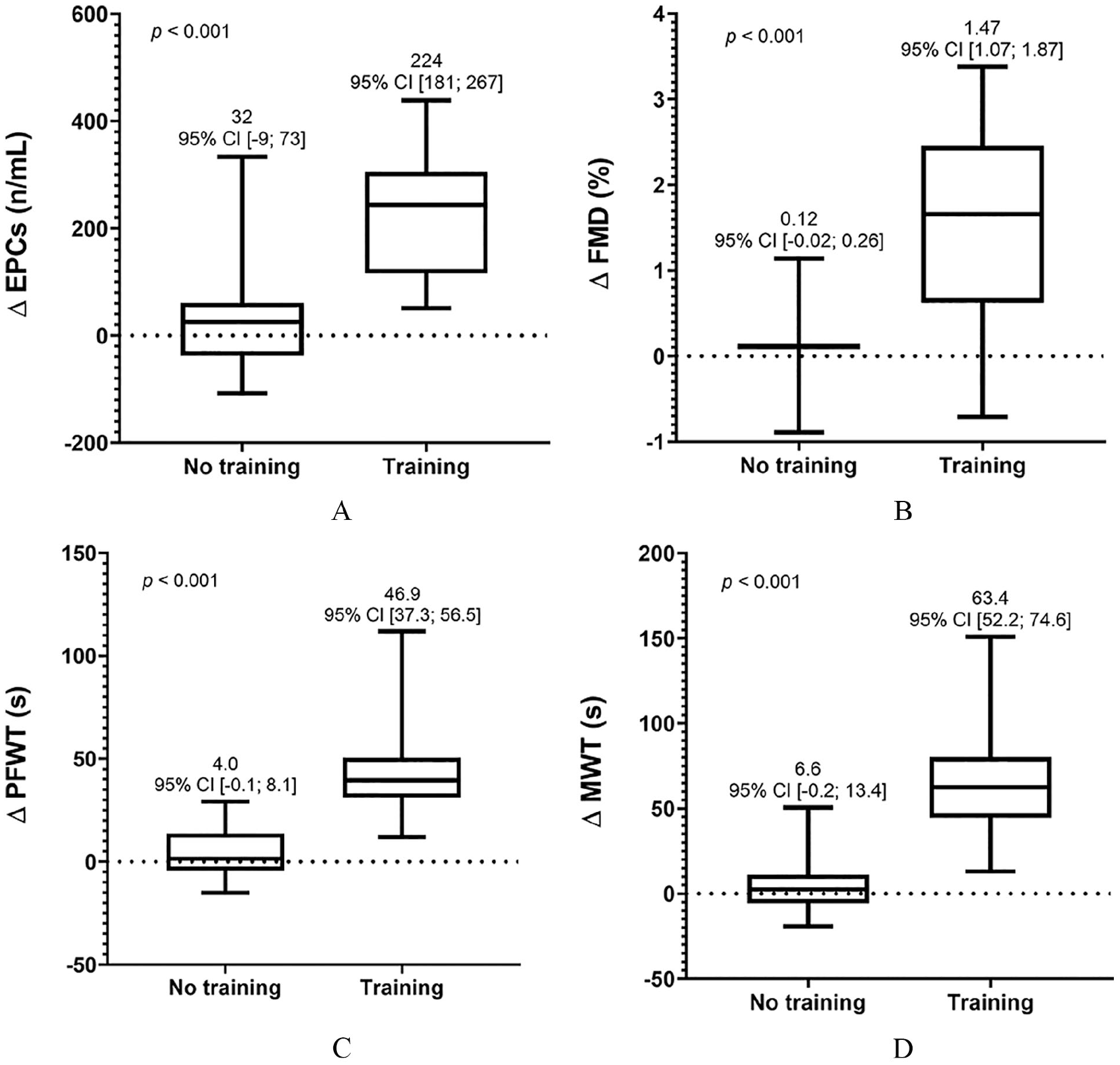
Comparison of change in (
A significant positive correlation was found between the change in EPCs and the change of FMD, MWT, and PFWT in the training group (Table 2). The change of FMD was also significantly correlated with both MWT and PFWT (r = 0.506, p = 0.004 and ρ = 0.567, p = 0.001).
Correlations of change in endothelial progenitor cells after 12 weeks in the training and nontraining groups.
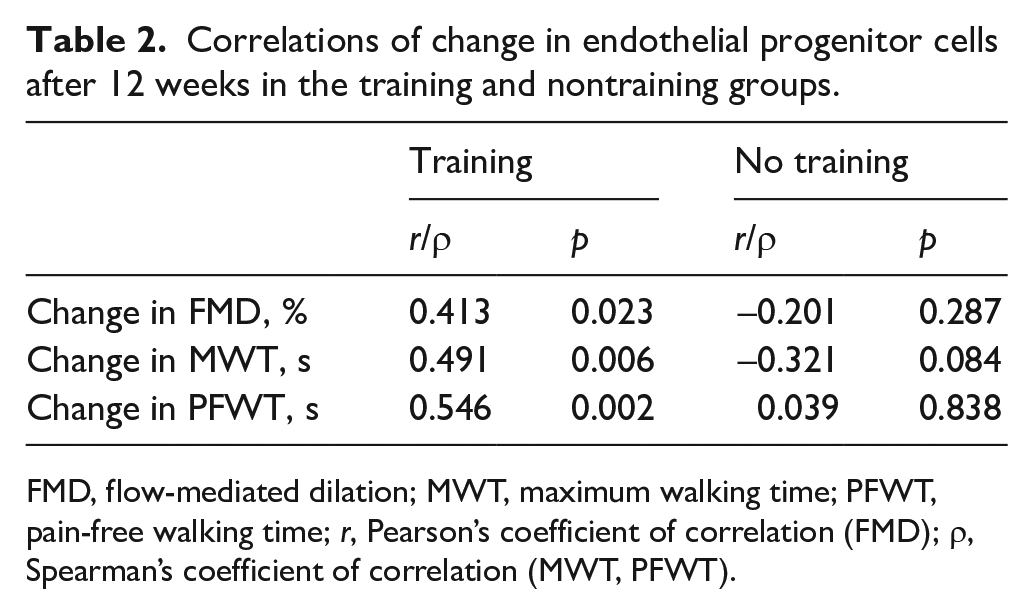
FMD, flow-mediated dilation; MWT, maximum walking time; PFWT, pain-free walking time; r, Pearson’s coefficient of correlation (FMD); ρ, Spearman’s coefficient of correlation (MWT, PFWT).
Performing multiple regression analysis model including the change of EPCs and FMD, EPCs were independent determinants of the increase in PFWT, but not of MWT, without significant interaction (Figure 4).
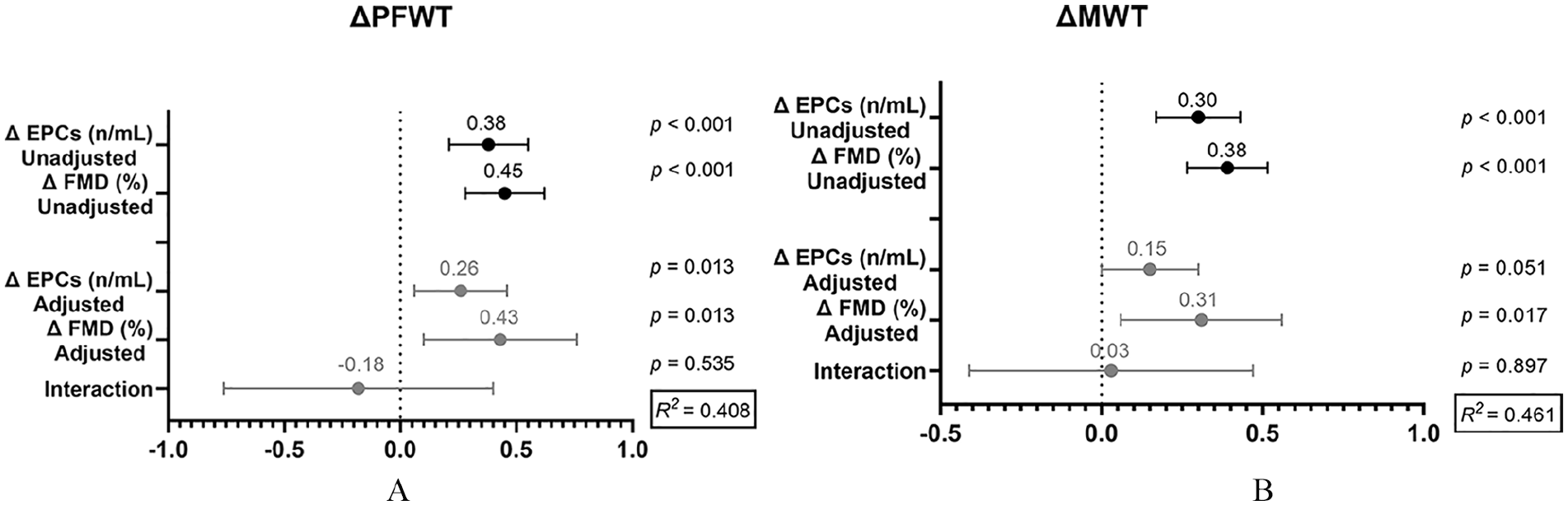
Multiple linear regression models for changes in (
Discussion
The results show that a 12-week structured home-based training program can induce a significant increase in the number of circulating EPCs, associated with an improvement of brachial FMD and walking capacity, in patients with IC.
Furthermore, we observed that a single-effort ischemic exercise is associated with an increase in circulating EMPs, followed by an increase in EPCs. Interestingly, the increase of EMPs and EPCs was inversely correlated with the PFWT, suggesting that the exercise-induced ischemia could be the triggering factor for the endothelium activation. This hypothesis is in line with recent evidence, pointing out the role of hypoxia as a possible triggering factor for EMPs release. 20
Moreover, these variations correlate with each other, suggesting that an activation of endothelium occurs soon after physical effort, subsequently leading to a recruitment of EPCs from the bone marrow.
There is increasing evidence that EMPs could take an active part in cell-to-cell communication 21 and vascular repair. 22 Little is known about the role of microparticles in PAD pathophysiology, except that EMPs are increased in patients with PAD. 23 Previous studies in healthy individuals reported no change, or even a reduction, of circulating EMPs after a single moderate to high-intensity physical effort,24,25 whereas physical inactivity is associated with their increase. 26 In patients with coronary artery disease, both no change or an increase has been described.21,27 To the best of our knowledge, no previous study has explored the effects of acute physical activity on EMPs in PAD.
Several authors proposed that physical activity is an inductor of EPCs mobilization from the bone marrow. 28 Multiple mechanisms have been proposed to explain this effect, including the effect of endothelial nitroxide synthase and hypoxia-inducible factor 1 on bone marrow,29,30 inhibition of pro-inflammatory and myelosuppressive cytokines, 31 and up-regulation of β2-adrenergic receptors. 32 Sandri et al. 33 observed an increase in EPCs count in patients with PAD after a 4-week exercise training program. However, no previous study has reported a significant association between EPCs mobilization and the improvement of walking capacity in patients with PAD.
According to our results, we can hypothesize that the mobilization of EPCs might contribute to the improvement in walking capacity in IC patients undergoing structured physical training. EPCs could contribute to the improvement of walking capacity through promoting neo-angiogenesis, 34 or restoring proper function in damaged endothelium. 35
In a murine model, endothelial-derived microparticles are able to increase the capacity of EPCs to regenerate the endothelial layer and to restore the endothelial production of NO. 36 Our results are consistent with this experimental evidence, and support the hypothesis of an interaction between EMPs and EPCs, to restore the proper endothelial function.
In this regard, a significant correlation between the increase in EPCs count and the FMD was observed in the training group, supporting a possible relationship between EPCs mobilization and FMD improvement in patients with IC, undergoing structured physical training. However, the multiple regression analysis did not show any significant interaction between the change in EPCs and FMD. Interestingly, after correction for the increase in EPCs count, the improvement of FMD was the only significant determinant of MWT improvement. It is still debated whether endothelial function has a role in mediating the improvement of walking capacity after physical training.37,38
According to our results, the improvement of FMD could have a relevant role, and would also be independent from EPCs mobilization. Notably, we measured FMD in the brachial artery, which usually shows a lower grade of atherosclerotic disease than lower limb arteries. Thus, we cannot exclude a possible different regional effect of EPCs mobilization in arteries with more severe atherosclerotic disease.
The home-based approach to physical activity prevented us from exerting strict control over patient compliance to exercise training; however, the improvement in walking capacity was consistent with previous studies 2 and this suggests that adherence to the training program was adequate. Furthermore, the small sample size could have prevented us from detecting significant correlations and performing subgroup analyses. In particular, the correlation between the variation of EMPs and EPCs after the acute ischemic physical activity was observed only in the general group of enrolled subjects; however, the lack of significance in the patient group can be attributed to a sample size effect, since the relative weight of the control group is negligible. The sample size is small for the female sex, accounting for 25% of patients and 20% of controls. Other authors have proposed that females with PAD are less likely than males to seek medical advice and tend to receive an appropriate diagnosis when overt critical limb ischemia occurs. 39 In our study, the presence of critical limb ischemia is an exclusion criterion and this could explain the lower prevalence of females in the study population.
Finally, no experimental evidence that the CD34+/KDR+ cells could actually maturate into endothelial cells is provided; however, the association between CD34+/KDR+ cells and severity of PAD has been previously described. 40 The described protocol has been employed in previous research, producing consistent results.12,41
Conclusion
Our results suggest that EPCs mobilization contributes to the improvement of walking capacity in patients with IC undergoing structured physical training. A number of different, partly independent, mechanisms are involved in this process, and our results highlight the potential role of EMPs release and endothelial function improvement. Considering the above-mentioned limitations, and the lack of previous studies on this topic, we cannot state any hypothesis on the relationship between these factors. Therefore, further studies are needed.
Supplemental Material
sj-docx-1-vmj-10.1177_1358863X211020822 – Supplemental material for Effects of structured home-based exercise training on circulating endothelial progenitor cells and endothelial function in patients with intermittent claudication
Supplemental material, sj-docx-1-vmj-10.1177_1358863X211020822 for Effects of structured home-based exercise training on circulating endothelial progenitor cells and endothelial function in patients with intermittent claudication by Leonella Pasqualini, Francesco Bagaglia, Stefano Ministrini, Maria Rosaria Frangione, Christian Leli, Donatella Siepi, Rita Lombardini, Ettore Marini, Meisam Naeimi Kararoudi, Alexander Piratinskiy and Matteo Pirro in Vascular Medicine
Supplemental Material
sj-docx-2-vmj-10.1177_1358863X211020822 – Supplemental material for Effects of structured home-based exercise training on circulating endothelial progenitor cells and endothelial function in patients with intermittent claudication
Supplemental material, sj-docx-2-vmj-10.1177_1358863X211020822 for Effects of structured home-based exercise training on circulating endothelial progenitor cells and endothelial function in patients with intermittent claudication by Leonella Pasqualini, Francesco Bagaglia, Stefano Ministrini, Maria Rosaria Frangione, Christian Leli, Donatella Siepi, Rita Lombardini, Ettore Marini, Meisam Naeimi Kararoudi, Alexander Piratinskiy and Matteo Pirro in Vascular Medicine
Footnotes
Acknowledgements
Miss Helen Walsh, Freelance English Language Editor (Lancashire, UK), is thanked for her editorial assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by ‘Fondo per la Ricerca di Base 2018’ of Università degli Studi di Perugia. The funder did not have any role in the design/conduct of the study.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
