Abstract
The role of endothelial progenitor cells (EPCs) in peripheral artery disease (PAD) remains unclear. We hypothesized that EPC mobilization and function play a central role in the development of endothelial dysfunction and directly influence the degree of atherosclerotic burden in peripheral artery vessels. The number of circulating EPCs, defined as CD34+/KDR+ cells, were assessed by flow cytometry in 91 subjects classified according to a predefined sample size of 31 non-diabetic PAD patients, 30 diabetic PAD patients, and 30 healthy volunteers. Both PAD groups had undergone endovascular treatment in the past. As a functional parameter, EPC colony-forming units were determined ex vivo. Apart from a broad laboratory analysis, a series of clinical measures using the ankle–brachial index (ABI), flow-mediated dilatation (FMD) and carotid intima–media thickness (cIMT) were investigated. A significant reduction of EPC counts and proliferation indices in both PAD groups compared to healthy subjects were observed. Low EPC number and pathological findings in the clinical assessment were strongly correlated to the group allocation. Multivariate statistical analysis revealed these findings to be independent predictors of disease appearance. Linear regression analysis showed the ABI to be a predictor of circulating EPC number (p=0.02). Moreover, the functionality of EPCs was correlated by linear regression (p=0.017) to cIMT. The influence of diabetes mellitus on EPCs in our study has to be considered marginal in already disease-affected patients. This study demonstrated that EPCs could predict the prevalence and severity of symptomatic PAD, with ABI as the determinant of the state of EPC populations in disease-affected groups.
Introduction
Diseases of the vascular system, including cardiac, cerebrovascular and peripheral artery disease are a major health burden and leading causes of death and disability. Cardiovascular risk factors damage the intima vessel layer by promoting premature aging and deterioration of the endothelial function. 1 The endothelial monolayer hosts anti-thrombotic, vasodilatation and anti-inflammatory functions, which, when lost, lead to occlusive artery disease. 2 Bone marrow-derived endothelial progenitor cells (EPCs) play an important role in the homeostasis of the endothelial monolayer and are related to cardiovascular morbidity and mortality. 3 Circulating peripheral EPCs contribute to a regulation and reparation process in damaged blood vessels including angiogenesis and re-endothelialization of denuding lesions in the intima.2,4–6 Until recently, the common understanding of endothelial regeneration believed the turnover of peripheral adjacent endothelial cells to be the leading mechanism. In recent years a growing body of evidence points to EPCs as a crucial promoter of the endogenous endothelial regeneration process. 7 EPCs co-express surface markers of both hematopoietic (CD34) and endothelial cells (KDR), which can be quantified by fluorescent activated flow cytometry. 8 Moreover, the functionality of EPCs can be determined by in vitro cell proliferation assays.
The state of endothelial health may be assessed by measuring the thickness of the vessel wall (carotid intima–media thickness, cIMT), the reacting capacity of the vessel to increased flow (flow-mediated dilatation, FMD), or blood pressure gradients over the peripheral artery system (ankle–brachial index, ABI). In healthy individuals, the ability of EPCs to form colonies correlates with FMD. On the other hand, several diseases, the presence of cardiovascular risk factors, and impaired endothelial function are associated with impaired EPC number and function. 3 The aim of the present study was to define surrogate parameters that determine the severity of the atherosclerotic disease based on clinical and laboratory findings to give a more substantive understanding of pathophysiological mechanisms. We hypothesized that the number and function of circulating EPCs are inversely correlated with the severity of peripheral artery disease (PAD). Furthermore, we assumed a negative impact of concomitant diabetes mellitus (DM) on the progenitor cell population.
Methods
Study subjects
The recruitment of study subjects was arranged by the outpatient clinic of the Division of Clinical and Interven-tional Angiology, Inselspital, University Hospital Berne, Switzerland. A total of 91 patients were consecutively selected according to a predefined sample size of 31 non-diabetic PAD patients, 30 diabetic PAD patients (PAD+DM), and a group of 30 healthy volunteers with an ABI > 0.9 and no history of cardiovascular disease or DM serving as a control group. The PAD and the PAD+DM subjects had angiographically documented severe atherosclerosis and prior endovascular lower extremity revascularization.
Medical history including cardiovascular events and risk factors, current drug treatment (glucose-control medication, anticoagulation, beta-blocker, ACE-/AT-antagonists, statins, diuretics, Ca2+-antagonists, hormones, immunosuppressors), and clinical parameters such as the body mass index (BMI) or the waist-to-hip ratio were assessed during a personal interview and from medical files.
The cantonal ethics committee (Kantonale Ethikkommission Bern) approved the study protocol (Ref.-Nr- KEK-BE 029/06), and all study subjects gave written informed consent before entering the study.
Risk factor and comorbidity definitions
The presence of DM was defined by fasting blood sugar >120 mg/dl, glycosylated hemoglobin >7%, or if the patient was on glucose-control medication. Artery hypertension was defined by systolic blood pressure >140 mmHg and/or diastolic blood pressure >80 mmHg, or if the patient consumed any antihypertensive drug. Hyperlipidemia was defined by a total cholesterol level >193 mg/dl, high-density lipoprotein (HDL) cholesterol <38 mg/dl, triglycerides >177 mg/dl, or if the patient consumed any lipid-lowering drug. According to Doll et al., 9 we classifiedcurrent smokers (> 5 cigarettes a day) or those who stopped <10 years ago into the high-risk group. Non-smokers and those who stopped >10 years ago were classified in the low-risk group.
Coronary heart disease was defined as a history of myocardial infarction, unstable angina, and previous percutaneous or surgical coronary revascularization. Cerebrovascular disease was defined as a history of minor or major stroke and transient ischemic attack.
Exclusion criteria
Exclusion criteria were the presence of renal failure (serum creatinine >1.7 mg/dl), severe liver dysfunction (> two-fold increase of serum AST, ALT), an atherothrombotic event within the last 6 months prior to recruitment (acute coronary syndrome, stroke, transient ischemic attack), missing informed consent, or age <50 or >75 years.
Blood sampling
All blood samples were collected from the fasting subjects (14 h overnight). Besides routine analyses (lipid profile, creatinine, glucose, HbA1c), high-sensitivity C-reactive protein (hs-CRP) and D-dimer were determined by the University Hospital Berne Laboratory using in vitro diagnostic quality kits. In addition, a range of preselected cardiovascular biomarkers (sE-selectin, sP-selectin, MCP-1, PAI-1, sICAM-1, sVCAM-1, sCD40-L, MMP-3, MMP-9, IL-6) was analysed by the F. Hoffmann-La Roche research laboratories.
Fluorescent-activated cell sorting (FACS)
Flow cytometric analysis was applied to determine the number of circulating EPCs according to their surface antigens KDR and CD34, as previously described.6,10 Mononuclear cells (MNC) were isolated from peripheral venous blood samples by density centrifugation and processed for the expression of cell-surface antigens with direct two-color analysis, as described previously. 11
Briefly, the samples were incubated at 4°C with a mouse anti-human KDR antibody (1:20 dilution; Sigma-Aldrich Chemie, Buchs, Switzerland) for 15 minutes. After a single washing step the FITC-labeled secondary antibody (Becton Dickinson, Allschwil, Switzerland) was applied for 15 minutes. Following an additional washing step and subsequent blocking with phosphate-buffered saline (PBS)-1% Top Block-0.1% NaN3, cells were stained for CD34 (15 minutes) using a PE-conjugated mouse anti-human CD34 antibody (Becton Dickinson). All incubations were done at 4°C and in a dark chamber, respectively. The samples were then washed twice for a total of four washing steps, solved in washing buffer, and analysed immediately. A negative control was conducted in parallel for every sample by substitution of the primary antibodies with FITC-labeled and PE-labeled anti-human IgG1 (Becton Dickinson). Cell counts were acquired over a period of 5 minutes (recorded cells per sample: mean ± SD) with an analytical cytometer using an auto sampler device (LSR-II; Becton Dickinson). The number of cells positive for CD34 and KDR (CD34+/KDR+) was assessed in each sample and standardized for a total of 0.5 million events with FlowJo™ Version 7.2.2. (Tree Star, Inc., Ashland, OR, USA). The results given are CD34+/KDR+ per 500,000 cells analysed and corrected for the negative control.10–14
Colony forming units-endothelial progenitor cells (CFU-EPC)
Blood samples of patients were collected into BD Vacutainer® CPT™ Cell Preparation Tubes (Becton Dickinson) and processed according to the manufacturer’s instructions for the isolation of mononuclear cells as reported previously. 10 After purification and lysis of remaining erythrocytes, the samples were solved in endothelial cell basal medium (EBM-2; Clonetics, San Diego, CA, USA) supplemented with EGM-2 MCV SingleQuots (Clonetics) and 5% FCS (Serum Supreme; Lonza Ltd, Basel, Switzerland) for plating in six-well plates coated with human fibronectin (Chemicon, Temecula, CA, USA) at a density of 10 million cells per well. After 48 hours in culture, non-adherent cells were separated and cultured for another 4 days in a 24-well plate (1 million cells/well) with Endocult™ medium (StemCell Technologies Inc., Grenoble, France). Hereon, the number of CFU-EPC in each well was assessed by two blinded investigators in accordance with previously published classifications of EPC colony-forming units.3,15
Flow-mediated dilatation (FMD)
Non-invasive determination of endothelial-dependent vasodilatative function was determined by measurement of the brachial artery FMD by high-resolution ultrasonography (Sequoia 512; Acuson, Mountain View, CA, USA). 10 The dilatation was induced by a temporary increase in shear stress following occlusion distal to the site of measurement, which boosts the endothelial nitrous oxide (NO) production.10,16
To reduce the observer error as a source of variability, the same investigator (LB) performed all the measurements, based on stored ultrasonography pictures.
Carotid intima–media thickness (cIMT)
The thickness of the intima and media layer of the extracranial carotid artery approximately 2 cm proximal to the bifurcation level was noninvasively measured by high-resolution B-mode ultrasound. Three measurements were taken manually in near and far walls (in reference to the ultrasound instrument) of the carotid artery. For subjects who had already manifesting plaque formation, the measurements are never done at the plaque site. The mean cIMT was defined as the mean of all measurements on both common carotid arteries; on average three on each side. Since there are important differences in measurements between different institutions, all the measurements were performed by the same investigator (LB) to minimize variability. 17
Ankle–brachial index (ABI)
Measurements and calculations were performed accord-ing to the recommendations of the American Heart Association. 18 After a 5-minute rest in the supine position with the upper body as flat as possible, the blood pressure cuff was used to measure systolic blood pressure (SBP) in the brachial artery of both arms. The SBP of the tibial arteries of the legs was determined with a Doppler ultrasound device and a blood pressure cuff, which was inflated over the level of SBP. The measurements were taken on deflation. During the study, only raw blood pressure values were recorded. Statisticians calculated the ABI values post hoc. The ABI for each leg equals the ratio of the higher of the two systolic pressures (anterior tibial and posterior tibial) to the higher reading of the right and left brachial artery pressures.19–22
Statistical analysis
We present data as mean ± standard deviation. For categorical variables, we report p-values derived from the chi-squared test and for continuous variables, p-values are reported from the Kruskal-Wallis test. Univariate correlations were calculated using the Pearson correlation coefficient when data were normally distributed. Multivariable linear regression models were fitted to identify independent variables associated with the mean of EPC, CFU-EP, cIMT and FMD changes in study subjects. Results were considered statistically significant when p < 0.05. The analysis was performed using Stata (version 11) for Windows.
Results
Baseline characteristics of study subjects
We enrolled a total of 91 study subjects, of which there were 31 patients with manifest PAD, 30 patients with additional DM, and 30 healthy volunteers.
Table 1 summarizes the clinical conditions and laboratory findings of all study subjects. The healthy subjects differ significantly with regard to most baseline characteristics as compared to both disease-affected groups. As expected, clinical assessment was normal in healthy subjects, with a definite lower cardiovascular risk profile. An important finding is the high prevalence of incidental hyperlipidemia in healthy subjects, which was not treated by any lipid-lowering agent. In the disease-affected groups, 20 subjects presented with an ABI barely over 0.9, most likely due to precedent therapeutic endovascular intervention.
Patient characteristics.
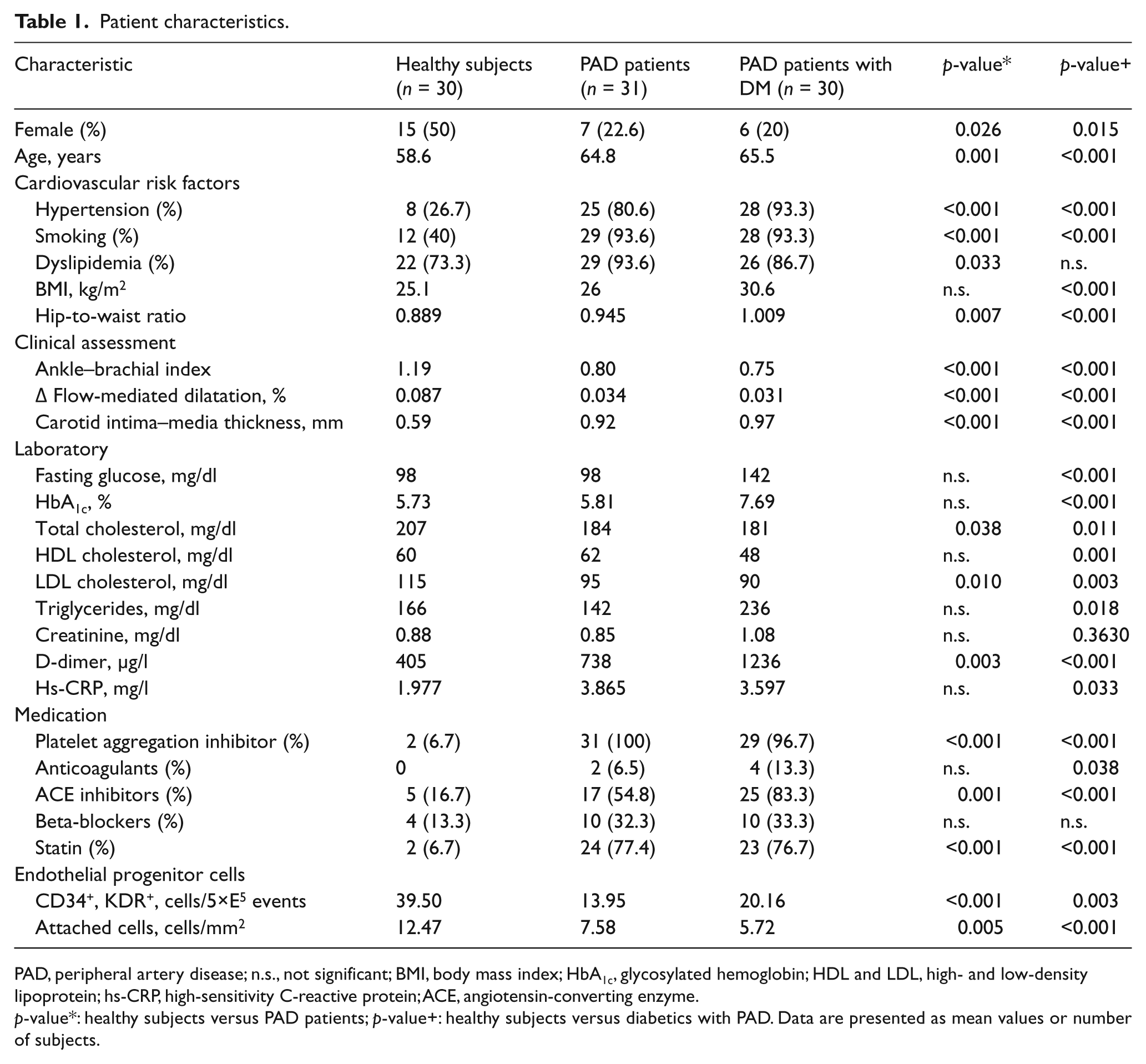
PAD, peripheral artery disease; n.s., not significant; BMI, body mass index; HbA1c, glycosylated hemoglobin; HDL and LDL, high- and low-density lipoprotein; hs-CRP, high-sensitivity C-reactive protein; ACE, angiotensin-converting enzyme.
p-value*: healthy subjects versus PAD patients; p-value+: healthy subjects versus diabetics with PAD. Data are presented as mean values or number of subjects.
EPC distribution
Figure 1 shows the circulating EPC number and EPC function (as in vitro colonies) to be more scattered within the group of healthy subjects compared to the disease-related groups. Within the disease-related groups the population of stem cells is homogenously at a low level. In figures, the range for the healthy group was from 5.5 to 109.5 cells/5×E5 events with a mean of 39.5 ± 28.6, whereas the range for the PAD group was 0.5 to 45 cells/5×E5 events with a mean of 13.9 ± 12.5, and the PAD+DM group was 1.0 to 52.5 cells/5×E5 events with a mean of 20.1 ± 12.5. The divergence in EPC functionality was similar, with a range for the healthy group from 2 to 34 cells/mm2 with a mean of 12.47 ± 7.3, whereas the functionality for the PAD group was 2 to 21 cells/mm2 with a mean of 7.6 ± 5.2 and the PAD+DM group was 1 to 17 cells/mm2 with a mean of 5.7 ± 3.9.
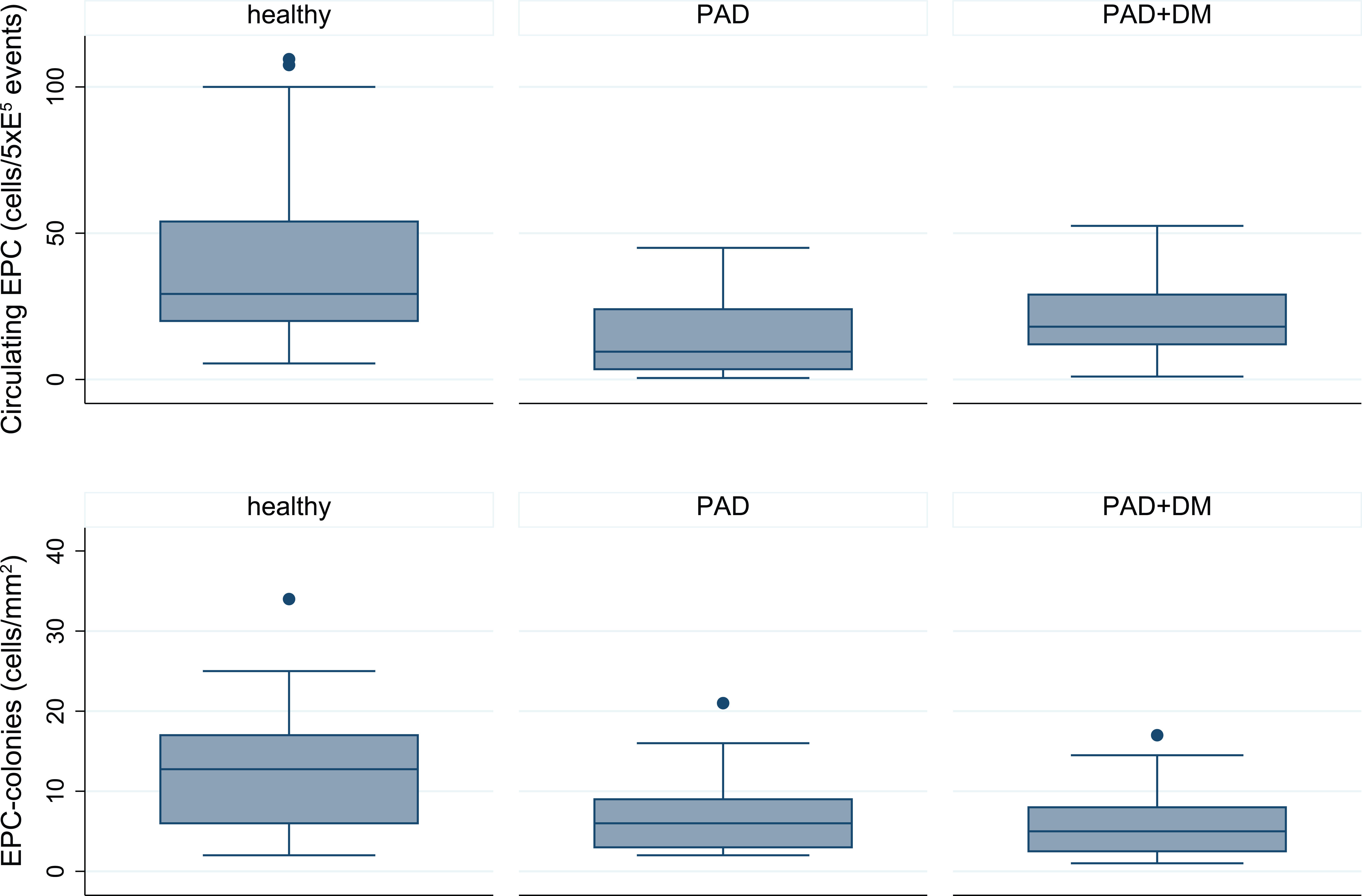
Bar graph illustrating the number distribution (top) and function (bottom) of circulating endothelial progenitor cells (EPCs) in patients with peripheral artery disease (PAD), PAD and diabetes mellitus (DM) and healthy controls. The number and function of EPCs were both significantly higher in the healthy group compared to the disease-affected groups. Compared with disease-related groups, the healthy group showed a highly scattered distribution pattern.
Multivariate linear regression analysis
We correlated clinical PAD parameters and biochemical EPC markers with the group allocation by adjusting the values for cardiovascular risk factors (age, gender, hypertension, dyslipidemia, smoking, waist-to-hip ratio). The linear regression shows statistical significance throughout the analysis, and is summarized in Table 2.
Results of multivariate linear regression analysis.
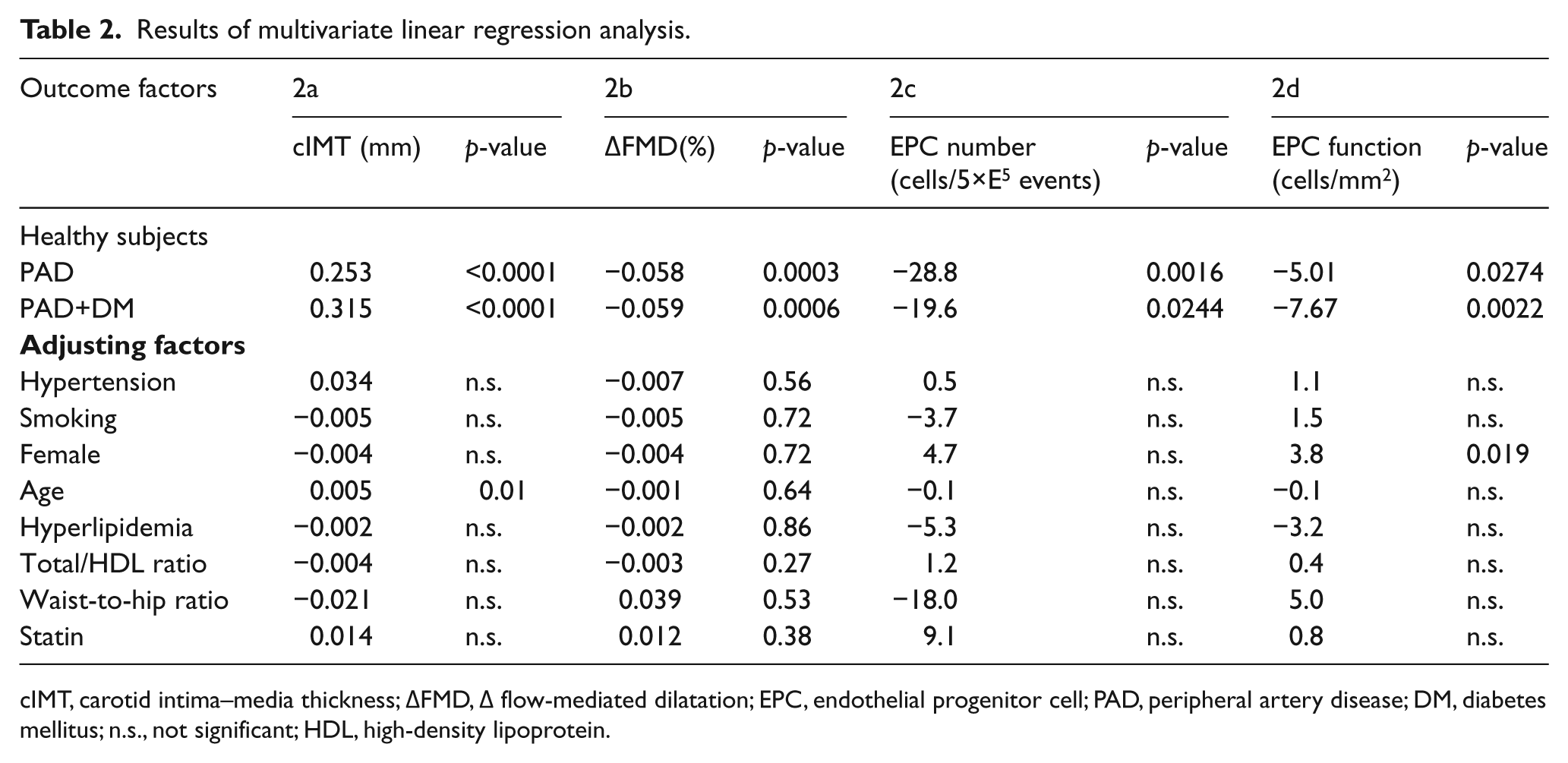
cIMT, carotid intima–media thickness; ∆FMD, ∆ flow-mediated dilatation; EPC, endothelial progenitor cell; PAD, peripheral artery disease; DM, diabetes mellitus; n.s., not significant; HDL, high-density lipoprotein.
Ultrasonographic imaging of the cIMT and assessment of endothelial reactivity (FMD) showed a highly significant correlation (p<0.0001) to the appearance of PAD (Table 2a and 2b), with further aggravation of the findings with concomitant DM. The number of circulating EPCs was correlated statistically significantly (p=0.0016/p=0.0244, respectively) to group allocation. We noticed a considerable decrease in disease-affected groups, with a meandifference of −28.86/−19.66 cells/5×E5 events in the PAD and PAD+DM groups, respectively (Table 2c).
Surprisingly, the additional occurrence of DM in disease-affected groups did not worsen the number of circulating EPCs. Rather, we observed slightly higher EPC numbers in patients with DM. However, this observation has no statistical significance.
We found the ability of EPCs to form colonies to be statistically significantly impaired in both PAD (p=0.027) and PAD+DM patients (p=0.0022). In this functional assessment, superposed DM is worsening the potential of isolated EPCs to spawn. The multivariate linear regression analysis (Table 2a–d) adjusted for the common cardiovascular risk factors showed these findings to be independent of gender, hyperlipidemia, smoking, hypertension, statin therapy, waist-to-hip ratio and the total cholesterol/HDL ratio.
Correlation of biochemical and clinical measures
In the patient groups (n=61), a Pearson’s product-moment correlation based on Fisher’s transformation was applied to characterize the impact of biochemical EPC markers on the clinical PAD manifestation. We found the number of circulating EPCs correlated with severity of PAD using the ABI as a clinical reference (p=0.027; correlation = 0.286). The lower the ABI, the fewer the circulating EPCs (Figure 2). Moreover, we found a statistically significant correlation (p=0.017; correlation = −0.4324) of cIMT with EPC functionality (Figure 3).
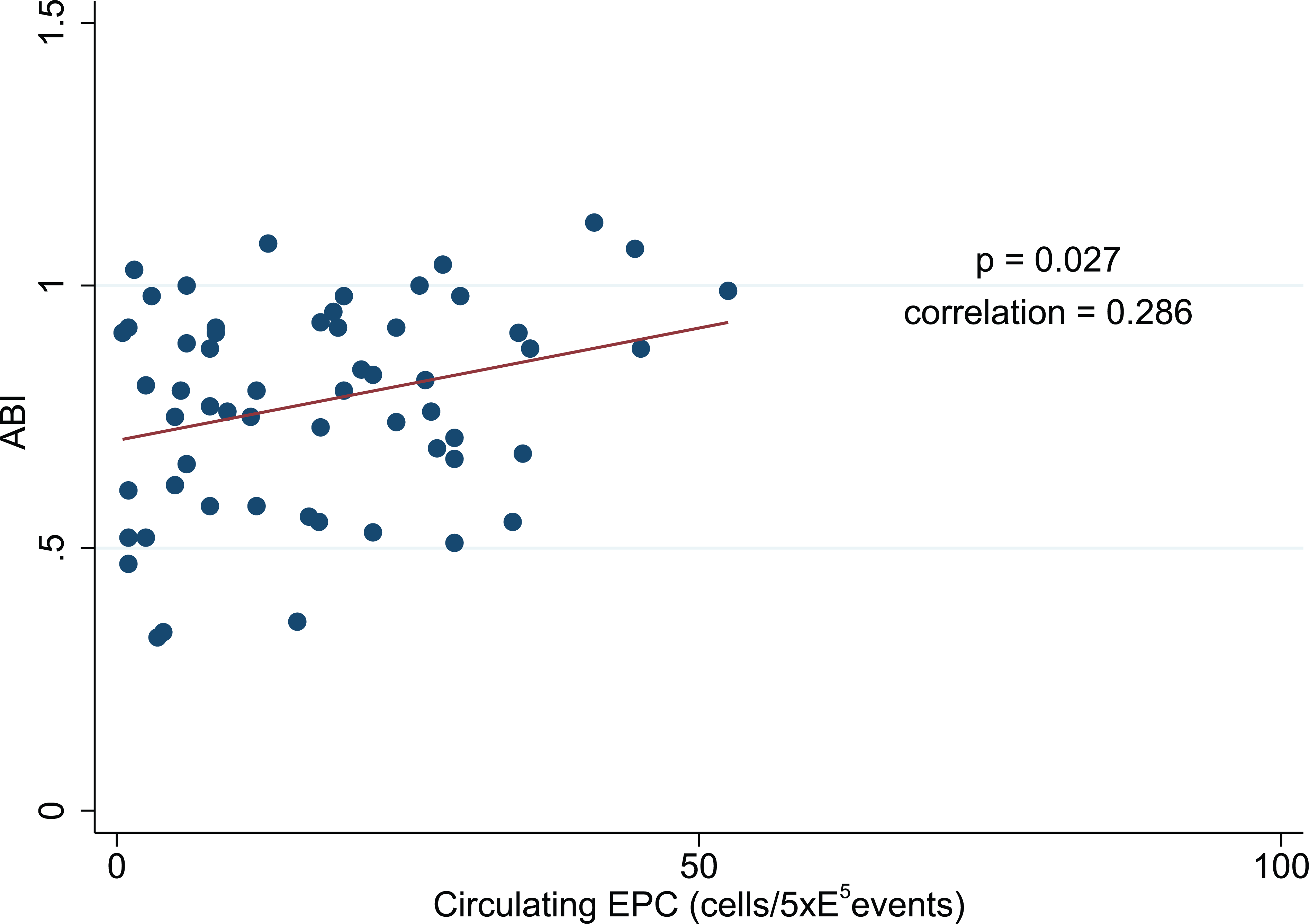
Linear progression analysis demonstrating a positive correlation between the ankle–brachial index (ABI) and the number of circulating endothelial progenitor cells (EPCs) includes peripheral artery disease (PAD) patients and PAD with diabetes mellitus (PAD+DM) patients (60 observations).
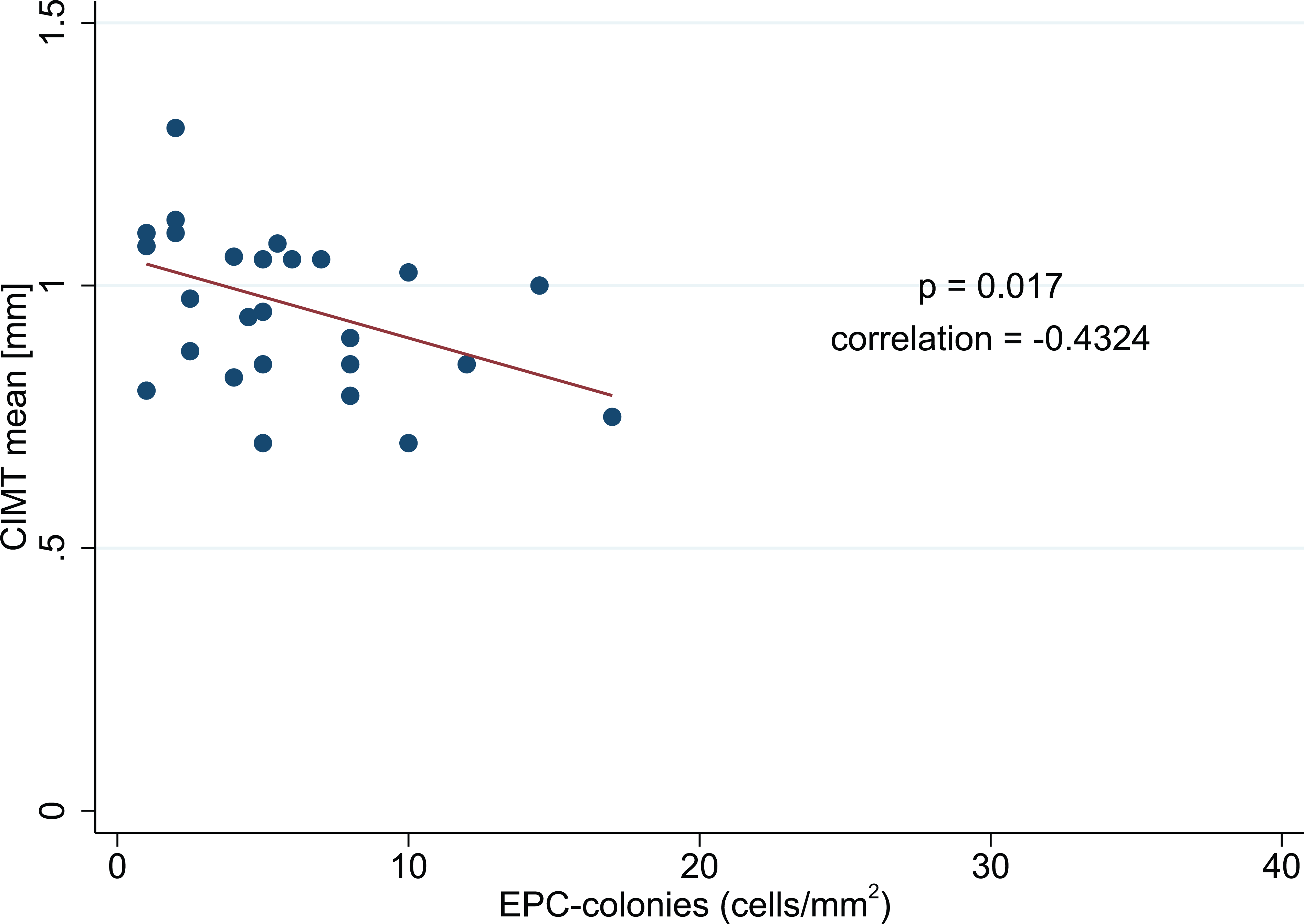
Linear regression analysis representing a negative correlation between carotid artery intima–media thickness (cIMT) and functionality of endothelial progenitor cells (EPCs) presented as colony-forming units includes patients with peripheral artery disease and diabetes mellitus (PAD+DM) only (30 observations).
The linear regression analysis that correlates ABI and EPC function is borderline not statistically significant, most likely due to previous endovascular interventions. Figure 4 shows the correlation shown by Pearson’s product-moment correlation of ABI and EPC function based on Fisher’s transformation (p=0.061; correlation = 0.243). No statistical significance could be found for correlations with FMD and EPC parameters.
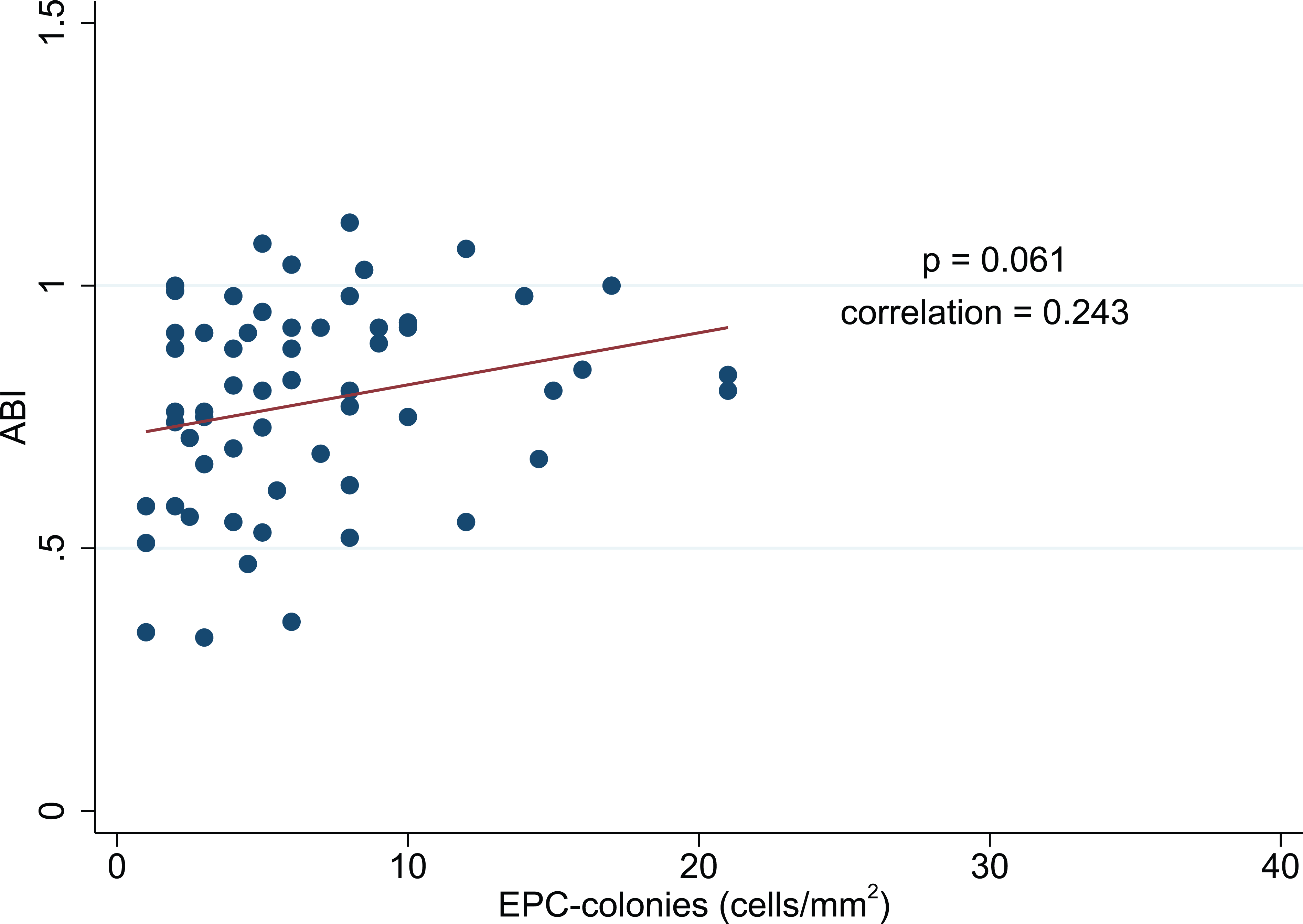
Linear progression analysis indicating a positive correlation between ankle–brachial index (ABI) and the endothelial progenitor cell (EPC) function includes peripheral artery disease (PAD) patients and patients with PAD and diabetes mellitus (PAD+DM) (60 observations).
Biomarker Analysis
Descriptive statistics showed statistically significant differences in selected biomarker distributions (Table 3). D-dimers were almost doubled or tripled in PAD and PAD+DM groups, respectively, compared to healthy volunteers (p=0.003/p<0.001). Relative to hs-CRP, a statistical significance was found only when healthy volunteers were compared to the PAD+DM group (p=0.033). However, multivariate analysis did not disclose linear correlations between biomarkers and circulating EPCs, EPC function or clinical PAD indices (data not shown).
Biomarker analysis.
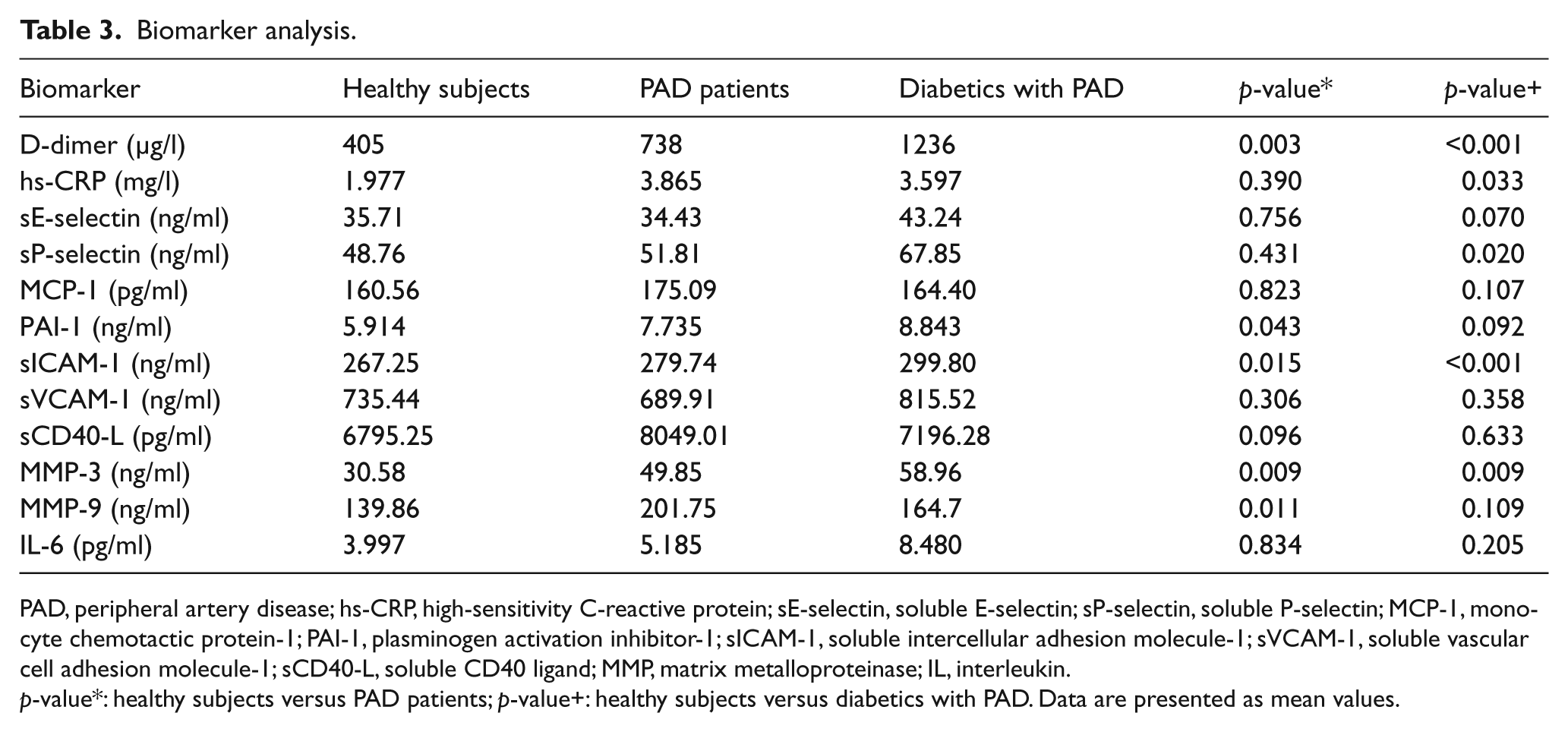
PAD, peripheral artery disease; hs-CRP, high-sensitivity C-reactive protein; sE-selectin, soluble E-selectin; sP-selectin, soluble P-selectin; MCP-1, monocyte chemotactic protein-1; PAI-1, plasminogen activation inhibitor-1; sICAM-1, soluble intercellular adhesion molecule-1; sVCAM-1, soluble vascular cell adhesion molecule-1; sCD40-L, soluble CD40 ligand; MMP, matrix metalloproteinase; IL, interleukin.
p-value*: healthy subjects versus PAD patients; p-value+: healthy subjects versus diabetics with PAD. Data are presented as mean values.
Discussion
In the present study we describe a numeric depletion as well as a malfunction of EPCs correlating to clinical signs of endothelial dysfunction in patients with PAD. Compared to a group of healthy volunteers, we found an altered state of progenitor cell population in disease-related groups. The statistical analysis shows a significant correlation of mean circulating EPCs and their ability to proliferate in a culture medium, as previously demonstrated for other cardiovascular diseases. 3 These data support the involvement of EPCs in alterations to the state of endothelial function, as their depletion may cause impairment of regenerating processes 22 and progression of cardiovascular diseases.
A low ABI is an accurate 23 and reliable 24 marker of generalized atherosclerosis. It is a valuable predictor of subsequent cardiovascular morbidity and mortality.25–27 Our data allow us to postulate that EPCs are a reliable biological marker of PAD, as a low ABI correlates to a low number of circulating EPCs (Figure 2). Concordantly, EPC number is inversely correlated to cardiovascular risk factors. 28 Interestingly, this depletion is not only related to environmental or clinical factors, as heritable factors have recently been described to contribute even more substantially to the progenitor phenotype. 29 Owing to the inclusion of patients who had already benefitted from percutaneous endoluminal angioplasty with immediate ABI improvement, the statistical analysis was more difficult. We decided not to exclude these patients, as this represents real clinical life. Also, we considered the atherosclerotic burden, endothelial dysfunction and the state of circulating EPCs enduring stability, in particular >6 months after an endovascular intervention. Therefore, we assume this correlation to be even stronger than statistically suggested. The same consideration applies for the correlation of EPC function and ABI (Figure 4), where the correlation is borderline not significant (p=0.061).
The EPC distribution within the three groups is, in our opinion, of particular interest. EPC number and function in healthy subjects was scattered over a wide range, which reflects the inter-individual state of endothelial integrity and on-going repair processes. In disease-affected groups, we demonstrated a narrowing of the variability to almost the same amount in both groups (Figure 1). According to these results, we hypothesize a dynamic state of the EPC population. The postulated influence of the disease is reducing the dynamic responsiveness of the system to injury. A depleted, malfunctioning and narrowed population is not able to perform the same repairing processes, and presumably leads to further progression of the disease. The hindered migration of EPCs to the sites of endothelial lesions produces disequilibrium between progressive vascular damage and insufficient vascular repair, leading to atherosclerosis and finally to vascular disease.
Patients with PAD and DM represent a particularly high-risk population for cardiovascular morbidity and mortality. 30 The assessment of diabetic patients in their own group allowed us to study the effects of concomitant DM separately. Recent studies described a numeric depletion of the EPC population in diabetic patients.31–33 In contrast, in our study, this effect of DM on the severity of PAD did not apply to the EPC population. The number of EPCs in the DM group was significantly higher compared to the PAD group. As expected, the functionality was impaired, but without reaching statistical significance.
A possible effect of DM on EPCs is a reduced adherence on sites of endothelial damage, and therefore the inability to complete the physiological effect on the endothelium. 33 Nevertheless, we have to take account that DM has other pathophysiological ways to aggravate cardiovascular disease than acting on stem cells.
One of the key concerns in PAD is the increased risk of cardiovascular ischemic events due to the systemic nature of the atherosclerotic process. Owing to the concomitant cardiovascular and cerebrovascular disease, PAD is considered a coronary disease risk equivalent, as it confers equal risk for a future major vascular event, as a previous myocardial infarction.23,34 We support this well-accepted thesis by correlating EPC function to cIMT. Statistical significance (p=0.017) was found in a post hoc analysis in the PAD+DM group, even though patients were asymptomatic for cIMT thickening (Figure 3). Indeed, cIMT represents an intermediate phenotype for early subclinical atherosclerosis, and has been included in the list of organ damage conditions in the European Guidelines on Cardiovascular Disease Prevention as a high-value risk population marker.17,35 This correlation points out the relevance of EPCs in the general pathophysiological concept of atherosclerosis.
Atherosclerosis is considered a chronic vascular inflammatory disease and the activity of inflammation, measured by the level of hs-CRP, has been related to cardiovascular risk 36 and age. 29 In our study, we found significant D-dimer elevation to almost doubled (PAD group) and tripled (PAD+DM group) values when compared to healthy volunteers. These findings may reflect the pro-thrombotic and pro-inflammatory activity of damaged endothelium in atherosclerotic disease.
However, in multivariate analysis we could not document linear correlations to circulating EPC number, function or clinical PAD indices. The correlation between EPCs and inflammatory markers remains controversial. While hs-CRP provided prognostic information for the development of symptomatic PAD in addition to ABI, 37 D-dimer was associated with an increased risk of myocardial infarction but not with PAD progression. 38 Analysis of other specialized biomarkers showed no abundant statistical significance, which may be due to the relatively small sample size and can therefore be accorded only poor significance.
Limitations
Limitations of the study are the small size of the three groups and the heterogeneity of the healthy volunteers compared to the patient groups. In addition, our volunteers despite being clinically healthy, presented with incidentally altered laboratory findings, especially dyslipidemia. Another considerable factor is the before-mentioned inclusion of patients who had already benefitted from prior endovascular therapeutic interventions. These issues could mostly be compensated for by multivariate statistical analysis. Apart from these clinical issues, the identification of such a rare cell type like EPCs remains challenging and controversial. Since their first description in 1997, EPC characterization has been under intense scientific investigation. However, as no specific antibody still exists for these cells, inconsistency persists about the use of the ideal antibody composition to identify circulating EPCs. In our case, we have reverted to the established combination of KDR and CD34 cell positivity.10,39,40 Other research prefers additional staining for CD45 as well as the use of live cell markers. Further restrictions concern the use of directly labeled antibodies to improve the accuracy of FACS analysis.
Hill et al. first described the use of colony-forming units as a functional analysis of EPCs. 3 Ideally, assessment of EPC function should not rely on sole colony formation as colony numbers may not correlate directly with endothelial-like function. Oxidized low-density lipoprotein uptake or integration into tubules, such as in matrigel assays of cultured or fresh EPCs, represent more reliable measures of endothelial function. In particular, as recent research by Yoder et al. demonstrated that tube formation in matrigel assays allows the identification of those EPCs that participate in the structural development of new blood vessels. 41 However, owing to the high volume of sample material in our setting, the implementation of these in vitro assays was simply not realizable.
Conclusions
In summary, our study goes beyond previous observations of an association between EPCs and PAD. 42 We provide statistical evidence that the number and function of circulating EPCs correlates with the appearance and severity of PAD using the ABI as a clinical reference. These results support the pathophysiological and clinical importance of circulating EPCs as a biomarker for PAD. The narrowed EPC distribution in disease-affected groups compared to the high variability in healthy volunteers suggests progenitor cells are a dynamic cell population, with a dedicated state of reactivity. The narrowing of cell numbers and reduced activity alone might lead to cardiovascular disease or, alternatively, may indicate exhaustion of the endogenous repair mechanisms, which then leads secondarily to disease aggravation.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study is supported in part by the Swiss National Foundation (No. 3200B0-114100 to CK), the Swiss Heart Foundation to CK and the University of Bern, Switzerland (Grant-in-aid to CK).
