Abstract
Peripheral artery disease (PAD) represents a major health care burden. Despite the advent of screening and interventional procedures, the long-term clinical outcomes remain suboptimal, especially in patients with chronic kidney disease (CKD). While CKD and PAD share common predisposing factors, emerging studies indicate that their co-existence is not merely an association; instead, CKD represents a strong, independent risk factor for PAD. These findings implicate CKD-specific mediators of PAD that remain incompletely understood. Moreover, there is a need to understand the mechanisms underlying poor outcomes after interventions for PAD in CKD. This review discusses unique clinical aspects of PAD in patients with CKD, including high prevalence and worse outcomes after vascular interventions and the influence of renal allograft transplantation. In doing so, it also highlights underappreciated aspects of PAD in patients with CKD, such as disparities in revascularization and higher peri-procedural mortality. While previous reviews have discussed general mechanisms of PAD pathogenesis, focusing on PAD in CKD, this review underscores a need to probe for CKD-specific pathogenic pathways that may unravel novel biomarkers and therapeutic targets in PAD and ultimately improve the risk stratification and management of patients with CKD and PAD.
Introduction
Approximately 8.5 million adults in the United States and 200 million adults worldwide are affected by peripheral artery disease (PAD).1–4 The prevalence of PAD continues to rise and has grown by 13.1% and 28.7% in high income and low to middle income countries, respectively. 4 This high prevalence of PAD has wide-reaching implications for morbidity, mortality, quality of life, and health care costs. While a multitude of risk factors can contribute to PAD, chronic kidney disease (CKD) has emerged as a significant independent risk factor. Intriguingly, the prevalence of PAD is as high as 24–37% in patients with CKD.5–10 Patients with CKD exhibit unique nuances in various aspects of PAD that are worthy of special consideration and constitute the rationale for this review. Covering clinically relevant points in PAD, among other aspects, the current review expands on outcomes of PAD in patients with CKD after vascular interventions and CKD-specific mechanisms of PAD pathogenesis.
Clinically, ankle–brachial index (ABI) is commonly used to diagnose PAD. Generally, an ABI of 0.9–1.4 is considered normal. An ABI less than 0.9 is considered abnormal, and supports a diagnosis of PAD. The results of ABI tests in patients with CKD must be interpreted carefully, as CKD, and associated comorbidities such as diabetes, can lead to increased vascular calcification, resulting in vessel stiffness or noncompressible vessels. 8 The incompressibility of the vessels can lead to elevated ABIs (> 1.4) or falsely normal ABIs. In this setting, the use of toe pressures and toe–brachial indices can help provide a better estimate of PAD than the use of ABI alone, as toe vessels are less often affected by the calcification.11,12 Thus, we recommend the addition of toe pressures and toe–brachial indices when screening for PAD in the CKD population. Severity of PAD is graded by two widely accepted classification systems: the Rutherford classification (categories 0–6) 13 or Fontaine classification (stages I–IV) (Tables 1A and 1B).14,15 In both of these classification schemas, increasing grade or stage corresponds to the severity of disease. 16
Rutherford classification for chronic limb ischemia.

AP, ankle pressure; PVR, pulse volume recording; TM, transmetatarsal; TP, toe pressure.
Five minutes at 2 mph or 3.2 km/h on a 12% incline.
Fontaine classification system for chronic limb ischemia.

Patients with CKD have higher prevalence and more severe PAD
The prevalence of PAD is higher in the CKD population compared to the general population. Foley et al. examined 1,091,201 patients from the Medicare database and noted a 9.6% prevalence of PAD in patients without CKD, and a threefold higher prevalence of 32.6% in CKD patients. 17 Similarly, analysis of the United States Renal System database of 1,249,076 patients revealed that the prevalence of PAD in any CKD patient was 24.9%. 18 Increasing severity of CKD is associated with higher prevalence of PAD. For example, patients with CKD stage 4 had a 20% prevalence of PAD compared to 30% prevalence of PAD in patients with CKD stage 5 on hemodialysis.8,18,19 Tables 2 and 3 demonstrate a striking increase in the prevalence of PAD with CKD.5–8,17–23
Prevalence of PAD by CKD status in various studies.
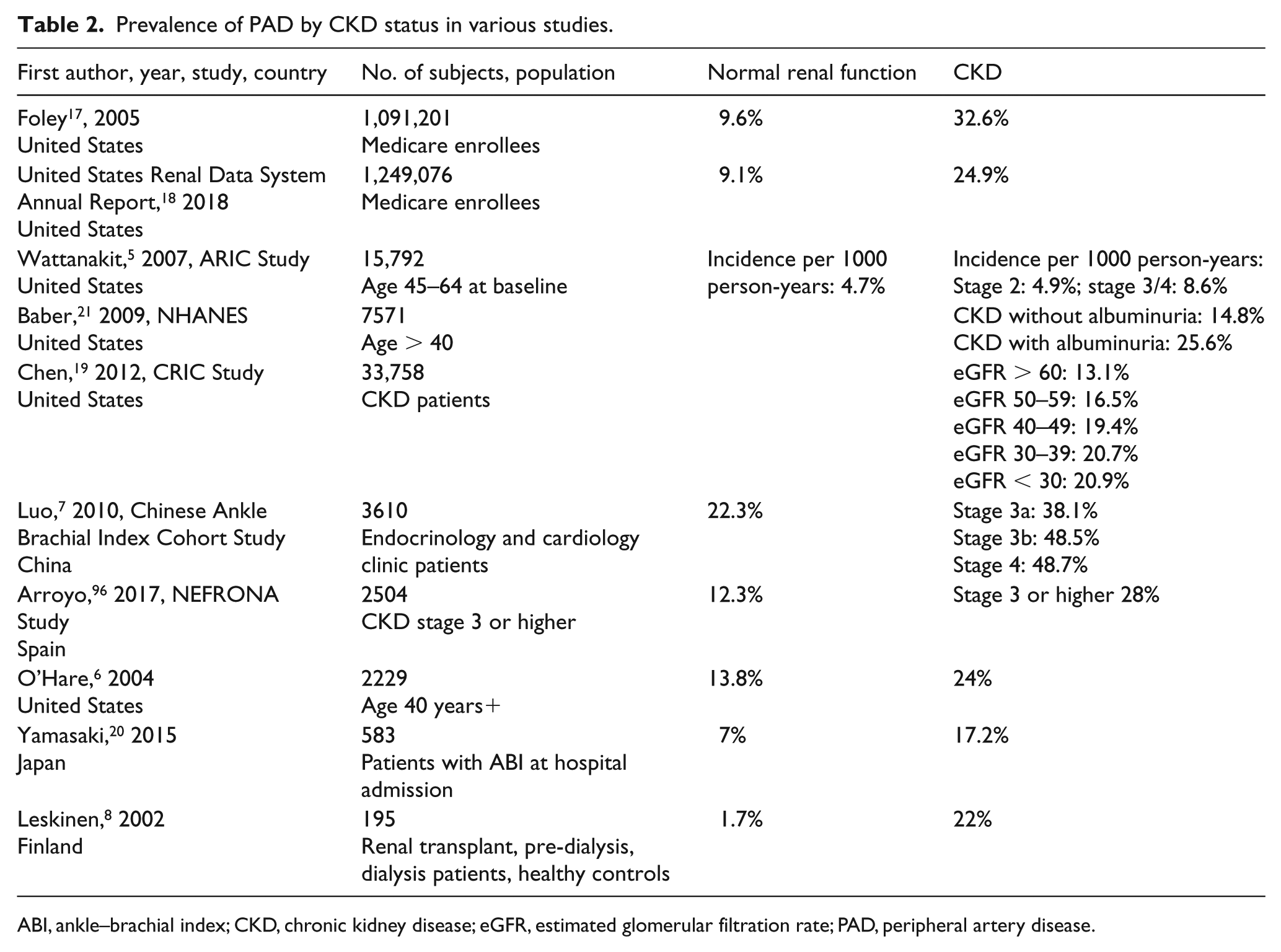
ABI, ankle–brachial index; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; PAD, peripheral artery disease.
PAD prevalence in hemodialysis and peritoneal dialysis patients.
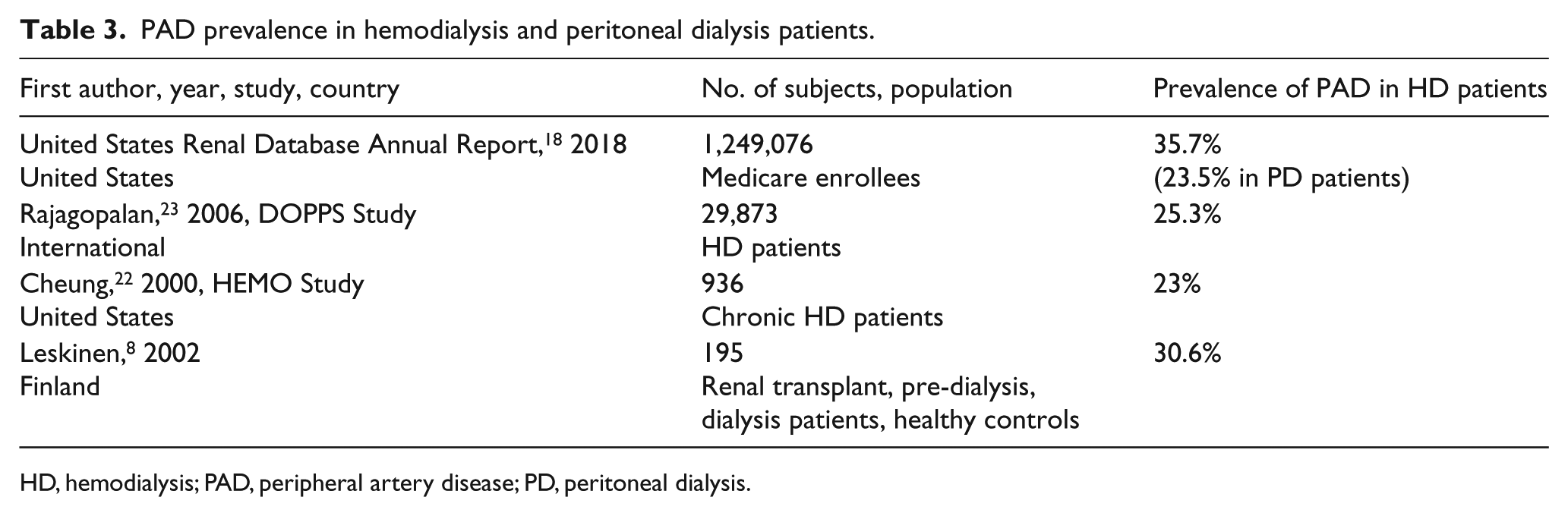
HD, hemodialysis; PAD, peripheral artery disease; PD, peritoneal dialysis.
This high prevalence of PAD in patients with CKD is also reflected in the inpatient population and is associated with higher cost and mortality. One quarter of patients hospitalized with PAD have CKD. 24 Additionally, these patients incur 15% more health care expenditures related to their hospital stay and have a 21% longer length of stay. Patients with advanced CKD who were hospitalized for PAD exhibit an increased risk of mortality (OR 1.84, 95% CI 1.02–3.32, p = 0.44) even after adjusting for Rutherford stage of PAD. 25
As the risk of PAD increases with CKD stage, so does the risk of chronic limb-threatening ischemia (CLTI). CLTI is a severe manifestation of PAD that presents with ischemic rest pain (pain of the foot or ankle at rest) and/or tissue loss (ischemic ulceration or gangrene). 26 In patients undergoing bypass grafting with autologous saphenous vein, the prevalence of CLTI increased with higher stage of CKD (77.6% with CKD stage 1/2 to 100% with CKD stage 5). 27 Those PAD patients at the higher stages of CKD were more likely to present with significant tissue loss and had more compromised limb wounds.
In patients admitted to the hospital for CLTI, patients with CKD, compared to those with normal renal function, had longer hospital stays, more bleeding complications (16% vs 4.26%, p < 0.01), more wound infections (11.6% vs 3.31%, p < 0.01), and required earlier re-interventions (20.5% vs 11.8%, p = 0.028). 28 Moreover, these outcomes were worse in CKD patients who developed superimposed acute kidney injury (AKI). Zlatanovic et al. reported that after various vascular procedures there was double the risk of mortality in patients with CKD with AKI compared to patients without AKI. 28 Taken together, the above data underscore the importance of CKD as a strong and independent risk factor of PAD and CLTI.
Patients with CKD undergo fewer revascularization procedures for PAD
There are multiple strategies, such as open surgical or endovascular procedures, to revascularize an ischemic limb in the setting of PAD. 26 Surgical revascularization options include bypass grafts and endarterectomy. Endovascular revascularization options include angioplasty, atherectomy, and stenting, etc., among others.29,30 Several parameters including lesion characteristics and patient-related factors affect the choice of intervention. While endovascular procedures have been largely recommended for patients with significant comorbidities, there is not yet a consensus on what revascularization technique is best. 31 The ‘Best Endovascular vs. Best Surgical Therapy in Patients with Critical Limb Ischemia (BEST-CLI)’ and ‘Bypass versus Angioplasty in Severe Ischemia of the Leg-2 (BASIL-2)’ trials are currently underway and will help to guide decision making for patients with CLTI.32,33 Since these studies include patients with CKD, they will shed further light on the best possible management of CLTI in this population.
Despite the high prevalence of PAD and CLTI in this population, several reports suggest that patients with CKD may be offered fewer revascularization procedures than patients without CKD despite comparable severity of PAD. Using the Veterans Affairs National Patient Care Database, O’Hare et al. evaluated 6227 newly diagnosed PAD patients with CLTI between 2002 and 2004. 34 Despite representing 34% of the entire patient cohort, patients with CKD made up only 28% of those who underwent revascularization and were 45% of those who underwent amputation in the first 6 months after diagnosis of PAD. Even with the adjustment for age, race, and comorbidities, with increasing severity of CKD there were decreasing odds of undergoing revascularization compared to patients with normal renal function. Decreased utilization of revascularization procedures was reported in a European cohort as well. Using a large, German insurance database, Lüders analyzed 41,882 PAD patients, 20.2% of whom had CKD. They reported that at a comparable severity of PAD, 67.2% of patients without CKD underwent revascularization, while this percentage dropped to 48% in the patients with CKD stage 4 or 58.2% in stage 5. 35 Both of these studies lacked data on confounders, such as the ambulatory status, extent of tissue loss, specifics of the lesions such as site and severity of lesion (distal vs proximal) or the presence of gangrene etc., 36 all of which are important considerations that can affect the choice of procedures.
The observed trend of lower rates of interventions in patients with CKD may also be driven by a multitude of factors, such as higher rates of complications in patients with CKD, poor functional or nutritional status, and advanced wounds that are deemed unsalvageable. A possible deterrent for offering endovascular intervention is concern for contrast-induced nephropathy. Strategies such as CO2 contrast angiography, pre-hydration, N-acetyl cysteine and sodium bicarbonate may help to avoid contrast-induced nephropathy.37–39 Taken together, more studies are needed to understand the clinical reasons behind a decrease of revascularization procedures in CKD patients despite higher prevalence and severity of PAD.
Patients with CKD experience worse outcomes after interventions despite comparable primary patency rates
Patients with CKD represent one-quarter of patients that undergo revascularization procedures for PAD. In patients with CKD, several studies strongly indicate worse outcomes after revascularization, such as failure of limb salvage and increased risk of amputation, despite similar or better graft patency (Table 4).27,40–45 In patients with CKD who underwent infrapopliteal bypass for CLTI, Kumada et al. reported that patients on hemodialysis had a 4.92-fold higher risk of limb loss compared to patients with CKD not on hemodialysis, and decreased rates of 5-year amputation-free survival (43.6% vs 78.8%, p = 0.0033). 41 This increased risk of amputation occurred despite nearly equivalent rates of revascularization of the target limb (12.4% vs 12.2%). Using prospectively collected data from the Vascular Study Group of Northern New England database, Simons et al. evaluated patients who underwent elective infrainguinal lower extremity bypass for CLTI for clinical failure, which was defined as amputation or persistence or worsening of ischemic symptoms despite graft patency. 46 At 1-year postprocedure, patients who were on dialysis had a 3.7-fold increased risk of clinical failure compared to patients with normal renal function.
Postintervention overall survival, primary patency rates, and limb salvage in CKD patients.
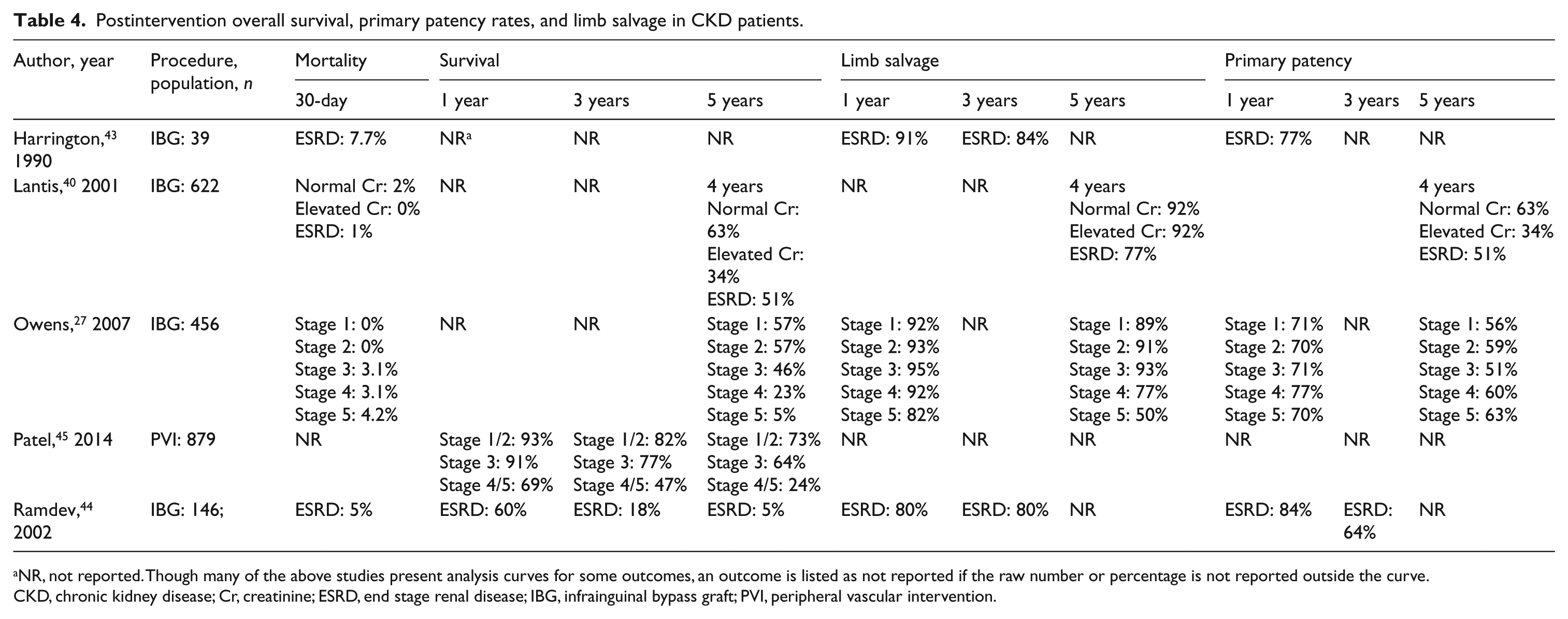
Though many of the above studies present analysis curves for some outcomes, an outcome is listed as not reported if the raw number or percentage is not reported outside the curve.
CKD, chronic kidney disease; Cr, creatinine; ESRD, end stage renal disease; IBG, infrainguinal bypass graft; PVI, peripheral vascular intervention.
Poor limb salvage rates in patients with CKD have been observed after endovascular interventions as well. Patel et al. reported that the presence of CKD stage 5 in patients undergoing percutaneous endovascular interventions increased the risk of late amputation despite having patent bypass grafts. They also showed an increased need for re-interventions in CKD patients at 1 year and 3 years. 45
In all the above studies, worsened limb salvage after a successful intervention can be defined as a ‘clinical failure.’ Some groups have attempted to evaluate for clinical factors that affect amputation risk in CKD patients. Using the COhorte des Patients ARTériopathes (COPART) registry, a prospective multicenter, observational study of patients hospitalized with PAD in France, Lacroix et al. observed an increased rate of amputation with CKD patients compared to patients without renal impairment. 25 The overall 1-year amputation rate was 26.3%, while for patients with an estimated glomerular filtration rate (eGFR) < 30 mL/min per 1.73 m2 that number increased to 40.2%. However, after adjusting for cardiovascular history (such as history of stroke or heart failure) and treatments (such as statins or antiplatelet therapy) in addition to comorbidities, the odds of amputation were nearly equivalent amongst groups. This study points to the contribution of non-CKD-related factors, such as cardiovascular health and comorbidity burden, to clinical failure despite technically successful intervention. However, other factors such as nutritional and functional status, increased rates of infection and wound dehiscence, etc., may be at play. Also, CKD-specific mediators and factors in compromising limb salvage remain a strong possibility. For example, studies have reported microvascular rarefaction resulting in impaired microcirculation in various vascular beds including skeletal muscles in CKD patients and animal models, which may contribute to this phenomenon.47–50 Collectively, several studies indicate a distinct problem of clinical failure despite technically successful revascularization in CKD patients, which underscore an urgent need for more work in this specific area.
Patients with CKD have an increased risk of perioperative complications
Using the Veterans Affairs (VA) Clinical Assessment, Reporting, and Tracking (CART) program, Xie et al. reported that patients with CKD have a 1.57-fold higher risk of death after peripheral vascular interventions compared to those with normal or mildly reduced renal function. 51 Using data from the VA National Surgical Quality Improvement Program (NSQIP) for patients who underwent lower extremity revascularization, O’Hare et al. observed that patients with CKD had increased rates of 30-day postoperative mortality compared to those with normal or mildly reduced renal function (4–10% vs 2%, p < 0.001). 52 This increased risk of mortality persisted even after adjusting for several confounders in these patients. Studies using various databases have also demonstrated increased risk of postoperative mortality in patients with CKD after both open surgical revascularization and endovascular interventions compared to others.52–55 Patients with end stage renal disease (ESRD) experience an increased rate of cardiovascular complications after vascular interventions. Using data from the VA NSQIP for patients who underwent lower extremity revascularization, O’Hare et al. observed that even after adjusting for confounders, those with an eGFR of 30–59 and an eGFR < 30 mL/min/1.73 m2 had higher rates of 30-day postoperative myocardial infarction (4% vs 1%, p < 0.001). Patients with moderate CKD undergoing revascularization procedures were significantly at higher risk for prolonged intubation after vascular intervention. Using data from the VA NSQIP for patients who underwent lower extremity revascularization, O’Hare et al. observed that those with an eGFR < 30 mL/min/1.73 m2 were at a 2.7-fold higher risk of prolonged intubation (> 48 hours) compared to patients with an eGFR > 60 mL/min/1.73 m2. 52 This relationship of increased risk for respiratory compromise persisted after adjusting for confounders.
In general, in comparison to patients without PAD, patients with PAD have a two to three times elevated risk of cardiovascular morbidity and mortality, including myocardial infarction, stroke, and mortality related to coronary artery disease. 3 In particular, patients with CKD and PAD have elevated rates of coronary artery disease and cerebrovascular disease compared to CKD patients without PAD. 56 One study showed a co-prevalence of 65% of coronary artery disease in patients with CKD who have PAD. 56 Patients with CKD have increased rates of congestive heart failure compared to patients with PAD but no CKD.35,40,57,58 Thus, it is likely that the increased risk of perioperative events in patients with CKD can be due to co-prevalence of significant cardiac disease.
PAD in the renal transplant patient
Management of PAD prior to renal transplantation
PAD is not considered a contraindication to renal transplantation. However, if PAD is known, both the American Society of Transplantation and the American Heart Association recommend a preoperative noninvasive screening for coronary artery disease given the high rate of concurrence of PAD and coronary artery disease. However, these institutes recommend intervention only in the setting of symptomatic PAD disease.59–61
Since the renal allograft is anastomosed to the iliac vessels, presence of significant aortoiliac calcifications or atherosclerosis may affect the ability to have a successful anastomosis. Franquet et al. examined patients with CKD who underwent elective aortic bypass grafting for aortoiliac calcification in preparation for renal transplant. 61 Of the 21 patients who underwent this procedure, only 11 patients could receive successful renal transplant. Moreover, four patients died while waiting for a transplant and five were removed from the list for poor health. Given the high rate of postoperative complications, this group recommended against a two-staged procedure, but rather if vascular bypass is deemed necessary, it should be performed at the time of the renal transplant. Because of the untoward risk of aortic bypass, as well as the increased peri- and postoperative risks for these ESRD patients on the renal transplant waiting list, aortic bypass should be reserved for treatment of aneurysmal disease or symptomatic PAD. Currently, the American Society of Transplantation does not recommend pre-emptive treatment of asymptomatic aortoiliac disease in preparation for transplant. 60
Patients with PAD have increased risk for renal allograft failure after renal transplantation
A review of kidney transplant recipients from the Mayo Clinic showed that low ABI (< 0.9) was a significant and independent predictor of allograft failure (odds ratio (OR) 2.77, 95% CI, 1.68–4.58, p < 0.001). 62 Similarly, other studies have also shown more than twice the increased risk of graft failure in the PAD population. 63 The mechanisms underlying increased risk of allograft failure in patients with PAD remain unclear, though factors such as the presence of atherosclerotic disease and the inflammatory state of PAD may play a role.
Renal transplantation may improve PAD progression
Analysis of the United States Renal Data System database revealed that after adjusting for other confounders, the relative risk of developing PAD in kidney transplant recipients decreased by 23% compared to those ESRD patients on dialysis on the waiting list. 64 Because those chosen as renal transplant candidates are CKD patients with less comorbidity burden and good health, the possibility of a selection bias cannot be ruled out. However, comparison of PAD between renal transplant recipients and CKD patients on the transplant list may reduce this confounding effect. Taken together, these data suggest that renal allograft transplantation may retard the progression of PAD in CKD patients, possibly by the reduction or removal of non-dialyzable mediators of PAD. Immunosuppressive medications, such as tacrolimus, and anti-metabolites, such as mycophenolate mofetil, can potentially contribute to slowing the progression of PAD after transplantation through anti-proliferative effects on processes such as neointimal hyperplasia.65–67
Renal transplantation potentially confers survival advantage to CKD patients with PAD
Multiple studies have shown a reduction in risk of mortality in PAD patients after renal transplantation.68,69 Renal allograft transplantation resulted in a twofold reduction of 5-year mortality for PAD patients compared to those remaining on the waiting list (hazard ratio (HR) 0.440, p < 0.001). This potential improvement in the overall mortality is overshadowed by the poor outcomes after vascular intervention. Many clinical studies evaluating the effect of underlying renal disease on outcomes after vascular interventions exclude renal transplant recipients given the change in physiology and the possible effect of various pharmacologic therapies. However, Grisafi et al. compared the outcomes after vascular interventions for PAD in hemodialysis patients to those who had received transplantation, and found surprisingly that transplant recipients had worse limb salvage rates and amputation-free survival compared to hemodialysis patients (22% vs 82%, p < 0.02). 70 The authors point to the possible contributory effect of immunosuppressive agents on wound healing and postoperative recovery. Nonetheless, more work is needed to understand the effect of renal transplantation on outcomes after vascular intervention.
Pathogenesis of PAD in CKD
Inflammation and oxidative stress
CKD is a state of global inflammation, as demonstrated by several animal models and human studies.71,72 Inflammation contributes to the progression of cardiovascular diseases by inducing the release of cytokines such as interleukin (IL)-1, IL-6, IL-1β, IL-8, tumor necrosis factor (TNF)-α, TGF-β, and high-sensitivity C-reactive protein and fibrinogen, all of which induce pro-fibrotic and atherothrombotic processes. CKD-induced inflammation is linked to perpetual amplification of the further underlying inflammatory state, which exacerbates vascular calcification and endothelial dysfunction, setting the stage for PAD.71,73 Because inflammation and oxidative stress are integral components of the pathogenesis of PAD, it strongly argues for their roles in the pathogenesis and progression of PAD in CKD.19,74 This contention is supported by a recent study involving 3758 CKD patients from the Chronic Renal Insufficiency Cohort (CRIC). 69 The results showed significant associations of PAD and several risk factors, such as high-sensitivity C-reactive protein, white blood cell count, uric acid, glycosylated hemoglobin and fibrinogen, insulin resistance and cystatin C. Collectively, an exhaustive literature supports the role of inflammation and oxidative stress in PAD pathogenesis in CKD patients.
Angiogenesis
The development of new vessels from existing ones in the setting of arterial occlusion is critical to prevent ischemic manifestations of PAD. Angiogenesis is controlled by the balance between proangiogenic and anti-angiogenic factors. Patients with CKD have reduced levels of proangiogenic mediators, such as the circulating vascular endothelial growth factor (VEGF) ligand and endostatin and circulating endothelial cells in peripheral blood. 75 CKD is also associated with the rarefaction of microcirculation in various vascular beds, including skeletal muscles, which is a reflection of defective angiogenesis in PAD and contributes to poor outcomes after vascular intervention.47,49,50 Molecularly, this defect is characterized by a decrease in the levels of hypoxia-inducible factor (HIF)-1α and its target genes, such as Angpt-2, TIE-1 and TIE-2, Flkt-1, and MMP-9, indicating an impaired hypoxia-driven angiogenesis. While there is ample evidence of impaired angiogenesis in CKD milieu, the specific mediators and pathways involved remain elusive. This represents a major area in need of investigation in CKD patients.
Uremic toxins
CKD is characterized by the retention of several solutes, which begin to accumulate in the early stages of CKD.76–78 Because these solutes unleash toxicity on various cells, they are appropriately termed as uremic toxins (see the online supplementary table). An exhaustive literature supports the association of some uremic toxins with various cardiovascular complications, including accelerated atherosclerosis, cardiac fibrosis, and endothelial dysfunction in animal models of CKD and in human patients.79–82
Uremia is a potent prothrombotic environment.83,84 Emerging evidence has uncovered prothrombotic properties of indolic uremic toxins, such as indoxyl sulfate (IS) and acetate and kynurenine. These toxins induce postintervention thrombosis by upregulating tissue factor in the vessel wall to trigger extrinsic coagulation cascade and by enhancing platelet reactivity.84–88 These toxins exert oxidative stress and inflammation, and inhibit angiogenesis, all of which along with pro-atherothrombotic effects can contribute to PAD and to the poor outcomes after vascular interventions in CKD patients.84,89–92
Various studies have examined the association of uremic toxins, such as p-cresyl sulfate (PCS), IS, and beta-2 microglobulin (B2M), with PAD in patients with CKD.93–95 Levels of PCS and IS have been associated with the incidence of PAD and are elevated in PAD patients. 94 In a small group of 72 hemodialysis patients, PCS correlated with new onset PAD. Lin et al. enrolled 100 patients on hemodialysis at a single medical center and screened them for PAD recorded as ABI and brachial–ankle pulse wave velocity (PWV) and correlated them with the serum levels of PCS and IS levels.87,93,
Despite their several modes of toxicity, which can potentially contribute to the prevalence of PAD in CKD, there is a dearth of clinical and mechanistic studies directly examining the role of uremic toxins in the induction and progression of PAD or response to vascular interventions. This area is in need of further research.
Conclusion
Overall, the problem of PAD in CKD is a growing public health concern that requires further research in several areas highlighted in this review.
The higher risk of incident PAD and CLTI in patients with CKD presents a compelling argument to increase PAD screening and awareness of the confounding effect of underlying uremic vascular disease on ABI in this population. More work is needed to understand provider and patient factors that lead to decreased utilization of interventions for revascularization in patients with CKD and PAD. Yet, at this time, patients with CKD and PAD have worsened outcomes with increased postprocedural morbidity and mortality. The increased CLTI and poor limb salvage rates, despite vascular patency in this population, highlight the need for a comprehensive analysis of vascular beds beyond the macrovasculature and the need for further work to probe for the mediators that drive this perplexing problem.
Supplemental Material
Niki_Final_Uremia_PAD_Review_12-12-18_final_supp_table_1 – Supplemental material for Unique aspects of peripheral artery disease in patients with chronic kidney disease
Supplemental material, Niki_Final_Uremia_PAD_Review_12-12-18_final_supp_table_1 for Unique aspects of peripheral artery disease in patients with chronic kidney disease by Nkiruka V Arinze, Andrew Gregory, Jean M Francis, Alik Farber and Vipul C Chitalia in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was funded in part by NCI R01CA175382, NIH R01HL132325, and the Boston University Evans Faculty Merit award (VCC); and a T32 training grant in immunobiology of trauma: T32 GM086308-06A1 (NVA).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
