Abstract
The role of vascular smooth muscle cells (VSMCs) in vascular calcification, which is related to chronic kidney disease (CKD), has been studied in greater detail in the major arteries relative to the peripheral arteries. We compared the calcifying characteristics of peripheral VSMCs relative to non-pathologic major VSMCs in patients with severe peripheral artery disease (PAD). We isolated peripheral VSMCs from the posterior tibial artery of 10 patients with CKD who underwent below-knee amputation for critical limb ischemia (CLI). Using normal human aortic VSMCs as a control group, we cultured the cells in normal and high phosphate media for 10 days, and subsequently tested by immunofluorescence staining. We compared the calcification levels between the two groups using various assays, tests for cell viability, and scanning electron microscopy. As a result, calcification of pathologic peripheral VSMCs increased significantly with time (p = 0.028) and was significantly higher than that in human aortic VSMCs in calcium assays (p = 0.043). Dead cells in the pathologic VSMC group were more distinct in high phosphate media than in human aortic VSMCs. In conclusion, VSMCs from the peripheral artery of patients with severe CKD and CLI who underwent amputation surgery showed marked calcifying characteristics compared to normal human aortic VSMCs.
Keywords
Introduction
Vascular smooth muscle cells (VSMCs) are known to play active roles in vascular calcification formation in patients.1–3 Vascular calcification does not occur singly; rather, it is a complex pathological process by VSMCs, which may differ according to histological location in the blood vessel and anatomical location in the arterial tree. 2 A recent study has demonstrated that the biology of arterial beds, and thus the involvement of VSMCs in vascular calcification, differs between anatomical sites such as the carotid and coronary arteries.4,5
Vascular calcification is observed more in patients with chronic kidney disease (CKD), which affects kidney structure or function and is assessed using variables such as the glomerular filtration rate or the presence of proteinuria. 6 Large observational studies have shown a relationship between vascular calcification and CKD. 2 In response to high extracellular phosphate in CKD, dysregulation of VSMCs is known to facilitate vascular calcification. 2 However, the results of multiple trials to regress or prevent vascular calcification in patients with CKD have been disappointing. 7
Meanwhile, peripheral artery disease (PAD) is caused by insufficient blood flow to the limbs due to the occlusion or narrowing of peripheral arteries. CKD is a significant risk factor for PAD, and the prevalence of PAD is elevated in patients with CKD by up to 37%. 8 Patients with PAD often show symptoms such as ischemic pain, claudication, and ulcers. PAD may lead to debilitating conditions such as critical limb ischemia (CLI), in which patients may require amputation. 9 The limited options for the treatment of PAD are all mechanical modalities such as surgery, or intervention such as balloon angioplasty. Vascular calcification acts as a potential driver of PAD.10–13 It also has been demonstrated to be a risk factor for the restenosis of endovascular stent therapy and increases the risk of limb amputation. 14
However, previous research regarding the role of VSMCs has been mainly based on major arteries rather than on peripheral arteries; nevertheless, PAD is no less clinically significant than diseases of major vessels, such as coronary artery disease.4,15 Previous studies on PAD focused mostly on clinical diagnosis or treatment and not on the biologic characteristics. 16 Surprisingly, studies that compare the calcifying characteristics of VSMCs in major and peripheral arteries have been limited in both number and scope. 17 Thus, we sought to identify the calcifying characteristics of peripheral VSMCs in patients with CLI, which related to CKD, under a high phosphate concentration relative to those of nonpathologic VSMCs from major arteries. We hypothesized that there are some similarities as well as differences between the two kinds of VSMCs under in vitro calcifying conditions.
Materials and methods
This study was approved by the Institutional Review Board of the Seoul National University Hospital and Myongji Hospital, Hanyang University College of Medicine. Informed consent was obtained from all subjects who participated in this study, which was performed in agreement with the principles outlined in the Declaration of Helsinki.
Patients, who met following all these criteria, were included in the study: (1) patients with CLI who showed necrosis of the foot and required below-knee amputations for severe PAD; (2) patients with CKD who had an estimated glomerular filtration rate < 60 mL/min/1.73 m2; and (3) patients with vascular calcification in the lower leg as diagnosed by simple radiographs (Figure 1). We excluded patients who (1) had a history of medial ankle injury from which we would harvest, or (2) simultaneously had other diseases which were likely to affect the outcome of vascular biopsy, such as vasculitis.
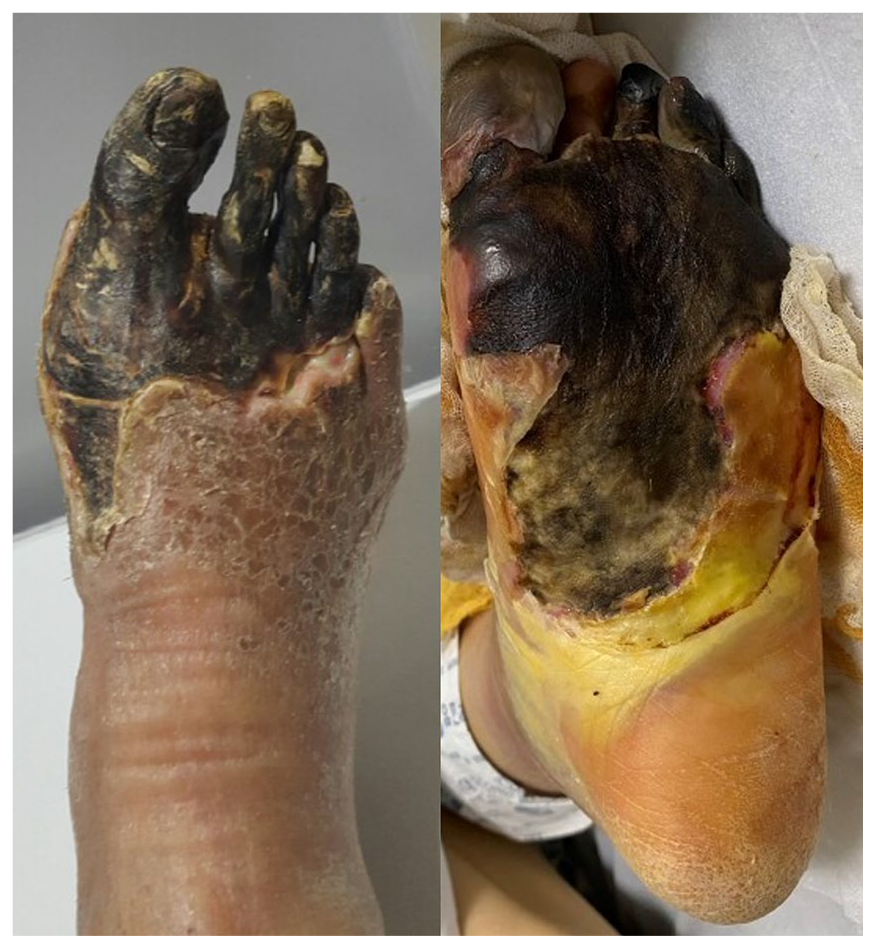
The feet of patients with PAD and CLI severe enough to undergo amputation surgery.
Final samples were obtained from 10 patients (n = 10) who underwent below-knee amputations at either the Seoul National University Hospital (n = 8) or Myongji Hospital, Hanyang University College of Medicine (n = 2).
Histology of the pathologic vessels
After a macroscopic examination, all pathologic vascular specimens were prepared in an undecalcified manner for embedding using the regular paraffin method, followed by embedding in plastic. Subsequently, the embedded samples were cut to obtain serial sections to prevent artificial damage. 18 The hematoxylin and eosin (H&E) and Masson–Goldner trichrome stains were used for light microscopy analysis. 19
Isolation and culture of human VSMCs
Vascular biopsies showing pathologies in the amputated legs of patients were obtained at the time of surgery. For isolation of pathologic peripheral VSMCs, we excised ~5 cm of the posterior tibial artery from the amputated ankle (Figure 1). Isolation and culture of the VSMCs were performed by the enzymatic tissue digestion method. 4
Human vascular aortic smooth muscle cells (catalog no. C-12533) were purchased from PromoCell (Heidelberg, Germany) or isolated from human aortas using an in-house protocol. Briefly, the arterial tissues were removed from the adventitia by scraping of the endothelium. Arterial strips were washed three times with 1× phosphate-buffered saline to remove blood, following which they were digested using 2 mg/mL of collagenase type Ⅱ enzyme (Sigma-Aldrich, Saint louis, MO, USA) for 1 h at 37°C in Dulbecco’s modified Eagle medium (DMEM; Gibco, Grand Island, NY, USA). Next, the cell suspension obtained was centrifuged, and the pelleted VSMCs were collected and seeded in six-well plates containing smooth muscle cell growth medium (SMC-GM2; PromoCell, Heidelberg, Germany), to which was added 1% antibiotic-antimycotic (Gibco). The seeded cells were incubated at 37°C in a humidified atmosphere containing 5% CO2. The VSMCs were propagated during the subsequent passages in SMC-GM2, and had an average incubation time of 7 days. Cell viability was assessed using trypan blue after each passage. All assays were conducted with subconfluent cells from passages one to three. 20 For the calcification experiments, VSMCs from both groups were grown using different media depending on the experiment; for the control group, smooth muscle cell growth medium was used, and inorganic phosphate (Pi) concentrations were adjusted using stocks of 3 mM Na2HPO4/NaH2PO4 (pH 7.4).20,21 The media was changed every 2 days in all the cultures.
Immunofluorescence staining
We performed immunofluorescence staining although did not use a cell sorting method. 22 Cells were seeded in six-well plates and grown to 50–70% confluence. Next, the confluent cells were washed twice with cold 1× phosphate-buffered saline and fixed with 4% paraformaldehyde for 10 min at room temperature. Purification of VSMCs was confirmed by immunohistological staining with monoclonal rabbit anti-alpha smooth muscle actin antibody (Abcam, Cambridge, UK), and nuclear staining was performed with the monoclonal mouse anti-transgelin antibody (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) and Hoechst 33258 (Invitrogen, Carlsbad, CA, USA).23,24
The visualization of calcified deposits
The outlook, microstructure, and crystal structure of the calcium deposits were characterized by field emission scanning electron microscopy (FE-SEM) (S-4700; Hitachi, Japan).25,26 Samples for SEM observations were prepared by fixing with 2.5% glutaraldehyde and coating them with gold or carbon. The samples were scanned on a FE-SEM in a range of 10–20 kV. 25
Calcification assays
To visualize the degree of calcification, cells were seeded in 24-well plates and grown to 70–80% confluence; cells were treated with 3 mM Pi and then incubated for 1, 3, 5, 7, and 10 days.27,28 Briefly, cells were fixed with 4% paraformaldehyde for 10 min at room temperature and stained for 10 min with 40 mM Alizarin Red S (pH 4.2; Sigma-Aldrich) at room temperature. The calcium deposits were quantified using a colorimetric kit from BioAssay Systems (Hayward, CA, USA) after solubilization of the deposits for 4 h at room temperature with 0.6 N HCl. 29 The quantity of calcium deposits was expressed as microgram per milliliter. The deposition of calcium was quantified in a separate set of identically treated cells that were decalcified with HCl after experimentation. Calcium was assayed using the QuantiChrom Calcium Assay Kit (BioAssay Systems) according to the manufacturer’s recommendations.30,31
Cell viability assay
Cell viability was analyzed using a LIVE/DEAD Cell Imaging Kit R37601 (ThermoFisher Scientific, Waltham, MA, USA) following the manufacturer’s instructions. Cells were seeded in 24-well plates and cultured for 24 h. Cells were prepared in 24-well plates overnight, and 70–80% confluent cells were treated with or without 3 mM Pi and then incubated for 10 days. After Pi treatment, LIVE/DEAD assay working solution was added to each well and these were maintained at room temperature for 15 min. Subsequently, cells were observed using a fluorescence microscope (DMi8; Leica, Hesse, Germany), and green and red fluorescence signals indicated the presence of viable cells and dead cells, respectively.30,32
Statistical analysis
The quantified data are presented as mean ± SD. Each experiment was replicated three times. The Wilcoxon test was used to compare the quantity of calcification between the 1st and the 10th day. The Mann–Whitney test was performed for comparison between two independent groups at the final day of culture in calcium assay. Statistical significance was assumed at the 95% confidence level (p < 0.05). All statistical analyses were conducted using IBM SPSS, version 19.0 (IBM Corp., Armonk, NY, USA).
Results
The mean age of patients was 71.4 years old (56–86) and all of them were men. Six had diabetes, while four had coronary artery disease vessel disease. One patient had chronic liver disease and another had cerebral infarction history.
The status of the posterior tibial arteries we harvested were variably diseased – from partially occluded to grossly not specific. H&E stain and Masson–Goldner trichrome staining revealed diffuse and thick deposits (blue-green color) in the tunica media of the undecalcified samples of peripheral arteries harvested from the posterior tibial artery of patients (Figure 2A).
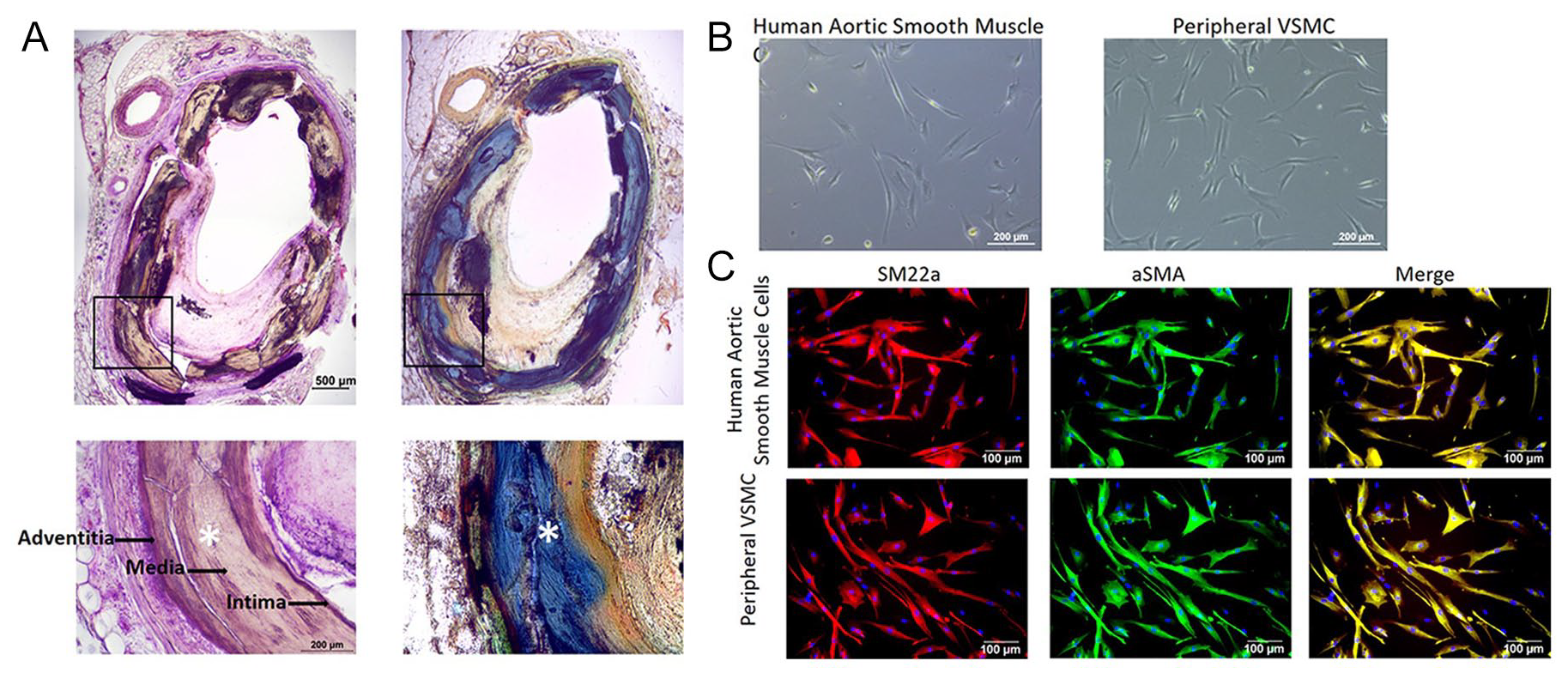
(A) H&E stain showing dense deposits (asterisks) in tunica media. Calcified matrix (blue-green color) is observed with Masson–Goldner trichrome stains. (B) VSMCs after 10 days of the culture process in general media – their morphology is elongated and sometimes triangular in shape. (C) Through immunofluorescence staining for SM22α and α-SMA, the VSMCs are confirmed as the cells we intended to culture.
After 10 days of culturing, we observed VSMCs with elongated and triangular (scarce) morphology in both groups (Figure 2B). We confirmed that the cultured cells were, in fact, the targeted VSMCs by immunofluorescence staining for SM22α and α-SMA (Figure 2C).
On the first day of cell culture under high phosphate conditions, we observed VSMCs clearly in both groups using a scanning electron microscope (Figure 3). With time, most cells died, and cell debris with calcium accumulated in the calcifying media (Figure 3).

Scanning electron microscopy of normal human aortic VSMCs and pathologic VSMCs in calcifying media obtained with a magnification of 5000 at 1 keV.
We examined calcification in pathologic VSMCs and human aortic VSMCs using Alizarin Red S staining and found that calcification was more prominent and significantly higher in pathologic VSMCs from peripheral arterial cells than in human aortic VSMCs when grown in calcification-inducing media, using the calcium assay (p = 0.043) (Figure 4A,B). Calcification of pathologic VSMCs after 10 days of culture in calcifying media was significantly elevated relative to that on day 1 (p = 0.028) and showed a significant difference between cells grown in calcifying media relative to normal media after 10 days of culture (p < 0.001) (Figure 4C). In contrast, calcification of pathologic VSMCs and human aortic VSMCs grown in normal media showed no difference after 10 days of culture (p = 0.735) (Figure 4C).
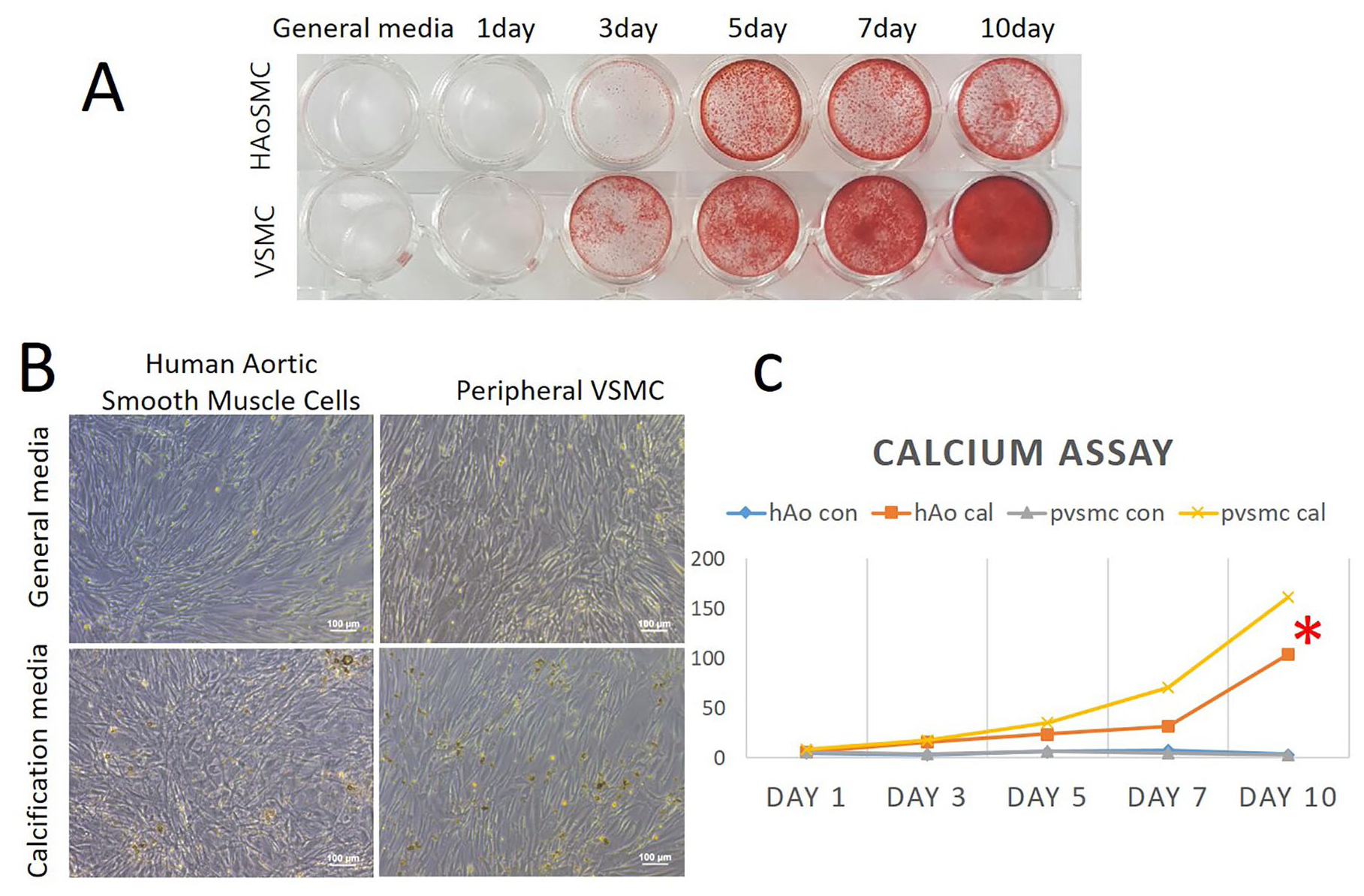
Calcium assay and quantitative analysis. (A) Alizarin Red S staining showed outstanding red stain in pathologic VSMC compared to HAoSMC in calcification media. (B) The specific shapes of VSMCs, observed in general media, was lost in calcification media. Calcium deposit in calcification media was observed more in the peripheral VSMC group. (C) Pathologic VSMCs in calcifying media showed the most calcification among four groups, followed by human aortic VSMC in high phosphate media. Asterisk indicates a significant difference of calcification between pathologic VSMC and human aortic VSMC in calcification media. Calcification of pathologic VSMC and human aortic VSMC in normal media was minimal.
Dead cells from the pathologic VSMC group in calcifying media noticeably increased compared to the other three groups: similarly grown human aortic VSMCs in general media, in calcification media, and the pathologic VSMC in general media (Figure 5A). We noted that the number of viable VSMCs from peripheral arterial cells grown in calcification media showed a slight decrease on day 7 of culture (Figure 5B).
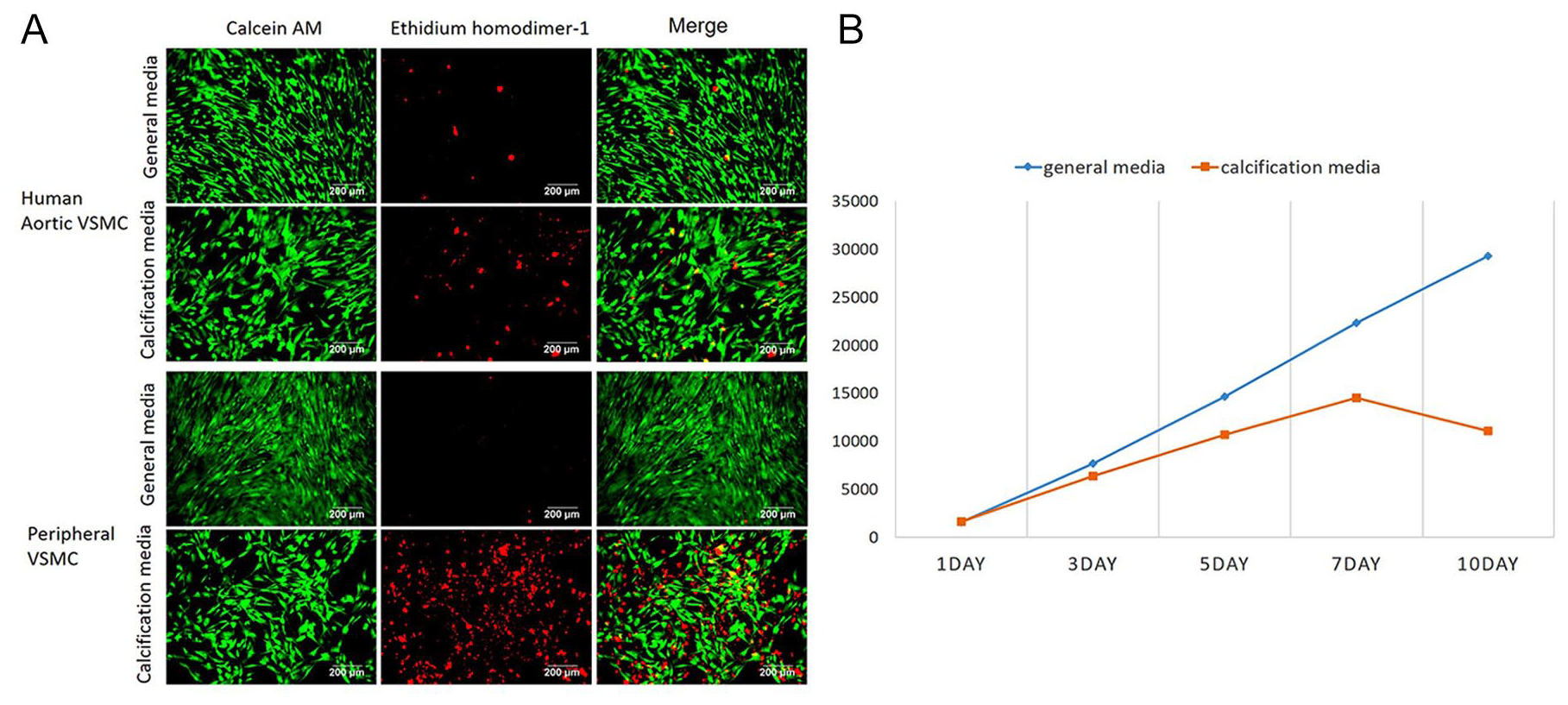
VSMCs of pathologic VSMC and human aortic VSMC in both general media and calcification media. (A) They were incubated with calcein-AM, EthD-1, and annexin V-Alexa Fluor® 647 conjugate (Life Technologies, Carlsbad, CA, USA) to label live cells (green) and dead cells (red). (B) We noted that living VSMCs in calcification media slightly decreased past 7 days.
In summary, we observed prominent medial wall calcification in samples harvested from the posterior tibial artery of CLI patients with CKD. VSMCs from the pathologic peripheral artery showed pro-calcifying characteristics relative to normal human aortic VSMCs, even after several culture passages. Further, the viability of pathologic VSMCs from the peripheral artery was poorer than that of normal aortic VSMCs when grown in calcification media.
Discussion
In vitro and in vivo studies have shown that VSMCs play an active role in generating osteochondrogenic precursors by transdifferentiation in calcifying arteries. 33 Previous clinical studies have been focused on vascular calcification in coronary arteries, carotid arteries, the aorta, or its branches. 34 The few reported studies regarding vascular calcification of the peripheral arteries, especially in patients with CKD and diabetes, have focused on the characteristics of atherosclerotic plaques in the peripheral arteries. One study reported plaque characteristics in patients with PAD undergoing primary endarterectomy, which showed a time-dependent shift towards a less lipid-rich lesion and reduced intraplaque hemorrhage. 16 Another evaluated arterial pathology in the entire length of the lower extremities. 35 There might be several differences in many aspects, including clinical presentation or calcification between major arteries and peripheral arteries. 36 However, we revealed that the pro-calcifying characteristics of VSMCs in patients with PAD were similar to those of in vitro studies reporting on VSMCs in major vessels. 9
It has been proved that elevated serum phosphate salt is a risk factor for vascular calcification among CKD populations in clinical studies.37,38 In fact, calcification of VSMC can be triggered by high phosphate treatment, by which mechanism remains unclear, and is still studied in various ways. 37 Thereafter, high phosphate-inducing methods for calcification of VSMC have been widely used for exploring the mechanism of vascular calcification. 39 We also used this method to observe the calcifying character of peripheral VSMC harvested from CKD patients.
It is well known that VSMC in pathological conditions can dedifferentiate into a synthetic phenotype and facilitate vascular calcification. There are many studies exploring various pathways involving this mechanism. 40 In contrast, this study focused on the comparison of behavior between two kinds of cells (diseased peripheral VSMC and normal major VSMC) under the same pathologic condition (high phosphate). Interestingly, cells cultured from diseased peripheral VSMC made much more calcification compared to cells cultured from normal major VSMC, although the two groups had common features of VSMCs confirmed by several results, such as cell morphology or immunofluorescent staining. Cells cultured from patients’ peripheral VSMC may have synthetic phenotypes, which enabled those outcomes. Future study would be required to explore which phenotype the cells from pathologic VSMC activated.
Under various stressful insults, VSMC has mainly three options: autophagy as a fight, senescence as an adaption, or die as an apoptosis. 41 The choice of each of these strategies likely depends on the strength of the stimulus, the time of exposure, and the state of VSMCs. 41 Previous studies suggested that a high phosphate condition acts on VSMC, by promoting vascular calcification, stimulating osteogenic/chondrogenic differentiation, inducing vesicle release, and causing apoptosis.2,42,43 We observed that the growth rate of pathologic peripheral VSMCs under high phosphate media was lower than that under general media. Moreover, the number of viable VSMCs from peripheral arterial cells grown in high phosphate media showed a slight decrease on day 7 of culture, which means increasing apoptosis of grown VSMCs.
Both medial wall calcification within the smooth muscle layer and atherosclerosis, two distinct forms of vascular calcification, are significant risk factors for cardiovascular events, altered bone and mineral metabolism, or death. 7 Vascular calcification, in the lower extremities of patients with CKD and diabetes, is reported to occur mostly in the tunica media. Approximately 20% of patients who underwent the ankle–brachial index test for CLI were found to have noncompressible arteries due to tibial artery calcification. 44 Our results from experiments using the posterior tibial artery were consistent with these studies that identified vast amounts of calcified deposits in the medial wall.
Limitations
This study has several limitations. First, we did not compare cells from the same origin, which might have biased our results. Analyzing samples from major vessel such as the aorta of the same patient from whom we harvested peripheral vessels would have provided more insights. However, it is not possible to obtain such samples due to ethical issues. So, we performed subgroup analysis for a fair comparison of the calcifying character of cells – a culture of the same specimen of each group in both general media and calcifying media. We believe that the outcome of this study could be at least a basis for further studies exploring pathologic peripheral artery, which might be different from major artery in calcifying character. To the best of our knowledge, this is the first study on the characteristics of vascular calcification in the peripheral limb arteries of patients with CKD who underwent amputation surgery for CLI. Second, the sample size of this study is small as recent medical advances have made limb salvage possible, thus reducing the need for amputation surgery. However, the characteristics of VSMCs examined in this study showed features similar to those of previous studies. Third, we could not guarantee the purity of VSMC in culture, although we employed the previous known methods as described. Moreover, it would not have significantly affected our outcomes had there been a lack of purity because we think that the majority of calcification generated in our culture was from VSMCs.
Conclusions
We observed marked calcification in VSMCs from the peripheral arteries of patients with severe CKD and CLI who underwent amputation surgery, when compared to calcification levels of normal human aortic VSMCs. We believe our study provides a basis for further research to help identify the distinct characteristics of peripheral VSMCs, especially those related to vascular calcification as a major risk factor in PAD. Thus, further studies are needed to explore the link between the pro-calcifying characteristics of peripheral VSMCs and its triggering signals.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2018R1D1A1B07046823).
