Abstract
Mondor disease is characterized by an acute painful thrombophlebitis occurring at specific anatomical sites. Data on its incidence, characteristics of clinical presentation, and course are unavailable to date. We studied the course of Mondor disease in patients diagnosed and followed at the University Hospital Zurich (Switzerland) between 2004 and 2020. The primary study outcomes were a diagnosis of active cancer either at the time of clinical diagnosis of Mondor disease or within 1 year, as well as 1-year all-cause death and recurrent Mondor disease. We included 45 patients and classified them into one of the three Mondor disease subgroups: thoracic (n = 26), penile (n = 12), or axillary (n = 7). The median age was 39 (Q1–Q3: 30–45) years and 44% of patients were men. Surgery was the likely cause of Mondor disease in 53.8% of patients with a thoracic form, 41.7% of those with a penile location, and all of those with an axillary location. Known active cancer was present in nine (20%) of 45 patients at baseline. One-year follow-up was available for 43 patients (median 94 months), whereas 6-month data were available for the remaining two patients. During the available follow-up, no patient had a new diagnosis of cancer. In conclusion, one in five patients with Mondor disease had known cancer at the time of diagnosis. During follow-up, the rate of new cancer diagnosis and death was negligible, providing reassurance about the good prognosis of this condition. Based on these preliminary data, extended cancer screening besides what is recommended by current guidelines for the general population might not be necessary in patients with Mondor disease.
Keywords
Introduction
Mondor disease (or Mondor’s disease) is characterized by thrombophlebitis at specific anatomical sites, including the anterior chest wall, penis, and axilla. Its occurrence has been frequently described in association with local triggers, such as breast fibroadenomas, surgery, infections, or trauma.1–5 Data on its annual incidence and long-term prognosis are not available to date. As is the case for other rare disorders of thrombosis and hemostasis,6,7 available information necessarily and primarily comes from isolated reports or small case series. There is a similar paucity of data regarding the characteristics of its clinical presentation and treatment patterns.1–4
As for venous thromboembolism,8,9 it has been postulated that Mondor disease may represent the signal of an underlying cancer and, therefore, support its early diagnosis.1,5 Current evidence does not support the implementation of extensive cancer screening after unprovoked venous thromboembolic events 10 due to its relatively low prevalence/incidence. Nevertheless, efforts are being undertaken to identify the patient population, which possibly benefits from such a strategy. In Mondor disease patients, this decision can solely be based on a subjective decision of the treating physician in the absence of data from longitudinal studies.
In this analysis, we aimed to study the prevalence and 1-year incidence of active cancer diagnosis and death in patients with confirmed Mondor disease evaluated at a single center, as well as its clinical characteristics and treatment patterns.
Methods
The present study was conducted as a retrospective analysis of medical charts of patients with Mondor disease. We performed an initial screening of all patients with suspected Mondor disease who had been treated at the University Hospital Zurich (USZ) over a 16-year period (2004–2020). The final data analysis was performed in September 2020.
The final study population consists of adult patients with an established diagnosis of Mondor disease, as defined by the presence of a painful (in most cases), palpable, cord-like induration on body surfaces after the exclusion of other local pathological conditions. Cases of Mondor disease were included if they could be classified according to the three typical presentations: (i) ‘classic presentation’, involving the anterolateral thoraco-abdominal wall or breast; (ii) ‘penile presentation’ with dorsum and dorsolateral involvement of the penis; and (iii) the ‘axillary web syndrome’ with the mid-upper arm involved.
The primary outcomes of this study were: (i) the diagnosis of active cancer either at the time of clinical diagnosis of Mondor disease or within 1 year of diagnosis; (ii) death from all causes within 1 year from diagnosis; and (iii) recurrent Mondor disease within 1 year from diagnosis. Active cancer was defined as cancer diagnosed or treated ⩽ 6 months before Mondor diagnosis and recurrent or metastatic cancer – including locally or regionally advanced cancer other than basal cell or squamous cell skin cancer.
The secondary outcomes of this study were the prevalence of comorbidities known at the time of clinical diagnosis and the use of anticoagulants, antiplatelets, pain killers, and topic therapy.
Follow-up data were collected in a pseudonymized fashion from available medical reports (both outpatient and inpatient discharge letters) recorded in the online patients’ charts after the diagnosis of Mondor disease. No deviation from standard of care procedures concerning the collection of new clinical data, invasive procedures, laboratory analysis, telephone contact, or storage of blood material were necessary. After 2015, a general consent for the use of personal data for research purposes was implanted at our institution. Personal data of patients diagnosed between 2004 and 2015 could be used if the patient could not be reached to retrospectively collect a signed general consent (article 34 of the Human Research Law, HFG). Personal data of patients diagnosed after 2015 could not be used in the absence of a written general consent. The study protocol was approved by the Ethical Commission on 16 April 2020 (ID 2020-00292).
Descriptive analyses of the baseline characteristics used counts and percentages for categorical data, whereas continuous data were expressed as mean (SD) or median (Q1–Q3). The prevalence at baseline and 1-year incidence of the outcomes of interest were presented together with the corresponding 95% CI to provide readers with a measure of precision for our estimates. IBM SPSS version 25.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis.
Results
Characteristics of the study population
Our search of the institutional electronic patient database (2004–2020) by the keyword ‘Mondor’ identified 62 cases in whom the diagnosis of Mondor disease was suspected. Of those individuals, the diagnosis was confirmed in 45, who were classified in one of our predefined subgroups: thoracic (n = 26, 57.8% of total), penile (n = 12, 26.7%), or axillary (n = 7, 15.6%). The other 17 patients were initially identified by that keyword, but not included in the final analysis, as it was reported that ‘the diagnosis of Mondor disease was excluded’. No patient was excluded due to lack of a general consent before 2015.
Complete 1-year follow-up data were available for all patients, excluding two who had been diagnosed with Mondor disease approximately 6 months before the final data analysis. Owing to the investigation period 2004–2020, a follow-up analysis was possible in the range 6 months to nearly 16 years, depending on the date of initial diagnosis. Median follow-up was 94 (Q1–Q3: 61–133) months.
Baseline characteristics of the total study population and subgroups are summarized in Table 1. In brief, 20 patients (44%) were men and the median age was 39 (Q1–Q3: 30–45) years. In the table, we also provided a descriptive synthesis of the prevalence of comorbidities and concomitant medications at diagnosis across Mondor disease subgroups. Of note, two patients (4.4%) had a history of deep vein thrombosis: one patient had isolated distal deep vein thrombosis and the other had recurrent lower-extremity deep vein thrombosis.
Demographics and baseline characteristics of patients diagnosed with Mondor disease.
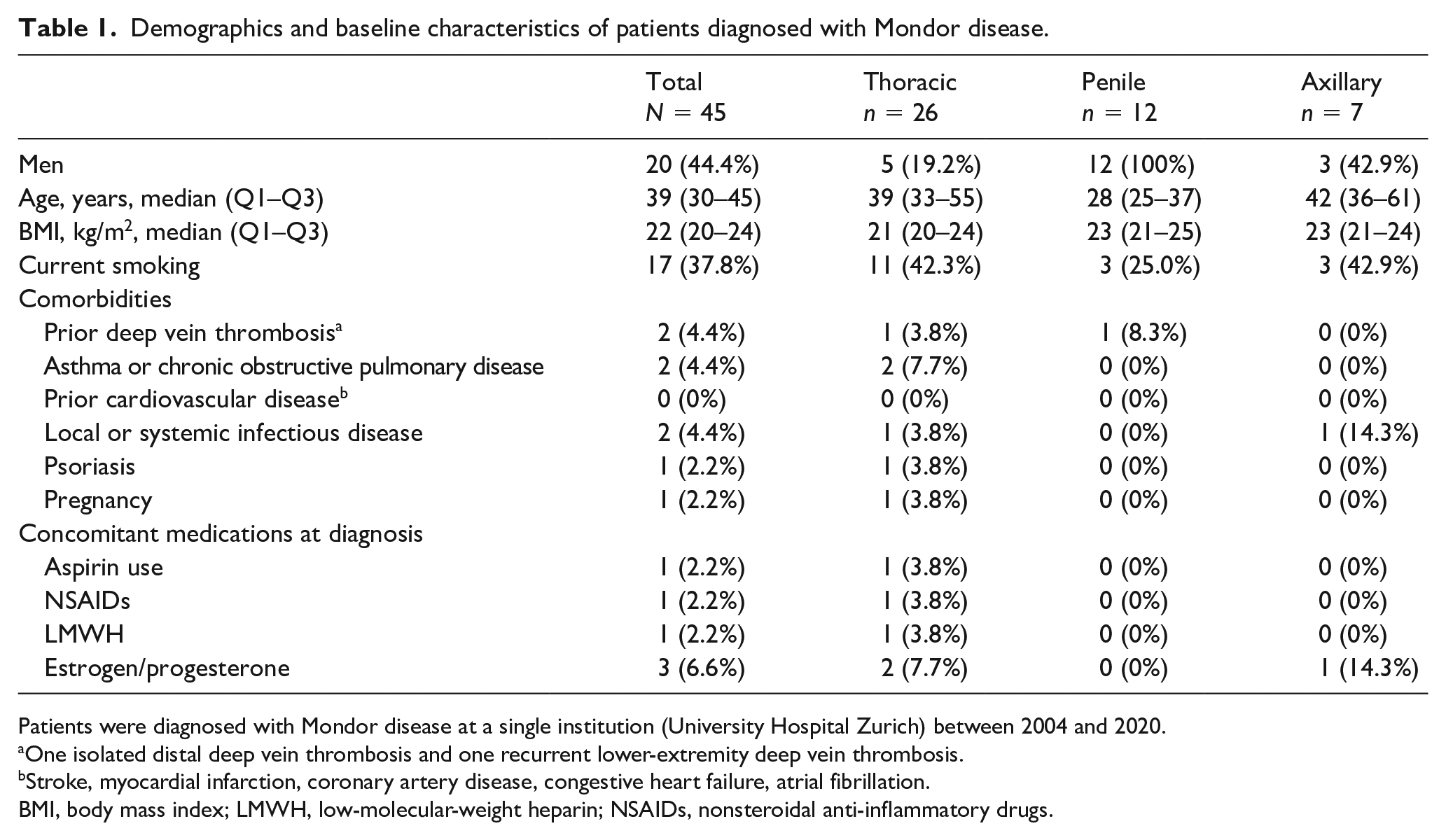
Patients were diagnosed with Mondor disease at a single institution (University Hospital Zurich) between 2004 and 2020.
One isolated distal deep vein thrombosis and one recurrent lower-extremity deep vein thrombosis.
Stroke, myocardial infarction, coronary artery disease, congestive heart failure, atrial fibrillation.
BMI, body mass index; LMWH, low-molecular-weight heparin; NSAIDs, nonsteroidal anti-inflammatory drugs.
In the overall population, the median hemoglobin level was 134 g/L (Q1–Q3: 120–140; n = 17 missing). Median values of platelet count and white blood cells were 254 g/L (Q1–Q3: 215–284; n = 17 missing) and 7 g/L (Q1–Q3: 5.8–8.1; n = 17 missing), respectively. Renal function was within normal values (estimated glomerular filtration rate (eGFR) > 60 mL/min) in all patients.
Initial clinical presentation and treatment
The most prevalent initial signs and symptoms were a palpable cord in 32 patients (71.1%) and pain in 25 patients (55.6%), respectively (Table 2 and Figure 1). An imaging test to complement the clinical diagnosis or due to another medical indication was found for 25 patients (55.6%), consisting of ultrasonography in one-third of cases. Of note, two patients (4.4%) received computed tomography (CT) or positron emission tomography (PET)-CT scans, respectively, for other reasons (cancer staging).
Clinical presentation of Mondor disease, diagnostic tools, and medical treatment.
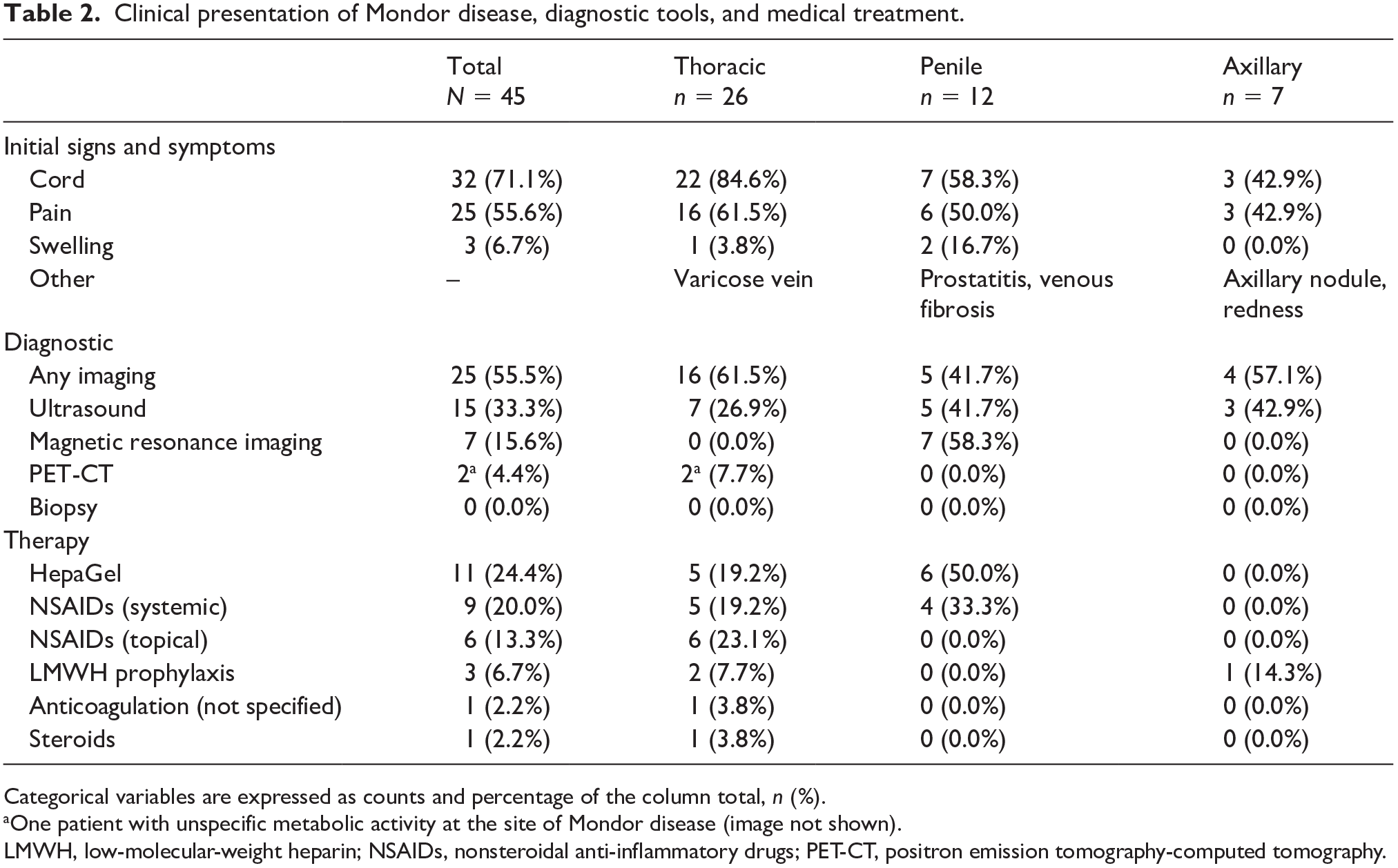
Categorical variables are expressed as counts and percentage of the column total, n (%).
One patient with unspecific metabolic activity at the site of Mondor disease (image not shown).
LMWH, low-molecular-weight heparin; NSAIDs, nonsteroidal anti-inflammatory drugs; PET-CT, positron emission tomography-computed tomography.
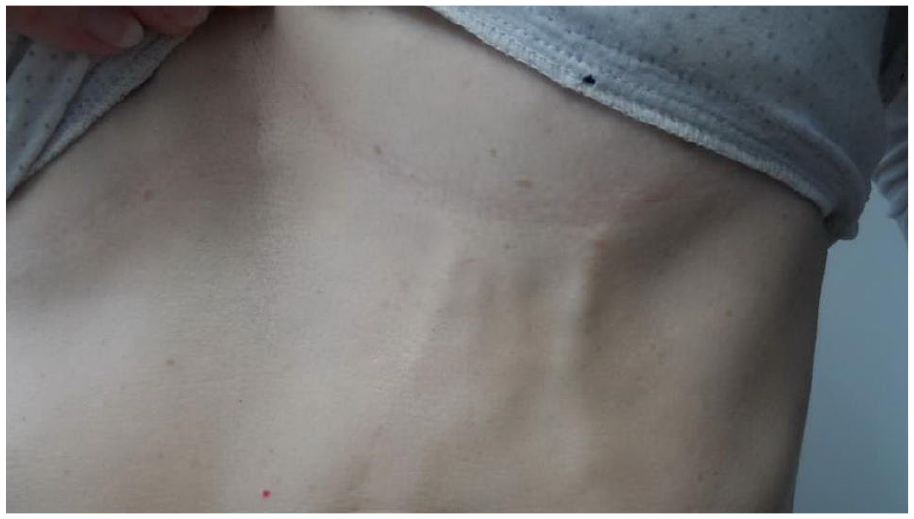
Example of the typical clinical appearance of Mondor disease (thoracic variant) in a young woman.
Concerning treatment, two patients (4.4%) had been receiving aspirin or low-molecular-weight heparin (LMWH) before the diagnosis of Mondor disease. The acute therapy of Mondor disease consisted of heparin compounds in 11 patients (24.4%). Systemic nonsteroidal anti-inflammatory drugs (NSAIDs) were used in nine patients (20%) and topical NSAIDs in six patients (13.3%). LMWH (three patients, 6.7%), systemic anticoagulation (one patient, 2.2%), and steroids (one patient, 2.2%) were rarely used (Table 2). No patient required an escalation of the medical therapy due to worsening of symptoms.
Prior surgery was identified by the treating physicians who managed the patients as the most likely cause of Mondor disease in 14 patients (53.8%) with thoracic, in five patients (41.7%) with penile, and in seven patients (100%) with axillary location. The most common type of surgery was reconstructive/aesthetic breast surgery, which was performed in eight (30.8%) out of 26 patients of the thoracic subgroup. Other potential causes, as described previously in the literature,1–5 are listed in Table 3. One patient was diagnosed with thoracic Mondor’s disease and had a history of psoriasis (that affected the scalp and nails). In the group with the penile form, three patients (25%) were MSM (men who have sex with men). One patient was found to have prostatitis. In the group with the axillary form, one patient had prior blood draw. Another patient had an acute HIV infection.
Plausible causes of Mondor disease.
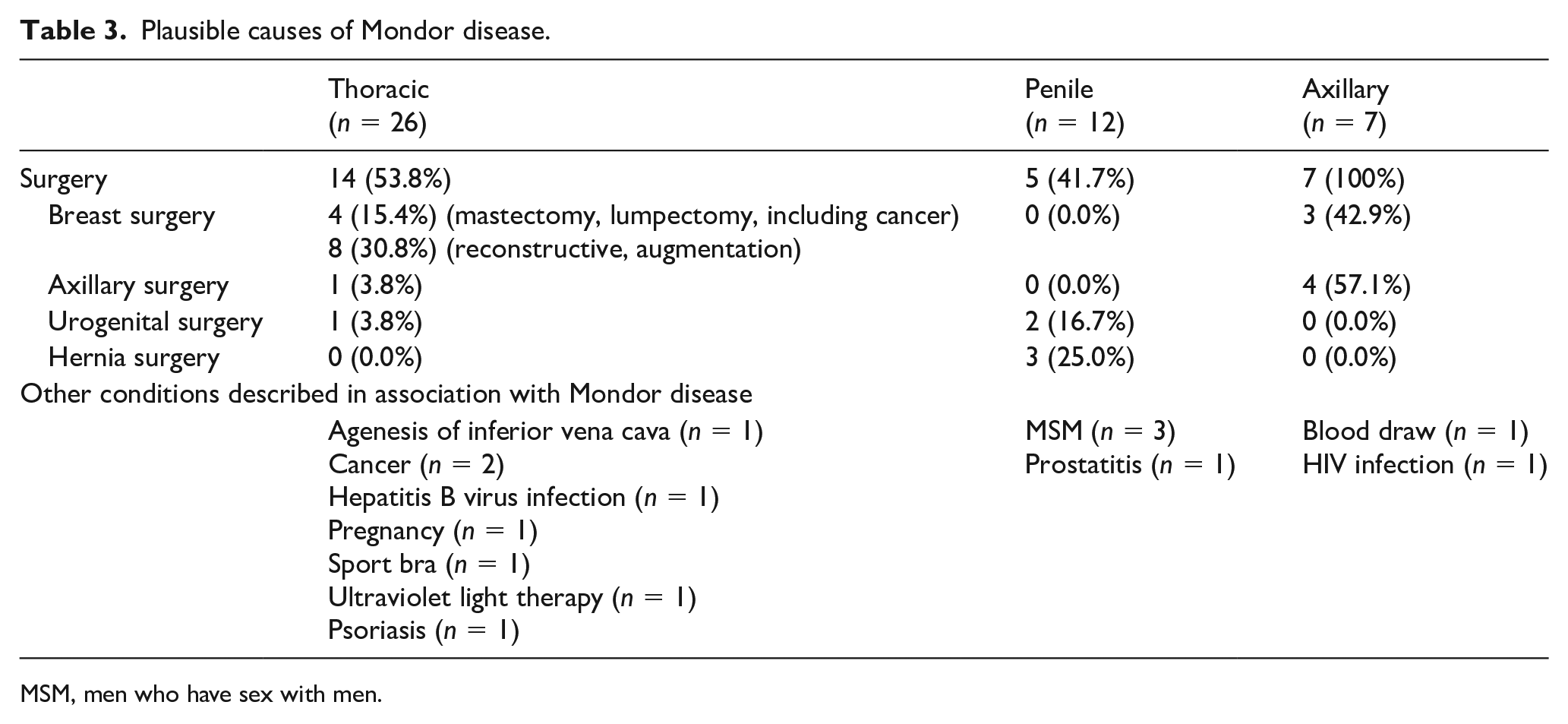
MSM, men who have sex with men.
Study outcomes
Known active cancer was present in nine (20%) of 45 patients at baseline, consisting of breast cancer in five patients (11.1%), melanoma in two patients (4.4%), and pancreatic cancer and germ cell carcinoma in one patient each (2.2%) (Table 4). The median age of cancer and noncancer patients was 49 and 36 years, respectively. Of nine patients with cancer, eight (89%) underwent surgery compared with 11 patients (31%) without cancer.
Baseline prevalence and 1-year rate of cancer and mortality.
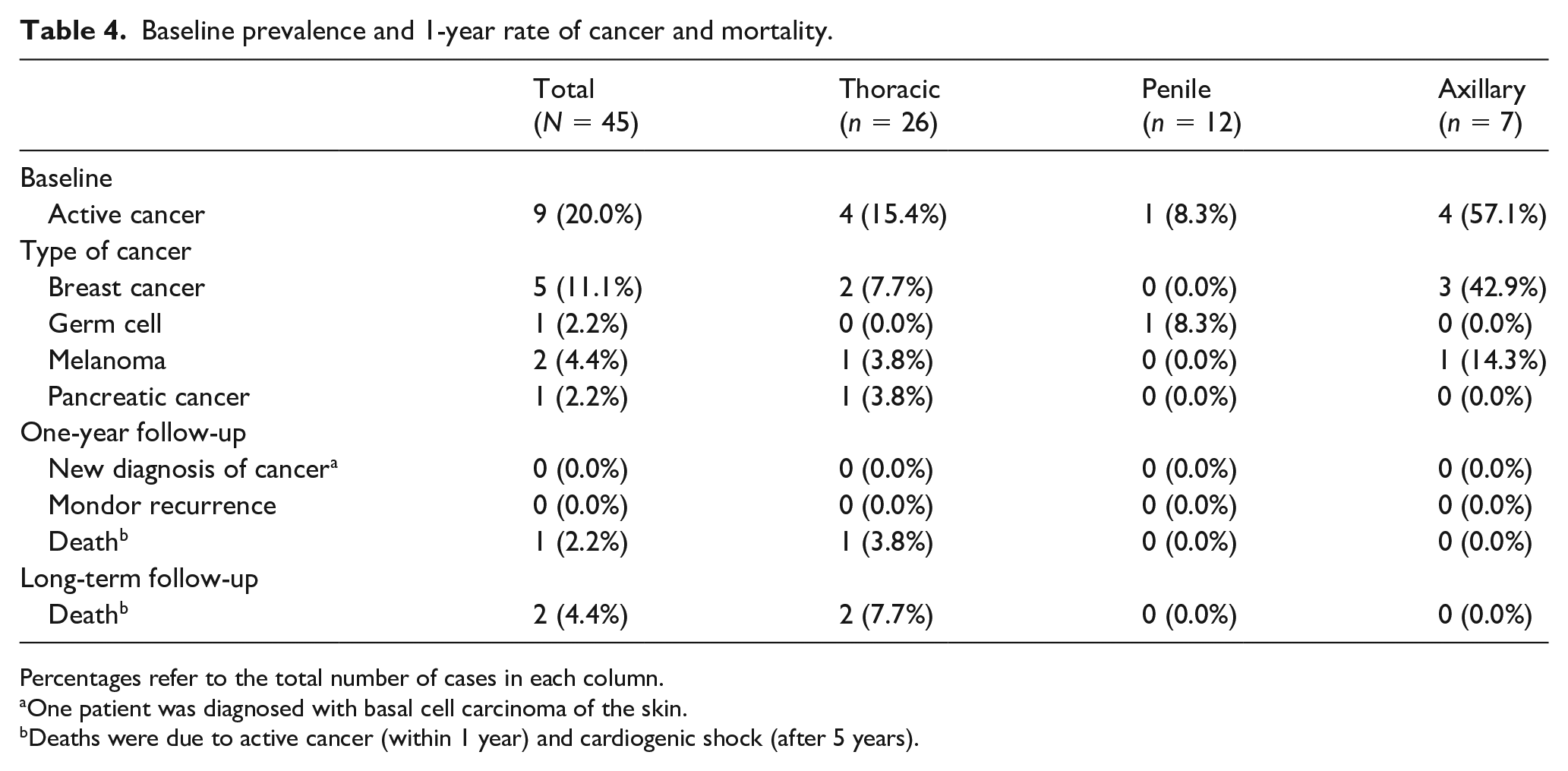
Percentages refer to the total number of cases in each column.
One patient was diagnosed with basal cell carcinoma of the skin.
Deaths were due to active cancer (within 1 year) and cardiogenic shock (after 5 years).
During 1-year follow-up (6 months only in n = 2), no patient had a new diagnosis of cancer. One patient was diagnosed with basal cell carcinoma of the skin, not included in the definition of active cancer, during the first year.
None of the patients had an episode of recurrent Mondor disease. Two patients with thoracic Mondor disease (4.4% of total, 7.7% of those in the thoracic group) died during long-term follow-up, of whom one (2.2% of total) within 1 year due to pancreatic cancer already known at baseline. The second patient died of cardiogenic shock 5 years after Mondor disease diagnosis.
Discussion
To the best of our knowledge, this represents one of the largest cohort studies of Mondor disease in the contemporary era and the first one with long-term follow-up data on the incidence of new cancer diagnosis and all-cause death after the diagnosis of Mondor disease. Our results contribute to better define the clinical presentation and treatment patterns of this condition and provide initial answers to long-standing questions concerning long-term management, notably cancer screening and long-term prognosis. Indeed, the results of our retrospective study must be carefully interpreted, as these patients had not been regularly examined for cancer after the diagnosis of Mondor disease. In our patient cohort, we found that the prevalence of known cancer at baseline (at the time of Mondor disease diagnosis) was substantial, corresponding to one-fifth of the patients. However, all cases of (active) cancer were already known and no new diagnosis of cancer was made during follow-up, whereas overall mortality was negligible. As preliminary as these data are, we suggest that Mondor disease should not be seen per se as a marker of poor prognosis and might not necessitate additional imaging evaluation deviating from any age- and sex-specific risk-based screening suggested by national/international guidelines.
Our findings of the prevalence of cancer at the time of Mondor disease diagnosis are partly in line with prior studies. In a cohort study from the 1980s, the authors reported a 12% prevalence of breast cancer among thoracic Mondor disease. 5 Other case reports reported on this association.11,12 Taken together, these observations led to speculation that mammography may be recommended to patients with Mondor disease of the chest wall and had been uncritically reported for the following decades. While we confirmed that the conditional probability of having cancer is high in patients diagnosed with Mondor disease, we could not demonstrate the opposite, namely that Mondor disease is a marker of occult cancer.
Our data on long-term follow-up confirmed that the prognosis of this group of patients is favorable, with only two deaths recorded over a 10-year follow-up, corresponding to a cumulative survival of more than 95%. No cases of Mondor disease recurrence had been recorded, suggesting that Mondor disease is probably related to transient changes or local complications of a recent surgery, more than a pro-coagulable status reflecting the severity of the underlying disease. Consistently, an asymptomatic course of the disease was observed, although only a minority of patients received systemic anticoagulation or anti-inflammatory therapy.
The pathophysiology of Mondor disease remains largely unknown, but possibly does not differ from elements of the classic Virchow’s triad, which are presented alone or in combination by most patients, which were present in most patients and included typical risk factors for venous thrombosis, such as prior surgery, trauma or local mechanical triggers, infections or inflammation, and cancer. Owing to the lack of a proper control group, this assumption on potential risk factors remains speculative.
Study limitations
Our study has limitations. First, no systematic cancer screening was performed in this study population. We partially accounted for this fact by evaluating the 1-year rate of cancer diagnosis and death, and further extending this time window to the latest timepoint available (up to 10 years in some patients). Having evaluated the electronic data from a large institutional database, including all the specialist visits, admissions, laboratory analyses, and imaging tests available for the individual patient, we are reassured that no new diagnoses or novel clinical manifestations of a neoplastic disorder had been missed. Second, this study was conducted in a relatively small cohort of patients, limiting the precision of our estimates. Based on the precision of our estimates, we cannot exclude that the 1-year rate of occult cancer diagnosis may be as high as 7%, although we observed no new cases. Finally, the definition of Mondor disease was necessarily based on what was reported by the treating physicians: from this perspective and in the absence of predefined criteria for diagnosis, both under- and overdiagnosis are possible. Indeed, we cannot exclude that a few cases of Mondor disease could have been missed (underdiagnosis), especially in patients with severe concomitant conditions, notably cancer, which may have been prioritized. One more limitation is the fact that the search parameters were based on the keyword ‘Mondor’, so other similar diagnoses like ‘penile thrombophlebitis’ or ‘abdominal wall thrombophlebitis’ may have been missed. Vice versa, it is conceivable that some of the patients may not have had Mondor disease, but, instead, an alternative diagnosis: for instance, this may be the case of sclerosing lymphangitis of the penis, a benign condition consisting of a firm cord-like swelling, which may be clinically indistinguishable from ‘penile’ Mondor disease.
Conclusion
In this single-center study including all patients with Mondor disease diagnosed at an academic institution over 16 years (2004–2020), we observed a substantial prevalence of known active cancer and recent surgery at the time of Mondor disease diagnosis. During a follow-up of at least 6 months for all patients and at least 1 year for 43 of 45 patients, no new diagnoses of (occult) cancer were made, providing reassurance about the benign course of this condition. Based on these preliminary retrospective data, an extended cancer screening, besides what is recommended by current guidelines for the general population, might not be necessary in patients with Mondor disease.
Footnotes
Declaration of conflicting interests
Stefano Barco has received congress and travel payments from Daiichi-Sankyo, Boston Scientific, and Bayer HealthCare, institutional grants from Sanofi and Bayer HealthCare, and personal fees and honoraria from Bayer HealthCare, LeoPharma, Boston Scientific, and Daiichi-Sankyo. The other authors have no conflicts of interest to report.
Funding
The work of Stefano Barco was partly supported by the German Federal Ministry of Education and Research (BMBF 01EO1003 and 01EO1503).
