Abstract

Keywords
Hereditary hemorrhagic telangiectasia (HHT) is a rare autosomal dominant disease, affecting approximately 1 in 8000 and complicated by vascular malformations (VMs).1,2 Patients are at risk of life-threatening complications from VMs in the lungs and brain, which can be treated preventatively. 3 We report here the current routine screening practice for lung and brain VMs across North American HHT Centers, with the goal of raising awareness of current standard practices. This is particularly relevant given the existing international controversy regarding brain VM management.
We designed a brief survey (see online supplemental material) to collect information about screening practices for pulmonary arteriovenous malformations (AVMs) and brain VMs in adult and pediatric (age < 18) patients with HHT. The survey, open from September 17 to 20, 2019, was circulated by email to Center Directors at all 28 North American HHT Centers of Excellence (See www.curehht.org). Research ethics board approval for this research was not required, as no patients were involved.
Center Directors from all 28 HHT Centers of Excellence responded (100% response rate). A total of 32 Directors responded, as four Centers have co-directors. For those sites, results agreed and were combined. Directors reported a mean HHT patient experience of 13.4 years (SD = 8.0 years) and were following a mean of 621 patients with HHT (range 70–3500).
Survey results are presented in Table 1. For adult patients with HHT, 100% of Center Directors routinely screened for pulmonary AVMs and brain VMs, with transthoracic contrast echocardiography (TTCE) and brain MRI, respectively. For children, 82% of Center Directors routinely screened for pulmonary AVMs using a variety of protocols including clinical evaluation, supine and upright pulse oximetry, chest radiography, or TTCE, and 89% routinely screened for brain VMs, using MRI.
Survey results regarding screening practices for lung AVMs and brain VMs from 28 North American HHT Centers of Excellence.
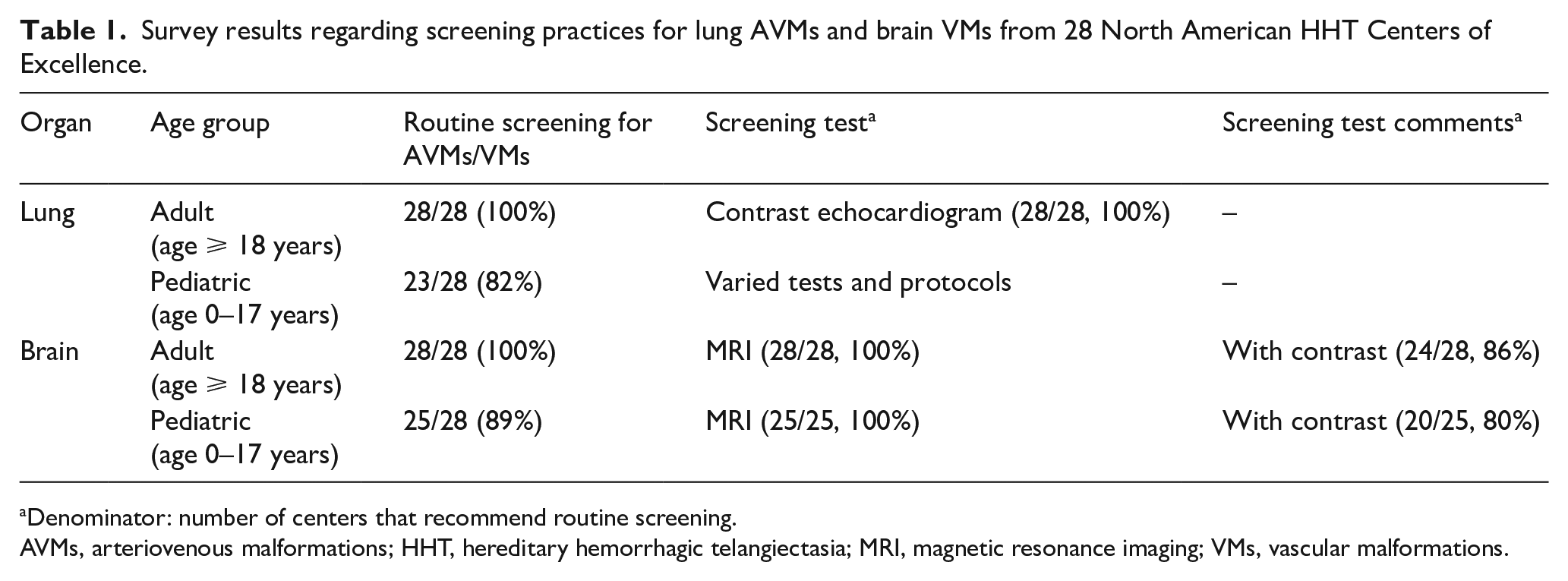
Denominator: number of centers that recommend routine screening.
AVMs, arteriovenous malformations; HHT, hereditary hemorrhagic telangiectasia; MRI, magnetic resonance imaging; VMs, vascular malformations.
For adults, routine pulmonary AVM screening is recommended at all HHT Centers, using TTCE, as per HHT guidelines. 4 This demonstrates that HHT guidelines recommendations have clearly translated into standard clinical practice across North American HHT Centers of Excellence. In addition, we report that 82% of North American HHT Centers are routinely recommending pulmonary AVM screening for children, in keeping with international guidelines. 4 The centers not routinely recommending screening for pulmonary AVMs in children reported that they were not routinely caring for pediatric patients. Consistent with guidelines 4 and subsequent literature, 5 protocols for screening varied. However, the majority of centers included contrast echocardiography in their screening protocol for children.
At the time of the first HHT International Guidelines (literature review and consensus process in 2006), 4 there were international disparities in expert opinion and in practice with regards to screening for brain VMs, with North American HHT experts supporting a recommendation for screening, but European experts not recommending. The discussions at that time reflected a cultural difference around the role of screening when preventative therapy is not recommended in all brain VM patients. 6 This is not surprising and has been described in other clinical situations, such as breast and other cancer screening – where there is higher reported uptake in Americans than in Europeans. 7 Contributing factors may include cultural values around screening and risk, health systems structure, and screening costs. Recently, the European position was reiterated in a position statement from the European Reference Network for Rare Vascular Diseases (VASCERN) on brain VM screening in adults and children with HHT. 8
A 2017 systematic review and meta-analysis concluded that the prevalence of brain VMs in patients with HHT is approximately 10%. 9 The controversy around brain VM screening stems from the risks of treatment for brain VMs, including hemorrhage, neurological deficit, and infection.4,6 As such, preventative intervention is not recommended for all patients; the risks of the treatment must be balanced against the risk of spontaneous intracranial hemorrhage without treatment.8,10–13 Since publication of the HHT International Guidelines, 4 the controversy regarding brain VM screening has continued to evolve. Non-HHT brain VM studies, such as the ARUBA trial, have suggested that sporadic brain VMs, if asymptomatic, may be best left untreated. 14 However, the ARUBA trial excluded patients with HHT, and its design has raised many concerns.15–17 Thus, its relevance to the HHT brain VM controversy is uncertain. In HHT-specific studies, the average risk of intracranial hemorrhage from brain VMs is estimated to be ‘low’ at 0.5–2% per year.8,11,13 However, most of these adult studies are limited by survivor bias, and therefore likely underestimate risk of intracranial hemorrhage. A systematic review and meta-analysis of literature regarding brain VMs in HHT found that a high proportion of patients with HHT with brain VMs will experience symptoms (50%) or AVM-related hemorrhage (20%), further highlighting the role of screening in patients with HHT, which may allow for intervention prior to symptoms or complications. 9 Patients with HHT with evidence of previous rupture have the highest risk of prospective hemorrhage, in the range of 10% per year. 13 Other factors, such as imaging phenotype12,18 and VM size, may predict hemorrhage, though large prospective studies are pending.
Evidently, interpretation of the literature and perceptions of the risks and benefits related to brain VM screening and treatment differs significantly between Europe and North America; this is also evident in clinical practice. All North American HHT Centers recommend routine screening for brain VM in adult patients with HHT, whereas only one out of eight VASCERN HHT Centers routinely does so. 8
We report here that the current standard practice across North American HHT Centers is to routinely recommend screening for pulmonary AVMs and brain VMs in adults and children. Preventative management is currently recommended for pulmonary AVMs. 4 For brain VMs, routine screening at North American HHT Centers allows for the identification of patients at highest risk of intracranial hemorrhage and for a multidisciplinary team to engage the patient in a risk–benefit discussion regarding brain VM treatment. Future research is needed to identify predictors of intracranial hemorrhage amongst patients with HHT with brain VMs, as well as the outcomes of brain VM treatment in HHT, to help guide clinical decision-making regarding screening and treatment. In addition to brain VM and pulmonary AVM screening, the role of routine screening for hepatic AVMs is a topic of research and consideration. As such, a repeat survey regarding organ VM screening practice, including center-specific reporting, may help characterize changes in clinical practice over time. Moreover, future research should address determinants of quality care for patients with HHT, such as undergoing organ VM screening and considering patient, social, center, and health services factors.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X20974452 – Supplemental material for Screening for pulmonary and brain vascular malformations is the North American standard of care for patients with hereditary hemorrhagic telangiectasia (HHT): A survey of HHT Centers of Excellence
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X20974452 for Screening for pulmonary and brain vascular malformations is the North American standard of care for patients with hereditary hemorrhagic telangiectasia (HHT): A survey of HHT Centers of Excellence by Alexandra Kilian, Marianne S Clancy, Scott Olitsky, James R Gossage and Marie E Faughnan in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
