Abstract
Limited data exist that comprehensively describe the practical management, in-hospital outcomes, healthcare resource utilization, and rates of post-hospital readmission among patients with submassive and massive pulmonary embolism (PE). Consecutive discharges for acute PE were identified from a single health system over 3 years. Records were audited to confirm presence of acute PE, patient characteristics, disease severity, medical treatment, and PE-related invasive therapies. Rates of in-hospital major bleeding and death, hospital length of stay (LOS), direct costs, and hospital readmission are reported. From January 2016 to December 2018, 371 patients were hospitalized for acute massive or submassive PE. In-hospital major bleeding (12.1%) was common, despite low utilization of systemic thrombolysis (1.8%) or catheter-directed thrombolysis (3.0%). In-hospital death was 10-fold higher among massive PE compared to submassive PE (36.6% vs 3.3%, p < 0.001). Massive PE was more common during hospitalizations not primarily related to venous thromboembolism, including hospitalizations primarily for sepsis or infection (26.8% vs 8.2%, p = 0.001). Overall, the median LOS was 6.0 days (IQR, 3.0–11.0) and the median standardized direct cost of admissions was $10,032 (IQR, $4467–$20,330). Rates of all-cause readmission were relatively high throughout late follow-up but did not differ between PE subgroups. Despite low utilization of thrombolysis, in-hospital bleeding remains a common adverse event during hospitalizations for acute PE. Although massive PE is associated with high risk for in-hospital bleeding and death, those successfully discharged after a massive PE demonstrate similar rates of readmission compared to submassive PE into late follow-up.
Introduction
Pulmonary embolism (PE) is a significant public health problem, representing a leading cause of cardiovascular death, and is attributable to a rising number of hospital admissions and associated health care costs. 1 Acute PE manifests along a spectrum of clinical severity, from asymptomatic minor PE to life-threatening, massive PE. However, over half of patients admitted with pulmonary embolism present with features that represent an intermediate or high risk of 30-day complications.2,3 Survivors of PE suffer many chronic health issues, including persistent right ventricular (RV) dysfunction, reduced exercise capacity, and poor health status.4,5 Despite advances in risk stratification and care coordination for PE, treatment variability and in-hospital mortality rates remain high.6,7
The primary objectives for PE treatment are reduction in acute RV strain, symptom improvement, and reduction in long-term complications, including chronic thromboembolic pulmonary disease, functional impairment, and death. While single-arm and randomized treatment studies provide an important basis for current recommendations for PE management, the outcomes of PE treatment are represented by very few patients given the overall burden of disease. Furthermore, important exclusion criteria limit generalizability of these results. The societal burden of PE includes not only the high costs of acute care, 8 particularly among those requiring admission to the intensive care unit (ICU), 9 but also the costs of disease recurrence, readmission, and late complications. 10 Outcomes from long-term post-hospitalization follow-up are also lacking from many seminal PE treatment trials, leaving important questions about the true burden of PE unanswered. Therefore, the impact of evolving treatments on outcomes for submassive and massive PE remain uncertain in routine clinical practice.
We sought to comprehensively describe the in-hospital outcomes and resource utilization among a large cohort of consecutive patients with acute submassive and massive PE that were hospitalized in a single metropolitan health system. Patient and treatment characteristics were evaluated for association with a composite outcome of in-hospital bleeding and mortality as well as rates of readmission and in-hospital resource utilization, including costs of care.
Materials and methods
Study design and population
All consecutive adult patients admitted to Saint Luke’s Hospital of Kansas City and Saint Luke’s East Hospital of the Saint Luke’s Health System (SLHS) between January 1, 2016 through December 31, 2018 with an International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) code for PE (see online supplementary Table 1) on the hospital discharge summary were retrospectively identified and considered for study inclusion. All charts were manually audited to confirm details of index hospitalization for PE, including date and time of PE diagnosis, vital signs at diagnosis, invasive treatment modality, if performed, and assignment of PE subtype. Patients confirmed to have acute PE during index hospitalization were included for analysis in the Saint Luke’s Outcomes of Pulmonary Embolism (SLOPE) study (Figure 1). The Saint Luke’s Institutional Review Board reviewed the study protocol and approved the study. Informed consent was waived given the retrospective nature of the analysis.
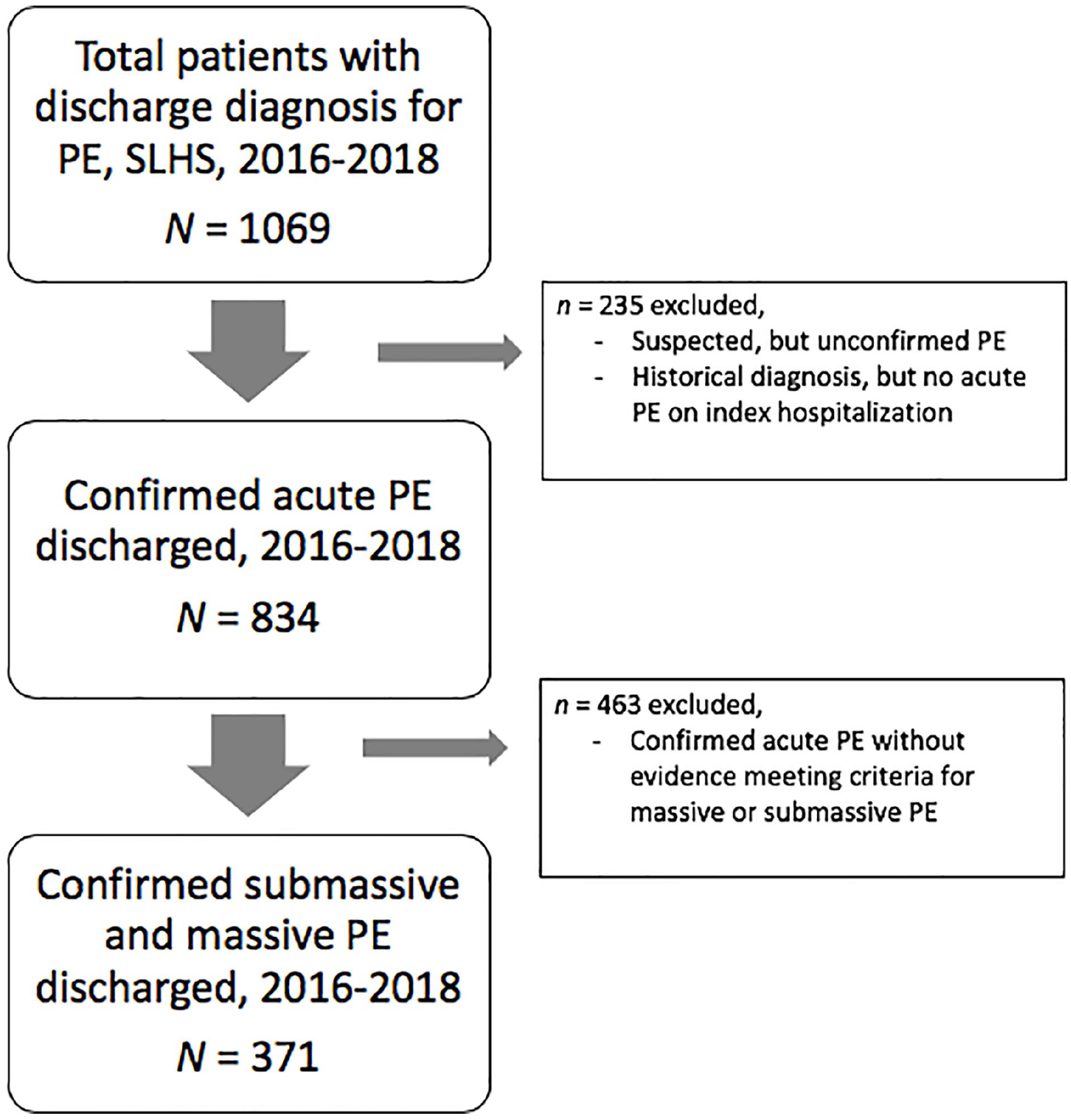
Study flow diagram of acute PE in the SLHS.
Manually abstracted clinical details of the hospitalization were entered into an institutionally licensed Research Electronic Data Capture (REDCap) online database (Vanderbilt University, Nashville, TN, USA) hosted at SLHS.11,12 Institutional databases were screened for all-cause hospital readmissions and all-cause death through March 31, 2019, ensuring at least 3 months of chart follow-up for all subjects.
Study population
Patients were eligible if they were > 18 years of age, had a radiologic- or echocardiographic-confirmed acute PE, and had clinical features indicating submassive or massive PE. Diagnostic features of primary PE included a new filling defect within the pulmonary arteries on chest computed tomography (CT) angiogram, high-probability ventilation/perfusion (VQ) scan, or visualized thrombus-in-transit with evidence of RV strain on echocardiogram.
Definitions
Massive PE was defined as acute PE associated with cardiac arrest or systemic hypotension. Systemic hypotension was defined as systolic blood pressure < 90 mmHg of > 15 minutes in duration and requiring vasopressors. Submassive PE was defined as acute PE without hemodynamic compromise or cardiac arrest, but with any of the following criteria: evidence of RV strain, including CT angiography-assessed right ventricular-to-left ventricular ratio (RV/LV) of > 0.9, or echocardiographic evidence of RV dilatation or RV hypokinesis; an elevated N-terminal-pro-brain natriuretic peptide (NT-pro-BNP) level (defined as ⩾ 500 pg/mL); or an elevated troponin-I (defined as ⩾ 0.12 ng/mL). Cases of acute PE that did not meet the above criteria for massive or submassive PE, or did not undergo such testing for further classification, were excluded from this analysis.
Baseline characteristics, pulmonary embolism (PE) diagnosis, and treatment
Baseline characteristics included patient demographics, past medical history (PMH), primary discharge diagnosis category of index hospitalization, admission status, primary health insurance, vital signs at diagnosis of PE, including body mass index (BMI), laboratory data, imaging modality for PE diagnosis, and antiplatelet and any anticoagulant (i.e. prophylaxis or therapeutic) directly preceding PE diagnosis. Administrative claims codes were used to identify PMH diagnoses of malignancy, heart failure, chronic lung disease, prior PE, and prior deep vein thrombosis (DVT) (see online supplementary Table 1). Primary health insurance was categorized as commercial, Medicare, Medicaid, or other. Admission status was categorized as elective, emergency, urgent, or trauma according to the assigned Medicare Provider Analysis and Review (MedPAR) inpatient admission status. 13
The primary discharge diagnosis was assumed to represent the primary reason for hospital admission, categorized into six broad groups based on relevant primary discharge diagnosis: (1) PE (with above ICD-10-CM codes); (2) DVT; (3) hematology/oncology; (4) sepsis/infection; (5) cardiovascular; and (6) other. The ‘other’ discharge diagnosis category included a broad array of neurologic conditions, alimentary conditions, trauma or orthopedic conditions, among others. The complete list of all primary discharge diagnoses and associated categorizations are summarized in online supplementary Table 2. Primary discharge diagnoses identified as PE or DVT were more broadly categorized as VTE, thereby accounting for patients primarily admitted for a VTE event compared with patients not primarily admitted for a VTE event.
The time and date of acute PE was recorded to allow for evaluation of admission time to diagnosis, laboratory data closest to the time of diagnosis, evaluation of medication administration after diagnosis, and evaluation of post-diagnosis bleeding outcomes, among other comparisons. The time of diagnosis was defined as the time of reporting of the radiographic confirmation of acute PE. To perform additional risk stratification, vital signs nearest the time of diagnosis were recorded and used to calculate the Pulmonary Embolism Severity Index (PESI) 14 and simplified PESI (sPESI) 3 scores.
Treatments of acute PE included systemic anticoagulation (AC), systemic thrombolysis (ST), and invasive procedures. Subcutaneous forms of non-weight-based heparin or low-molecular-weight heparin (LMWH) dosed in a manner appropriate for VTE prophylaxis were excluded in the analysis of post-PE anticoagulation as such forms are subtherapeutic. All other forms of therapeutic AC were considered equivalent. The times from diagnosis to AC were obtained from the timestamp of PE diagnosis relative to medication administration records. Invasive procedures included catheter-directed thrombolysis (CDT), CDT with ultrasound-assisted thrombolysis (USAT) using the EKOS® Catheter (Boston Scientific, Marlborough, MA, USA), endovascular mechanical thrombectomy, and surgical pulmonary embolectomy. Two forms of endovascular thrombectomy were utilized during the study period, including FlowTriever® (Inari Medical, Inc., Irvine, CA, USA) and the Indigo® System (Penumbra, Inc., Alameda, CA, USA). Placement of an inferior vena cava (IVC) filter was considered separately from other invasive procedures for PE as the procedure does not reduce thrombus burden within the pulmonary artery.
Primary outcome
A composite primary outcome of all-cause in-hospital mortality plus a combined bleeding endpoint of intracranial hemorrhage and major bleeding was used to evaluate differences in populations and treatment strategies. Intracranial hemorrhage was identified by ICD-10-CM code I62.9 and subsequently adjudicated by chart review. Major bleeding was defined as any transfusion of red blood cells during hospitalization that followed the time-stamped diagnosis of PE. Transfusion events were identified by searching an institutional database, which included administration time and date records of all transfusions.
Secondary outcomes
Hospital characteristics
Hospital length of stay (LOS), ICU admission rate, ICU LOS, and standardized direct costs were obtained from institutional administrative databases. ICU care was attributed to the hospitalization during which a PE occurred, although admission to the ICU was not necessarily related to PE. Direct costs included a summary of institutionally reported expenses associated with professional services, supplies, and medical facilities. In our health system, such information is routinely shared to the Vizient Clinical Data Base/Resource Manager™ (Vizient, Inc., Irving, TX, USA) administrative claims database for external comparison of our performance relative to other academic health centers, populated by hundreds of health systems and community hospitals nationwide. 15 Standardized direct costs reflect an estimated cost of care, harmonized with local market factors, including regional differences in labor costs by location of the hospital using the US Department of Commerce area wage index. Standardized patient care costs thereby provide generalizable cost estimates beyond our institution. Factors associated with PE care were further analyzed in tertiles of standardized direct cost. This manuscript was reviewed by Vizient, Inc. per established licensing agreements.
Readmission
All patients discharged from the SLHS with a diagnosis of PE were screened for all-cause readmission and the date of readmission was recorded to allow for time-to-event analysis. Patients were censored for death. Institutional databases were screened for vital status and all-cause hospital readmissions to a SLHS facility through March 31, 2019, ensuring at least 3 months’ follow-up for all subjects.
Statistical analysis
Categorical variables are summarized as counts and percentages. Continuous variables are summarized as means with SD or median with IQR. Between-group differences were assessed using chi-squared tests or Fisher’s exact test for categorical variables and Student’s t-test or Wilcoxon rank-sum test for continuous variables. Kaplan–Meier curves were used to illustrate the cumulative risk of all-cause readmission. Between-group differences of all-cause readmission were compared among PE subgroups using the log-rank test. A two-sided p-value less than 0.05 was considered statistically significant. All statistical analyses were performed using SAS, version 9.4 (SAS Institute Inc., Cary, NC, USA) and R, version 3.5.3 (Vienna, Austria). 16
Results
Patient characteristics
Of 1069 patients identified by administrative claims to have a hospitalization that included treatment for acute PE, 371 patients met study criteria and were further analyzed within cohorts of massive (11%) or submassive (89%) PE (Figure 1). Baseline characteristics of this population are presented in Table 1.
Baseline characteristics during treatment for acute massive and submassive PE.
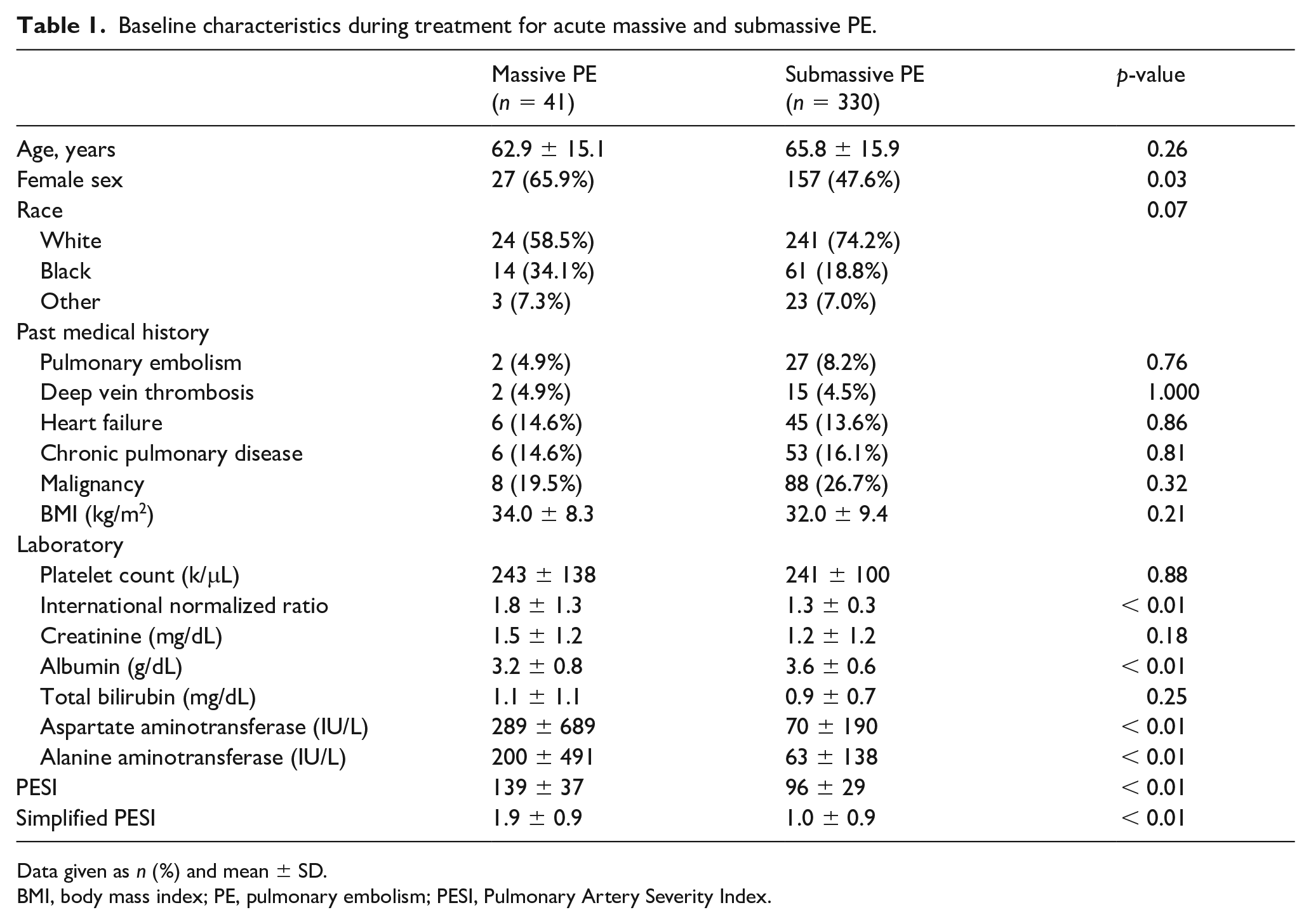
Data given as n (%) and mean ± SD.
BMI, body mass index; PE, pulmonary embolism; PESI, Pulmonary Artery Severity Index.
As expected, patients with massive PE, compared to submassive PE, had a higher mean PESI score (139 ± 37 vs 96 ± 29, p < 0.01) and higher sPESI (1.9 ± 0.9 vs 1.0 ± 0.9, p < 0.01). Massive PE was more commonly diagnosed among females (66% vs 48%, p = 0.03), although was not associated with other baseline differences in age, race, BMI, or in past medical history, including malignancy, prior PE or DVT. Patients with massive PE were less frequently discharged with a primary VTE diagnosis (32% vs 66%, p < 0.01). In fact, massive PE, compared to submassive PE, was diagnosed three times more frequently during hospitalizations for infection or sepsis (26.8% vs 8%, p < 0.01).
Treatment of submassive and massive PE
Therapeutic AC was offered to the majority of patients diagnosed with PE (89.8%). However, the median time from diagnosis to AC was longer for patients with massive PE (5.7 hours (IQR, 1.3–21.8) vs 2.6 hours (IQR, 1.0–7.0), p = 0.015) (Table 2). Systemic thrombolysis was used infrequently (1.8%) and was more commonly administered to cases of massive PE (8.1% vs 1.0%, p = 0.01).
Treatment characteristics and clinical outcomes of acute massive and submassive PE.
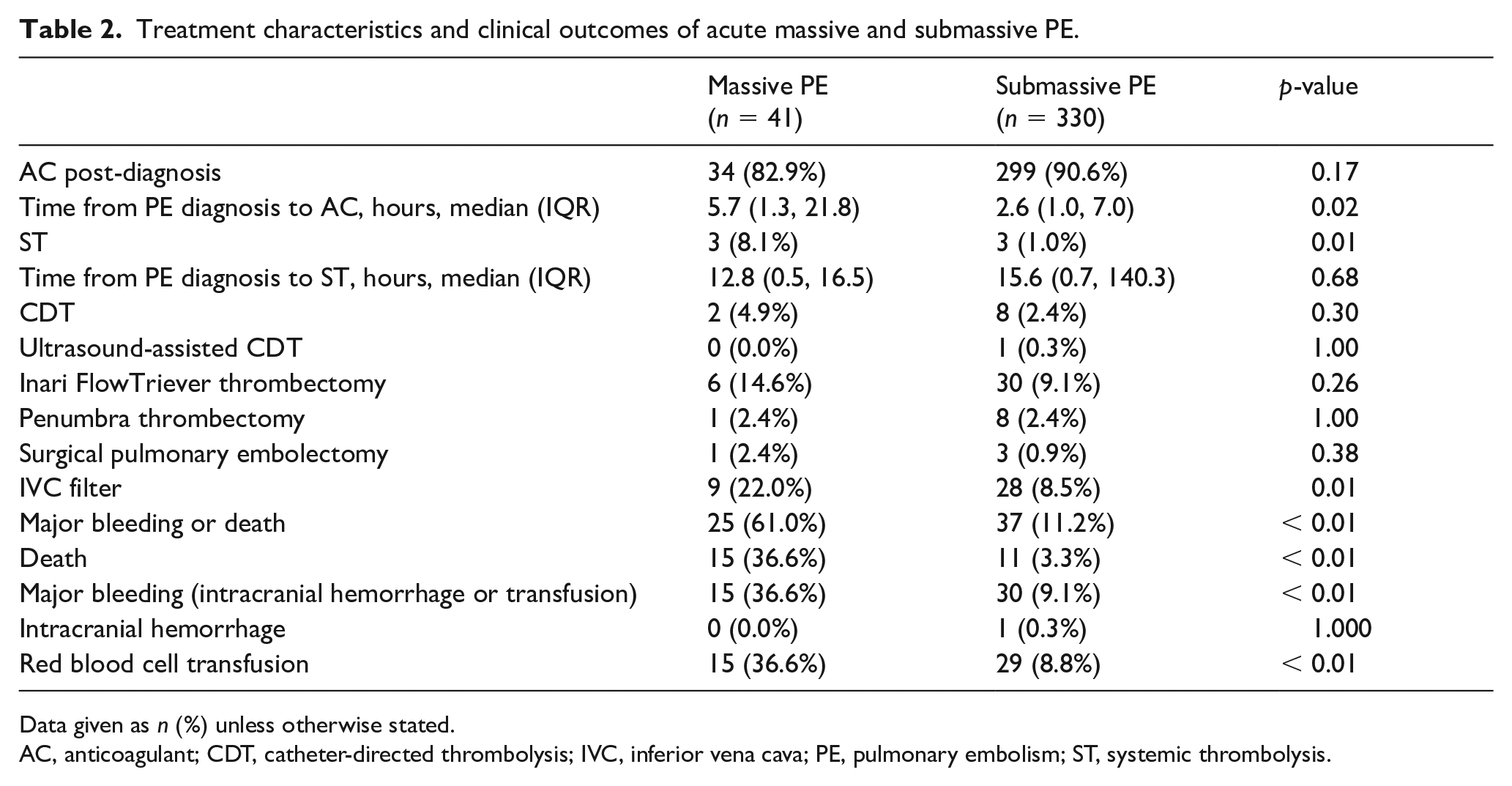
Data given as n (%) unless otherwise stated.
AC, anticoagulant; CDT, catheter-directed thrombolysis; IVC, inferior vena cava; PE, pulmonary embolism; ST, systemic thrombolysis.
Those with massive PE received invasive procedures at greater rates than those with submassive PE, though the difference was not statistically significant (22% vs 12%, p = 0.08). There were no differences in frequency of individual invasive treatments that address pulmonary thrombus (i.e. CDT, thrombectomy). However, IVC filters were more commonly implanted among patients with massive PE (22% vs 8.5%, p = 0.01).
In-hospital outcomes following acute PE
The primary composite endpoint of in-hospital bleeding and death was observed among 17% of patients. The component endpoints for the overall population included in-hospital death (7%), packed red blood cell transfusion (12%), and a single intracrancial hemorrhage (0.3%). Massive PE, compared to submassive PE, was associated with significantly higher rates of major bleeding (36.6% vs 9.1%, p < 0.01) and death (36.6% vs 3.3%, p < 0.01) (Table 2).
Among the 62 patients that suffered in-hospital bleeding and death, we evaluated several characteristics of patients and hospitalization beyond the PE subtype (see online supplementary Table 3). Those that suffered a primary bleeding or death event, compared to event-free hospitalizations, demonstrated no significant differences in age, BMI, or baseline past medical history, including malignancy, heart failure, chronic lung disease, or history of PE. Similarly, no differences were noted in treatment with anticoagulation (89% vs 90%, p = 0.77), systemic thrombolysis (1.8% vs 1.8%, p = 1.00), and overall invasive procedure use (12.9% vs 13.3%, p = 0.94). However, the median time to anticoagulation was significantly longer among patients suffering a primary bleeding or death event (4.6 hours (IQR, 1.2–14.2) vs 2.6 hours (IQR, 0.9–6.9), p < 0.01). IVC filters were more frequently implanted during hospitalizations that incurred a primary bleeding or death event, compared to hospitalizations without such clinical events (24% vs 7.1%, p < 0.01).
Venous thromboembolism (VTE) vs non-VTE hospitalization
Most hospitalizations that included care of an acute PE were associated with a primary discharge diagnosis of VTE (63%), including PE (60%) and DVT (2.7%). Therefore, hospitalizations for trauma-related conditions (15.6%), sepsis/infection (10.2%), and non-PE-related cardiovascular disease (8.9%), among others, represented the remaining 37.5% of hospital admissions during which PE occurred.
Patients hospitalized primarily for VTE were more commonly admitted emergently (77% vs 55%, p < 0.01), were represented primarily by submassive PE (94%), and had lower PESI (97 ± 30 vs 107 ± 37, p < 0.01) scores. Accordingly, massive PE was more common among admissions related to a non-VTE primary discharge diagnosis (20% vs 5.6%, p < 0.01). Regardless of PE subtype, patients hospitalized primarily for VTE, relative to hospitalizations primarily for other causes, had shorter median time to anticoagulation (2.3 hours (IQR, 0.9–6.9) vs 3.4 hours (IQR, 1.1–11.1), p = 0.03) and were more frequently offered invasive procedures to treat PE (17.2% vs 6.5%, p < 0.01). Patients hospitalized primarily for VTE experienced a significantly lower composite primary endpoint of in-hospital bleeding and death (9.1% vs 30%, p < 0.01). The component endpoints of in-hospital death (3.0% vs 13.7%, p < 0.01) and major bleeding (6.0% vs 22.3%, p < 0.01) were commensurately reduced.
Hospitalization details and resource utilization
Hospitalizations for acute PE were most commonly associated with an emergent (69%) or urgent (26%) admission status (Table 3). The median hospital LOS for patients treated for PE was 6.0 (IQR, 3.0–11.0) days and 47.2% required admission to the ICU. Patients with massive PE utilized the ICU more frequently (78% vs 43%, p < 0.01).
Hospital characteristics during treatment for acute massive and submassive PE.
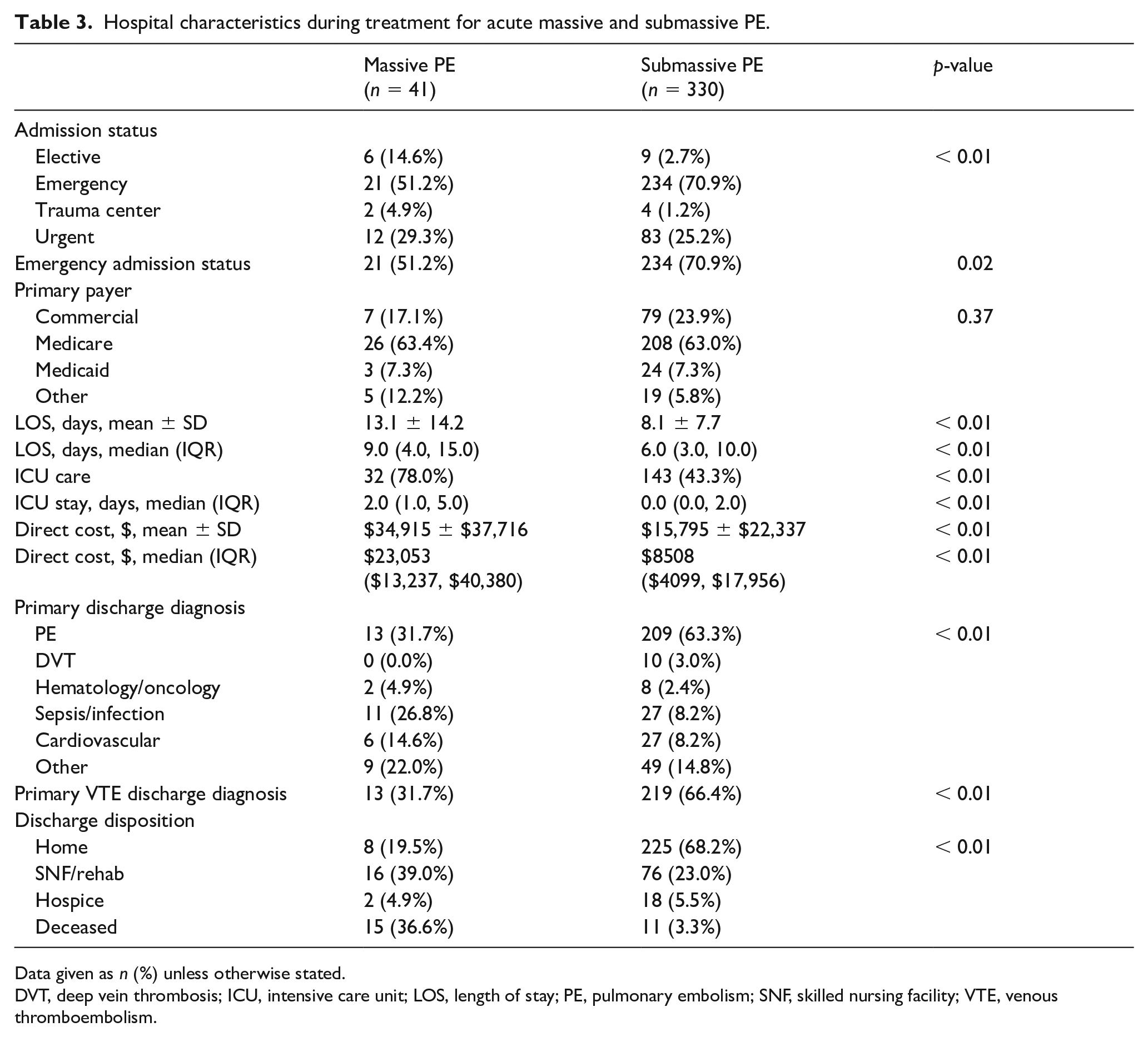
Data given as n (%) unless otherwise stated.
DVT, deep vein thrombosis; ICU, intensive care unit; LOS, length of stay; PE, pulmonary embolism; SNF, skilled nursing facility; VTE, venous thromboembolism.
The standardized mean direct costs for PE care were $17,908 ± $25,168. Median direct costs were $10,032 (IQR, $4467–$20,330) overall, although those that required ICU care incurred a much higher median cost of $18,354 (IQR, $10,957–$35,337). Hospitalizations during a massive PE event cost nearly three times more than those for submassive PE ($23,053 (IQR, $13,237–$40,380) vs $8508 (IQR, $4099–$17,956), p < 0.001). Medicare was the most common payor type (63.1%) and did not differ between PE subtypes (p = 0.37) (Table 3), nor those that suffered primary events (p = 0.21) (see online supplementary Table 3).
Following the positive trends for in-hospital outcomes, patients discharged with a primary VTE diagnosis (62.5%) had a shorter median LOS (4.0 days (IQR, 3.0–8.0) vs 10.0 days (IQR, 6.0–17.0), p < 0.01), lower rates of ICU care (38.8% vs 61.2%, p < 0.01), and corresponding lower median direct care costs ($6385 (IQR, $3352–$13,306) vs $17,579 (IQR, $9715–$37,348), p < 0.01). Such patients were also more likely to be discharged to home (75.4% vs 41.7%, p < 0.01).
Hospitalizations during which a primary major bleeding or in-hospital death occurred were associated with a longer median LOS (11.0 days (IQR, 6.0–22.0) vs 5.0 days (IQR, 3.0–10.0), p < 0.01), higher frequency of ICU care (75.8% vs 41.4%, p < 0.01), and higher median direct care costs ($30,081 (IQR, $16,123–$60,556) vs $7978 ($4064–$16,179), p < 0.01) (see online supplementary Table 3). Hospitalizations that incurred a primary bleeding or death endpoint event were also less commonly associated with a primary VTE discharge diagnosis (33.9% vs 68.3%, p < 0.01); primary discharge diagnoses related to infection/sepsis (19.4% vs 8.4%, p < 0.01) and hematologic/oncologic (8.1% vs 1.6%, p = 0.01) more commonly represented in that circumstance.
Tertiles of direct care costs
Characteristics of hospitalization were further analyzed by tertiles of acute care costs (see online supplementary Table 4). Patient characteristics of BMI, PESI score, and PE subtype were unique across cost tertiles. Patients that presented via the emergency room and that had a primary discharge diagnosis of PE most frequently represented the lowest cost tertile. Most patients in the lowest cost tertile were submassive PE; three patients with massive PE were classified in the low-cost subgroup, although all died shortly after admission. As expected, adverse clinical outcomes, LOS, and ICU care were significantly lower in the lowest cost subgroup.
Readmissions
Readmission-free survival was not different between submassive and massive PE subgroups over a mean follow-up of 19.3 months (Figure 2). Overall, readmission rates at 1 month, 3 months, 6 months, and 12 months were 14.0%, 22.5%, 28.0%, and 33.6%, respectively. Kaplan–Meier estimated rates of early readmission were not different at 1 month (14.3% vs 15.7%, p = 0.88) and at 3 months (24.4% vs 37.4%, p = 0.24) between submassive and massive PE subgroups, respectively. Characteristics of patients readmitted at 30 days and 90 days are reported in online supplementary Table 5.
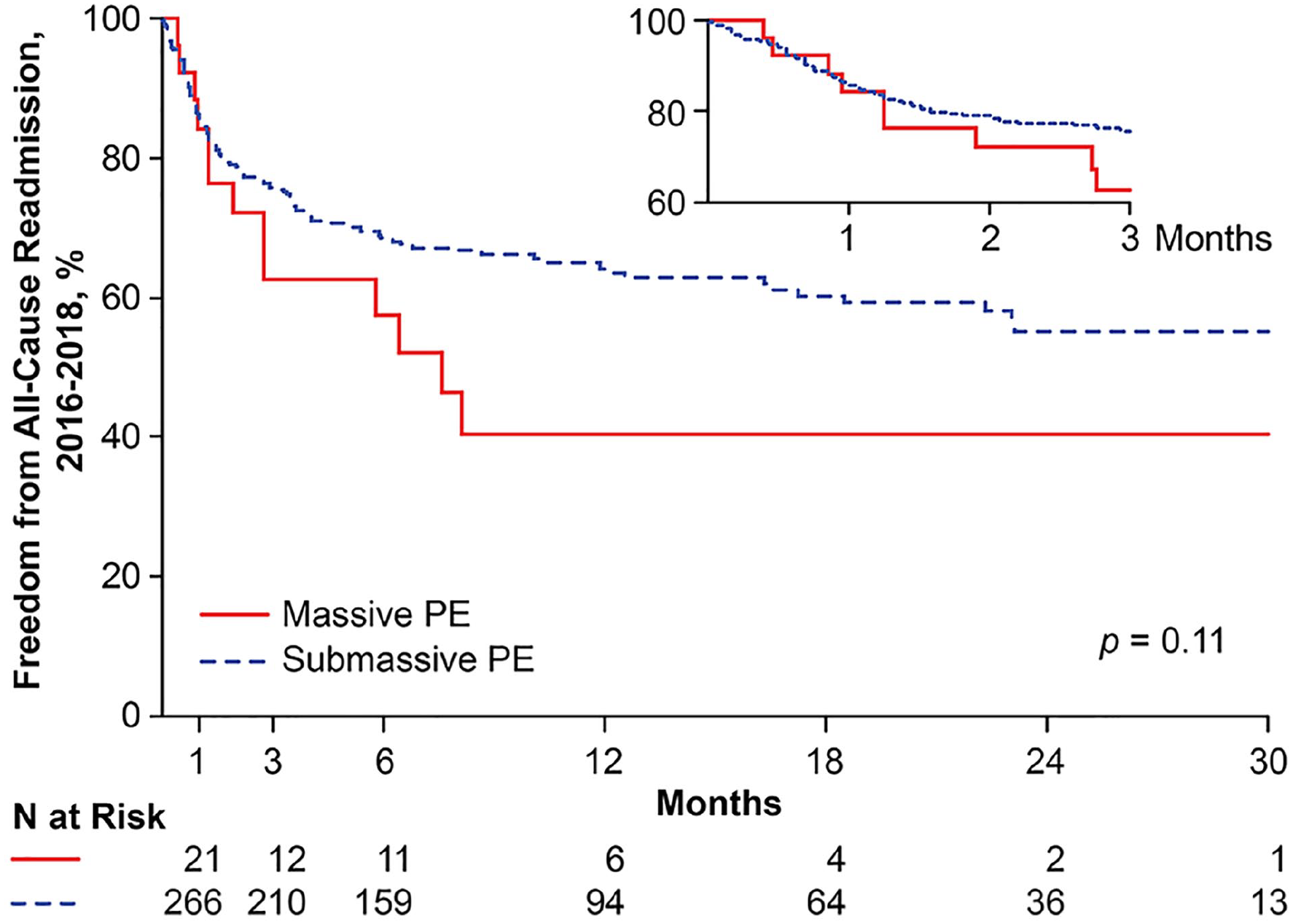
Readmission-free survival following hospitalization for acute massive and submassive PE.
Discussion
This analysis provides several noteworthy details of care among a contemporary, consecutive cohort of patients with intermediate or high-risk PE. First, despite low utilization of thrombolysis, in-hospital bleeding remains a frequent adverse event during hospitalization for massive and submassive PE (36.6% vs 9.1%, p < 0.01). Second, rates of in-hospital mortality (36.6% vs 3.3%, respectively) remain high. Together, in-hospital bleeding or death complicated one in six hospitalizations. Third, delays in administration of anticoagulation were noted during hospitalizations for massive PE, and for those hospitalizations listing a primary discharge diagnosis unrelated to VTE (e.g. sepsis), highlighting the complexities of routine care for acute PE. Lastly, despite patients with massive PE experiencing a considerably higher risk of bleeding and death, those with massive PE that were successfully discharged show no significant difference in readmission rates compared to submassive PE into late follow-up. This finding reinforces the importance of rapid diagnosis and consistent, coordinated, life-saving treatments, especially to those with high-risk PE.
Despite advances in treatment, in-hospital mortality rates for PE remain substantial. In the historic International Cooperative Pulmonary Embolism Registry (ICOPER) of 108 massive and 2284 non-massive PE cases studied from 1995 to 1996, the rate of death at 90 days was 51.9% and 14.5%, respectively. 17 A contemporary analysis from the Massachusetts General Hospital Pulmonary Embolism Response Team (PERT) database reported rates of in-hospital death of 8.3% overall, including rates of 32.6% and 4.5% for massive and submassive PE, respectively. 18 These rates are consistent with the observed in-hospital mortality of 7.0% in our analysis, as were the subgroup mortality rates in our cohort of 36.6% vs 3.3% for massive and submassive PE, respectively.
Contemporary expert consensus suggests short-term mortality may represent the optimal measure of in-hospital clinical effectiveness for various treatments of high-risk PE. 19 In this analysis, all intermediate- and high-risk patients hospitalized with submassive and massive PE were evaluated, regardless of treatment strategy. Numerous factors known to influence the risk of early death after PE, including the variables that inform the PESI 14 and sPESI 3 scores were assessed. Patients were also stratified based on the clinical stratification scheme from the American Heart Association. 6 However, other patient- and treatment-related variables were considered.
While our analysis is hypothesis-generating, the influence of treatment efficiency for PE deserves additional prospective study. Notably, those that suffered a primary endpoint of in-hospital death or bleeding had a median 4.6 hours of time from diagnosis to anticoagulation, which was nearly twice as long among those without such an endpoint. Time to treatment may be an indicator of adverse in-hospital outcomes, and perhaps as influential as the actual treatment provided. In the aforementioned analysis by Secemsky et al., the median time from diagnosis to PERT consultation was 50 (IQR, 23–197) minutes, although the study did not assess the impact of response time on outcomes. 18 This is an area of considerable interest and we intend to perform additional analysis to assess the impact of time to treatment, among other factors, on our primary bleeding and in-hospital death outcome as well as health resource utilization.
Significant knowledge gaps remain in the comparative effectiveness of various treatments, particularly in the context of routine care of intermediate and high-risk PE. Systemic thrombolysis provides benefits of reduced mortality and lower recurrence of PE among high-risk populations, although at the expense of bleeding complications, including a 1–3% risk of intracranial hemorrhage.20–22 Our experience with ST was limited, with only 1.8% of the total population receiving the treatment, including 10.8% of the massive PE cohort. From other reports, the overall utilization rates of ST are low and range from about 5% to 30% depending on the population studied.17,18,23 Whereas invasive treatments, including CDT,24–27 USAT,9,28–30 and mechanical thrombectomy, 31 have been shown to be effective in treating intermediate and high-risk PE, the use of such therapies in our cohort was relatively low (13.2%). In fact, the use of IVC filters was more common than ST or other invasive treatments. However, we believe IVC filter use in our population was more commonly provisioned as an option of ‘last resort’ among patients unsuitable for anticoagulation or ST. Complexities in management of massive PE were also manifested by differences in care, including a longer time to anticoagulation and lower overall rates of therapeutic anticoagulation. Accordingly, the cohort receiving IVC filters may have been biased as a particularly high risk for clinical outcomes. Moreover, during the study period, SLHS did not have a standing protocol to evaluate for residual DVT at the diagnosis of acute PE, a factor associated with increased risk, 32 which may identify a cohort most likely to benefit from temporary IVC filters. The use of IVC filters has declined as our experience using mechanical thrombectomy has grown. 33
Our study underscores the heterogeneity of patients treated for PE. This analysis highlights the routine clinical presentations and variability in care that may not be well represented in tightly controlled clinical trials of submassive or high-risk PE. One-third of patients suffering PE were hospitalized primarily for conditions unrelated to VTE, including conditions that confer a high bleeding risk. Such non-VTE hospitalizations, including those for sepsis or infection, were associated with higher-risk PE, as evidenced by higher PESI scores. In our experience, patients primarily admitted for a reason other than VTE experienced delayed time to anticoagulation, and invasive treatments were less frequently performed. This observation highlights the challenging balance of timely treatment while also mitigating bleeding risk, among other factors, that surround routine PE care.
Total admissions and hospital charges for PE have increased over the past two decades. 34 The observed median direct care cost of $10,032 is similar to other reports, although our analysis highlights other important drivers of acute care expenses.10,35,36 Factors associated with increased costs included longer LOS, ICU care, and hospitalizations primarily associated with a non-VTE diagnosis – a group that demonstrated higher PESI scores at time of diagnosis. These findings highlight the compounding challenges of PE, particularly when PE occurs during the hospitalization of non-VTE conditions.
The economic burden of DVT and PE in direct medical cost is large, as is the high rate of hospital readmission.10,37 We found similarly high rates of readmission at 1 month and 3 months. Importantly, despite substantially higher rates of in-hospital death and bleeding, patients discharged after treatment of massive PE were not more likely to be readmitted at 1 or 3 months compared to those with submassive PE. This finding suggests that successful management of high-risk patients with PE may also reduce their long-term risks of complications contributing to future readmission.
Study limitations
As a single-institution, retrospective, observational analysis, our study has several important limitations and these results should be interpreted as hypothesis-generating. The process to identify patients based on administrative claims codes could have missed some patients with PE that were not coded correctly. Massive PE was diagnosed more frequently during hospitalizations concurrent with sepsis. It is possible that sepsis could have confounded hypotension at the time of PE diagnosis, although when PE was diagnosed and if hypotension was present and treated, we labeled such patients as massive PE. However, the amount of hypotension attributable to sepsis versus PE would be difficult to parse, even in a prospective study. Transfusions were reported for any cause, so some transfusions may be unrelated to PE treatment, although all transfusions occurred after PE diagnosis. In addition to intracranial hemorrhage, our major bleeding definition included any red blood cell transfusion event after PE diagnosis. Our intention was to highlight the frequency of transfusion events, although this transfusion criteria was less stringent than some other criteria and increased the frequency of defined major bleeding events. As we individually adjudicated each chart, our data set includes meticulous sampling of details around acute PE care, including a time stamp of diagnosis and vital signs at that time, as well as details of invasive treatments. We also leveraged systematic reporting from our health system’s electronic health record data tables to collect robust, time-stamped details of validated medication utilization and laboratory data. Owing to the single electronic health record used in this analysis, we did not have the ability to identify patients that were readmitted or died outside of our health system. Standardized direct costs reflect an estimated cost of care, although do not provide a precise breakdown of expense components. Validation studies comparing standardized direct costs across Vizient centers have not been reported. Lastly, within this analysis, multiple comparisons were made, and some results indicating significance may have been due to chance.
Despite these limitations, the principle findings are congruent with other studies and have fostered development of our VTE treatment program. Importantly, this study provides focus for improvement in PE care. Our health system is currently implementing pathways for systematic risk stratification and care pathways to ensure all patients receive the potential benefit of advanced treatments, when appropriate. We intend to perform additional multivariable modeling of our primary bleeding and death outcomes, readmissions, and further analysis of cost as details of indirect, direct, procedural, and pharmacy costs become available.
Conclusions
Despite low utilization of thrombolysis, in-hospital bleeding remains a frequent adverse event associated with submassive and massive PE, thereby contributing to high healthcare resource utilization. When PE complicates the hospitalization of conditions unrelated to VTE, the severity of PE and outcomes are worse. Patients with massive PE demonstrate higher risk for in-hospital bleeding and death, although those successfully discharged demonstrate similar rates of readmission compared to submassive PE into late follow-up.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X20967415 – Supplemental material for Bleeding, death, and costs of care during hospitalization for acute pulmonary embolism: Insights from the Saint Luke’s Outcomes of Pulmonary Embolism (SLOPE) study
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X20967415 for Bleeding, death, and costs of care during hospitalization for acute pulmonary embolism: Insights from the Saint Luke’s Outcomes of Pulmonary Embolism (SLOPE) study by Matthew C Bunte, Kensey Gosch, Ahmed Elkaryoni, Anas Noman, Erin Johnson, Philip Jones, Mahathi B Indaram and Sravanthi Vupputuri in Vascular Medicine
Footnotes
Acknowledgements
The authors wish to acknowledge Ms Nivedha Madhan, Ms Rosann Gans, and Drs Divya Vundamati, Islam Abdelkarim, and Dushyant Ramakrishnan, in support of data collection. Thanks to Mr Jose Aceituno for support creating the central illustration. Special thanks go to Mr Lynn D Miller and Ms Michelle Terriquez for assistance with reporting from health system databases. The authors thank Ms Marci Kennedy for administrative support with the institutional REDCap database.
Declaration of conflicting interests
Dr Bunte has received research support from Inari Medical, Inc. None of the other authors has any conflicts to disclose.
Funding
Statistical analysis was supported through an investigator-initiated research grant from Inari Medical, Inc. However, the authors solely collected and retained the data and independently examined the data, and have submitted the manuscript for publication without data interpretation from the sponsor.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
