Abstract
Tyrosine kinase inhibitors (TKIs) of the BCR-ABL fusion protein have dramatically changed the mortality of chronic myeloid leukemia (CML) but they carry a risk of serious vascular morbidity. While TKIs do not cure CML, daily oral administration of a TKI can control CML and TKIs are chronic medications. Interestingly, vascular complications can occur at any time a patient is on a TKI. Therefore, it is imperative that all care team members and patients are aware of and watching for possible vascular complications. In the following review, a case of arterial thrombosis secondary to the TKI ponatinib is presented as well as a discussion of thrombotic and vascular adverse events reported with TKIs. TKIs are metabolized through the cytochrome P450 system and important drug interactions to consider are reviewed. Finally, we present a multidisciplinary approach to the management of patients with CML on TKIs.
Keywords
Case review
A 70-year-old female with past medical history significant for hypertension, hyperlipidemia, renal cell carcinoma in 1995 treated with nephrectomy, chronic kidney disease, fibromyalgia, hot flashes, and chronic myeloid leukemia (CML) since 1997 presented to cardio-oncology clinic for cardiovascular optimization prior to starting ponatinib (ICLUSIG; ARIAD, Cambridge, MA, USA).
The patient’s prior treatments for CML included imatinib (GLEEVEC; Novartis, East Hanover, NJ, USA), nilotinib (TASIGNA; Novartis), dasatinib (SPRYCEL; Bristol-Myers Squibb, Princeton, NJ, USA), and later bosutinib (BOSULI; Pfizer, New York, NY, USA), but she suffered severe muscle spasms, lack of bone marrow response, pleural effusion, and acute renal failure that prompted the need to consider use of ponatinib. At the time of her initial cardio-oncology clinic visit, her blood pressure, electrocardiogram, and echocardiogram were normal. She reported excellent functional status without any cardiac symptoms. She was on several medications which had the potential for drug interaction with ponatinib or increased risk of thrombosis and/or fluid retention. Atorvastatin was changed to rosuvastatin, diltiazem changed to carvedilol, and meloxicam and estradiol were discontinued. Our clinic visit focused on education about the potential vascular risk, optimization of her cardiovascular status prior to initiation of ponatinib, as well as medication adjustment.
Within 1 week of starting ponatinib, she presented to an outside hospital (OSH) with a severe headache, hypertensive emergency, and altered mental status. She was diagnosed with an acute embolic stroke by brain magnetic resonance imaging (MRI), showing three distinct focal ischemic areas in bilateral cerebral hemispheres. At the OSH she had no evidence of atrial fibrillation, which was confirmed on a follow-up 30-day event monitor, no cardioembolic source was identified on transesophageal echocardiogram, and carotid ultrasound showed moderate bilateral stenosis (50–69% stenosis, internal carotid). A presumptive diagnosis of ponatinib-related stroke was made and ponatinib was stopped. She was treated conservatively with anticoagulation, and focal deficits resolved. We believe that patient and family education about stroke warning symptoms contributed to the patient’s decreased morbidity from the stroke. In the following article, we provide a review of the common cardiovascular effects of tyrosine kinase inhibitors (TKIs) and a pragmatic approach to co-management of patients with CML on BCR-ABL TKIs.
Currently approved BCR-ABL TKIs
CML was the first malignancy to be linked to a chromosomal abnormality called the Philadelphia (Ph) chromosome. The Ph chromosome is present in nearly all cases of CML. 1 The truncated chromosome 22, called Ph chromosome, results from a reciprocal translocation, t(9;22)(q34;q11), that leads to juxtaposition of the breakpoint cluster region (BCR) gene from the long arm of chromosome 22 and Abelson murine leukemia viral oncogene homolog 1 (ABL1) gene from the long arm of chromosome 9. The resultant BCR-ABL1 fusion gene eventually translates into the BCR-ABL1 protein, which has persistently enhanced tyrosine kinase activity. Through various downstream pathways, the oncoprotein causes enhanced proliferation, differentiation arrest, and resistance to cell death, leading to endless division of immune precursors in leukemia cells.1,2
TKIs are a class of anticancer therapy that have dramatically improved survival in CML. In 1975, the observed 5-year survival rate in CML was 17.5% and in 2011 it was 75.4%. The National Cancer Institute Surveillance, Epidemiology and End Results (SEER) Program estimates the current 5-year survival with CML to be 69.2%. 3 Imatinib mesylate, the first TKI for Ph chromosome positive CML, was approved in 2001 by the US Food and Drug Administration (FDA). 4 Based on imatinib resistance, second and third generation inhibitors of BCR-ABL tyrosine kinase have subsequently been developed to mitigate resistance mutations and deepen responses (Table 1).2,5–9 The International Randomized Study of Interferon and ST1517 (IRIS) was the phase 3 landmark study which showed imatinib was superior to traditional therapy. 10 Ten-year follow-up of IRIS revealed that the estimated overall survival rate for imatinib-treated patients was 83.3%. 11 Similarly, the CML-study IV reported at 10 years that the overall survival of patients on imatinib was 84%. 12
Approved BCR-ABL TKIs.
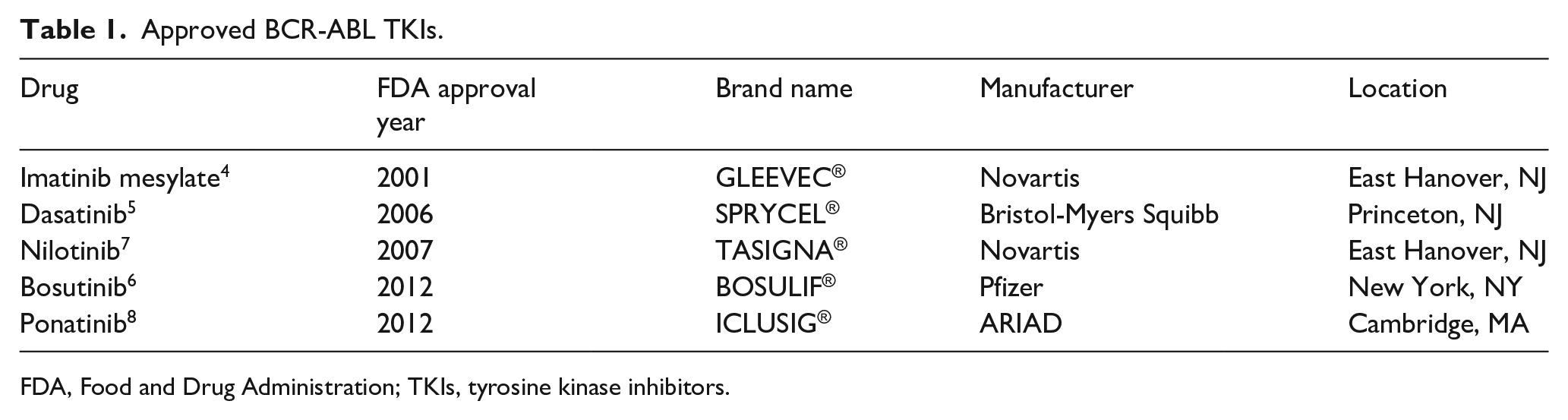
FDA, Food and Drug Administration; TKIs, tyrosine kinase inhibitors.
Patients with CML on a TKI require careful follow-up to predict when alternative TKIs, stem cell transplantation or treatment discontinuation should be considered. 13 The standard means of monitoring response is quantitative polymerase chain reaction measurement of BCR-ABL1 transcripts in peripheral blood. 14 A trial of TKI discontinuation is an acceptable option in patients who achieve a durable, deep molecular response (> 4-log reductions in BCR-ABL1 transcript levels for at least 2 years) and are expected to comply with the demands of frequent testing and follow-up. 15 Patients may seek to discontinue a TKI because of significant side effects, cardiovascular events, convenience, cost of medication, desire for pregnancy, or other reasons. 16 However, a TKI is resumed if there is evidence of loss of major molecular response (< 3-log reduction in BCR-ABL1 transcript level). Approximately half of patients who discontinue a TKI remain in a treatment-free remission (TFR). 17 Currently, nilotinib has the most prospective data on discontinuation for TFR. 17 Since TFR is feasible, more data are needed to determine how the incidence of cardiovascular events changes in patients with a durable molecular response who discontinued their TKI.
Differences in cardiovascular endpoints in CML clinical trials
While generally well tolerated, serious cardiac and vascular complications of BCR-ABL TKIs can occur. Significant vascular events including ischemic heart disease, ischemic cerebrovascular events, and peripheral artery disease (PAD) are reported with TKIs, but more commonly in the second and third generation TKIs: nilotinib and ponatinib. 18 TKI clinical trials for the treatment of CML have failed to report these events accurately due to the lack of uniform definitions of vascular events and limited insight into the pathophysiology. For example, an arterial ischemic event may occur from thrombosis, vasospasm or potentially vasculitis. 19 Current oncology clinical trials and the FDA utilize the Common Terminology Criteria for Adverse Events (CTCAE) in order to standardize reports of adverse events across clinical studies. Unlike many large cardiovascular trials, the CTCAE lacks adjudication of events by an independent panel and thus current CML clinical trial data may under-represent cardiovascular toxicities and lack insight into the pathophysiology of cardiac events.19,20
Additionally, many of the initial clinical trials are limited by the differences in design and baseline characteristics and risk factors. Thus, the inclusion and exclusion criteria of the patient populations studied should be considered when evaluating for cardiovascular toxicities. 21 For example, in the phase 3 ENESTnd trial of nilotinib, patients with pre-existing cardiac dysfunction were excluded and thus the studied population was lower risk than the general population.16,22,23 In contrast, in the PACE trial of ponatinib, the primary cardiac exclusion criterion was a corrected long QT interval, making it a potentially higher cardiovascular risk population. 24 Interestingly, the FDA temporarily withdrew ponatinib from the market in 2013 due to cardiovascular toxicity seen at the 24-month interval analysis. 20
Understanding that TKI trials in patients with CML had inherently different patient populations is critical when analyzing the number of cardiovascular events. Additionally, it is imperative to understand that many of these trials excluded patients with underlying cardiovascular disease and thus under-represent the general CML population. Therefore, clinicians treating patients with CML with TKIs need to remain vigilant, as the number of cardiovascular events in a real-world population is likely much higher than reported in clinical trials. Furthermore, unlike clinical trials with relatively short follow-up (5–10 years), patients with CML can be on TKIs indefinitely and transition from one to another if they develop resistance – thus, long-term cardiovascular monitoring is crucial.
Cardiovascular toxicities
Arterial thrombotic events
Nilotinib is associated with increased cardiovascular events including ischemic heart disease, ischemic cerebrovascular events, and PAD. In a 5-year follow-up of the ENESTnd trial, a phase 3 study comparing nilotinib to imatinib for first-line treatment of chronic phase CML, cardiovascular events occurred in 7.5% of patients on nilotinib 300 mg twice daily and 13.4% of patients receiving nilotinib 400 mg twice daily compared to only 2.1% in patients on imatinib 400 mg daily. 16 Six-year follow-up for patients receiving nilotinib 400 mg twice daily as first-line therapy reported 15% (11/73) of patients had atherosclerotic adverse events, including acute myocardial infarction (MI), stable angina, carotid stenosis, and stroke, with a median time to event of 42 months (range, 24–76 months). Management of atherosclerotic adverse events included coronary stenting, vascular surgery, amputation, and discontinuation of nilotinib. Cardiac events were more common in older patients and those with baseline cardiac risk factors. 25
Ponatinib carries a black box warning for arterial occlusion. In the phase 1 and 2 clinical trials of ponatinib, arterial occlusions, including serious and fatal events, occurred in at least 35% of patients. 8 Events included fatal MI, stroke, stenosis of large cerebral vessels, and severe peripheral artery disease. Ponatinib can cause recurrent or multisite vascular occlusion. Patients in the phase 1 and 2 ponatinib clinical trials were treated with revascularization when indicated. In these early trials, arterial occlusive events were more common with increasing age and in patients with prior ischemia, hypertension, diabetes or hyperlipidemia. 8
A 5-year follow-up of the PACE trial revealed that 25% (111/449) of patients on ponatinib had at least one arterial occlusive adverse event. Patients with more than two cardiac risk factors, history of ischemic or nonischemic cardiac disease, or venous thromboembolism (VTE) had a relative risk of 2.2 (95% CI, 1.5–3.3) of developing a serious arterial occlusive event; those with one risk factor carry a relative risk of 0.8 (95% CI, 0.5–1.2); and those with no risk factors have a relative risk of 0.4 (95% CI, 0.2–0.7). 26 In a real-world study of 85 patients with CML treated at 17 Italian centers, investigators showed that patients with a systematic coronary risk evaluation (SCORE) risk > 5% had a significantly higher rate of arterial occlusive events. SCORE risk is based on gender, age, systolic blood pressure, total cholesterol, and tobacco use. 27
Heart failure
Ponatinib also carries a black box warning for heart failure (HF). At 48 months of follow-up of the phase 2 PACE trial, 9% (39/449) of patients on ponatinib developed any grade of heart failure or left ventricular dysfunction, with 6% (29/449) developing fatal or serious heart failure or left ventricular dysfunction. Diagnosis of HF should lead to initiation of HF medical therapy and drug interruption, and in some cases discontinuation. 8 With regard to the other TKIs, initially there was concern that imatinib caused cardiomyopathy based on in vitro studies but this has not played out in clinical practice. Clinicians should be aware that cardiomyopathy can result from nilotinib, bosutinib, and dasatinib as well, although less frequently than with ponatinib. 18 For example, 1.6% of patients receiving dasatinib developed cardiomyopathy, congestive heart failure, diastolic dysfunction, fatal MI, and left ventricular dysfunction in the pivotal clinical trials. 5
Hypertension
Hypertension most commonly occurs with ponatinib but also is seen with nilotinib, imatinib, bosutinib, and dasatinib. Sixty-eight percent (306/449) of patients developed an increase in blood pressure at the 48-month follow-up of the phase 2 PACE study of pontinib, with cases of hypertensive crisis reported. There were 132 patients with stage 1 hypertension at baseline and 67% (88/132) progressed to stage 2 hypertension on ponatinib. 8 At the 5-year follow-up, 32% (142/449) of patients developed hypertension from ponatinib. 26 In cases of labile or treatment-resistant hypertension, ponatinib should be held and consideration should be given to evaluating for renal artery stenosis. 8
The 5-year follow-up of the ENESTnd trial revealed hypertension in 10.4% of patients on nilotinib 300 mg twice daily, 8.3% in patients on nilotinib 400 mg twice daily, and 4.3% in patients on imatinib. 16 Analysis of bosutinib trials revealed 7.8% of patients developed hypertension from the drug. Unlike ponatinib, though, most patients (89.2%) with a history of hypertension did not develop worsening hypertension. 28 In clinical trials, dasatinib resulted in hypertension in less than 10% of patients. 5
Pulmonary arterial hypertension
Pulmonary arterial hypertension (PAH) is a rare, but potentially lethal, complication most often seen with dasatinib. In 2011, dasatinib received an FDA warning after Bristol-Myers Squibb reported 12 cases of PAH related to dasatinib therapy confirmed by right heart catheterization. 29 Data derived from the 5-year follow-up study of DASISION showed that 5% of patients treated with dasatinib developed PAH compared to 0.4% in the imatinib arm. 30 Direct toxicity to the pulmonary vascular endothelium via production of mitochondrial reactive oxygen species is one proposed mechanism. 31
Currently, it is recommended that patients with dyspnea on dasatinib are screened with a chest X-ray and echocardiogram. 20 Doppler flow studies obtained during an echocardiogram may be an adequate noninvasive evaluation for potential PAH in the presence of pleural effusions. Right heart catheterization after a full noninvasive workup for dyspnea has been completed may be required to definitively establish a diagnosis of PAH. Once PAH is diagnosed, immediate cessation of dasatinib is advised since the current literature suggests the majority of patients’ PAH symptoms improve with cessation of therapy.32,33
Venous thrombosis
Ponatinib also carries a black box warning for VTE. Deep vein thrombosis, pulmonary embolism, superficial thrombophlebitis, and retinal vein occlusion with vision loss were reported in 6% (25/449) of patients treated with ponatinib.8,26 There is concern that there may have been unrecognized vascular events in the original trials of TKIs since venous vascular adverse events are not traditional oncologic adverse events. 34
Metabolic toxicities
Hyperglycemia
Since insulin receptors are tyrosine kinases, it is not surprising that TKI treatment is associated with changes in glycemic control and endocrine metabolism. Insulin stimulates the c-ABL kinase, which subsequently activates an insulin receptor-dependent metabolic effect leading to Akt/GSK-3beta phosphorylation and glycogen synthesis. Inhibition of c-ABL kinase is hence proposed to promote increased post-receptor insulin resistance.35,36 A systematic literature review showed that nilotinib had a higher incidence of hyperglycemia compared to dasatinib or imatinib. 37 In a study of 846 patients investigating the safety profile of nilotinib, 41% developed hyperglycemia within 3 months of starting therapy. 22 Therefore, monitoring glycemic control on nilotinib is recommended.
Hyperlipidemia
A retrospective cohort study of 1280 patients between 2006 and 2014 demonstrated that patients treated with nilotinib were 1.75 times more likely to develop hyperlipidemia compared to patients treated with dasatinib. 38 Patients being considered for nilotinib should be screened for lipid abnormalities before and early on during therapy. There should be a low threshold for recommending lifestyle intervention and/or initiating medical therapy.
In summary, the potential cardiovascular toxicities with various TKIs can be anticipated and risk factors optimized to improve cardiovascular outcomes. Table 2 highlights pragmatic clinical recommendations for tailored cardiovascular monitoring of patients receiving BCR-ABL1 TKIs.
Pragmatic clinical recommendations for tailored cardiovascular monitoring of patients with CML receiving BCR-ABL TKIs.
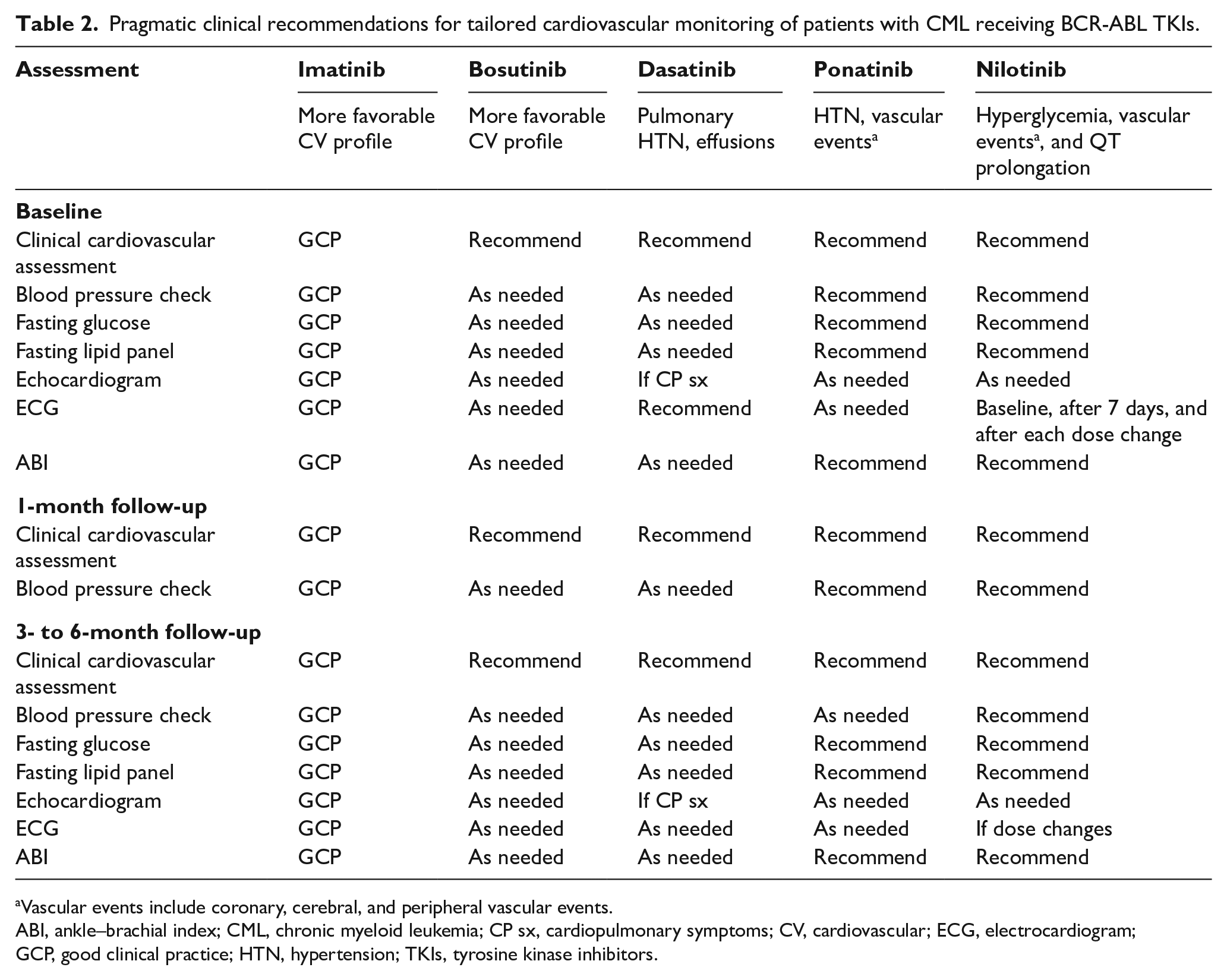
Vascular events include coronary, cerebral, and peripheral vascular events.
ABI, ankle–brachial index; CML, chronic myeloid leukemia; CP sx, cardiopulmonary symptoms; CV, cardiovascular; ECG, electrocardiogram; GCP, good clinical practice; HTN, hypertension; TKIs, tyrosine kinase inhibitors.
Drug interaction
Common medical drugs and CYP interactions
All TKIs are extensively metabolized by the cytochrome P450 (CYP) isoenzyme CYP3A4.4–8 Concomitant administration of strong CYP3A4 inhibitors increases TKI serum concentration significantly, whereas administration of strong CYP3A4 inducers reduces TKI serum concentrations. 39 A list of common CYP3A4 inducers, inhibitors, and substrates is presented in Figure 1. Interactions with drugs classified as CYP3A4 substrates or additionally as moderate CYP inhibitors or inducers are often undervalued in clinical practice, likely due to limited clinical data.40,41 For example, clearance of simvastatin (CYP3A4 substrate) is decreased by 70% when administered with imatinib. 42 By changing to a statin that is not a substrate of CYP3A4 (e.g. rosuvastatin or pravastatin), this interaction would be avoided. Commonly prescribed cardiac-related medications, including specific statins (e.g. atorvastatin, simvastatin), dihydropyridine calcium channel blockers, and antiarrhythmic drugs, often have clinically relevant drug–drug interactions that need to be evaluated on a case by case basis and medication adjustments made. Consultation with a clinical pharmacist is advised to monitor for drug interactions and educate patients about appropriate administration. 42
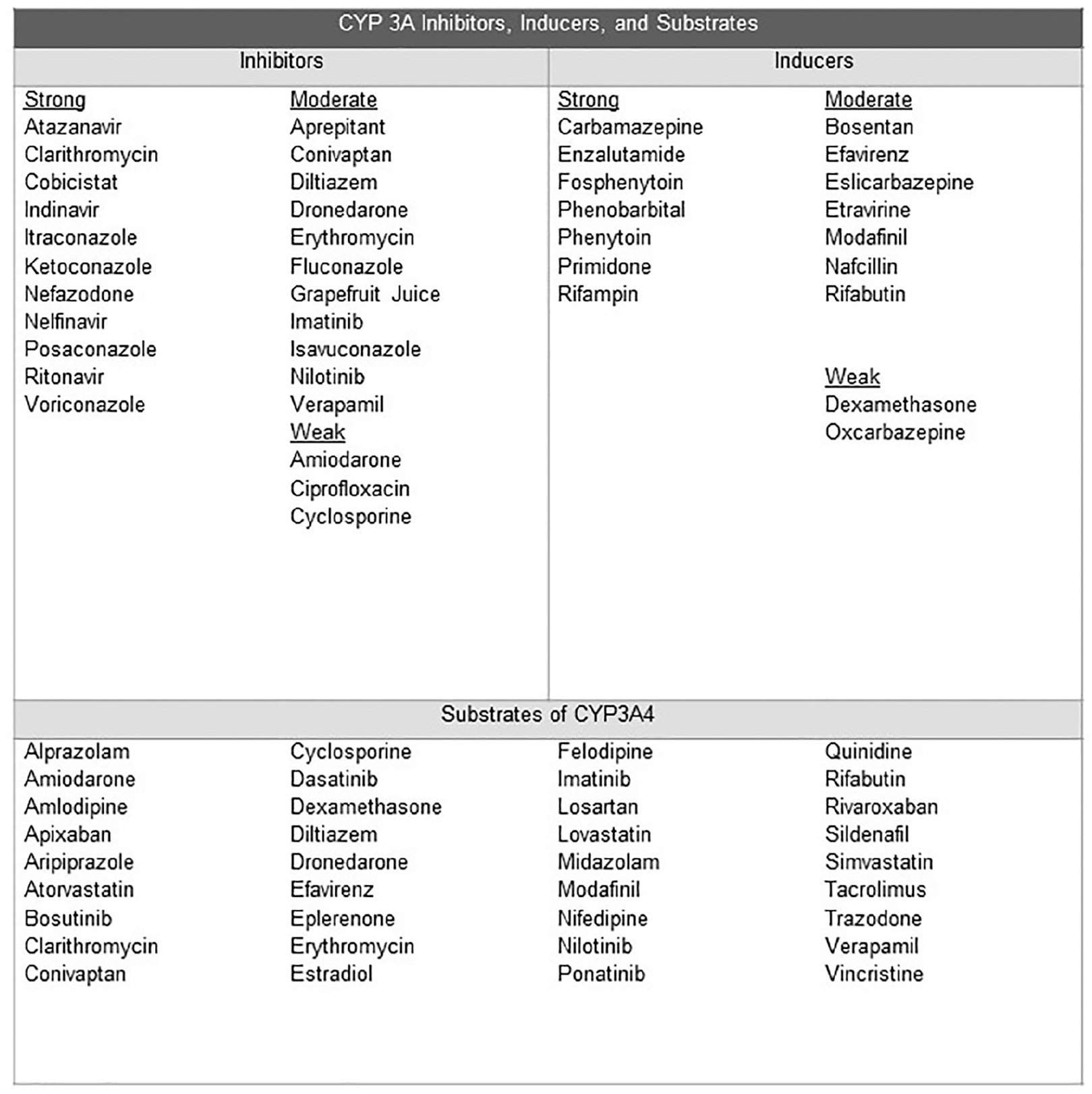
CYP 3A inhibitors, inducers, and substrates.
Collaborative management
Data clearly demonstrate that patients with CML treated with TKIs are at an increased risk of developing a variety of cardiac and vascular events. 18 Additionally, many patients with CML are treated for decades with TKIs and have underlying risk factors such as older age, hypertension, hyperlipidemia, diabetes, and obesity that also require management. 20 Owing to the increased awareness of cardiovascular events, we created a multidisciplinary team of providers to perform cardiovascular risk assessment and have developed a cardiovascular monitoring and management strategy for patients with CML treated with TKIs. Our collaborative team includes members from hematology-oncology, cardio-oncology, vascular medicine, clinical pharmacy, and nutrition. Currently, our hematology-oncology providers refer patients with CML with two or more cardiac risk factors who are starting a TKI to the cardio-oncology clinics for cardiac risk optimization and to develop a cardiac monitoring strategy. Vascular medicine assists with co-management of patients with progressive PAD and management of acute vascular events. The clinical pharmacist assists in identification of drug–drug interactions and potential alternatives, including statins, dihydropyridine calcium channel blockers, and antiarrhythmic medications. Nutritionists help personalize diet recommendations.
As a team, we discuss optimal strategies for patients who develop complications from TKIs and the best therapeutic approaches when alternative second and third-line TKI therapy is indicated due to either progression of CML or cardiovascular events. The clinical recommendations for cardiovascular monitoring depends on the TKI chosen, which often relies on a patient’s historical response to CML therapies, underlying cardiac risk factors, and cardiovascular disease. In general, patients with CML with underlying cardiovascular disease (coronary disease, heart failure, cerebrovascular events) are preferentially treated with imatinib and bosutinib due to a more favorable cardiovascular risk profile.19,23,28 Based on previously published data and clinical recommendations from the European LeukemiaNet, we have adopted the pragmatic clinical recommendations for tailored cardiovascular monitoring in patients with CML receiving treatment with TKIs seen in Table 2.18–20 Our practical approach for cardiovascular monitoring is tailored to the specific TKI with more intensive monitoring for cardiovascular events in patients treated with nilotinib or ponatinib.18–20 Owing to concern of possible PAH in patients treated with dasatinib, a baseline echocardiogram is reasonable in patients with a potential for elevated pulmonary pressures, including a history of lung disease, connective tissue disorders, thromboembolic pulmonary disease, or congenital heart disease. While a routine baseline echocardiogram is not recommended for patients prior to starting dasatinib, as mentioned before there is a low threshold for PAH evaluation in patients exhibiting any cardiopulmonary symptoms.18,30 While Table 2 offers a practical approach to cardiovascular monitoring and common cardiovascular-related events, providers must treat each patient independently. Even though cardiovascular events are relatively low in imatinib and bosutinib-treated patients, providers must remain vigilant as heart failure and vascular events have been reported and thus good clinical judgement is always indicated.
ABCDEs of prevention
TKIs have revolutionized the treatment of CML, making it a chronic disease. With a mindset of treating patients for extended periods with TKIs, and the natural prevalence of cardiovascular events that occur with an aging population, adopting a comprehensive cardiovascular checklist is important in treating patients with CML. While there is a paucity of literature describing how preventative strategies mitigate cardiovascular outcomes in patients with CML, we extrapolate current guidelines that are based on the general population. Although there are no prospective trials of cardiovascular prevention in patients with CML, there are several expert recommendations on cardiovascular surveillance and management of risk factors in patients with CML receiving TKIs.19,43,44 The basis of all these recommendations focus on the education and engagement of patients with CML regarding the potential for cardiovascular events and aggressive cardiac risk factor modification. Our institution has adopted the preventative ‘ABCDE’ checklist (Figure 2), outlined in several of Javid Moslehi’s papers, that focuses on pragmatic steps to prevent cardiovascular disease.19,20 The ‘ABCDE’ algorithm is an established means of cardiovascular reduction in a general population and commonly accepted in breast cancer survivors and prostate cancer patients.45–47 The ‘ABCDE’ checklist highlights awareness of potential cardiac and vascular events, controlling blood pressure, optimizing lipid profiles, controlling glycemic levels, engaging patients in appropriate diet and exercise guidelines, as well as monitoring for drug interaction.48,49 Clinicians are advised to collect information about the presence of cardiovascular risk factors, past cardiovascular history and check about medications that might have interactions with TKIs by using the ABCDE checklist at baseline and follow-up visits. The checklist serves as a template to allow providers to address the most concerning risk factors at each visit and also as a tool to enlist patients as active participants in the management of their chronic disease.

Preventative ‘ABCDE’ checklist for TKIs.
Return to the patient case
Returning to our patient case, we conclude with a summary of items addressed during our initial cardio-oncology visit using the ABCDE approach.
Follow-up: owing to our education about potential ischemia on ponatinib, the family promptly had the patient evaluated at the nearest stroke center and was treated. Fortunately, the patient had no residual sequlae from her stroke at the time of discharge. At her follow-up hematology-oncology appointment, she was evaluated for possible consideration for interferon versus omacetaxine. The patient had previously been on interferon without CML progression and thus opted to resume interferon treatments.
Conclusion
In summary, TKIs used in the treatment of CML increase the risk of serious thrombotic and vascular events. It is critical for providers and patients to be aware of these complications and manage modifiable cardiac risk factors. In truth, while managing modifiable cardiac risk factors is the current approach in preventative cardiology, little is known about the benefits of this approach in vascular oncology and cardio-oncology. We are currently following our patients with CML longitudinally to assess for cardiac and vascular outcomes to ultimately determine how to best care for their vascular health.
Footnotes
Acknowledgements
The authors thank Javid Moslehi MD for his mentorship, expertise in the field of TKIs. The authors also thank their patients, in particular ST, who are their best teachers.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
