Abstract
Background:
Venous thromboembolism (VTE) is a known complication of coronavirus disease (COVID-19) in patients requiring hospitalization and intensive care. We examined the association between extended pharmacological VTE prophylaxis and outcomes among patients hospitalized with COVID-19.
Methods:
This was a retrospective cohort study of patients with an index positive SARS-CoV-2 polymerase chain reaction (PCR) test at the time of, or during hospitalization. Patients who were prescribed extended pharmacological VTE prophylaxis were compared against patients who were not. Multivariable logistic regression was used to produce odds ratio (OR) estimates and Cox proportional hazard models for hazard ratios (HR) with 95% CI to examine the association between pharmacological VTE prophylaxis and outcomes of interest. Primary outcomes were 30- and 90-day VTE events. Secondary outcomes included 30- and 90-day mortality, 30-day superficial venous thrombosis (SVT), acute myocardial infarction (MI), acute ischemic stroke, critical limb ischemia, clinically significant bleeding, and inpatient readmissions.
Results:
A total of 1936 patients were included in the study. Among them, 731 (38%) were discharged on extended pharmacological VTE prophylaxis. No significant difference was found in 30- and 90-day VTE events among groups. Patients discharged on extended VTE prophylaxis showed improved survival at 30 (HR: 0.35; 95% CI: 0.21–0.59) and 90 days (HR: 0.36; 95% CI: 0.23–0.55) and reduced inpatient readmission at 30 days (OR: 0.12; 95% CI: 0.04–0.33) when compared to those without.
Conclusion:
Patients discharged on extended VTE prophylaxis after hospitalization due to COVID-19 had similar thrombotic events on follow-up. However, use of extended VTE prophylaxis was associated with improved 30- and 90-day survival and reduced risk of 30-day inpatient readmission.
See commentary by Cameron SJ and Carman TL. Thromboprophylaxis for COVID-19: Time to ask for an extension? Vasc Med 2023; 28, 340–341.
Introduction
Since its origin in late 2019, coronavirus disease (COVID-19) has infected over half a billion patients, leading to millions of hospitalizations and claiming 6 million lives worldwide so far. 1 Although COVID-19 primarily involves the respiratory tract, it has been shown to be a multisystem disease causing increased risk of thromboembolic complications, especially venous thromboembolism (VTE). 2 Studies have shown increased risk of VTE in patients hospitalized due to COVID-19, especially among those requiring intensive care. 3 Early initiation of prophylactic anticoagulation in the hospital has been associated with improved survival. 4 Though in-hospital VTE prophylaxis is very commonly used in patients with COVID-19 infection, the role of extended pharmacological VTE prophylaxis remains unclear.
VTE is an important cause of preventable morbidity and mortality in hospitalized patients. Pharmacological agents like unfractionated heparin (UFH) and low molecular weight heparin (LMWH) are well studied and commonly used to reduce risk of VTE in high-risk medical patients during hospitalization. 5 Autopsy studies have indicated endothelial injury and microvascular thrombosis are important pathophysiologic features in patients who died from complications of COVID-196,7 and, unsurprisingly, VTE in COVID-19 is associated with worse outcomes. 8 In a landmark study published by Tang et al., prophylactic heparin showed improved survival in patients hospitalized for COVID-19, making pharmacological VTE prophylaxis an important part of management strategy. 9 Owing to challenges associated with timely diagnosis of VTE, risk stratification and appropriate VTE prophylaxis became even more important in COVID-19 patients. 10
Early in the pandemic, when studies were lacking regarding how best to care for patients with COVID-19, many health systems developed institutional expert committees to provide clinical guidance. At our health system, institutional experts formed a COVID Clinical Content Group in April 2020, which provided guidance on key clinical questions regarding the care of patients with COVID-19. One area of active discussion is the role of extended VTE prophylaxis after hospitalization with COVID-19. At the time of discharge, providers were advised to assess the IMPROVE VTE score, 11 IMPROVE BLEEDING score12–14 (auto-calculated from electronic medical records data), and to review D-dimer levels during hospitalization. Criteria to consider prescribing extended VTE prophylaxis included if the IMPROVE BLEEDING score was < 7 and IMPROVE VTE score was 4 or more, or with an IMPROVE VTE score of 2–3 with a D-dimer level of twice the upper limit of normal during hospitalization. 2 Guidance was included in discharge order sets (Figure 1). The purpose of this analysis was to examine the association between extended VTE prophylaxis with outcomes among patients hospitalized with COVID-19.

Discharge order guidance.
Methods
Study population and data collection
This retrospective analysis included adult patients (⩾ 18 years) who needed hospitalization and had an index severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) polymerase chain reaction (PCR) positive test between March 2020 and December 2020, within a health system comprising of 11 hospitals in Minnesota and Western Wisconsin. Patients were excluded for any of the following: pregnancy at the time of hospitalization, death, diagnosis of VTE, major bleeding event during hospitalization, on therapeutic or prophylactic anticoagulation for any indication before hospitalization, and patients who declined use of their medical records for health research. Data were abstracted from electronic medical records to include demographics as well as clinical characteristics by members of the organization’s clinical research informatics team. This study was approved by the Allina Heath Institutional Review Board (study #1661097).
Study measures
Patients were placed in two study groups based on prescribed therapy at the time of discharge (extended prophylactic anticoagulation, no anticoagulation). Extended prophylactic anticoagulation was defined as any of the following medications: unfractionated heparin 5000 units subcutaneous twice or three times per day, enoxaparin 40 mg subcutaneous once or twice per day (if body mass index > 40), and rivaroxaban 10 mg once per day. The primary clinical outcomes of interest were VTE at 30 and 90 days posthospital discharge. Secondary outcomes of interest were mortality at 30 and 90 days, superficial venous thrombosis (SVT), acute myocardial infarction (MI), acute ischemic stroke, clinically significant bleeding (defined as any bleeding needing clinic, urgent care, or emergency department visit, a procedure to control bleeding, or any amount of packed red blood cell transfusion), and inpatient readmission all within 30 days of discharge. Demographic characteristics included age, sex, patient-reported race and ethnicity, and history of tobacco use. Additional clinical characteristics included weight, major comorbid conditions, medications prior to hospitalizations, anticoagulation use during hospitalization, in-hospital supplemental oxygen needs, admission to intensive care unit (ICU), extracorporeal membrane oxygenation (ECMO), laboratory values of D-dimer, C-reactive protein (CRP), ferritin within 72 hours of hospitalization, and the Severity of Illness (SOI) and Risk of Mortality (ROM) modifiers assigned by the All Patient Refined Diagnosis Related Groups (APR-DRG). The SOI is defined as ‘the extent of organ system loss of function or physiologic decompensation’ whereas the ROM is the likelihood of in-hospital mortality based on age, diagnoses, and procedures (both are coded as minor, moderate, major, and extreme).15,16
Statistical analysis
Patients were compared to examine differences in outcomes of interest between those prescribed prophylactic anticoagulation and those not prescribed prophylactic anticoagulation at discharge. Descriptive statistics were used to summarize baseline and hospital-related characteristics among all study participants. Chi-squared, Fisher’s exact, and Wilcoxon rank sum tests were used, as appropriate, to compare differences in baseline and hospital-related characteristics between study groups.
To examine the association between prophylactic anticoagulation at discharge and clinical outcomes of interest, multivariable Firth’s logistic regression models were used to produce odds ratios (OR) with 95% CI. Firth’s logistic regression was used to reduce biases in predictions due to the small number of events occurring in models and separation of data points.17,18 Additionally, we examined differences in all-cause mortality between study groups. Survival was calculated from the date of discharge to the date of death within the first 30 and 90 days. Multivariable Cox proportional hazard models were used to examine the association between prophylactic anticoagulation at discharge and risk of death within 30 and 90 days from hospitalization with COVID-19.
Covariate selection
Covariates for all the aforementioned multivariable models included age, sex, race, severity of illness, in-hospital anticoagulation, remdesivir use, steroid use, any oxygen supplementation during hospitalization, and discharge disposition. The variables age, sex, race, severity of illness index, in-hospital anticoagulation, and discharge disposition were selected as covariates a priori.19–22 The severity of illness index incorporates comorbidities related to the diagnosis and hospital course of care, and therefore individual comorbidities were not included in multivariable models. 15 In-hospital remdesivir and steroid use were included due to the significant differences in proportions of patients receiving these therapies between study groups and any potential effect these therapies may have on clinical outcomes.23,24 Oxygen supplementation was included as a potential marker for severity of COVID-19 infection and due to the potential effect on outcomes.20,25,26 Owing to a very small number of patients in our study who received tocilizumab and convalescent plasma, these variables were not included in multivariable models. Analyses were performed using SAS for Windows, Version 9.4 (Cary, NC, USA) and p-values less than 0.05 were considered statistically significant.
Results
A total of 1936 patients were included in the analysis; 731 patients (38%) had been prescribed prophylactic anticoagulation at the time of discharge (Table 1). With respect to baseline characteristics, patients who were prescribed prophylactic anticoagulation at discharge were older (mean age 65 vs 62 years) and had a higher prevalence of chronic obstructive pulmonary disease (COPD) (60.7% vs 51.5%) and obesity (32.8% vs 26.4%) when compared to those not prescribed prophylactic anticoagulation at discharge. Race distribution differed between the two groups but ethnicity and language did not. A higher number (p < 0.0001) of patients in the prophylactic anticoagulation group received in-hospital therapies of remdesivir, convalescent plasma, steroids, and anticoagulation. A higher proportion of patients in the prophylactic anticoagulation group required supplemental oxygen. Furthermore, patients in this group had higher CRP within 72 hours and higher severity of illness and risk of mortality scores. Those who were not on prophylactic anticoagulation at discharge had the highest likelihood of being discharged home (self-care).
Differences in the distribution of demographic and clinical characteristics among patients who underwent hospitalization for COVID-19, by prophylactic anticoagulation status at discharge.
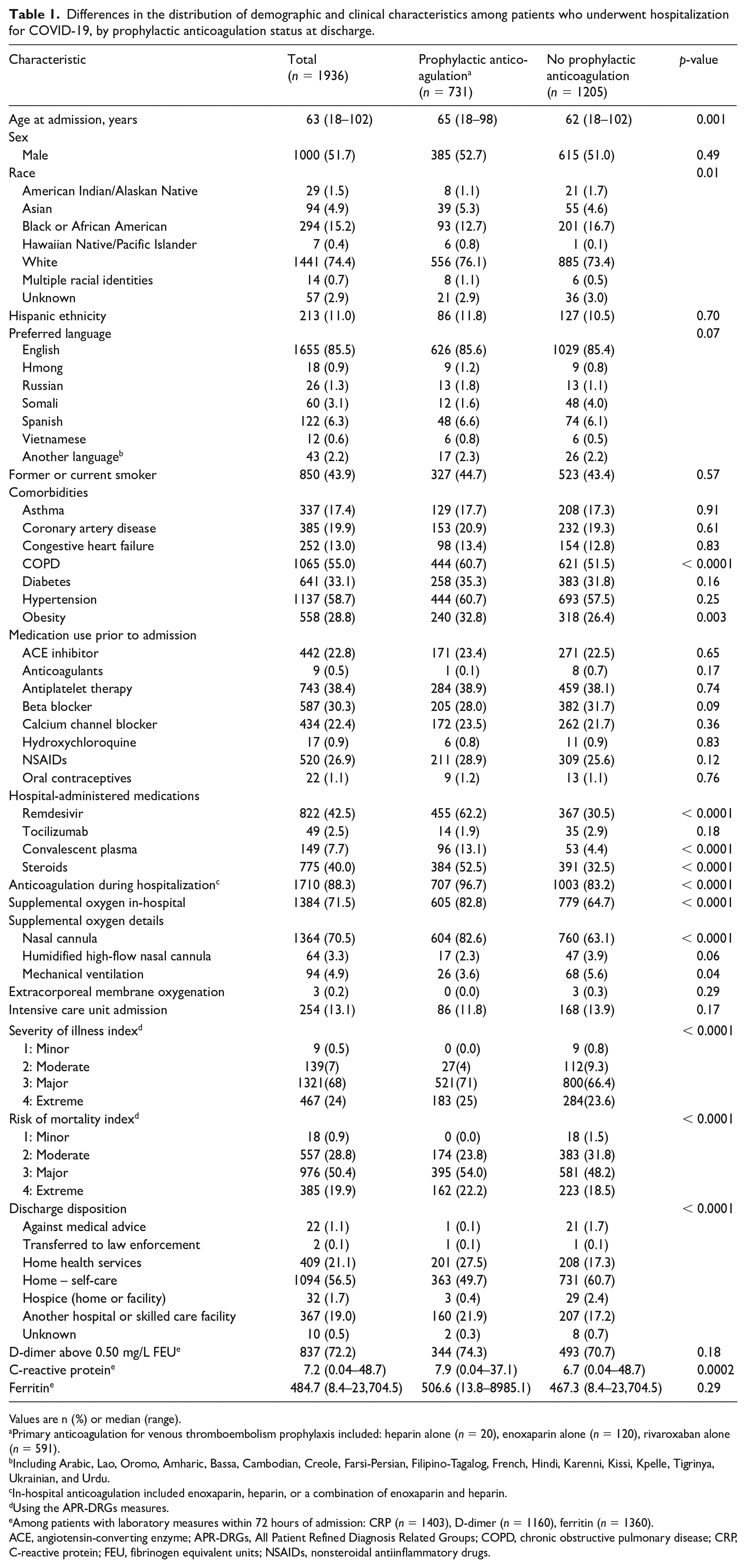
Values are n (%) or median (range).
Primary anticoagulation for venous thromboembolism prophylaxis included: heparin alone (n = 20), enoxaparin alone (n = 120), rivaroxaban alone (n = 591).
Including Arabic, Lao, Oromo, Amharic, Bassa, Cambodian, Creole, Farsi-Persian, Filipino-Tagalog, French, Hindi, Karenni, Kissi, Kpelle, Tigrinya, Ukrainian, and Urdu.
In-hospital anticoagulation included enoxaparin, heparin, or a combination of enoxaparin and heparin.
Using the APR-DRGs measures.
Among patients with laboratory measures within 72 hours of admission: CRP (n = 1403), D-dimer (n = 1160), ferritin (n = 1360).
ACE, angiotensin-converting enzyme; APR-DRGs, All Patient Refined Diagnosis Related Groups; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; FEU, fibrinogen equivalent units; NSAIDs, nonsteroidal antiinflammatory drugs.
After adjusting for potential confounding, there was no significant difference between the prophylactic anticoagulation groups and the primary outcomes of 30- or 90-day VTE (OR: 1.13; 95% CI: 0.52–2.49 and OR: 1.30; 0.67–2.52, respectively) when compared to those without prophylactic anticoagulation (Table 2). Patients prescribed prophylactic anticoagulation had a significantly lower inpatient services readmission rate within 30 days from discharge (OR: 0.12; 95% CI: 0.04–0.33). There were no significant differences between study groups with respect to SVT, ischemia, acute MI, acute stroke, or clinically significant bleeding. It should be noted that these were rare events reflected by the small numbers.
Differences in the distribution of clinical outcomes among patients hospitalized for COVID-19, by prophylactic anticoagulation status at discharge.
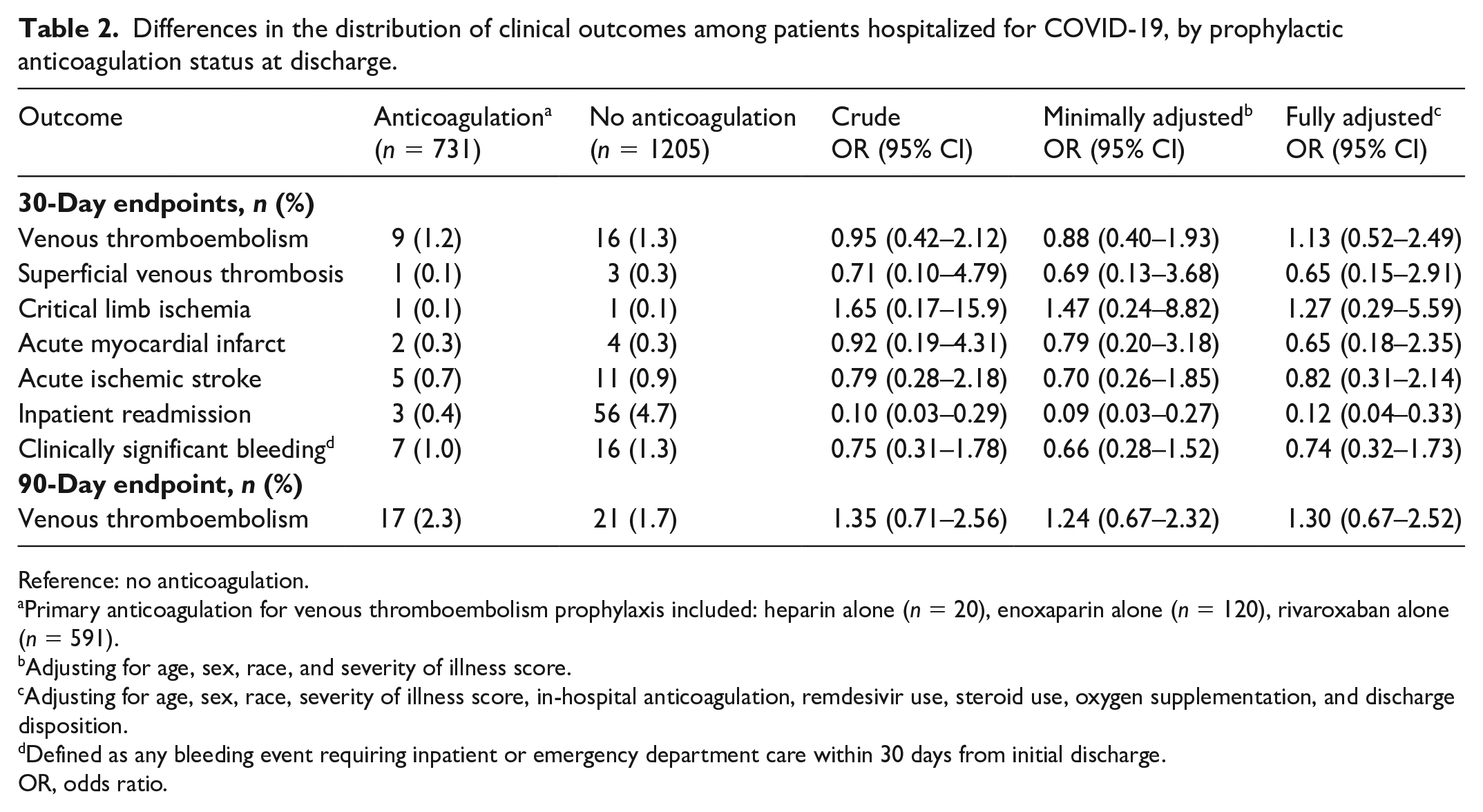
Reference: no anticoagulation.
Primary anticoagulation for venous thromboembolism prophylaxis included: heparin alone (n = 20), enoxaparin alone (n = 120), rivaroxaban alone (n = 591).
Adjusting for age, sex, race, and severity of illness score.
Adjusting for age, sex, race, severity of illness score, in-hospital anticoagulation, remdesivir use, steroid use, oxygen supplementation, and discharge disposition.
Defined as any bleeding event requiring inpatient or emergency department care within 30 days from initial discharge.
OR, odds ratio.
When examining the association between prophylactic anticoagulation and risk of death, patients in the prophylactic anticoagulation group had a 65% reduction in the risk of dying within 30 days (HR: 0.35; 95% CI: 0.21–0.59) when compared to those who were not prescribed prophylactic anticoagulation (Table 3, online Supplementary Figure 1). This association remained at 90 days postdischarge (HR: 0.36; 95% CI: 0.23–0.55) (Table 3, online Supplementary Figure 2).
Risk of dying within 30 and 90 days after hospitalization among patients with COVID-19, by prophylactic anticoagulation status at discharge.
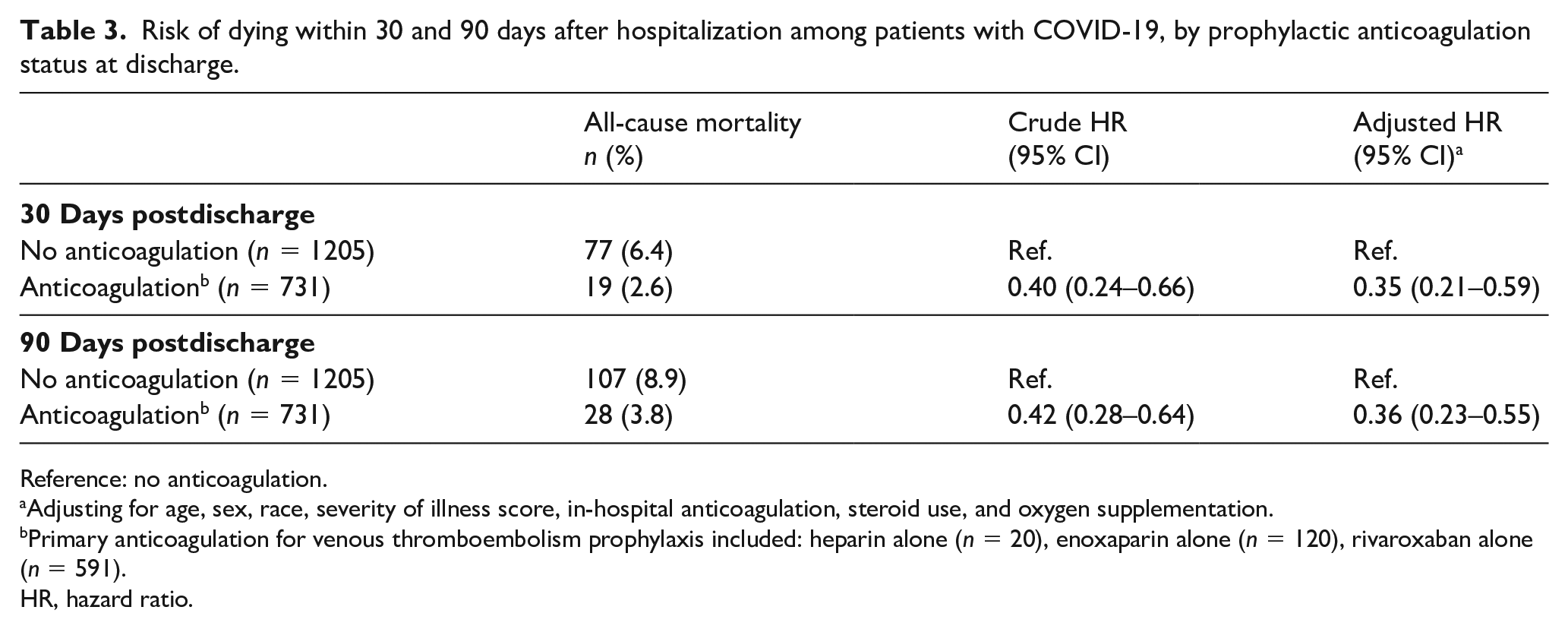
Reference: no anticoagulation.
Adjusting for age, sex, race, severity of illness score, in-hospital anticoagulation, steroid use, and oxygen supplementation.
Primary anticoagulation for venous thromboembolism prophylaxis included: heparin alone (n = 20), enoxaparin alone (n = 120), rivaroxaban alone (n = 591).
HR, hazard ratio.
Discussion
This study analyzes the real-world experiences of extended pharmacological VTE prophylaxis in patients hospitalized with COVID-19. We did not find significant differences in the primary outcome of VTE at 30 and 90 days. Our study did show improved survival at 30 and 90 days as well as reduction in 30-day readmissions in patients discharged on pharmacological VTE prophylaxis.
In most major studies showing benefits of pharmacological VTE prophylaxis in hospitalized medical patients, 6–14 days has been used as a standard duration for prophylactic anticoagulation. 27 In practice, the average duration of hospitalization for medical conditions is around 4–5 days, with risk of developing VTE persisting after hospital discharge. 28 Studies have shown a benefit of up to 6 weeks of extended pharmacological VTE prophylaxis after hospitalization for medical illness in selected patients.29,30 Based on results from the MAGELLAN and MARINER clinical trials, rivaroxaban has been approved by the US Food and Drug Administration (FDA) for extended VTE prophylaxis in selected medical patients.30,31 Similarly, based on findings of the APEX trial, betrixaban was previously approved by the FDA for VTE prophylaxis during hospitalization and after discharge for medical patients before it was later-on withdrawn from the market. 32
Risk of VTE after hospitalization for COVID-19 is well reported and can persist for weeks after discharge.33,34 Role of extended VTE prophylaxis after hospitalization for COVID-19 has remained an active area of discussion and research, and currently no consensus exists on this important clinical question.
A large prospective registry with 4906 postdischarge patients after hospitalization for COVID-19 highlighted the risk of thromboembolic complications and mortality after hospitalization, and advised the need for postdischarge surveillance including consideration of extended VTE prophylaxis. 35 Most current guidelines do not recommend routine extended VTE prophylaxis after COVID-19 hospitalization due to limited evidence and concern for bleeding risk associated with anticoagulant medications. 36
Overall, the rate of VTE at 30 and 90 days after hospitalization with COVID-19 was low in our study, which is consistent with published studies.33,35,37 Owing to low incidence of postdischarge VTE, the decision on prescribing extended prophylaxis is complicated, especially due to the high-risk nature of anticoagulant medications. However, bleeding complications in our study were low, which aligned with major trials evaluating extended VTE prophylaxis.30,31 Recently, a randomized controlled trial (MICHELLE) showed improved outcomes in high-risk patients (IMPROVE VTE score ⩾ 4, or 2–3 with D-dimer > 500 ng/mL) discharged on 35 days of rivaroxaban after hospitalization for COVID-19 without major bleeding in either group. 38 Our study adds to the current literature, demonstrating that in real-world experience extended VTE prophylaxis is well tolerated in patients with COVID-19 without excess bleeding risk, and showed improved patient outcomes and reduced 30-day readmissions.
The PREVENT-HD trial randomized 1284 symptomatic outpatients with positive COVID-19 testing and had at least one additional risk factor (age over 60 years; prior history of VTE, thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity or D-dimer > upper limit of normal) to receive rivaroxaban 10 mg daily or placebo for 35 days. The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, MI, ischemic stroke, acute limb ischemia, noncentral nervous systemic embolization, all-cause hospitalization, and all-cause mortality. The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical site and fatal bleeding. The trial was stopped early because of a lower-than-expected event incidence (3.2%) compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.39,40
The author attributed the low event rate to a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines. Results of the main intention-to-treat analysis (in 1284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
A recent meta-analysis that systematically searched for studies that evaluated the effects of extended thromboprophylaxis in postdischarge patients with COVID-19 until 13 June 2022, included eight studies involving 10,148 patients. The results confirmed that extended thromboprophylaxis, primarily prophylactic use of anticoagulants for < 35 days, was significantly associated with a reduced composite outcome of thromboembolism and all-cause mortality in high-risk postdischarge patients with COVID-19 (OR: 0.52; 95% CI: 0.41–0.67, p = 0.000). Interaction analysis revealed that the effect estimates were consistent between the randomized control trials (RCT) and observational studies (pinteraction = 0.310). Similar to our findings, extended thromboprophylaxis did not increase the risk of major bleeding events (OR: 1.64; 95 % CI: 0.95–2.82, p = 0.075). 41
Studies have shown that patients hospitalized after a COVID-19 positive test had a higher risk of dying up to 12 months after discharge compared to COVID-19 negative patients, 42 and that the majority of post-COVID-19 discharge mortality appears to occur within 30 days after discharge. 43 We continue to learn effects and impacts of post COVID-19 syndrome on different organ systems and potential complications. 44
In light of available literature, it is clear that the effects of COVID-19 persist beyond hospitalization and clinical management should take that into consideration. Our study addresses an area of management which currently has very limited evidence available, and shows the potential to impact outcomes after COVID-19-related hospitalization while we await results of additional clinical studies assessing extended VTE prophylaxis in COVID-19 patients.
In spite of having a higher incidence of comorbid conditions including COPD and obesity, higher severity of illness, and risk of mortality scores, patients discharged home with extended VTE prophylaxis showed improved survival at both 30 and 90 days and decreased risk of inpatient readmission at 30 days. Though the exact reasons remain elusive, several points are worth mentioning. First, to their counterpart, a higher (p < 0.0001) number of patients discharged home on extended VTE prophylaxis received remdesivir, convalescent plasma, and steroids (Table 1). It is certainly possible that these medications had an impact on survival and hospital readmission. Second, since our study included subjects admitted to the hospital in the month of December 2020 – a time during which COVID-19 vaccination was first made available to health care workers and high-risk patients, it is possible that some high-risk subjects had already received their first vaccination dose and this might have impacted disease severity and long-term mortality. Third, the lower mortality and readmission rates may be explained by the antiinflammatory and antiviral effects of heparin. 45
Study limitations
There are limitations of this study that should be noted. The nonrandomized design and retrospective nature is a major limitation. Although we had a large sample size of patients, the outcomes of interest were relatively rare. Future studies with larger sample sizes should continue to examine these outcomes. Our study spans over a time when our understanding of COVID-19 was evolving, and universal testing of all hospitalized patients was not done at our institution due to scarcity of tests. Additionally, some therapies (e.g., steroids) were not shown to improve survival yet. As such, some patients with COVID-19 may have been inadvertently excluded due to unknown status, and in-hospital treatments may have changed over time. After some consideration, we decided to include all hospitalized patients with index SARS-CoV-2 PCR positivity during the study period, which could have led to the inclusion of patients where COVID-19 positivity was asymptomatic. It is unknown if asymptomatic COVID-19 positivity may contribute to any additional VTE risk in hospitalized patients during and after hospitalization. Though education sessions and discharge order guidance (Figure 1) were available to providers about when to consider extended VTE prophylaxis, it was up to the discharging provider regarding to which patients to prescribe extended VTE prophylaxis and, as such, selection bias in prescribing extended VTE prophylaxis should be considered. Providers were guided to provide extended VTE prophylaxis based on the IMPROVE VTE score and D-dimer values outlined in the Introduction, but we did not measure whether patients actually received prophylaxis based on advice – so again, selection bias remains a limitation. Furthermore, despite our study being generally representative of the United States’ racial and ethnic populations, our study population was primarily non-Hispanic White, and future work with oversampling of patients from other racial and ethnic backgrounds will enhance the generalizability of results. 46 Finally, this study included subjects treated at the time when milder COVID-19 variants were not prevalent and when vaccination was not widely available for health care workers and high-risk patients. The impact of vaccination on the outcomes cannot evaluated.
Summary
In summary, this study did not show reduced clinical VTE incidence at 30 and 90 days in patients prescribed extended VTE prophylaxis after hospitalization with COVID-19. It did suggest extended pharmacological VTE prophylaxis can have beneficial effects in reducing mortality and 30-day readmissions in patients discharged after hospitalization with COVID-19 without increased bleeding risk. Future research should continue to examine the risk and benefit of extended prophylactic anticoagulation after a COVID-19 hospitalization among larger sample sizes to reproduce these findings.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231159945 – Supplemental material for Outcomes in patients discharged with extended venous thromboembolism prophylaxis after hospitalization with COVID-19
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231159945 for Outcomes in patients discharged with extended venous thromboembolism prophylaxis after hospitalization with COVID-19 by Love Patel, Ashley Stenzel, Christopher Van Hove, Abbey Sidebottom, Rajesh Kethireddy, Ngoc Ha, David Beddow, Jesse Manunga, Ghaziuddin Qadri, Justin Kirven and Nedaa Skeik in Vascular Medicine
Supplemental Material
sj-pdf-2-vmj-10.1177_1358863X231159945 – Supplemental material for Outcomes in patients discharged with extended venous thromboembolism prophylaxis after hospitalization with COVID-19
Supplemental material, sj-pdf-2-vmj-10.1177_1358863X231159945 for Outcomes in patients discharged with extended venous thromboembolism prophylaxis after hospitalization with COVID-19 by Love Patel, Ashley Stenzel, Christopher Van Hove, Abbey Sidebottom, Rajesh Kethireddy, Ngoc Ha, David Beddow, Jesse Manunga, Ghaziuddin Qadri, Justin Kirven and Nedaa Skeik in Vascular Medicine
Footnotes
Data availability
The data that support the findings of this study may be available upon reasonable request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
