Abstract
Patient-reported difficulties in affording health care and their association with health status outcomes in peripheral artery disease (PAD) have never been studied. We sought to determine whether financial barriers affected PAD symptoms at presentation, treatment patterns, and patient-reported health status in the year following presentation. A total of 797 United States (US) patients with PAD were identified from the Patient-centered Outcomes Related to TReatment Practices in Peripheral Arterial Disease: Investigating Trajectories (PORTRAIT) study, a prospective, multicenter registry of patients presenting to vascular specialty clinics with PAD. Financial barriers were defined as a composite of no insurance and underinsurance. Disease-specific health status was measured by Peripheral Artery Questionnaire (PAQ) and general health-related quality of life was measured by EuroQol 5 (EQ5D) dimensions at presentation and at 3, 6, and 12 months of follow-up. Among 797 US patients, 21% (n = 165) of patients reported financial barriers. Patients with financial barriers presented at an earlier age (64 ± 9.5 vs 70 ± 9.4 years), with longer duration of symptoms (59% vs 49%) (all p ⩽ 0.05), were more depressed and had higher levels of perceived stress and anxiety. After multivariable adjustment, health status was worse at presentation in patients with financial barriers (PAQ: –7.0 [–10.7, –3.4]; p < 0.001 and EQ5D: –9.2 [–12.74, –5.8]; p < 0.001) as well as through 12 months of follow-up (PAQ: –8.4 [–13.0, –3.8]; p < 0.001 and EQ5D: –9.7 [–13.2, –6.2]; p < 0.001). In conclusion, financial barriers are associated with later presentation as well as poorer health status at presentation and at 12 months.
Introduction
Estimates of the number of people without insurance in the US have declined from nearly 50 million in 2010 1 to approximately 28 million in 2015, but for those who are uninsured, there are persistent challenges. 2 The percentage of underinsured adults, has been stable at 22% from 2010 to 2014 – an estimated 31 million. 3 Lack of health insurance is associated with decreased use of health care services, more unmet health care needs, lack of access to medical care, and worse outcomes in general.4–7 However, having access to health insurance does not translate into access to optimal care. 7 Patients with health insurance who have difficulty affording health care are usually defined as underinsured. 3 The problem of underinsurance, although less well studied, has also been thought to have a similar adverse impact on patient care and outcomes in general.8,9
In the case of peripheral artery disease (PAD), estimated to affect approximately 8.5 million individuals above the age of 40 years in the US 10 and more than 200 million worldwide, 11 there are little data evaluating the associations of insurance status with PAD outcomes. 12 This poses a challenge, especially since PAD is often underdiagnosed and undertreated. 13 Previous work showed that, in 2006, under the Massachusetts Health Care Reform, after expansion of coverage to 98% of its residents, disparities in PAD severity and amputation and revascularization rates by race and socioeconomic status were no longer detected. 14 A further understanding of how financial barriers to care are associated with PAD symptom presentation, care, and outcomes in a real-world cohort is critical as it may highlight potential targets for supportive interventions that can help improve PAD outcomes.
To address this gap, we examined the associations of financial barriers, a composite of no insurance and underinsurance, with patient characteristics, patterns of care (including guideline-directed medical therapy, referral for exercise therapy, and smoking cessation counseling), psychosocial factors, and health status outcomes in the Patient-centered Outcomes Related to TReatment Practices in Peripheral Arterial Disease: Investigating Trajectories (PORTRAIT) study. An understanding of these associations may have important implications given the current debate on the coverage and affordability of health insurance at a national and state level.
Methods
The PORTRAIT study was a multinational study designed to examine the characteristics of patients presenting with new or worsening PAD symptoms to vascular specialty clinics, variation in practice patterns of evidence-based PAD treatments, and the impact on health outcomes. 15 The study recruited patients from centers in the US (n = 10), The Netherlands (n = 5), and Australia (n = 1) between June 2011 and October 2015. Patients aged 21 years or older, presenting to a PAD specialist (vascular surgeon, vascular medicine specialist, radiologist or cardiologist), patients with new-onset or recent exacerbation of exertional leg symptoms and an abnormal resting ankle–brachial index (ABI) (⩽ 0.90) 16 or a significant drop in post-exercise ankle pressure of ⩾ 20 mmHg 17 were included. Those with a non-compressible ABI, lower-limb revascularization in the same leg, critical limb ischemia, who did not speak English/Spanish/Dutch, who were hard of hearing, prisoners, patients previously enrolled in the PORTRAIT registry or those who were unable to provide informed consent were excluded. For the purposes of this study, patients from non-US sites and those with missing data on insurance and financial concerns were excluded; the former were excluded since insurance coverage differs by country. The PORTRAIT study was designed in accordance with the Helsinki Declaration and the protocol was approved by the institutional review boards of all participating centers. Written or telephone informed consent was obtained according to the approved protocol.
Classification and measurement of financial barriers
We compared US patients with financial barriers to those with no financial barriers. The presence of financial barriers was defined as: (1) no insurance; and (2) underinsurance, defined as having insurance but reporting financial concerns to care. 7 Financial barriers were assessed using a self-reported questionnaire wherein patients were asked three validated questions (Figure 1)10,18,19,20 assessing whether, due to cost, patients: (1) avoided obtaining health care services; (2) had not taken a prescribed medication in the past year; and (3) experienced difficulty in getting medical care when needed (Figure 2). A binary ‘yes’ or ‘no’ scale was used for the 1st question, with a 5-point Likert scale ranging from ‘always’ to ‘never’ for the 2nd and ranging from ‘extremely difficult’ to ‘not a problem at all’ for the 3rd question. Those who answered either ‘yes’ to the 1st question, or ‘always’/‘frequently’/‘occasionally’ to the 2nd question, or ‘extremely difficult’/‘moderately difficult’/‘somewhat difficult’ to the 3rd question were categorized as having financial concerns. This approach has been tested and studied in prior acute myocardial infarction (MI) cohorts.9,18,19
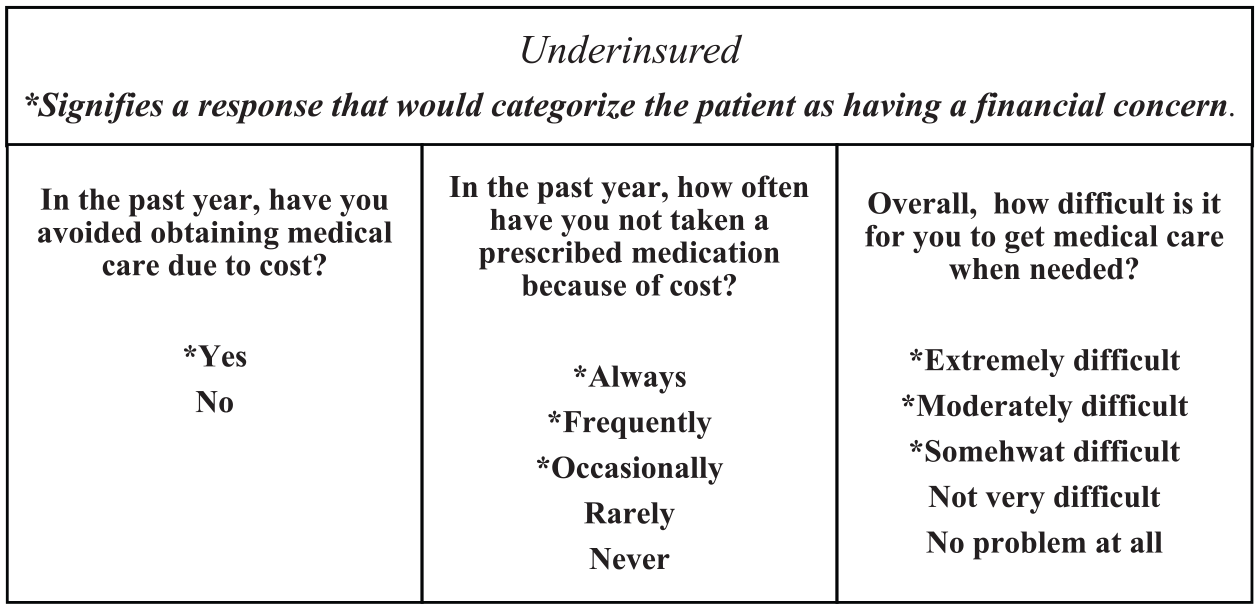
Financial barriers were assessed by asking patients a series of three questions.
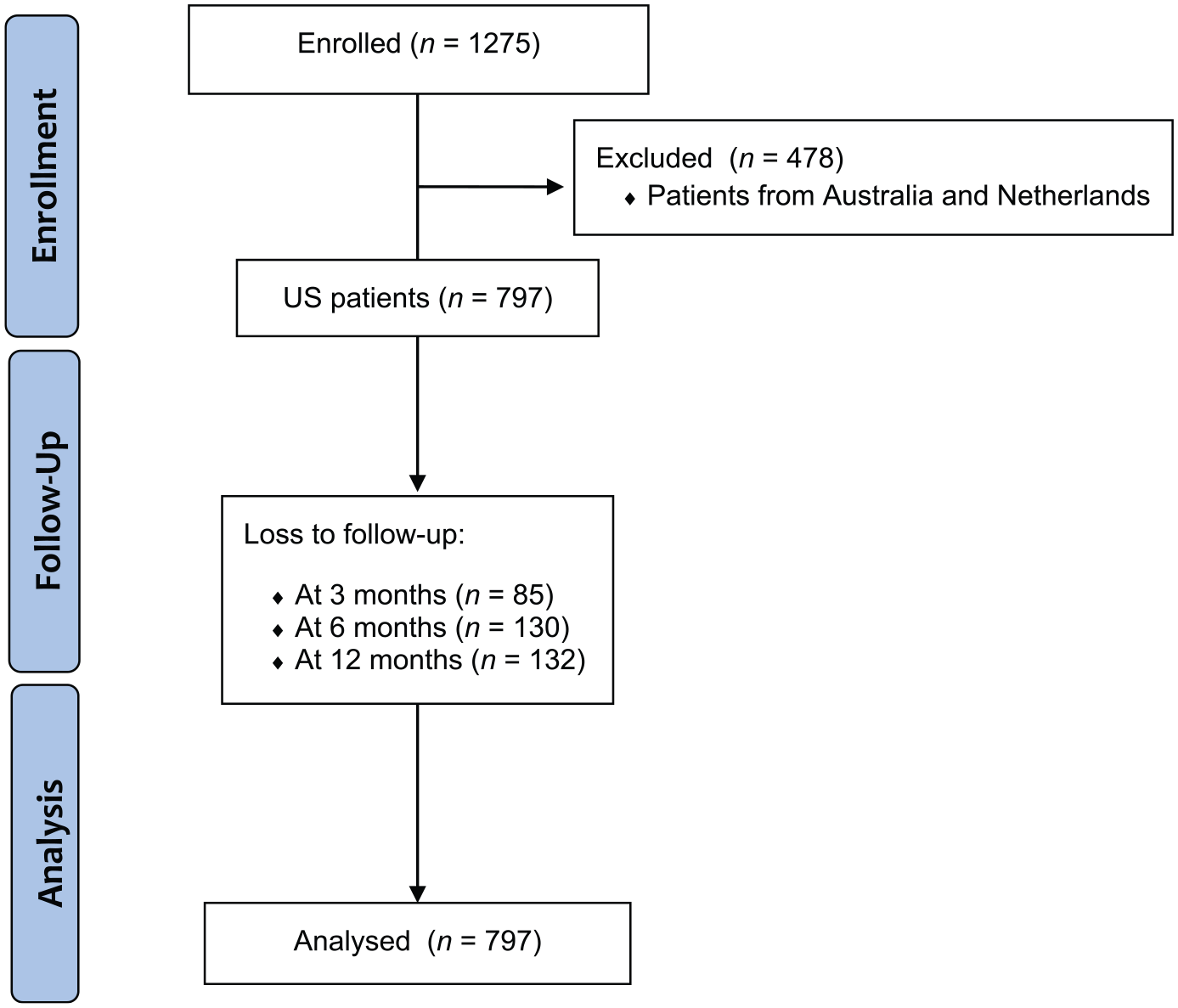
Consolidated Standards of Reporting Trials (CONSORT) flow diagram for enrollment and loss to follow-up at 3, 6, and 12 months.
Care patterns, health outcomes and psychosocial factors
Patterns of care were assessed by guideline-directed medical therapy (GDMT), referrals to supervised exercise therapy, and smoking cessation. These are part of the performance measure set 20 for PAD as well as part of the recent guidelines. 21 GDMT included use of antiplatelet, statin, and antihypertensive therapies. We also looked at provider specialty (varying between interventional cardiology, vascular surgery, interventional radiology, vascular medicine, and general cardiology).
Health status was measured at baseline using the Peripheral Artery Questionnaire (PAQ) and the EuroQol Visual Analogue Scale (EQ5D-VAS). The PAQ is a 20-item, disease-specific questionnaire that assesses six health status domains relevant to patients with PAD: symptoms, recent changes in symptoms, physical limitations due to claudication, treatment satisfaction, social functioning, and quality of life. 22 A summary score (range 0–100) is calculated as the average of scores from each domain, with higher scores indicating less functional limitation, fewer symptoms, better treatment satisfaction, higher social functioning, and better quality of life. 23 We supplemented this with information from the EQ5D-VAS. The EQ5D, which is a validated generic instrument that uses five different dimensions of health assessment (mobility, self-care, usual activities, pain or discomfort, and anxiety/depression), contains a visual analogue scale (VAS) that rates individuals’ perception of their overall health and is often used as a summarized index to measure health status from a patient’s perspective with a higher score denoting a better status. 24 PAQ and EQ5D-VAS scores were obtained at 3, 6, and 12 months to look at longitudinal changes in health status.
Psychosocial variables were obtained from the baseline patient interview and included depressive symptoms, anxiety, and perceived stress. Depressive symptoms were assessed using the Patient Health Questionnaire (PHQ-8), 25 an eight-item depression scale that has been established as a valid diagnostic and severity tool for depressive disorders in both clinical and large-population based studies. This measure quantifies the frequency of depressive symptoms experienced in the past 2 weeks based on the eight Diagnostic and Statistical Manual of Mental Disorders (4th edition) criteria for major depressive disorder. A PHQ-8 score ⩾ 10 has 88% sensitivity and specificity to detect major depression.
Anxiety was measured using the Generalized Anxiety Disorder scale (GAD-2), 26 a short screening tool that consists of the first two questions of the GAD-7 scale, which has good reliability, and factorial and structural validity for the diagnosis of generalized anxiety disorder. 27 In the GAD-2, participants are asked about how often they feel nervous and how often they are not able to stop worrying. A score of > 3 indicates possible anxiety disorder.
Perceived stress was measured using the Perceived Stress Scale (PSS-10), 28 a widely used instrument for measuring the perception of stress (i.e. the degree to which situations in life are perceived as stressful). It consists of 10 questions asking respondents about their feelings of stress during the last month. The PSS has been shown to have adequate internal and test-retest reliability and is one of the most widely used instruments for measuring non-specific perceived stress.
We abstracted clinical characteristics from the electronic medical records at baseline. This included age, sex, race, education, employment, living status, and insurance status. Clinical comorbidities included history of stroke, hypertension, angina, MI, percutaneous coronary interventions, coronary bypass surgery, chronic kidney and lung disease, diabetes, and smoking.
PAD specific characteristics were also abstracted. We included history of amputation and any intervention for PAD which was further categorized into surgical and endovascular interventions. As indicators of disease burden and severity, we collected information about patients’ symptom presentation, duration of pain, Rutherford category, 29 resting ABI, and lesion characteristics. The latter included lesion location (iliac/femoral/infrapopliteal/distal), lesion site (proximal/distal/both), and symptomatic leg (right/left/both, single/both).
Statistical analysis
We compared baseline characteristics, patterns of care, psychosocial factors, and health status between patients with and without financial barriers using Student’s t-test for continuous variables and chi-squared or Fisher’s exact tests for categorical variables. We used linear mixed-effects models to examine the longitudinal health status (PAQ summary score and EQ5D-VAS score) over time (baseline and 3, 6, and 12-month follow-up) in each financial barrier group, modeling the individual as a random effect and using an unstructured covariance structure to estimate correlations among repeated assessments within individuals. The unadjusted model included main effects of the financial barrier group and time point, as well as an interaction between the financial barrier group and time. The adjusted model expanded on the unadjusted model including 11 baseline patient characteristics: age, sex, race, education, working status, symptomatic leg, ABI, smoking status, and history of MI, percutaneous coronary intervention, and diabetes. We verified the interaction between receiving invasive treatment within 3 months (both endovascular and surgical) with financial burden, but since the interaction term for invasive treatment was not significant (p = 0.64), we did not adjust for invasive treatment in the final model. All analyses were performed with SAS software, Version 9.4 (SAS Institute Inc., Cary, NC, USA). A p-value of < 0.05 was considered statistically significant.
Results
After excluding patients not from the US (n = 478), the final analytical cohort consisted of 797 US patients. Figure 2 shows a CONSORT flow diagram from which the final analytical cohort was derived. These patients were enrolled from 10 US vascular specialty clinics. Patients from non-US sites were excluded because of differences in health care delivery systems potentially affecting insurance patterns. Baseline characteristics of the total sample stratified by presence/absence of financial barriers are shown in Table 1. In the total cohort, 21% (n = 165) reported financial barriers.
Insurance status, demographics, cardiac risk factors/comorbidities, PAD characteristics, and treatment patterns stratified by financial status.
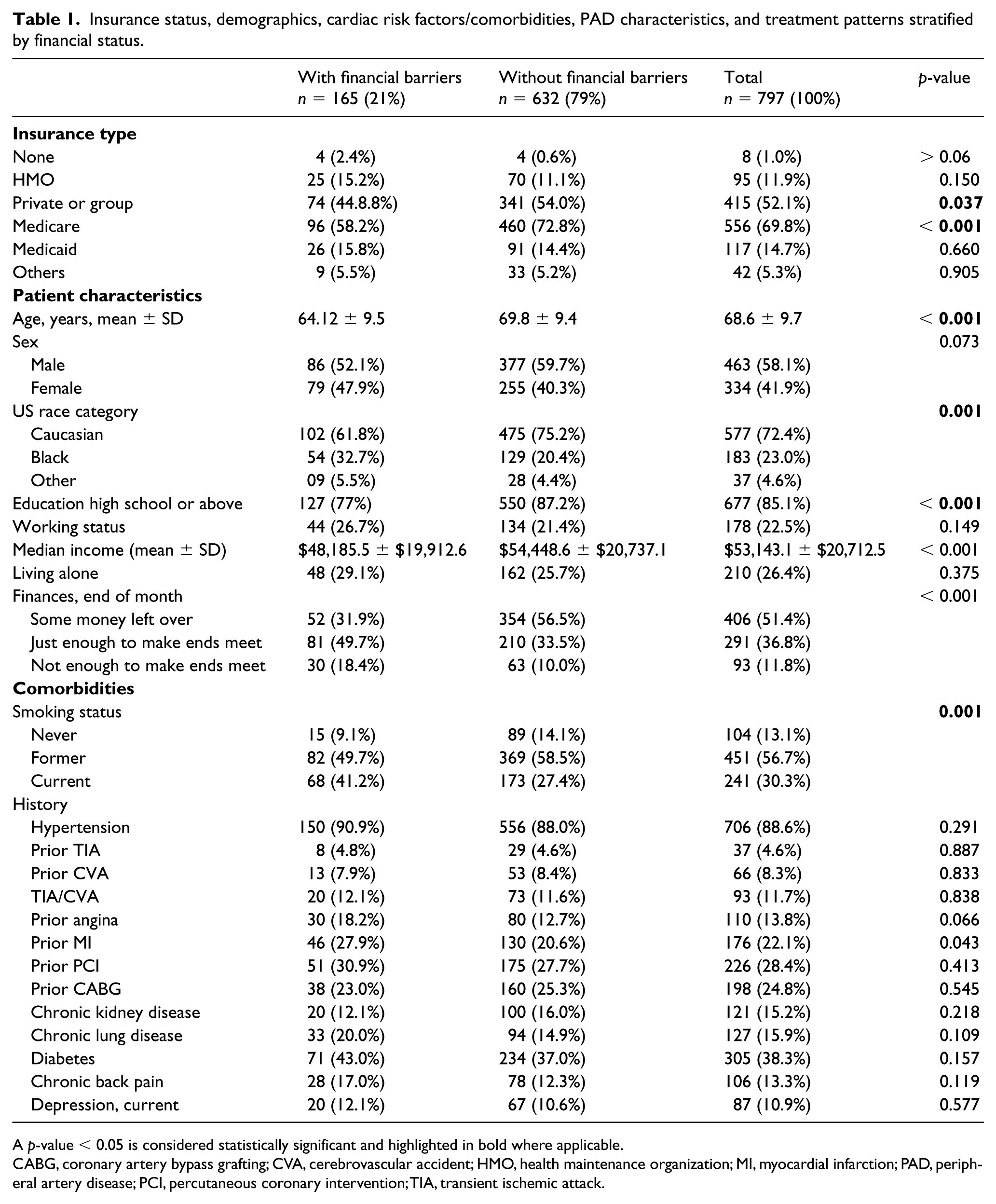
A p-value < 0.05 is considered statistically significant and highlighted in bold where applicable.
CABG, coronary artery bypass grafting; CVA, cerebrovascular accident; HMO, health maintenance organization; MI, myocardial infarction; PAD, peripheral artery disease; PCI, percutaneous coronary intervention; TIA, transient ischemic attack.
Demographics and socioeconomic status
The mean age of our patient population was 69 ± 10 years, with 58% males and 72% Caucasian (Table 1). Those with financial barriers were younger (64 ± 9.5 vs 70 ± 9.4 years, p < 0.001) and more likely to be black (32.7% vs 20.4%, p = 0.001). Overall, 85% had at least a high school education, while 78% did not work and 26% lived alone. Patients with financial barriers were less likely to have at least a high school education (77% vs 87%, p < 0.001).
Out of a total of 797 patients, 21% (n = 165) had financial barriers while 79% (n = 632) did not (Table 1). Among those 165 with financial barriers, the majority were underinsured (97.6%, n = 161), with only 2.4% being uninsured (n = 4). Of note, out of a total of four uninsured patients, eight patients did not report financial barriers to care and were allocated to the no financial barriers group. Of the 165 patients with financial barriers, 79% (n = 130) avoided obtaining health care services, 47% (n = 77) had not taken a prescribed medication in the past year, and 14% (n = 23) found it difficult to get medical care when needed (Figure 3). Among patients with financial barriers, 27% (n = 45) were categorized as having financial concerns based on their positive response to two questions and 5% (n = 8) based on their positive response to three questions (Figure 1). Patients with financial barriers had a significantly lower household median income compared to those without financial barriers, as extrapolated from the census database ($48,185.5 ± $19,912.6 vs $54,448.6 ± $20,737.1, p < 0.001). Additionally, patients with financial barriers were more likely to report that they either just had enough to make ends meet (49.7% vs 33.5%, p < 0.001) or did not have enough money to make ends meet at the end of the month compared with those without financial barriers (18.4% vs 10.0%, p < 0.001). Compared with those without any financial barriers, a smaller proportion of those with financial barriers had private insurance (44.8% vs 54.0%, p < 0.05) or Medicare (58.2% vs 72.8%, p < 0.001). There were no differences in those with Medicaid (15.8% vs 14.4%, p = 0.660).
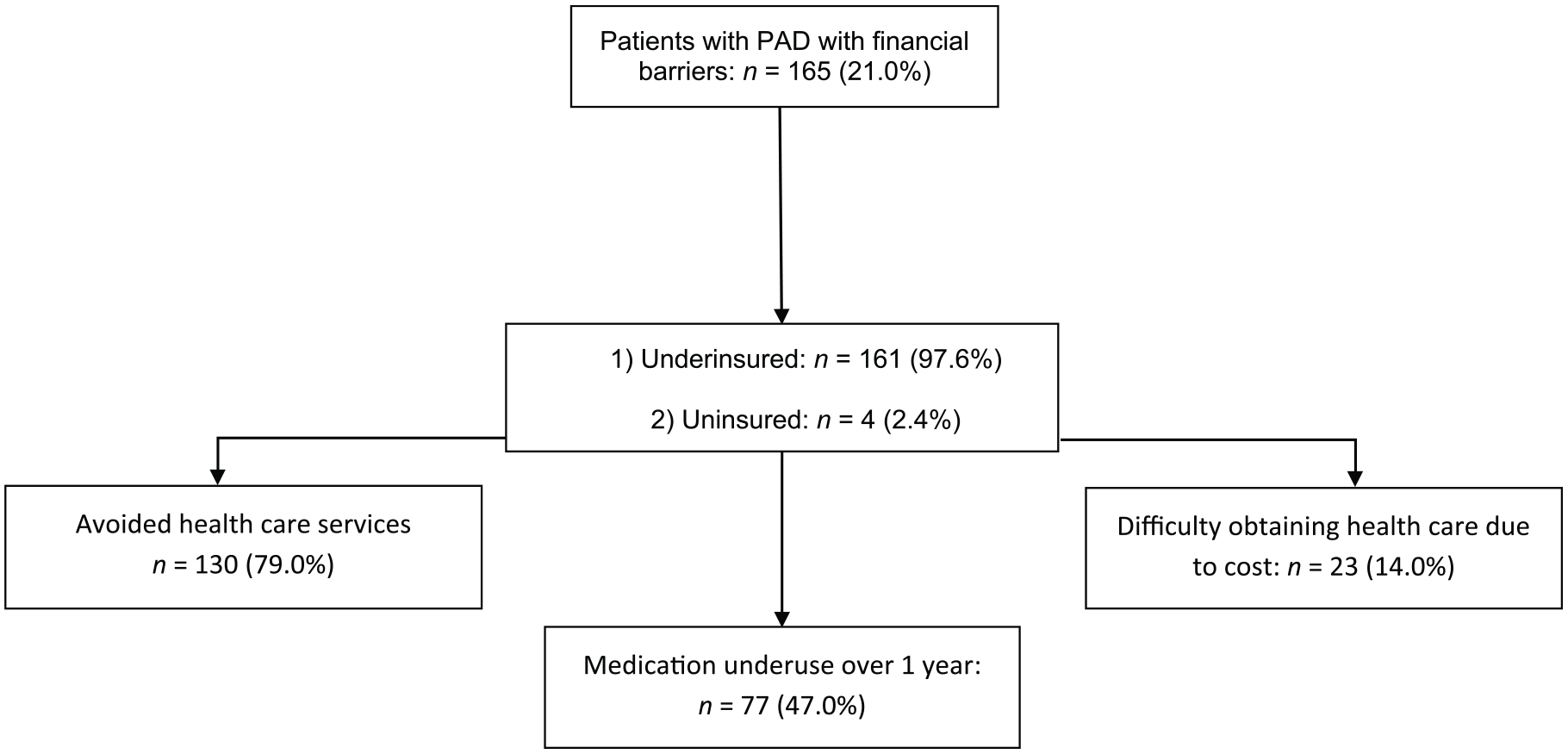
Patients with financial barriers reported different patterns of barriers to care including avoiding health care due to cost, medication underuse or difficulty obtaining care.
Cardiac risk factors/history and comorbidities
Those with financial burden had higher rates of current smoking (41.2% vs 27.4%, p = 0.001), but no differences were found in comorbidities/risk factors (hypertension, diabetes, history of MI etc) between the groups (Table 1).
PAD characteristics, vascular history, and lesion characteristics
The majority (88%) of patients presented with typical symptoms (Table 2) and those with financial barriers more often reported greater than 12 months of pain (59.5% vs 49.2%, p < 0.05). There were no statistically significant differences between the two groups in the mean resting ABIs, symptom severity, rates of amputation/any PAD intervention, lesion characteristics (location and site), laterality or primary provider for PAD.
PAD history, treatment, disease characteristics, and primary PAD specialty stratified by financial status.
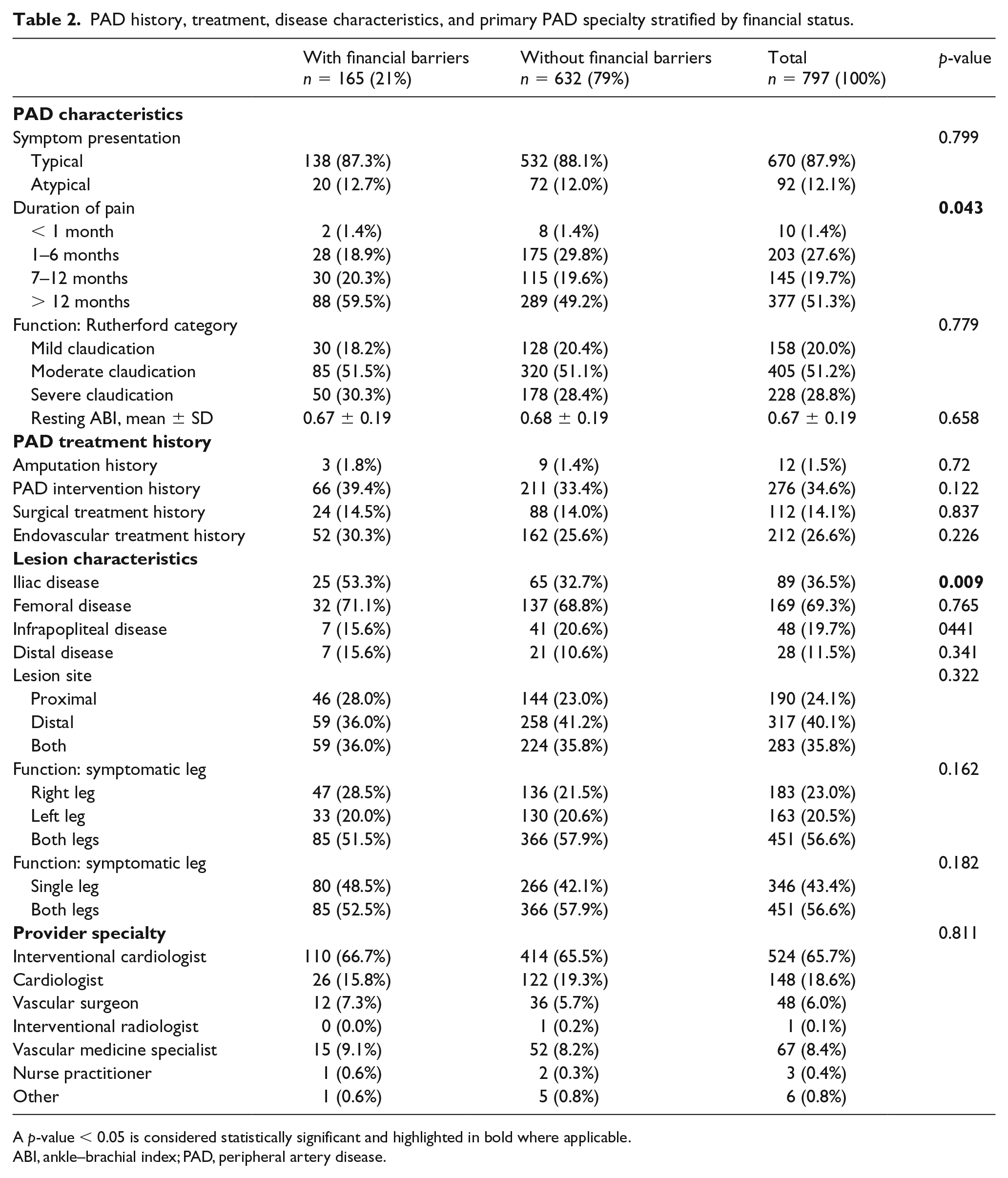
A p-value < 0.05 is considered statistically significant and highlighted in bold where applicable.
ABI, ankle–brachial index; PAD, peripheral artery disease.
Medications, smoking cessation, and exercise therapy and invasive treatment
Compared with patients without financial barriers, patients with financial barriers were just as likely to be on GDMT and receive exercise therapy referrals. As compared with those who did not have barriers, 81% and 90% of patients with financial burden were on statin and antiplatelet therapies, respectively (Table 3). Similar rates of beta-blockers, angiotensin-converting enzymes inhibitors and angiotensin receptor blocker were noted across patients who reported financial barriers versus those who did not. However, compared with patients who did not report financial barriers, fewer patients with financial barriers were on calcium channel blockers (34% vs 36%). There were no differences in referral for exercise therapy and smoking cessation counseling, nor were there any differences in rates of invasive (endovascular and surgical) treatment between the two groups within the first 3 months of presentation.
Quality of PAD care stratified by financial status (prior to and after PAD evaluation).
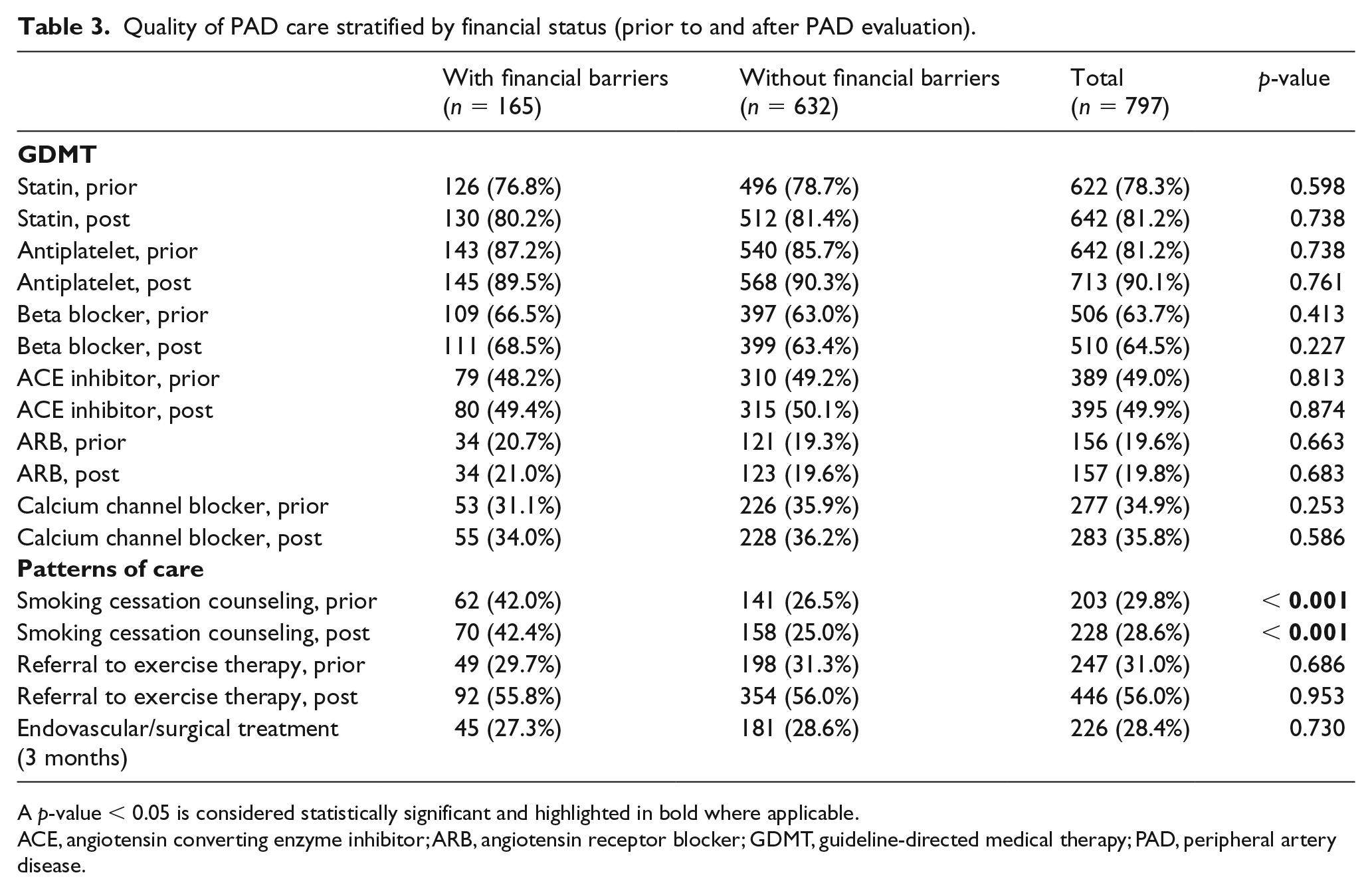
A p-value < 0.05 is considered statistically significant and highlighted in bold where applicable.
ACE, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; GDMT, guideline-directed medical therapy; PAD, peripheral artery disease.
Psychosocial factors
Psychosocial status as measured by assessment of depression (PHQ-8), perceived stress (PSS-10), and anxiety (GAD-2) were significantly different between the two groups (Table 4). Patients with financial barriers were reporting higher levels of depressive symptoms (7.0 ± 6.3 vs 4.2 ± 4.8, p < 0.001), had higher levels of perceived stress (5.9 ± 3.7 vs 3.8 ± 3.3, p < 0.001), and higher levels of anxiety (1.7 ± 1.9 vs 0.9 ± 1.5, p < 0.001) when compared with patients without financial barriers. These differences persisted at 12 months of follow-up.
Health status and psychosocial factors at baseline and follow-up, stratified by financial status.
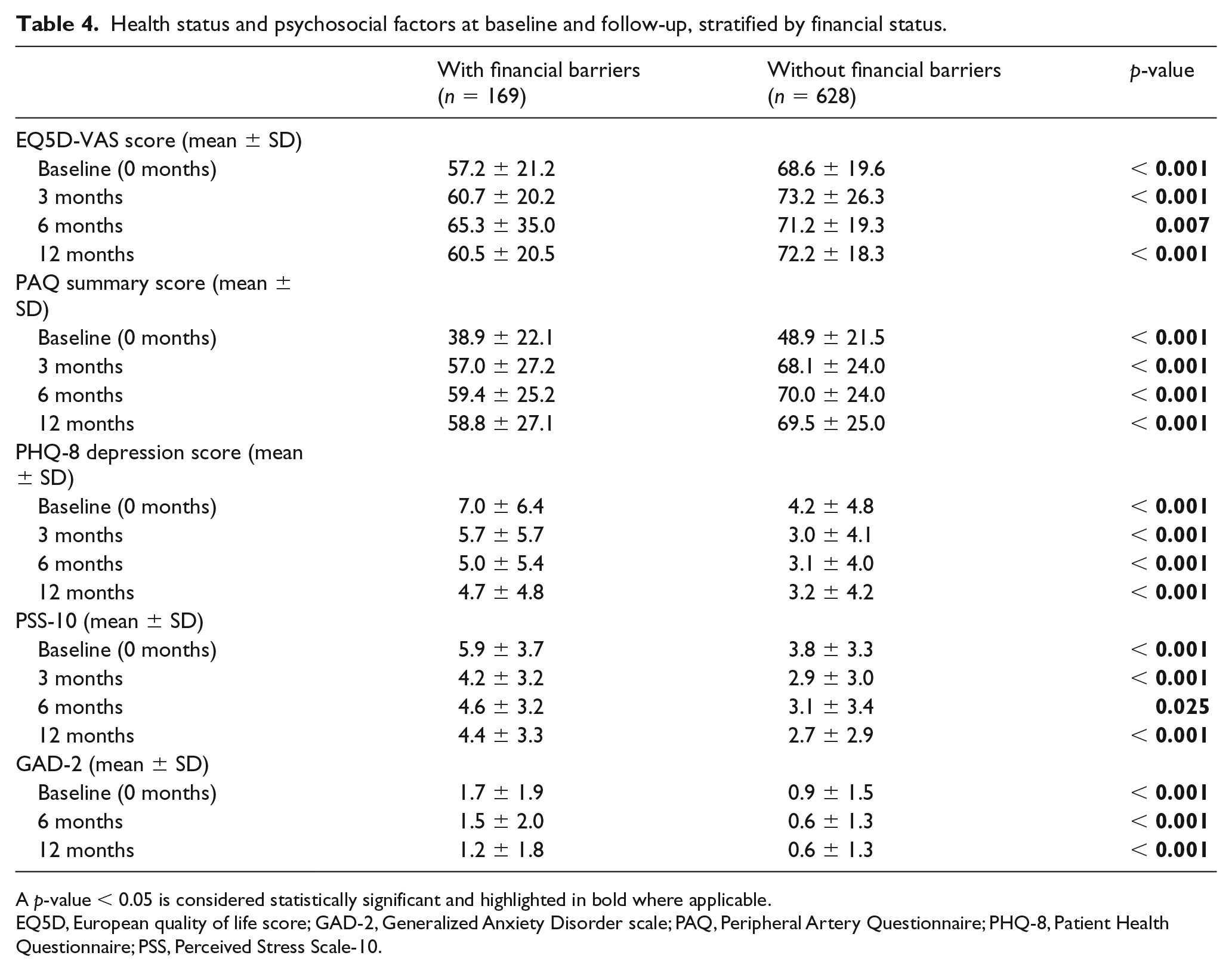
A p-value < 0.05 is considered statistically significant and highlighted in bold where applicable.
EQ5D, European quality of life score; GAD-2, Generalized Anxiety Disorder scale; PAQ, Peripheral Artery Questionnaire; PHQ-8, Patient Health Questionnaire; PSS, Perceived Stress Scale-10.
Health status outcomes
Health status was measured using the EQ5D-VAS and PAQ summary score (Table 4, Figure 4). In the unadjusted model, patients with financial barriers had worse generic health status at baseline as measured by the EQ5D (57.3 ± 21.1 vs 68.6 ± 19.6, p < 0.001), and this difference persisted at 3 and 12 months (60.7 ± 20.2 vs 73.2 ± 26.3, p < 0.001; 60.5 ± 20.5 vs 72.2 ± 18.3, p < 0.001). In the adjusted model using linear mixed models, these differences persisted and were statistically significant at baseline (EQ5D: –9.2 [–12.74, –5.8]; p < 0.001) and 12 months (–9.7 [–13.2, –6.2]; p < 0.001) (Table 4, Figure 4). In patients with financial barriers, when followed over a period of 1 year, there was a trend towards initial improvement in patient generic health status, an effect that plateaued at 1 year. Similar results were observed for disease-specific health status as measured by the PAQ. In the unadjusted model, patients with financial barriers had lower PAQ scores at baseline (38.9 ± 22.0 vs 48.9 ± 21.5), an effect which persisted at 3, 6, and 12 months of follow-up. These differences persisted in the adjusted models from baseline (PAQ: –7.0 [–10.7, –3.4]; p < 0.001) through 12 months of follow-up (–8.4 [–13.0, –3.8]; p < 0.001). In both the adjusted and unadjusted model for patients with financial barriers (Figure 4), health status as measured by the PAQ improved from baseline to 3 months and plateaued off until 12 months of follow-up. Similar observations were made for patients without financial barriers.
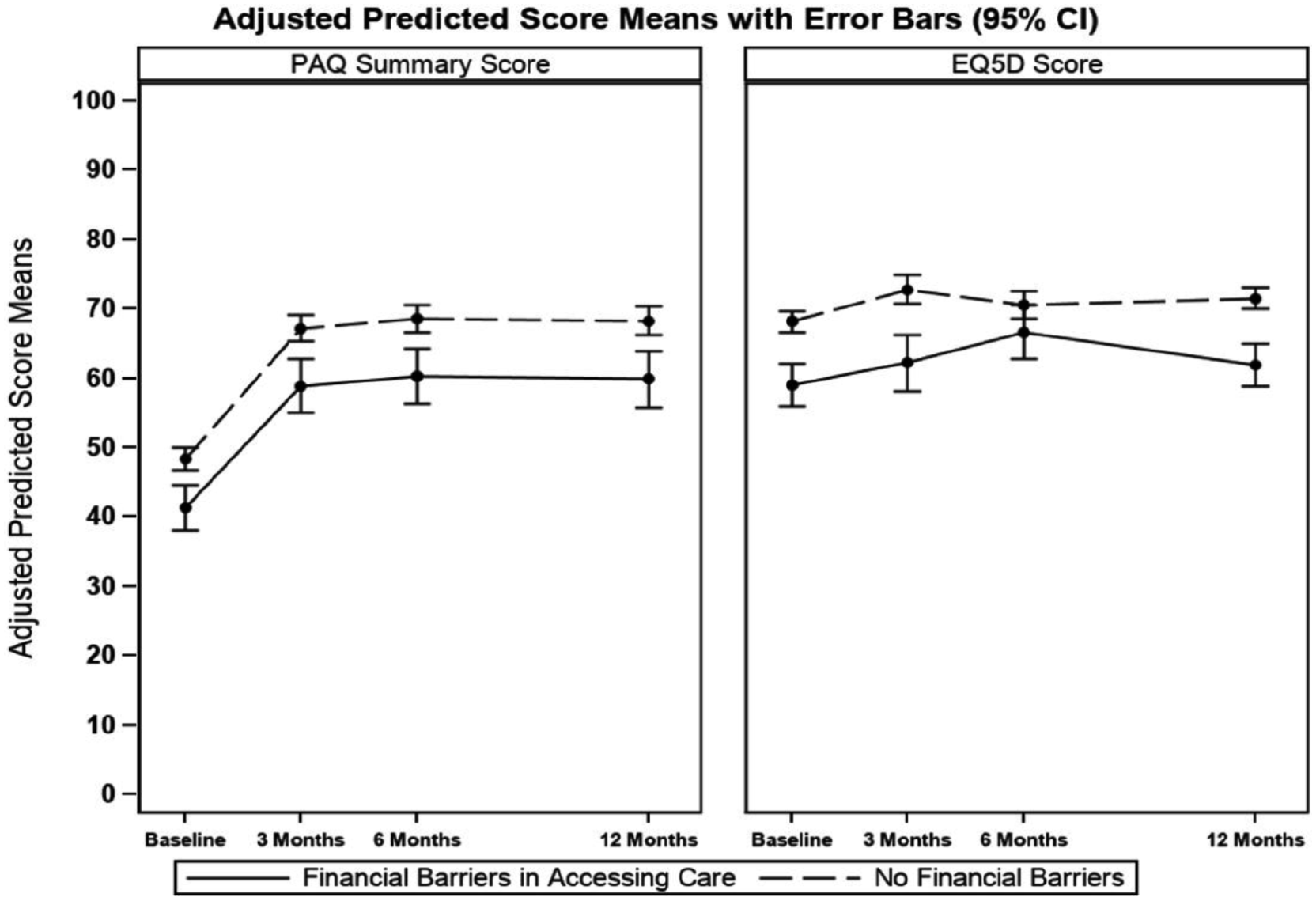
Adjusted health status mean score measures over time by financial concerns as determined by the PAQ and EQ5D-VAS questionnaires.
Discussion
In this prospective multicenter study of patients presenting with new or worsening PAD symptoms limited to patients in the US, we observed that ~ 99% of the patients seeking health care at a vascular specialty center had some form of insurance. This may in itself reflect a certain barrier to care in patients with PAD without insurance and needs to be studied further. In our study, one out of every five patients had financial barriers to accessing care. Those with financial barriers were more likely to present at a younger age and later in the course of the disease, with 60% presenting more than a year after the onset of symptoms. Those with financial barriers were also more likely to be noncompliant with prescribed medications. Additionally, they had worse health status at baseline including greater functional limitation, more symptoms, less treatment satisfaction, lower social functioning, and worse quality of life, along with worse perceptions of health at presentation. Additionally, those with financial barriers were more likely to be persistently depressed through 12 months of follow-up, starting at baseline, as well as having higher perceived stress and anxiety scores throughout the study. Despite similar care received, patients with financial barriers continued to have worse health status at 1 year.
To our knowledge, this is the first study to demonstrate a potential link between patient-centered financial barriers and health status outcomes. Using inpatient data from 2000 to 2005 from New York and Florida, Giacovelli et al. found that PAD individuals with Medicaid or without insurance were likely to present with life-threatening lower extremity ischemia, including rest pain, ulceration, and gangrene. 12 Our study extends the literature by highlighting adverse health outcomes in patients who are underinsured, which as of 2015 surpassed the number of uninsured nationally (31 million vs 28 million).2,3,30 Our study also highlights that despite receiving optimal medical therapy and an equal likelihood of receiving invasive treatment (endovascular and surgical), patients with financial barriers still had adverse health outcomes and worse depression and perceived stress. This study emphasizes the importance of examining the complete spectrum of financial barriers to a patient (i.e. from the uninsured to the underinsured), where the presence of insurance does not ensure access to optimal care and the associated outcomes. In our study, 165 patients were uninsured or underinsured. While underinsured patients were less likely to have Medicare or private insurance, rates of Medicaid coverage were similar. This is critical since it suggests that the problem of underinsurance is not limited to Medicaid cohorts, as suggested previously, 8 but can also be seen in Medicare and private insurance populations and with significant policy implications.
It is also worthwhile to note that in our study, 8 (1.0%) patients had no insurance while being seen in a specialty care. This has not been previously described in PAD populations and, given the small number, its significance remains unknown. This small number may imply that obtaining vascular specialty care for patients with PAD represents an insurmountable barrier and that our current documentation of financial barriers in PAD may just represent the tip of the iceberg. This hypothesis seems to be supported by a study in which uninsured patients with PAD were more likely to present with limb threatening ischemia (a more advanced stage of PAD) than claudication. 12 Overcoming access to care barriers due to insurance or underinsurance is one of the key basic areas that need to be addressed to avoid delays in presentation to obtain PAD care and to improve outcomes in populations at risk.
For our study we used a validated questionnaire to evaluate for financial concerns.9,18,19 We used detailed and structured interviews and identified patients who, due to cost, avoided obtaining health care services, had not taken a prescribed medication in the past year, or found it difficult to get medical care when needed. Similarly, we used validated questionnaires to examine health statuses from the patient’s point of view.22–24 This method of assessing patients’ perspectives on financial concerns and looking at patient-centered outcomes gives our study a significant advantage over any other administrative data source studies which use household income or zip code Census data as surrogates. While our study reliably elucidates financial concerns, more detailed investigation is needed to find out what concerns (high out of pocket copayments, premiums, coinsurance, deductibles; low lifetime health benefit ceilings; lack of catastrophic or stop-loss provisions) are driving these barriers.
The economic burden of PAD is high. Using linked Surveillance, Epidemiology, and End Results (SEER)–Medicare data for the year 2001, Hirsch et al. found that PAD-related costs account for 2.3% of total Medicare Part A and B expenditures. 31 Mahoney et al. found that in the REACH study, these expenditures were higher with increasing PAD severity. 32 In this current debate of increasing health care costs, one way of defraying the economic costs is ensuring adequate insurance which may help with early screening and diagnosis of the at-risk population and in providing early treatment. As seen from our study, lack of adequate insurance may result in more severe disease and thus more health care expenditure in the form of rehospitalizations and revascularizations, etc. It is well studied that patients with socioeconomic barriers tend to use less health care,4,33 are often diagnosed at an advanced stage of disease,34,35 are more likely to be hospitalized for ambulatory care-sensitive conditions,36–38 and are likely to use the emergency room as their main source of care,39,40 thus resulting in more expensive care with poorer outcomes. 41
While we saw that those with financial barriers were likely to have a worse health status at presentation, they continued to have a poorer status when followed up for a year, despite similar processes of care being measured. They continued to have more symptoms, increased physical limitations due to claudication, less treatment satisfaction, lower social functioning, higher depression rates, a higher level of stress/anxiety, and poorer quality of life. This is critical since this underscores the complexity of the issue at hand. A patient’s health status is determined by many factors; some of which can be influenced by PAD treatment. As an extension of these findings, we saw a higher psychosocial burden among those who had financial concerns, potentially adding to patients’ vulnerable risk profile.42,43 Our study clearly shows the need of focusing on these patient-centered disparities prior to their presentation to specialty care and ideally at the community, and then at a continuing level.
Our study also examined whether known racial disparities in PAD are related to financial barriers and health status outcomes in our cohort. Multiple studies have shown that non-Caucasians have higher rates of amputation and disease severity,44,45 are less often prescribed statin, antiplatelet, and antihypertensive therapies at baseline, 46 and are more likely to be economically disadvantaged as measured by a lower socioeconomic background or by frequencies of being under or uninsured.30,47,48 Our study adds additional evidence to this body of literature as we found that even when having gained access to vascular specialty care, racial disparities regarding whom is mostly affected by financial barriers, remain. African Americans experienced the highest rates of financial barriers in our cohort.
Broadening health care coverage is one area of improvement in the field of PAD as 45,000 deaths annually in the United States are attributable to lack of health insurance alone. 49 In addition, the role of community health centers, which provide a safety net for uninsured and underinsured patients, should be enhanced in early screening for patients at risk of PAD and early referral to specialists/specialty centers for treatment. Dedicated approaches to PAD awareness and treatment should be aimed at patients with socioeconomic barriers to care.
Limitations
Our study should be interpreted in the context of several potential limitations. Our patient cohort was mainly recruited from specialty centers across the US and our findings may not extend to settings outside of the network we recruited from. Additionally, while patients in our cohort were derived from vascular specialty clinics, this sample may not be representative of patients with more pronounced socioeconomic barriers, especially those without insurance or of health care settings with single-payer models or universal health care coverage. Another limitation is that we did not have detailed information as to what specific cost elements (e.g. copayments, deductibles, cost for medications) were contributing to the financial burdens related to patients’ care. Next, our study is observational in nature and we cannot establish directionality of the observed associations. As PAD is a debilitating disease, patients with advanced PAD may have more socioeconomic constraints given their functional limitations, which may impact their ability to work and generate income. Hence, we cannot rule out that the financial barriers reported in this study may in turn be due to advanced PAD itself.
Conclusion
In conclusion, financial concerns related to patients’ PAD care are a significant problem in patients who are seen in specialty care. A very small proportion of patients who are uninsured were seen at specialty PAD care centers we recruited from. Wider outreach to even more vulnerable populations without insurance to make sure PAD diagnostic and care needs are met is an important future focus. In addition, in general, at the policy level, systematic measures to promote affordability of health care is overdue, as financial concerns in accessing care continue to put enormous strain on our patient populations, our health care system, and society at large.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Spatz reports receiving support from the Centers for Medicare & Medicaid Services to develop and maintain performance measures used in public reporting programs. Dr Spertus is supported by grants from Gilead, Genentech, Lilly, Amorcyte, and EvaHeart, and has a patent for the Seattle Angina Questionnaire with royalties paid. Dr Desai is a consultant for Amgen, Boehringer Ingelheim, Cytokinetics, and Relypsa. Dr Smolderen is supported by an unrestricted research grant from Merck, Boston Scientific, and Abbott Vascular. Dr Mena-Hurtado is a consultant for Bard, Cook, Medtronic, Abbott, Boston Scientific, and Cardinal Health. The other authors report no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research reported in this manuscript was partially funded through the Patient-Centered Outcomes Research Institute (PCORI) Award (IP2 PI000573-01; CE-1304-6677) and an unrestricted grant by Gore.
The statements in this manuscript are solely the responsibility of the authors and do not necessarily represent the views of the Patient-Centered Outcomes Research Institute (PCORI), its Board of Governors or Methodology Committee.
All manuscripts for the PORTRAIT study are prepared by independent authors who are not governed by the funding sponsors and are reviewed by an academic publications committee before submission.
The funding organizations and sponsors of the study had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
