Abstract
To optimize cardiovascular health, the American Heart Association (AHA) has recommended ‘Life’s Simple 7 (LS7)’. We tested the hypothesis that greater adherence to the LS7 cardiovascular risk metric is associated with reduced risk of developing abdominal aortic aneurysm (AAA). A total of 14,375 black and white participants aged 45–64 years at the baseline visit of the Atherosclerosis Risk in Communities (ARIC) study cohort were included in this analysis. A 14-point summary score for LS7 was calculated, and participants were classified as having poor (0–4), average (5–9), or ideal (10–14) cardiovascular health. We also counted the number of ideal components. Poisson regression was used to calculate incidence rates for AAA, and Cox regression to calculate hazard ratios adjusted for age, race, sex, and socioeconomic status. Over 25 years of follow-up, we identified 545 clinically manifest AAA events. Incident rates per 1000 person-years declined markedly across LS7 categories: 3.4 for the ‘poor’ category, 2.2 for ‘average’, and 0.9 for ‘ideal’. Compared to individuals in the ‘poor’ LS7 category, individuals in the ‘average’ category had a 52% lower AAA risk (95% CI: 37% to 63%) and those in the ‘ideal’ category had an 80% lower risk (95% CI: 72% to 86%). For every additional ideal component, there was a 28% lower risk of AAA (95% CI: 23% to 33%). Greater adherence to the AHA’s LS7 cardiovascular risk metric is associated with a reduced risk of clinically manifest AAA. These findings support the recommendation to follow LS7 for primary prevention of AAA.
Keywords
Introduction
Abdominal aortic aneurysm (AAA) is a permanent, progressive, potentially fatal, localized dilation of the abdominal aorta. It is usually asymptomatic and is discovered incidentally or following rupture. 1 As many as one in three AAAs eventually rupture if left untreated. 2 The mortality following emergency surgery for individuals presenting with ruptured AAAs ranges from 30% to 60%.3–6
Screening provides a means for early identification of AAA cases and thus allows for institution of treatment aimed at slowing progression of AAA and prevention of possible rupture. The US Preventive Services Task Force (USPSTF) recommends one-time screening for AAA in men aged 65–75 who have ever smoked. 7 While there are significant benefits to screening for AAA, there are also limitations and harms associated with screening.1,7 Firstly, screening has no impact on reducing AAA incidence. Secondly, it often triggers surgical repair, which can be beneficial but carries some attendant morbidity and mortality. 7 Lastly, monitoring progression of AAA may cause psychological harm and reduce quality of life.1,8
The only strategy to reduce incidence of AAA is primary prevention through risk factor identification and control. Since 2010, the American Heart Association (AHA) has advocated following ‘Life’s Simple 7 (LS7)’ for general cardiovascular health. The LS7 cardiovascular risk metric provides ideal, intermediate, and poor criteria for seven risk factors or behaviors, namely smoking, body mass index (BMI), physical activity, diet, total cholesterol, blood pressure, and fasting serum glucose. Better adherence to an ideal LS7 is associated with reduced incidence of cardiovascular disease and cancer.9,10 However, the association between LS7 and AAA, specifically, is currently unknown.
Research has established several risk factors for AAA.11–16 For example, in the Atherosclerosis Risk in Communities (ARIC) study, Tang et al. documented that greater age, male sex, white race, current and former smoking, pack-years of smoking, hypertension, peripheral artery disease (PAD), greater height, greater total or low-density lipoprotein cholesterol (LDL-C), and lower high-density lipoprotein cholesterol (HDL-C) were risk factors for AAA. 17 Yet to our knowledge, no study has specifically examined the relation between adherence to LS7 and incident AAA. Understanding the relation between LS7 and clinically manifest AAA might help in simplifying the steps taken to prevent AAA and provide an additional means for individuals to monitor their risk of AAA. We therefore tested the hypothesis that greater adherence to LS7 is associated inversely with AAA incidence in a prospective, population-based cohort.
Methods
Study population
The ARIC study is a prospective community-based cohort study to identify risk factors for atherosclerosis and cardiovascular diseases. A total of 15,792 men and women aged 45–64 years were recruited between 1987 and 1989 from four communities in the United States (Washington County, MD; the northwest suburbs of Minneapolis, MN; Jackson, MS; and Forsyth County, NC). 18 Participants subsequently were examined in 1990–92, 1993–95, 1996–98, 2011–13, and 2016–17. Participants are also followed up by annual or semiannual telephone interviews. ARIC was approved by the institutional review board of each participating center, and all participants provided written informed consent for each study visit.
Of the 15,792 participants recruited at baseline, for this report, we applied the following exclusions: race other than white or black (n = 48), prior AAA surgery or angioplasty (n = 11), uncertain AAA status during follow-up (n = 30), missing LS7 data (n = 1328). A total of 14,375 participants were available for longitudinal analysis after these exclusions.
Measurement of Life’s Simple 7
As described in a previous publication, 19 we used the ARIC baseline visit, when participants were middle-aged, as the timepoint for assessment and classification of LS7 characteristics. Measurements included a food frequency questionnaire, physical activity, BMI, smoking, plasma total cholesterol, seated blood pressure after a 5-minute rest, and fasting blood glucose. 18 We classified each factor according to the AHA’s LS7 categories of ‘ideal’, ‘intermediate’, or ‘poor’.9,20
AAA ascertainment
ARIC identified incident AAAs by several strategies.17,21 In the annual telephone calls with ARIC participants, interviewers asked about any interim hospitalizations or identified deaths, and records for these outcomes were sought. ARIC also conducted surveillance of local hospitals to identify additional possible AAA hospitalizations or deaths. In addition, ARIC linked participant identifiers with fee-for-service Medicare data from the Centers for Medicare and Medicaid Services for the time period from 1991 to 2011 to find any missing hospital or outpatient AAA events for those over 65 years of age. We identified clinical AAAs as those with a hospital discharge diagnosis from any source or two Medicare outpatient claims that occurred at least 1 week apart, with International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) code 441.3 (ruptured AAA) or 441.4 (AAA without mention of rupture), procedure code 38.44 (AAA resection and replacement) or 39.71 (AAA endovascular repair); or a listed cause of death coded as ICD-9 441.3 or 441.4, or International Classification of Diseases, Tenth Revision code I71.3 (ruptured AAA) or I71.4 (AAA without mention of rupture). Although clinically detected, these AAAs include both symptomatic and asymptomatic AAAs that were medically documented. Because of our focus on AAA, we treated thoracic, thoracoabdominal, or unspecified aortic aneurysms as nonevents.
Statistical analysis
Baseline LS7 distributions were described for those who did or did not develop incident AAA. Categorical variables were presented as percentages. Continuous variables were presented as means and SD.
Using the LS7 components, we created two variables for analysis: (1) the number of ideal components of LS7 met by each participant and (2) a score in which each component was given points of 0, 1, or 2, which represent ‘poor’, ‘intermediate’, or ‘ideal’ health categories, respectively. 22 The sum of these scores in (2) across the seven components was categorized as ‘poor’ (0–4), ‘average’ (5–9), and ‘ideal’ (10–14) for cardiovascular health. We performed multivariable Cox regression to examine the relation of the two LS7 variables with time to incident AAA. Our main models were adjusted for age (years), sex (male, female), race (black, white), and socioeconomic status using baseline data on the maximum educational level (< high school, high school graduate, > high school, and unknown) and baseline annual income (< $12,000, $12,000–24,999, $25,000–49,999, $50,000+, and unknown). Poisson regression was used to calculate the crude incidence rates of incident AAA using these same LS7 variables. We also generated Kaplan–Meier failure plots, using Stata Statistical Software, release 12 (StataCorp., College Station, TX, USA). We repeated these analyses after excluding smoking from our LS7 summary score. Statistical significance was based on a two-sided type 1 error p-value of 0.05. The main statistical analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Among the 14,375 ARIC participants free of AAA at baseline, the mean (SD) age was 54.2 (5.8) years, 24.7% were black, and 55.3% were women. Over a median follow-up time of 22.6 years, from 1987–89 through 2011, there were a total of 545 incident AAA events. As shown in Table 1, participants who developed AAA were, at baseline, more likely to be older, male, and white compared to participants who did not develop AAA. Those who developed AAA had a higher prevalence at baseline of ‘poor’ blood pressure, diet, smoking, and total cholesterol, but a lower prevalence of poor BMI and glucose.
Demographic characteristics and LS7 components at baseline (1987–89) between participants who did and did not develop incident AAA (1987–89 through 2011), ARIC.
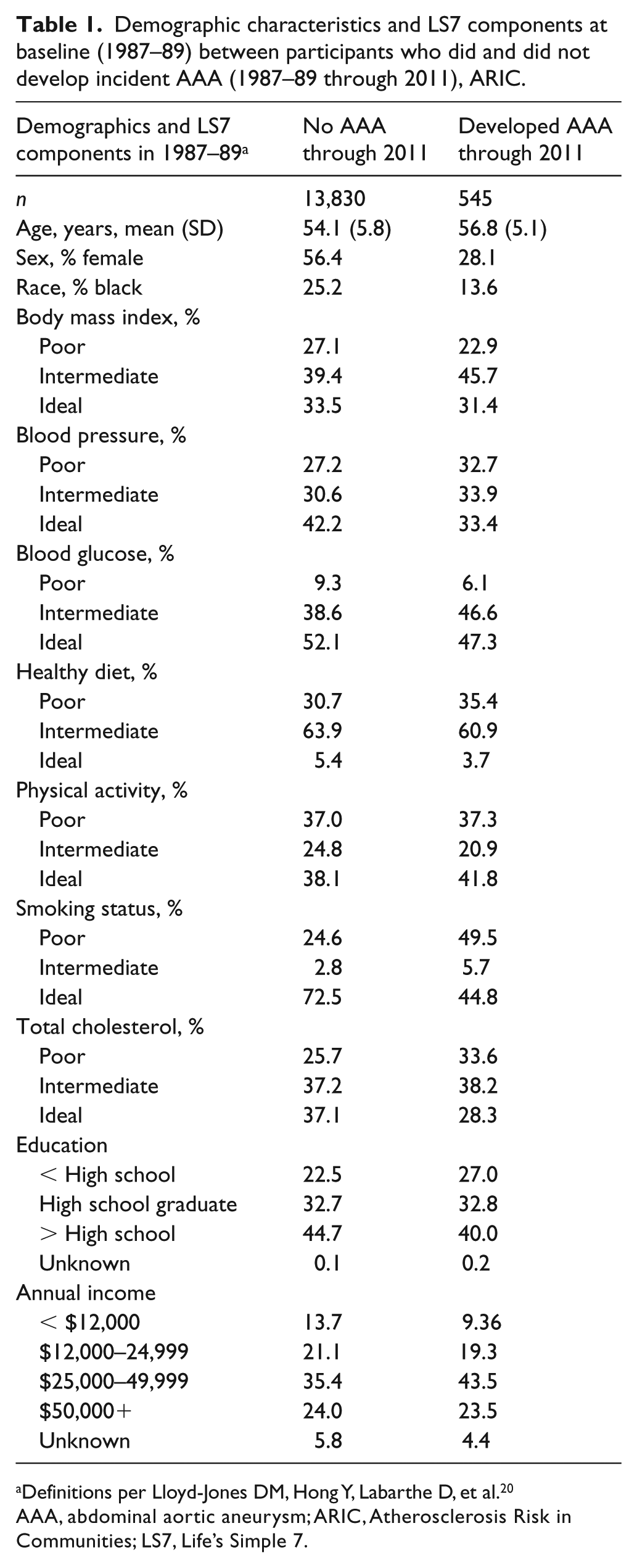
Definitions per Lloyd-Jones DM, Hong Y, Labarthe D, et al. 20
AAA, abdominal aortic aneurysm; ARIC, Atherosclerosis Risk in Communities; LS7, Life’s Simple 7.
As shown in Table 2, the crude incidence rate for incident AAA per 1000 person-years was 0.9 for the ideal LS7 summary category, 2.2 for the average category, and 3.4 for the poor category. Kaplan–Meier failure curves (Figure 1) also showed that the LS7 categories were inversely and strongly associated with the probability of incident AAA during follow-up (log-rank p-value < 0.0001). Hazard ratios (HRs) (95% CIs) adjusted for age, race, sex, and socioeconomic status (Table 2) were 0.20 (0.14, 0.28) for the ideal LS7 category and 0.48 (0.37, 0.63) for the average category, compared with the poor LS7 category as the reference (p-trend < 0.0001); this represents a 52% (95% CI: 37% to 63%) lower AAA risk for those in the average LS7 category and an 80% (95% CI: 72% to 86%) lower AAA risk for those in the ideal LS7 category.
Crude incidence rates and adjusted HRs with 95% CIs of clinically manifest AAA in relation to baseline LS7 categories, 1987–89 through 2011, ARIC.
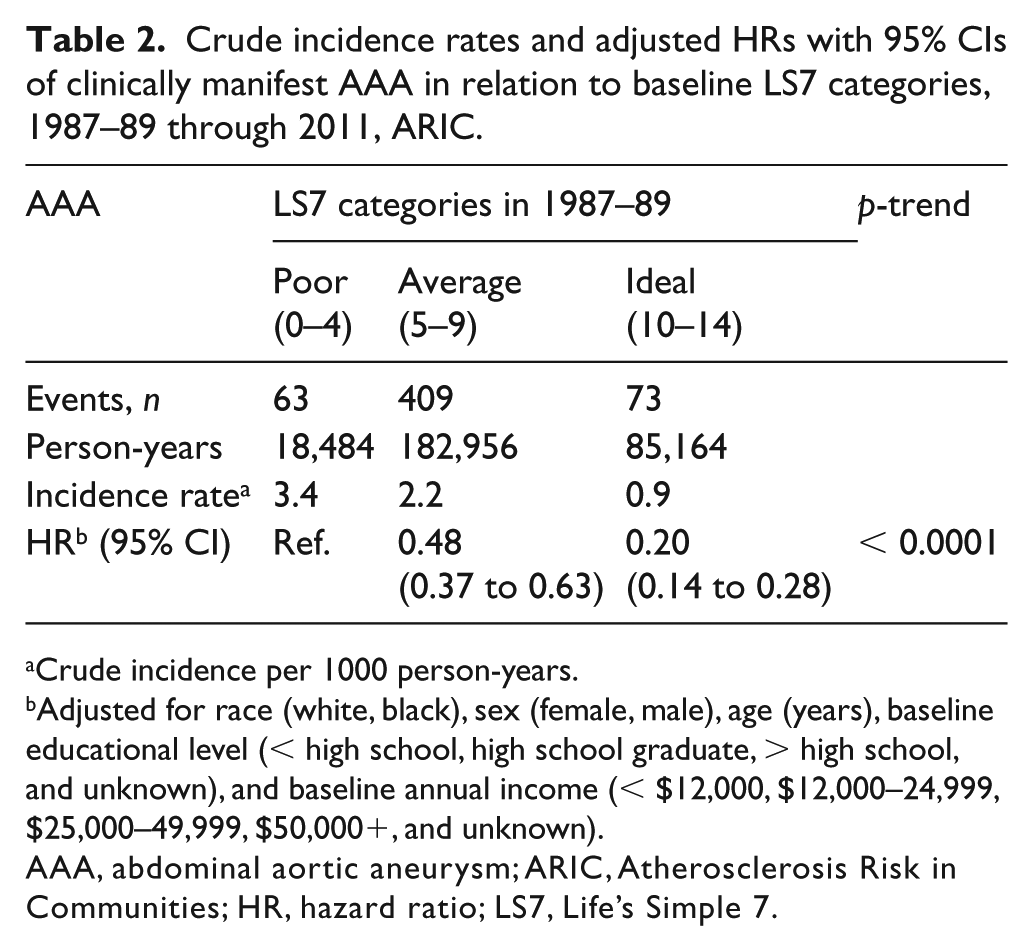
Crude incidence per 1000 person-years.
Adjusted for race (white, black), sex (female, male), age (years), baseline educational level (< high school, high school graduate, > high school, and unknown), and baseline annual income (< $12,000, $12,000–24,999, $25,000–49,999, $50,000+, and unknown).
AAA, abdominal aortic aneurysm; ARIC, Atherosclerosis Risk in Communities; HR, hazard ratio; LS7, Life’s Simple 7.
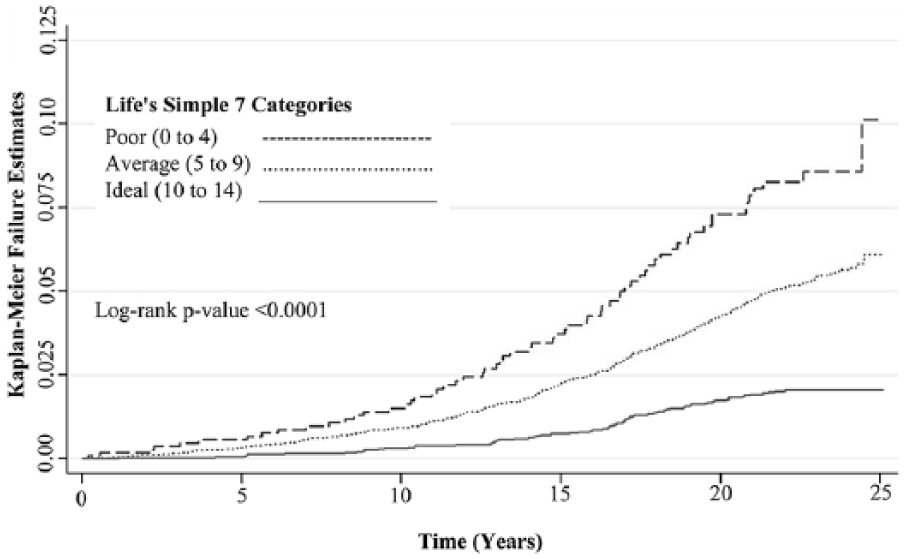
Kaplan–Meier failure curves for clinically manifest AAA, by baseline LS7 categories, 1987–89 through 2011, ARIC.
As Figure 2 shows, the inverse association between LS7 categories and incident AAA differed modestly by sex (p-interaction = 0.03) but not by race (p-interaction = 0.47). Compared to women in the poor category, women in the ideal LS7 category had 90% lower risk of AAA (95% CI: 80% to 95%). In men, risk was 76% (63% to 84%) lower in the ideal versus poor category.
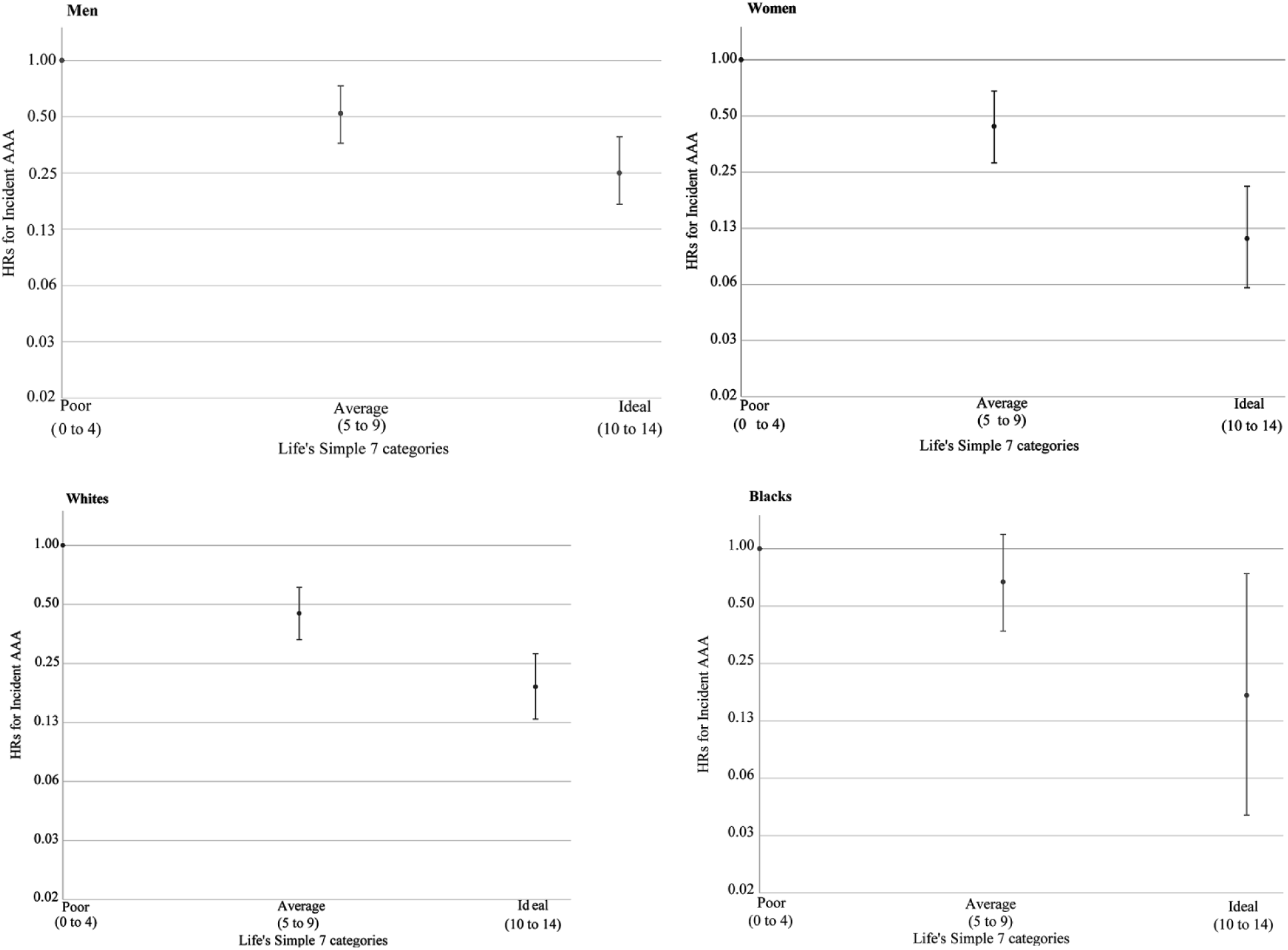
Sex and race-specific HRs for clinically manifest AAA in relation to LS7 categories at baseline, 1987–89 through 2011, ARIC.
As shown in Table 3, the number of ideal LS7 components was also inversely associated with incidence of AAA, ranging from 4.6 events per 1000 person-years in individuals with no ideal components to 0.5 events per 1000 person-years in individuals with six or more ideal components. Only 11.7% of the cohort had more than five ideal LS7 components, and they had approximately one-tenth the risk of AAA of those having no ideal components. Modeled as a continuous variable, for every additional ideal LS7 component attained, there was a 28% lower risk of incident AAA (95% CI: 23% to 33%). The association between our LS7 variables and AAA after excluding smoking from the summary scores (Supplemental Tables 1 and 2) were similar to the results presented in Tables 2 and 3. We also report the adjusted HRs for AAA for the individual components of LS7 (Supplemental Tables 3 and 4). Although, as expected, not smoking seemed the biggest LS7 contributor, better blood pressure and total cholesterol, and to a lesser degree healthy diet and physical activity, also contributed to lower AAA rates.
Crude incidence rates and adjusted HRs with 95% CIs of clinically manifest AAA in relation to number of ideal LS7 components at baseline, 1987–89 through 2011, ARIC.
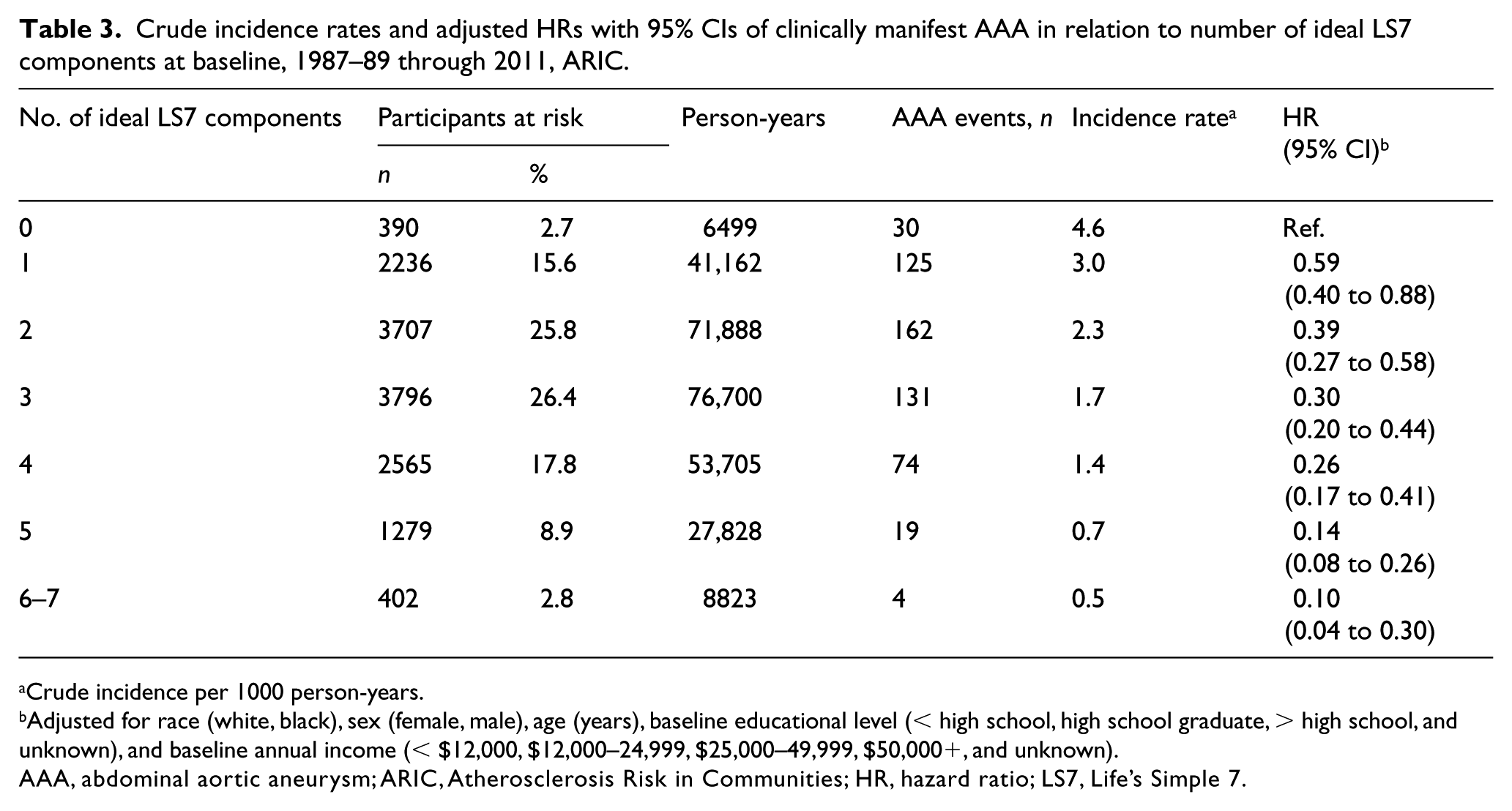
Crude incidence per 1000 person-years.
Adjusted for race (white, black), sex (female, male), age (years), baseline educational level (< high school, high school graduate, > high school, and unknown), and baseline annual income (< $12,000, $12,000–24,999, $25,000–49,999, $50,000+, and unknown).
AAA, abdominal aortic aneurysm; ARIC, Atherosclerosis Risk in Communities; HR, hazard ratio; LS7, Life’s Simple 7.
Discussion
In this large population-based longitudinal study, over a median follow-up time of 22.6 years, we found that greater adherence to the AHA’s LS7 metrics was associated with a lower risk of incident AAA in middle-aged black and white adults. The inverse association of LS7 components with incident AAA was strong and was independent of age, race, sex, and socioeconomic status, though stronger in women than in men. These findings suggest that adherence to LS7 recommendations in midlife can help in the primary prevention of incident AAA.
While screening high-risk individuals is an effective way for early identification of AAA and allows for early intervention, it is associated with a high financial cost, psychological harm, and it misses potentially fatal cases in individuals not labeled as high risk by the current screening guidelines. Over the years, some pharmaceutical options have been proposed as means to delay the progression of AAA and reduce the incidence of AAA rupture. Studies done to assess the benefits of these medications have shown: no benefit for the use of doxycycline in reducing the rate of AAA progression; 23 inconsistent results for the use of angiotensin-converting enzyme inhibitors to delay AAA growth;24,25 and a reduction in the risk of rupture for participants on statins compared to participants who have never used statins.26,27 Lifestyle advice for the management of AAA remains largely unchanged and has focused majorly on smoking cessation, blood pressure control, and the treatment of dyslipidemia. 3 Screening, statin use, and lifestyle interventions are targeted towards preventing progression and rupture of AAA and not preventing incident AAA in the first place. 3
The AHA’s LS7 guidelines aim to reduce the onset of elevated cardiovascular risk factors population-wide and thereby reduce the occurrence of major cardiovascular events and attendant morbidity and mortality. This population approach to primary prevention provides a potentially efficient and effective means to reduce the incidence of AAA and subsequent morbidity and mortality. Our study adds to the evidence, showing that adherence to LS7 reduces coronary disease, stroke, heart failure, 19 venous thromboembolism,22,28 cancer, 29 and other non-cardiovascular diseases 29 by demonstrating that greater adherence to LS7 in middle-age is associated with reduced incidence of AAA. Our representation of LS7 categories is similar to previous studies on the association of LS7 and various cardiovascular diseases,19,22,28 and allows for comparability of findings.
Of course, many studies have shown associations between components of the LS7 metric and incident AAA. These include positive associations of AAA with smoking,11,17 obesity,12,13 total cholesterol, 17 blood pressure, 30 and diets low in fruits, 31 low in both fruits and vegetables, 32 and low in energy. 33 In contrast, associations between physical activity and AAA have been inconclusive, and an inverse relationship between fasting serum glucose and AAA has been consistently reported.14–16 We were unable to find any study that examined the relationship of all the LS7 components, taken together, and incident AAA.
Limitations
One limitation of this study is that we used a single measure of LS7 components, precluding evaluation of AAA risk in relation to changes in these components or the overall score. Secondly, although the ARIC sample was community-based, our results generalize only to white and black individuals. Thirdly, we did not have information on other important risk factors, such as family history of AAA.34,35 Fourthly, although we excluded participants with prior AAA surgery at ARIC baseline, we did not screen the whole ARIC cohort to identify and exclude the relatively few prevalent AAAs we might have missed in the volunteers aged 45–64 years old. Lastly, we used ICD codes to ascertain the diagnosis of AAA from hospital records and death certificates. The validity of ICD codes is likely imperfect, and we certainly would have missed undiagnosed AAAs and AAAs that did not present to the hospital, although it seems unlikely that this should greatly affect the conclusions of our study.
Conclusion
Greater adherence to LS7 is associated with a reduced incidence of AAA. These findings provide evidence that adherence to LS7 may be useful for the primary prevention of AAA.
Supplemental Material
Supplementary_tables – Supplemental material for Association of Life’s Simple 7 with reduced clinically manifest abdominal aortic aneurysm: The ARIC study
Supplemental material, Supplementary_tables for Association of Life’s Simple 7 with reduced clinically manifest abdominal aortic aneurysm: The ARIC study by Abayomi O Oyenuga, Aaron R Folsom, Pamela L Lutsey and Weihong Tang in Vascular Medicine
Footnotes
Acknowledgements
The authors thank the staff and participants of the ARIC study for their important contributions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the National Heart, Lung, and Blood Institute (NHLBI) supported this research through R01HL103695 and ARIC contracts HHSN268201700001I, HHSN268201700002I, HHSN268201700003I, HHSN268201700004I, and HHSN268201700005I.
Supplemental material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
