Abstract
Little is known about whether markers of vitamin D metabolism are associated with the development of abdominal aortic aneurysm (AAA), though these markers have been linked to other cardiovascular diseases. We tested the hypotheses that risk of AAA is higher among individuals with low serum concentrations of 25-hydroxy vitamin D [25(OH)D], and among those with elevated concentrations of calcium, fibroblast growth factor 23 (FGF23), phosphorus, and parathyroid hormone (PTH) using data from a cohort of black and white individuals with long-term follow-up. Markers of vitamin D metabolism were measured using serum collected in 1990–1992 from ARIC study participants (mean ± SD age 56.9 ± 5.7 years, 43.2% male, 23.9% black). A total of 12,770 participants were followed until 2011 for incident AAA. Multivariable-adjusted Cox regression models were used. A total of 449 incident AAA events occurred over a median follow-up of 19.7 years. For the association between serum calcium and risk of incident AAA there was evidence of interaction by sex (p-interaction 0.02). Among women, in the fully adjusted model, the hazard ratio (95% confidence interval) comparing the highest to lowest quartile was 2.43 (1.25–4.73), whereas in men it was 1.01 (0.72–1.43). Not associated with risk of incident AAA were 25(OH)D, FGF23, phosphorus, and PTH. In this large prospective cohort, there was little evidence that markers of vitamin D metabolism are associated with risk of incident AAA. The positive association of calcium with AAA among women may warrant further investigation and replication in other populations.
Keywords
Introduction
Abdominal aortic aneurysm (AAA) is a condition in which a portion of the aortic wall undergoes progressive dilation and weakening, potentially leading to aortic rupture if untreated. 1 In the United States in 2014, aortic aneurysm (of which most are abdominal) was listed as the primary cause of 9863 deaths and a contributing cause in 16,242 deaths. 2
Key features thought to underlie the pathogenesis of AAA are the progressive degradation and remodeling of elastin and collagen fibers in the aortic wall.3,4 Relatively little is known about the etiology of AAA, though age, male sex, smoking status, and hypertension are important risk factors. Serum concentrations of 25-hydroxy vitamin D (vitamin D, 25(OH)D), calcium, parathyroid hormone (PTH), fibroblast growth factor 23 (FGF23), and phosphorus – all biomarkers in the vitamin D metabolic pathway – may potentially contribute to AAA development. Briefly, suboptimal 25(OH)D, PTH, and calcium concentrations have been linked to several AAA risk factors such as hypertension,5–11 inflammation,12,13 and vascular calcification.14,15 Elevated FGF23 has been related to aneurysmal development in both in vitro studies of human and rat aorta cells 16 and in rat experimental models. 17 High phosphate concentrations precede elevated FGF23 and have been associated with vascular calcification18–20 and myocardial fibrosis. 21
To date, no prospective epidemiologic studies have examined the association between serum markers of vitamin D metabolism and incidence of AAA. Using data from the population-based Atherosclerosis Risk in Communities (ARIC) cohort, we tested the hypotheses that risk of AAA is higher among individuals with low concentrations of 25(OH)D, and among those with elevated concentrations of calcium, FGF23, phosphorus, and PTH, over long-term follow-up.
Methods
Study design and population
The ARIC study 22 is a population-based prospective cohort, which in 1987–1989 enrolled 15,792 men and women, aged 45–64 years, from four US field centers: suburban Minneapolis, MN; Forysth County, NC; Washington County, MD; and Jackson, MS. Participants have taken part in several clinic visits, and have been followed continuously for health outcomes. Relevant to the present analysis, serum from visit 2 (1990–1992) was used for the measurement of markers of vitamin D metabolism. At each clinic visit participants provided informed consent. The ARIC study protocol was approved by institutional review boards at all study centers.
Of the original 15,792 participants, 14,348 attended visit 2, which is the baseline for the present analysis. We excluded participants with (a) baseline definite or probable AAA (n = 46), (b) race other than black or white (n = 42) and black individuals at the Minnesota and Maryland centers (n = 50) due to small numbers, as well as those who were (c) missing any biomarkers of interest (n = 1437) or (d) had no follow-up after visit 2 (n = 3). The final analytic sample was 12,770.
Biomarker measurement
Participants were asked to fast for 12 hours prior to the visit 2 blood draw. Serum was frozen at −70°C until analyzed in 2012–2013. Serum 25(OH)D2 and 25(OH)D3 were measured using a high sensitivity mass spectrometer (AB Sciex 5500, Atlanta, GA) at the University of Minnesota Molecular Epidemiology and Biomarker Research Laboratory, which for vitamin D is traceable to the US Centers for Disease Control reference measurement procedures. 23 Using samples split at the time of blood draw and sent to the lab 1 week apart, the blind duplicate coefficient of variation (CV) and Pearson correlation coefficients were as follows: 25(OH)D3 CV = 6.9%, r = 0.97; 25(OH)D2 CV = 20.8%, r = 0.98. Please note that these estimates encompass variability occurring as a consequence of sample processing, shipping, long-term storage and laboratory analysis. Serum calcium and phosphorus were measured on a Roche Modular P800 Chemistry Analyzer (Roche Diagnostics, Indianapolis, IN, USA) using a colorimetric method. Using the ARIC blind duplicates, the CVs were 2.4% and 3.0%, respectively. Serum PTH was measured on a Roche Elecsys 2010 Analyzer (Roche Diagnostics) using a sandwich immunoassay method (CV = 9.7%). Serum FGF23 was measured with a two-site enzyme-linked immunosorbent assay (Kainos Laboratories, Inc., Tokyo, Japan). The CV for FGF23 based on ARIC blind duplicate samples was 16.6%, while the CV from internal laboratory quality control samples was 8.8% at 41.4 pg/mL.
Outcome ascertainment
Incident AAA events accrued from baseline through December 31, 2011. Since baseline, ARIC study participants have been followed continuously for hospitalizations and deaths, through annual or semi-annual telephone calls to ARIC participants (or proxy), active surveillance of hospital discharge lists, and linkage to state and national death records. Additionally, for the period from 1991 to 2011, we linked participant identifiers with Medicare data from the Centers for Medicare and Medicaid Services (CMS) to find any missing hospital or outpatient AAA events for those aged over 65 years. Clinical AAAs were defined as those who had a hospital discharge diagnosis from any of the above sources, or two Medicare outpatient claims that occurred at least 1 week apart, with International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes of 441.3 (ruptured AAA), 441.4 (AAA without mention of rupture) or 441.02 (AAA dissection), or procedure codes of 38.44 (AAA resection and replacement) or 39.71 (AAA endovascular repair), or a listed cause of death coded as ICD-9 441.3 or 441.4 or ICD-10 code I71.02 (AAA dissection), I71.3 (ruptured AAA), or I71.4 (AAA without mention of rupture). 24 Notably, some of these clinical diagnoses would include asymptomatic AAAs that were captured through imaging for other indications.
Covariate information
Information on covariates was collected at the visit 2 clinic visit. Age, race, field center, sex, and smoking status and amount were self-reported. Pack-years were calculated. Participants brought to the visit all medications taken in the 2 weeks before the examination; medication names were transcribed and coded. Height, weight, and blood pressure were measured. Diabetes was defined by fasting blood glucose ≥126 mg/dL, non-fasting glucose ≥200 mg/dL, a self-report of physician diagnosis, or current medication use for diabetes. Plasma total cholesterol, triglycerides, and high-density lipoprotein cholesterol (HDL-C) were measured using standard ARIC procedures. 25 The estimated glomerular filtration rate (eGFR) was calculated using the 2012 CKD-EPI equation, which incorporates both cystatin C and creatinine. 26 The eGFR was categorized according to established clinical cut-points: ≥90, 60–89, and ≤59 mL/min/1.73 m2.
Data analysis
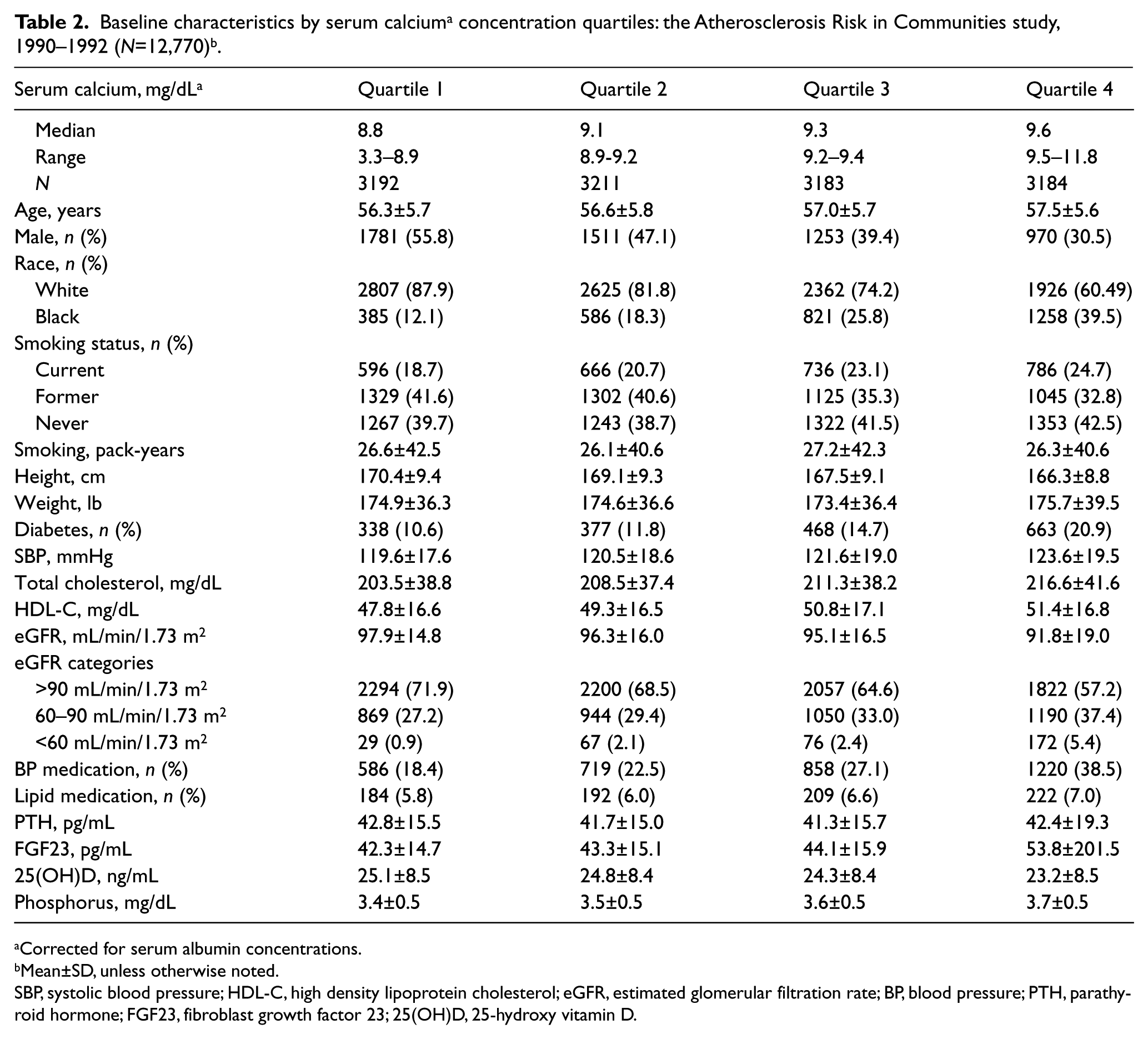
Serum 25(OH)D was the sum of 25(OH)D2 and 25(OH)D3. Concentrations were corrected to account for seasonality using a residuals approach. 27 Approximately 40% of non-skeletal calcium is bound to proteins, primarily albumin and globulin. 28 As such, serum calcium was corrected for serum albumin with the use of the following equation: measured total calcium (mg/dL) + 0.8 [4.0 − serum albumin (g/dL)]. 29 Albumin-corrected calcium was used for all analyses. Participant characteristics are presented as means and proportions, stratified by 25(OH)D commonly used clinical cut-points30,31 and serum calcium quartiles, respectively.
Cox proportional hazards models were used to calculate the hazard ratios (HRs) and 95% confidence intervals (CIs) of the association between markers of vitamin D metabolism and incident AAA. Person time accrued from the date of the visit 2 clinic visit until AAA diagnosis, death, loss-to-follow-up or 31 December 2011, whichever came first. Model 1 adjusted for age, sex, and race. Model 2 additionally adjusted for height, weight, smoking status (current, former, never), and lifetime smoking quantity [ln(pack years + 1)]. Model 3 further adjusted for total cholesterol, HDL cholesterol, systolic blood pressure, diabetes, antihyperlipidemic medication, antihypertensive medication, and eGFR categories. Models where PTH was the biomarker of interest also adjusted for season, with a 1-month lag: July – September, October – December, January – March, April – June. Linear trends were calculated by including the exposure categories in the models as an ordinal term.
Multiplicative interactions were tested by age, race, sex, smoking status, and eGFR (for FGF23 only). Subgroup analyses are reported, as appropriate. There were no meaningful violations of the proportional hazards assumption. SAS software, version 9.3 (SAS Institute Inc., Cary, NC, USA) was used for all analyses.
Results
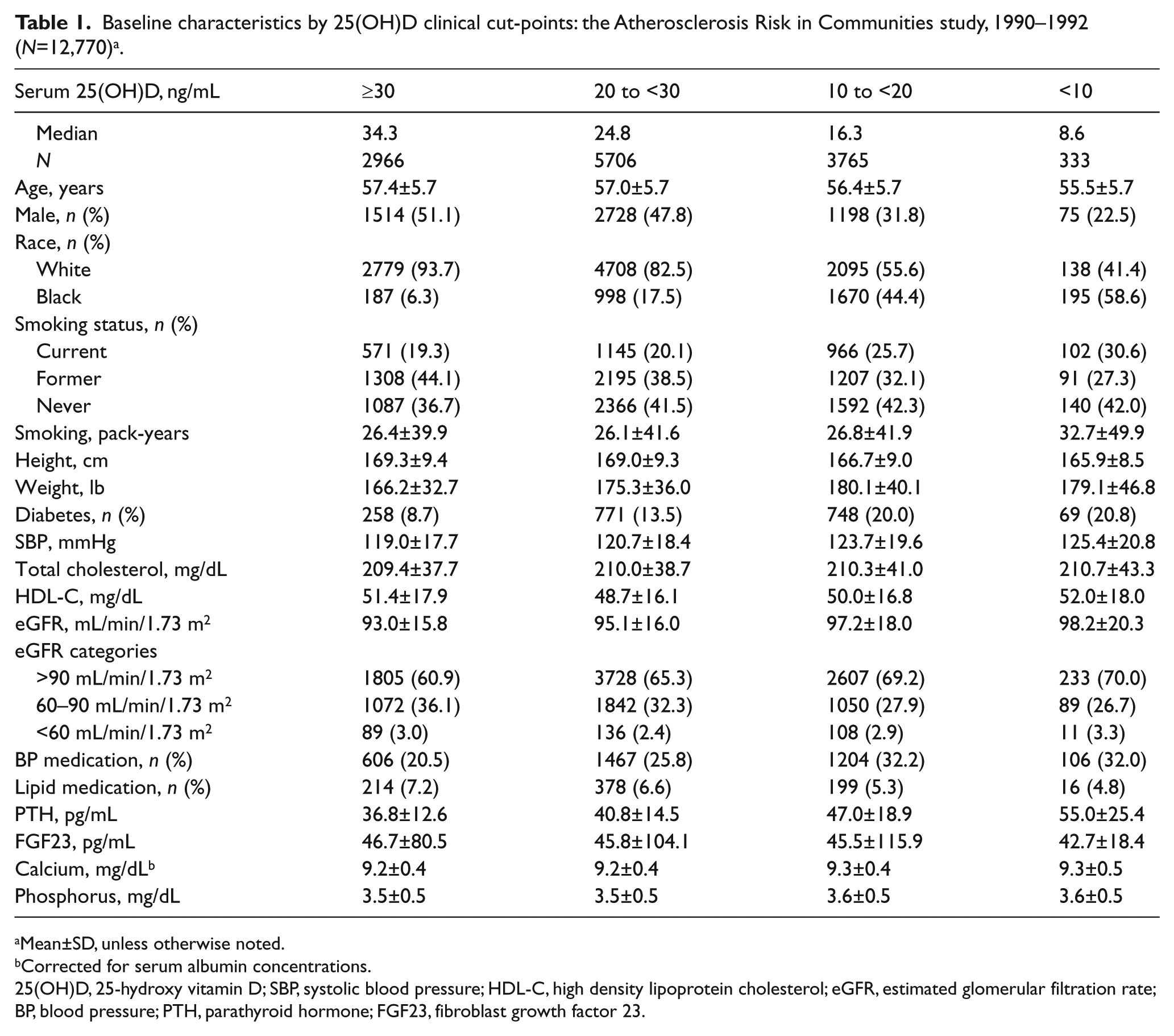
At baseline, participants were 43.2% male, 23.9% black, and on average (±SD) 56.9 ± 5.7 years old. The mean (±SD) concentrations of the vitamin D metabolism biomarkers are as follows: 25(OH)D = 24.4 ± 8.5 ng/mL, corrected calcium = 9.21 ± 0.41 mg/dL, phosphorus = 3.53 ± 0.48 mg/dL, PTH = 42.1 ± 16.5 pg/mL. FGF23 was highly skewed; the median (25th, 75th percentiles) was 41.9 (33.9–51.7) pg/mL. Table 1 presents participant baseline characteristics stratified by 25(OH)D categories, and Table 2 by calcium quartiles. As expected, black individuals had lower 25(OH)D concentrations than white individuals. In general, individuals with lower 25(OH)D and higher serum calcium had worse cardiometabolic profiles.
Baseline characteristics by 25(OH)D clinical cut-points: the Atherosclerosis Risk in Communities study, 1990–1992 (N=12,770) a .
Mean±SD, unless otherwise noted.
Corrected for serum albumin concentrations.
25(OH)D, 25-hydroxy vitamin D; SBP, systolic blood pressure; HDL-C, high density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate; BP, blood pressure; PTH, parathyroid hormone; FGF23, fibroblast growth factor 23.
Corrected for serum albumin concentrations.
Mean±SD, unless otherwise noted.
SBP, systolic blood pressure; HDL-C, high density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate; BP, blood pressure; PTH, parathyroid hormone; FGF23, fibroblast growth factor 23; 25(OH)D, 25-hydroxy vitamin D.
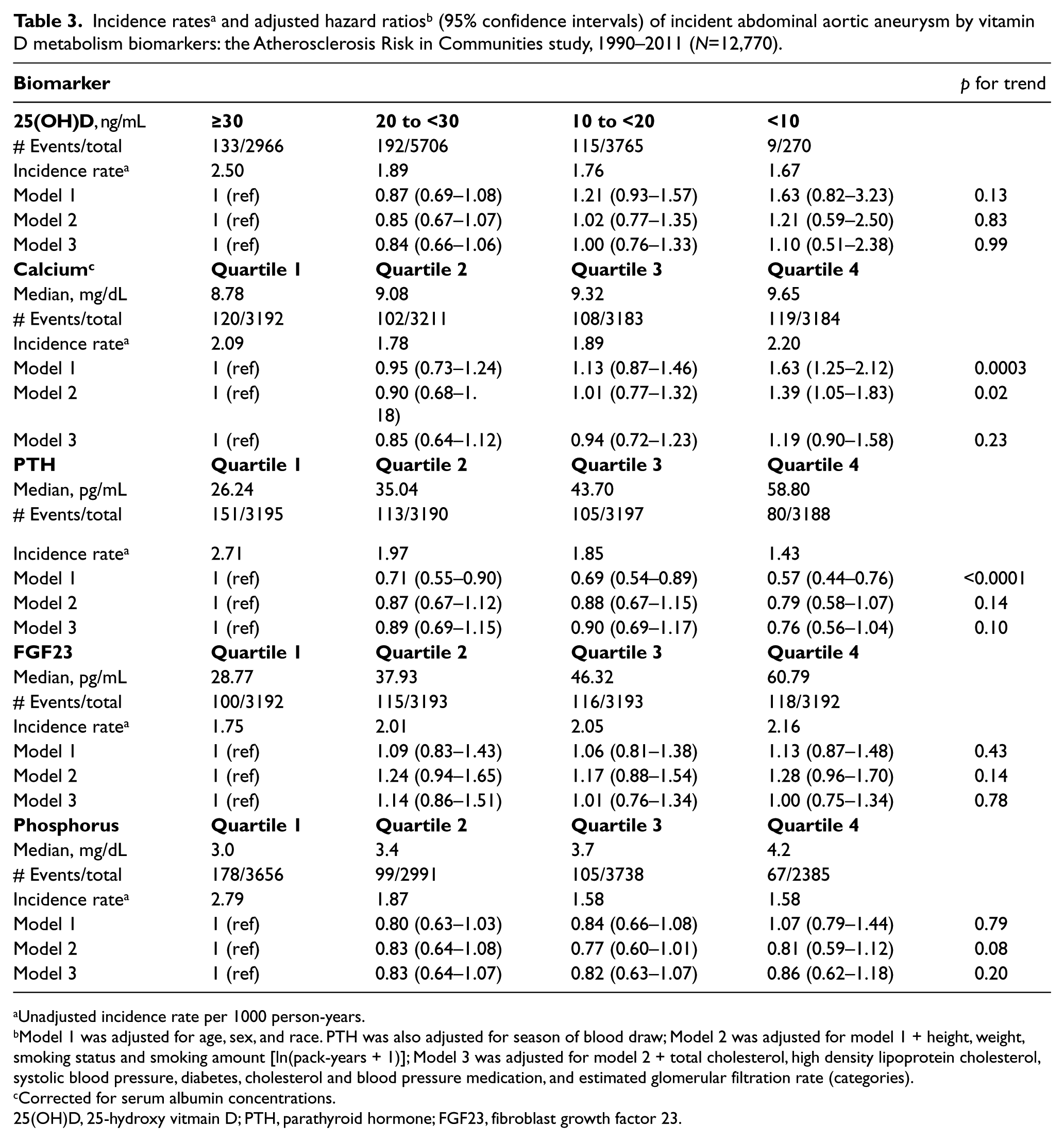
Over a median follow-up of 19.7 years (maximum 21.9 years), a total of 449 incident AAA events occurred. As shown in Table 3, 25(OH)D concentrations were not associated with risk of AAA. The HR (95% CI) for 25(OH)D concentrations <10 ng/mL versus ≥30 ng/mL was 1.63 (0.82–3.23) after accounting for demographics (model 1), 1.21 (0.59–2.50) after further adjusting for indices of anthropometry and smoking (model 2), and 1.10 (0.51–2.38) after additionally adjusting for cholesterols, blood pressure, diabetes, and eGFR (model 3). Findings were similar in analyses stratified by race. There was no evidence of interaction by age, race, sex, or smoking status (interaction p-values all >0.18).
Unadjusted incidence rate per 1000 person-years.
Model 1 was adjusted for age, sex, and race. PTH was also adjusted for season of blood draw; Model 2 was adjusted for model 1 + height, weight, smoking status and smoking amount [ln(pack-years + 1)]; Model 3 was adjusted for model 2 + total cholesterol, high density lipoprotein cholesterol, systolic blood pressure, diabetes, cholesterol and blood pressure medication, and estimated glomerular filtration rate (categories).
Corrected for serum albumin concentrations.
25(OH)D, 25-hydroxy vitmain D; PTH, parathyroid hormone; FGF23, fibroblast growth factor 23.
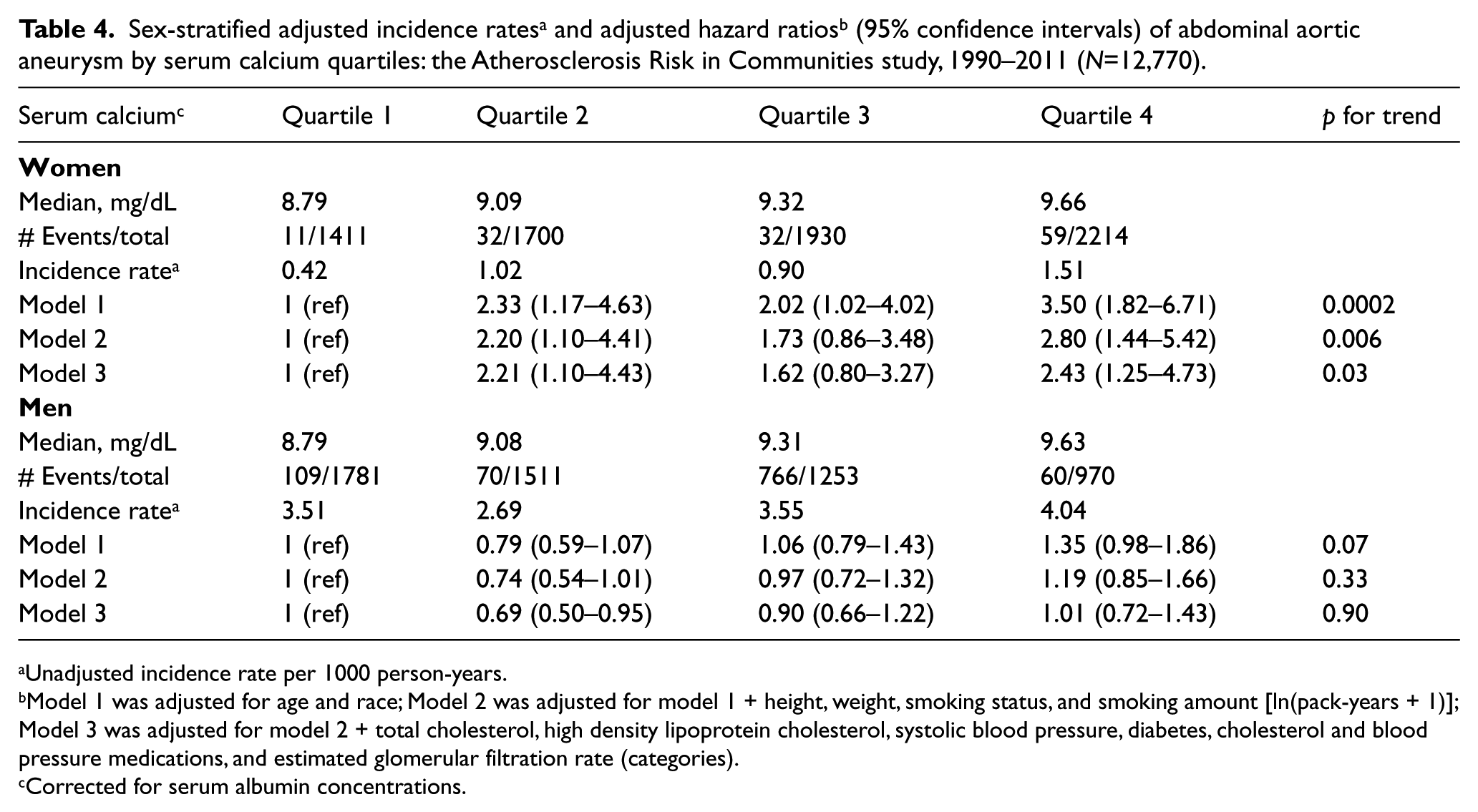
For serum calcium, participants in the highest versus lowest quartile were at higher risk of incident AAA after Model 1 adjustments [HR=1.63 (1.25–2.12)], but this association was attenuated with additional adjustment for anthropometrics and smoking [1.39 (1.05–1.83)] and became non-significant after adjustment for traditional cardiovascular risk factors [1.19 (0.90–1.58)]. There was evidence of interaction by sex (p for interaction in model 1 = 0.02; model 2 = 0.02; model 3 = 0.02), whereby the association appeared stronger in women than in men (Table 4). In the fully adjusted models, the women in the highest quartile of serum calcium (versus the lowest) had a HR for incident AAA of 2.43 (1.25–4.73), while in men the HR was 1.01 (0.72–1.43). Results were similar when sex-specific serum calcium quartiles were utilized (data not shown). There were no statistically significant interactions by age, race or smoking status.
Unadjusted incidence rate per 1000 person-years.
Model 1 was adjusted for age and race; Model 2 was adjusted for model 1 + height, weight, smoking status, and smoking amount [ln(pack-years + 1)]; Model 3 was adjusted for model 2 + total cholesterol, high density lipoprotein cholesterol, systolic blood pressure, diabetes, cholesterol and blood pressure medications, and estimated glomerular filtration rate (categories).
Corrected for serum albumin concentrations.
Neither FGF23 nor phosphorus was associated with risk of incident AAA in any of the models explored (Table 3). The demographic-adjusted (model 1) HR for the highest versus lowest quartiles of FGF23 and phosphorus were 1.13 (0.87–1.48) and 1.07 (0.79–1.44), respectively. For PTH, participants in the highest versus lowest quartile were at lower risk of incident AAA [0.57 (0.44–0.76)], after model 1 adjustments. However, this HR was attenuated with Model 2 adjustments [0.79 (0.58–1.07)]. There was no evidence of interaction by age, race, sex, or smoking status (interaction p-values all >0.12).
Discussion
In this large population-based prospective cohort there was little evidence that markers of vitamin D metabolism are associated with risk of incident AAA. For serum calcium, a statistically significant interaction by sex was observed. In analyses restricted to women, high versus low serum calcium was associated with an approximately doubling of AAA risk, even after accounting for traditional cardiovascular risk factors. There was no association between serum calcium and AAA risk in men. Other markers of vitamin D metabolism, namely 25(OH)D, FGF23, phosphorus, and PTH were not associated with risk of AAA. These findings provide the first prospective assessment of markers of vitamin D metabolism and future AAA risk in humans.
If suboptimal 25(OH)D or elevated serum calcium or PTH influence AAA risk, they likely do so by elevating established cardiovascular risk factors, namely hypertension,5–11 inflammation12,13 and vascular calcification.14,15 Our analysis did not provide convincing evidence that either 25(OH)D or PTH (which is downstream from 25(OH)D) are associated with AAA development. This is in contrast to the existing, albeit limited, literature. Evidence from animal models support the hypothesis that 25(OH)D may be associated with AAA risk. Specifically, it was recently reported that in an apolipoprotein E-knockout mouse model, treatment with calcitriol reduced dissecting AAA formation. 32 In humans, a cross-sectional study which screened older men for AAA reported a dose–response relationship between low 25(OH)D concentrations and greater abdominal aortic diameter. 33 Cross-sectional associations involving 25(OH)D should be viewed with caution, as reverse causation may occur since individuals with poorer health states often get less sunlight exposure and consequently have lower 25(OH)D concentrations. Higher PTH has been associated with impaired endothelial function, increased aortic pulse pressure, and decreased large artery elasticity. 34
Among ARIC participants, serum calcium was positively associated with incident AAA in women, but not in men. The potential role of calcium in cardiovascular disease, and more specifically in vascular calcification, is both controversial and complex.14,35–37 Emerging evidence suggests that initiation of calcification within the vasculature may be carefully regulated by osteoclast-like and osteoblast-like cells, in a process similar to that observed in bone. 36 Intake of calcium, in the form of dietary supplements, may transiently elevate serum calcium and by doing so disrupt homeostasis and promote vascular calcification.14,35 Our finding that serum calcium is associated with greater risk of AAA in women should be viewed as hypothesis generating and requires independent confirmation, as this sex interaction was unanticipated and may be due to chance. It is, however, notable that women take more dietary supplements than do men, 38 and among women taking calcium supplements 9% had evidence of hypercalcemia and 31% had hypercalcuria. 39 Whether calcium supplement intake may be leading to calcification of the abdominal aorta is unknown.
In the present analysis, we found no evidence that serum FGF23 or phosphorus were linked to AAA risk. This is counter to intriguing mechanistic evidence, which prompted us to test these hypotheses. Although much interest in FGF23 has been related to its potential role in left ventricular hypertrophy, 40 the co-receptor Klotho, which is mandatory to induce FGF23 signaling pathways, 16 is not expressed in the myocardium. It is, however, expressed in both human and rat aortas, as has been reviewed recently. 16 Additionally, in a rat experimental model of AAA, relative to controls, those exposed to a FGF23 antagonist had smaller aortic diameters and experienced a preservation of elastic fibers and smooth muscle cells. 17 FGF23 has also been associated with hypertension, 41 endothelial dysfunction42–44 and inflammation.45,46 Phosphate excess, which is upstream to high FGF23, has been associated with vascular calcification18–20 and myocardial fibrosis. 21
Strengths and limitations
The key strength of this study is its prospective assessment of AAA risk. Relatively few prospective studies have a sufficient number of AAA events to evaluate incidence. Additional strengths are the ascertainment of biomarkers using state-of-the-art assay methods, and the ARIC study’s extensive covariate information and active event surveillance. The key limitation of this study is that some AAA events were likely missed, perhaps particularly if the AAA event resulted in sudden death. ARIC did not conduct serial AAA screenings and therefore included in this analysis are only AAA recognized clinically, either due to the indication of AAA or being captured through imaging for another indication. 24 Misclassification of AAA case status would, however, likely be non-differential, and would therefore most likely bias results toward the null. Additionally, the biomarkers were only measured once, possibly not at the right time in the natural history of AAA. Lastly, at present it is unclear which biomarker is optimal for ascertaining vitamin D status. 47
Conclusion
In conclusion, despite evidence from in vitro animal and cross-sectional human studies suggesting a relation between vitamin D metabolism and the development of AAA, within the context of the prospective ARIC cohort markers of vitamin D metabolism were not substantially associated with AAA risk. The finding of greater AAA risk among women with high serum calcium concentrations requires verification in an independent cohort. AAA is an understudied cause of mortality; identifying factors which may be useful for the prevention of AAA, or for AAA screening, remains a priority.
Footnotes
Acknowledgements
The authors thank the staff and participants of the ARIC study for their important contributions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Institutes of Health (NIH) National Heart, Lung, and Blood Institute supported this research through R01 HL103695, R01 HL103706, and ARIC contracts HHSN268201100005C, HHSN268201100006C, HHSN268201100007C, HHSN268201-100008C, HHSN268201100009C, HHSN268201100010C, HHS-N268201100011C, and HHSN268201100012C. The NIH Office of Dietary Supplements also provided support through R01 HL103706-S1.