Abstract
The efficacy of biologic therapies in critical limb ischemia (CLI) remains elusive, in part, due to limitations in trial design and patient selection. Using a novel design, we examined the impact of complementing revascularization therapy with intramuscular JVS-100 – a non-viral gene therapy that activates endogenous regenerative repair pathways. In this double-blind, placebo-controlled, Phase 2B trial, we randomized 109 patients with CLI (Rutherford class V or VI) to 8 mg or 16 mg intramuscular injections of placebo versus JVS-100. Patients were eligible if they persistently had reduced forefoot perfusion, by toe–brachial index (TBI) or skin perfusion pressure (SPP), following successful revascularization with angiographic demonstration of tibial arterial flow to the ankle. The primary efficacy end point was a 3-month wound healing score assessed by an independent wound core laboratory. The primary safety end point was major adverse limb events (MALE). Patients’ mean age was 71 years, 33% were women, 79% had diabetes, and 8% had end-stage renal disease. TBI after revascularization was 0.26, 0.27, and 0.26 among the three groups (placebo, 8 mg, and 16 mg injections, respectively). Only 26% of wounds completely healed at 3 months, without any differences between the three groups (26.5%, 26.5%, and 25%, respectively). Similarly, there were no significant changes in TBI at 3 months. Three (2.8%) patients died and two (1.8%) had major amputations. Rates of MALE at 3 months were 8.8%, 20%, and 8.3%, respectively. While safe, JVS-100 failed to improve wound healing or hemodynamic measures at 3 months. Only one-quarter of CLI wounds healed at 3 months despite successful revascularization, highlighting the need for additional research in therapies that can improve microcirculation in these patients.
Keywords
Introduction
Critical limb ischemia (CLI), the most advanced form of peripheral artery disease (PAD), is associated with significant morbidity, major amputation, mortality, and poor quality of life due to persistent non-healing wounds.1,2 Over the last two decades, no new devices or adjuvant therapies have been approved for CLI despite a number of technical advances in revascularization methods.3–5 To that end, there is growing interest in developing biologic therapies with the prospect of offering new treatment or adjunctive strategies for advanced PAD. However, despite a basis in validated translational science and multiple clinical trials, the results collectively have been disappointing.6,7 Limitations of previous research include the common inclusion of the so-called ‘no-option’ patient, defined as individuals with a pattern of arterial occlusive disease that is considered non-reconstructible by surgical or endovascular therapy, or had failed revascularization attempts. Owing to their advanced CLI, along with complex comorbidities, such patients have no other reasonable treatment options available aside from the potential hope of biological therapy. Perhaps with unrealistic expectations for limb salvage, such studies have been doomed to inconclusive or frankly negative results. To date, no study has combined the mainstay of treatment, revascularization, with biologic therapy in CLI. Nor have any studies assessed post-revascularization perfusion to identify those patients most likely to benefit from adjunctive biologic therapy.
Stromal cell-derived factor 1 (SDF-1) is a naturally occurring chemokine protein that binds to the CXCR-4 receptor, activates endogenous regenerative repair pathways, promotes new blood vessel growth, prevents cell death when administered to damaged tissue, and causes remodeling of scar tissue.8–11 SDF-1 expression increases in response to tissue damage; however, this endogenous upsurge is transient and insufficient for wound healing in chronic ischemia states.9,12 Preclinical studies have shown that SDF-1 delivery was able to promote angiogenesis, increase vascular density, and accelerate wound healing.13–15 JVS-100 is a non-viral DNA plasmid-based therapy that encodes SDF-1 with an improved longevity and thus prolonged favorable effects. 16 We sought to investigate the efficacy and safety of JVS-100 as an adjunct to successful revascularization in a Phase 2B randomized, double-blind, placebo-controlled trial in patients with Rutherford class V and VI PAD who had abnormal perfusion as measured by toe–brachial index (TBI) post-revascularization.
Methods
Trial design
Study population
A total of 167 patients with CLI Rutherford class V and VI were enrolled. A qualifying ulcer was defined as the presence of tissue loss or dry gangrene for ⩾ 2 weeks in the foot of the index leg with a surface area of ⩾ 0.5 cm2 and ⩽ 25 cm2 (⩽ 10 cm2 if the target wound was located on the heel). 17 Alternatively, a minor amputation that was left open and performed within 12 days of the revascularization procedure may have been used as the index wound. Hemodynamically, all patients had to meet an inclusion criterion of an ankle systolic pressure ⩽ 70 mmHg, an ankle–brachial index (ABI) ⩽ 0.9, a toe systolic pressure ⩽ 50 mmHg, a TBI ⩽ 0.51, or dampened pulse volume waveforms with angiographic documented occlusion or severe stenosis (⩾ 70%) of all three tibial vessels. Subsequently and following a successful open or endovascular revascularization procedure in which angiographic flow to the ankle was demonstrated, assessment of forefoot perfusion using TBI or skin perfusion pressure (SPP), if TBI could not be performed, was completed. Patients demonstrating persistent forefoot malperfusion, as indicated by a TBI ⩽ 0.51, toe systolic pressure ⩽ 50 mmHg, SPP ⩽ 40 mmHg, or transcutaneous oximetry (TcPO2) ⩽ 40 mmHg post-intervention were then randomized (n = 109) to JVS-100 or placebo within 12 days of the above revascularization procedure (Figure 1). Of the 109 randomized patients, only one patient was lost to follow-up, one patient withdrew consent, and three patients died within 3 months of randomization. Major inclusion and exclusion criteria are shown in supplementary Appendix Table 1.
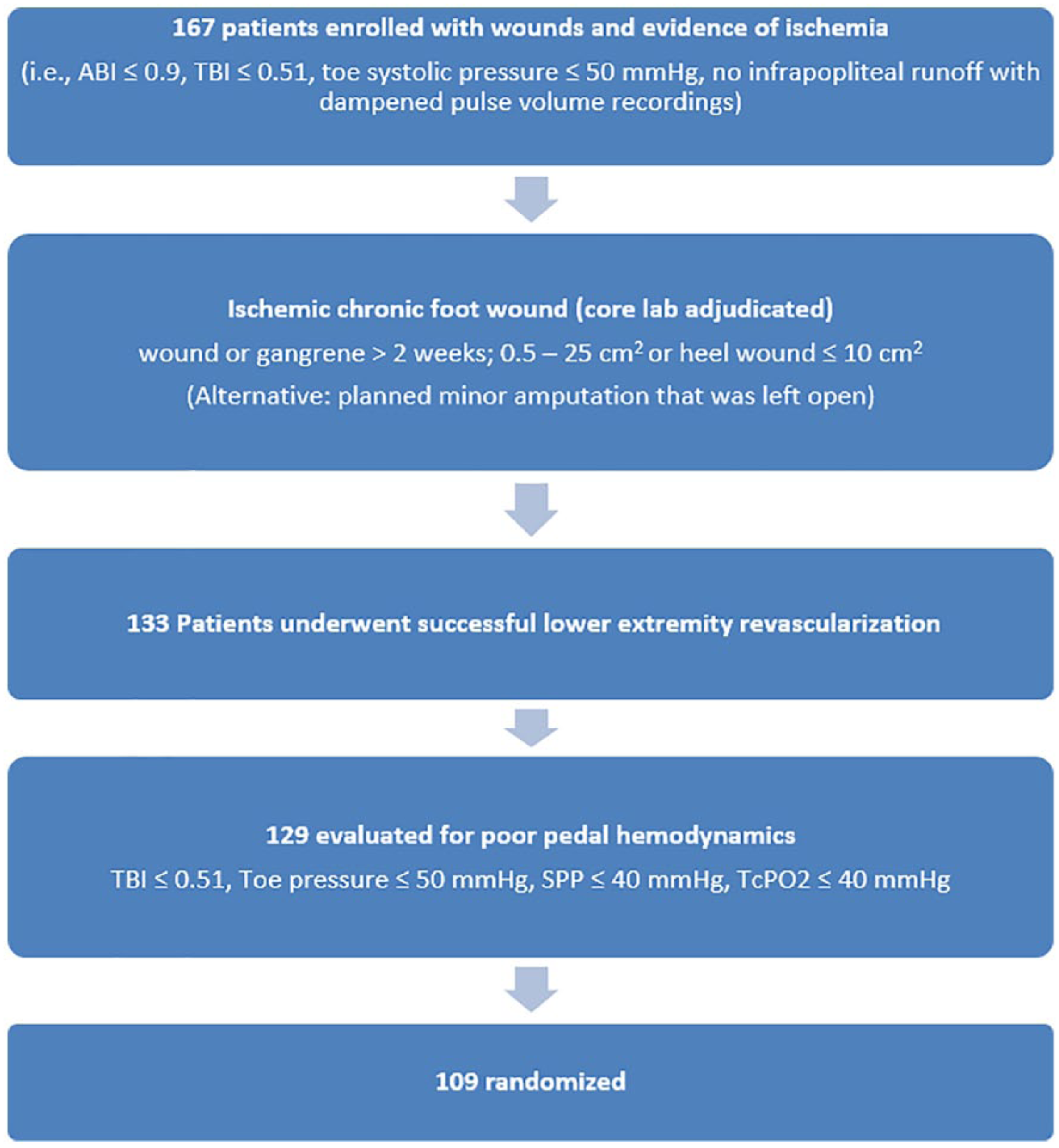
Flow chart of study design and enrollment. Patients with CLI Rutherford class V and VI were randomized to placebo or low- or high-dose JVS-100 only if they had persistent evidence of impaired pedal perfusion, by TBI or skin perfusion pressures, following successful revascularization with an angiographic demonstration of a tibial arterial flow to the ankle.
Randomization
Patients underwent randomization in a 2:1 ratio in a double-blinded fashion to receive 8 mg (8 × 1 mg/mL) or 16 mg (16 × 1 mg/mL) of JVS-100 or matching placebo intramuscular injections in the index leg and foot in a pattern shown in supplementary Appendix Figure 1. Randomization was performed with the use of an Interactive Web Response System (IWRS) and stratified by presence of diabetes and end-stage renal disease (ESRD). This study was approved by the United States Food and Drug Administration (FDA) and the Institutional Review Board of the participating centers. All participants provided written informed consent prior to enrollment and randomization (http://www.clinicaltrials.gov; registration number: NCT02544204).
Study drug blinding and administration
Pharmacists prepared JVS-100 or placebo injections by dividing the active drug or placebo vial contents into 8 (8 mg dose) or 16 (16 mg dose) syringes with a 25–30 gauge (physician’s discretion) needle attached. The syringes were delivered to the investigator in a blinded fashion for administration of 8 or 16 intramuscular injections into the index limb (Appendix Figure 1). All research personnel, including the site principal investigator, pharmacists, and those involved with wound care were blinded to randomization assignment. There was no report of any reaction at the injection site for any of the participating patients.
End points
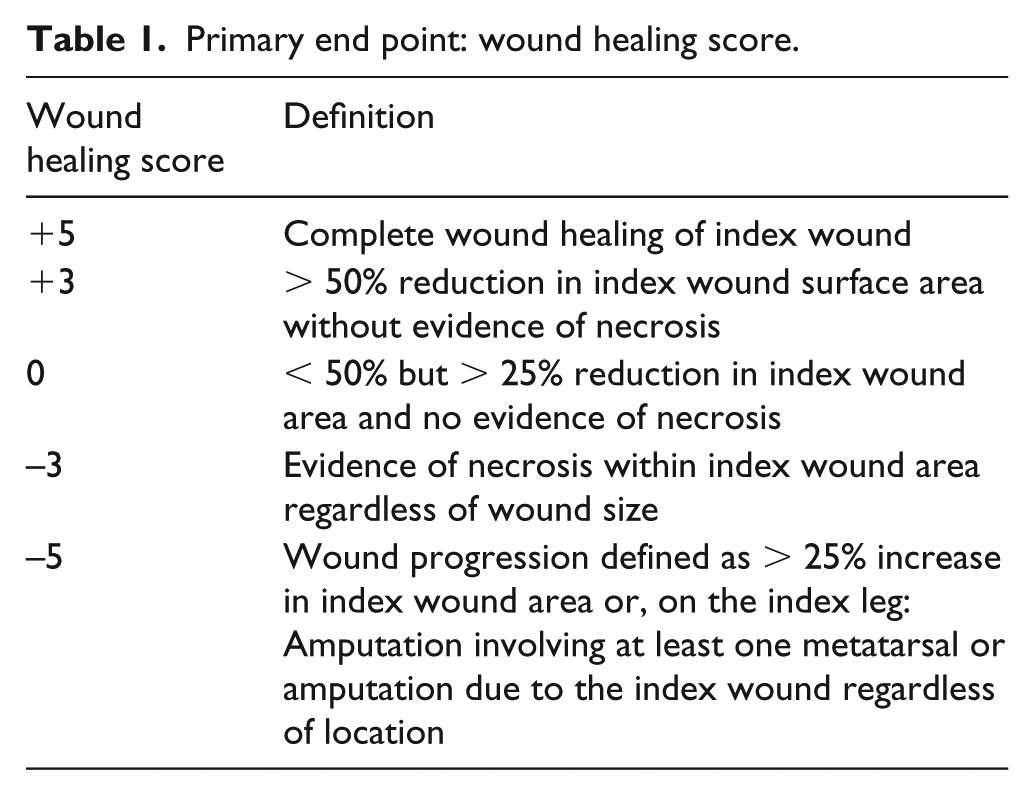
The primary efficacy end point assessed at 3 months was a wound healing score ranging from −5 (worst possible outcome of wound progression or amputation) to +5 (best possible outcome of complete wound healing) (Table 1). The primary safety end point was MALE (major adverse limb events) – a composite of major amputation plus clinically driven target lesion revascularization. Secondary end points included changes from baseline wound measures, incidence of complete wound healing, wound size, development of wound necrosis, major amputation, all amputations, and all-cause mortality. Hemodynamic measures including ABI and TBI were measured prior to revascularization and randomization, and at 3 months.
Primary end point: wound healing score.
Revascularization procedures and wound care
Patients underwent endovascular or open bypass procedures for lesions at or below the knee according to the local standard of care and the vascular specialist’s clinical judgment. At least one patent straight line blood flow to the foot was considered successful. There were no restrictions regarding the use of any approved devices for lower extremity revascularization. Wound care was left at the discretion of local enrolling sites; however, each site was required to identify a participating wound care specialist.
Statistical analysis
Sample size estimates for this trial are based on consultation with key opinion leaders in the field and data generated from registries composed of patients undergoing peripheral intervention plus standard of care. Furthermore, the trial is powered to statistically detect clinically meaningful differences in wound healing between groups of > 10%, similar to those reported in previous positive clinical trials,18–20 based on a difference of 1.8 in the mean with a pooled standard deviation of 3.0 for wound healing score at 3 months between the JVS-100 16 mg treatment group and placebo. Using the Wilcoxon rank sum test, a sample size of 80 patients (approximately 40 per treatment group) will provide 80% power to detect the difference between the JVS-100 treatment group and placebo using a two-sided 0.1 level of significance.
The change from baseline to that of composite end point at 3 months was tested using non-parametric analysis of covariance (ANCOVA), with a p-value (two-sided) of 0.1 considered statistically significant. For secondary end points, descriptive parametric or non-parametric summary statistics were used to evaluate continuous efficacy variables between the JVS-100 and placebo groups. All statistical analyses were performed using SAS software, Version 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
From November, 2016 through August, 2017, a total of 167 patients gave consent and were screened for randomization. Of these, 109 were randomized and followed. A total of three (2.8%) patients died, two (1.8%) had a major amputation, and 19 (17.4%) had any amputation within 3 months. The baseline characteristics of the patients were well balanced, with an equal number of individuals in each group with diabetes and ESRD on hemodialysis (Table 2). The mean post-procedure TBI was 0.26, which was not statistically different between the three groups. Similarly, the mean baseline wound area was 3.99 cm2, with no statistically significant difference between the groups.
Baseline demographic, wound, and angiographic characteristics (per protocol population).
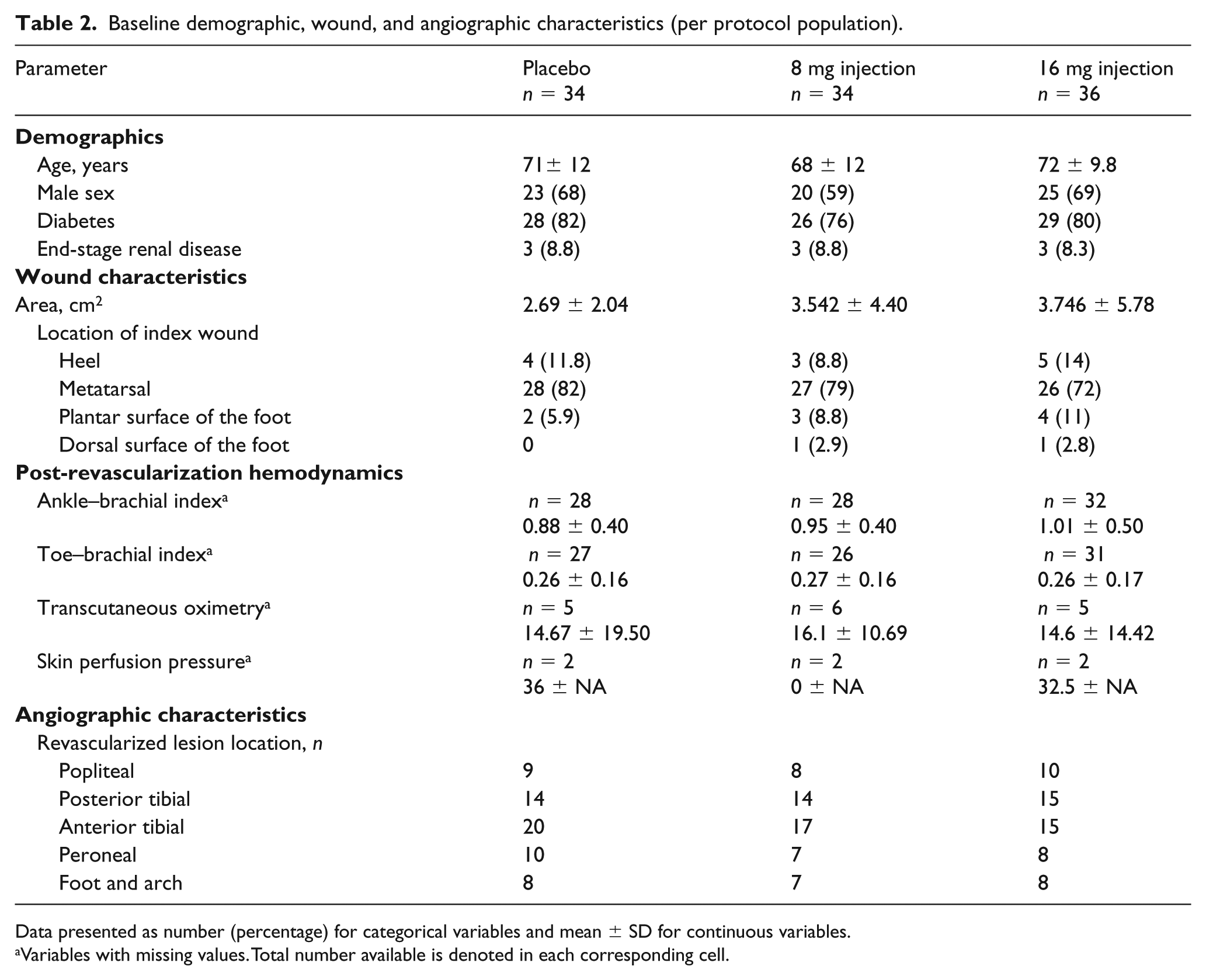
Data presented as number (percentage) for categorical variables and mean ± SD for continuous variables.
Variables with missing values. Total number available is denoted in each corresponding cell.
At 3 months, only 26% of wounds completely healed without any differences between the 3 groups (26.5% placebo, 26.5% 8 mg injection, and 25% 16 mg injection) (Figure 2). Similarly, no differences were seen among those with diabetes or ESRD. There was also no significant change in TBI between the groups (Table 2).
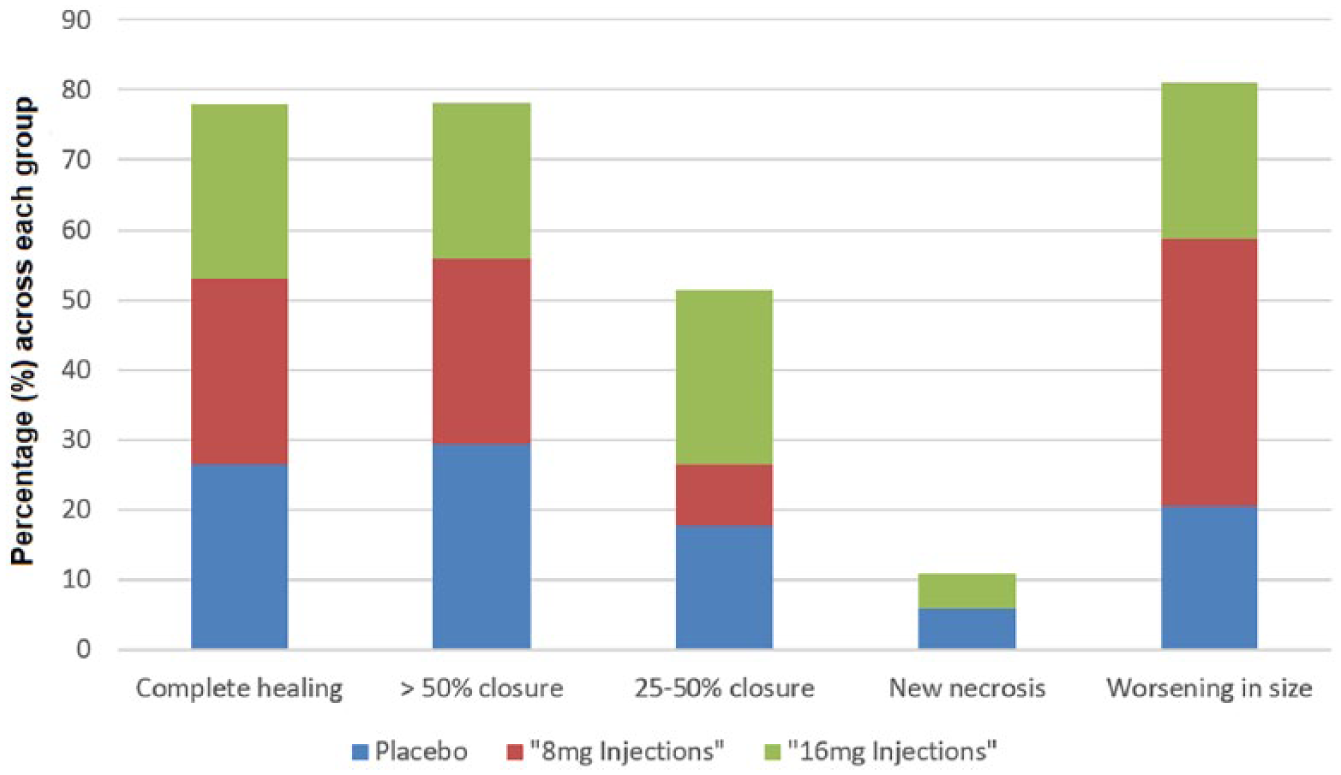
Primary efficacy according to wound healing score. All three groups had equivalent low rates (only one-quarter of patients in each group) of complete wound healing. Similarly, rates of ‘25–50%’, ‘new necrosis’, or ‘worsening in size’ and ‘> 50% closure’ were comparable among the three groups.
The total (sum) wound healing scores for the placebo, ‘8 mg injection’, and ‘16 mg injection’ groups were +34, +7, and +23, respectively (Table 3). The differences in MALE, major amputation, all amputations, wound area, and development of wound necrosis are shown in Table 3. None of the secondary end points, however, reached statistical significance. There were 24 wound infections: seven in the placebo group, five in the ‘8 mg injection’ group, and 12 in the ‘16 mg injection’ group.
Efficacy and safety outcomes at 3 months.
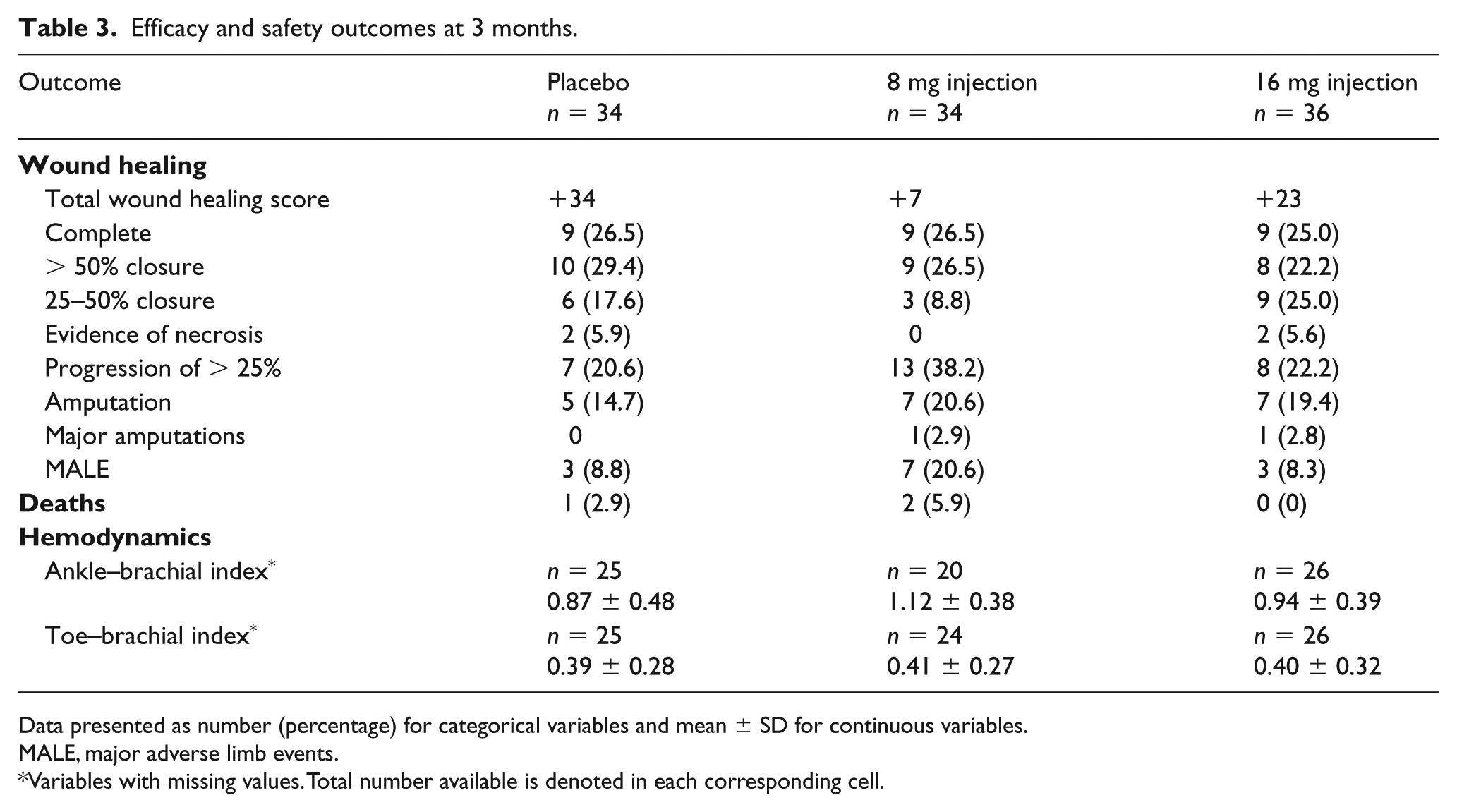
Data presented as number (percentage) for categorical variables and mean ± SD for continuous variables.
MALE, major adverse limb events.
Variables with missing values. Total number available is denoted in each corresponding cell.
Discussion
In the STOP-PAD trial, 8 mg and 16 mg doses of JVS-100 intramuscular injections, compared to placebo, failed to improve wound healing. Additionally, no improvement in foot perfusion was seen over time as measured by TBI. JVS-100 was safe, with similar rates of MALE and wound infections compared to placebo. Similarly, JVS-100 did not demonstrate any significant impact on secondary end points. However, despite proper wound care and follow-up only 25% of wounds healed within 3 months and there was a high rate of amputations.
Despite the many technical advances in treating CLI, including complex surgical and endovascular techniques to revascularize obstructed ankle and pedal arteries, the burden of disease including both macrovascular and microvasculature disturbance often prevents adequate distal tissue perfusion needed for timely wound healing and prevention of amputation.21–23 A minority of wounds in these patients heal within 3 months and persistent wounds are associated with hospital readmission, mortality, and MALE. 24 Furthermore, the presence of a wound is associated with increased cost, frequent follow-up, the need for offloading, and poor quality of life. 21 For these reasons, there has been continued interest in biologic therapy as a stand-alone or adjunctive to revascularization in patients with CLI. The hypothesis has been that revascularization will improve macrovascular perfusion and biologic therapies will enhance microvascular blood flow.
However, despite the many efforts using various pathways, including gene, cell-based, mesenchymal stem cells, and even marker-selected mononuclear cells, the results have been collectively disappointing.25,26 Indeed, after 20 years of research, no biologic therapy has been FDA-approved for the treatment of CLI. Several reasons have been identified and previously described. 27 In addition to finding the right biologic therapy or combination of therapies, previous studies have been limited by the selection of patients who are less likely to benefit from these treatments; for example, patients with claudication but without wounds, or, on the other end of the spectrum, the advanced ‘no-option’ CLI patients. In patients with claudication, the main perfusion issue is large vessel inflow disease (iliac and superficial femoral arteries); it is unlikely that the injections of biologic therapy in these patients will improve symptoms. Similarly, biologic therapy or any improvement in perfusion is unlikely to improve end-stage ‘no-option’ CLI where there is gangrene. Another important limitation of previous biologic therapies has been end points. Many of these studies relied on ABI as an end point; however, ABIs have many limitations and do not reflect foot perfusion.28,29 Other end points, such as walking distance, are unlikely to be impacted by improvement in microvascular circulation and there is significant variability in amputation-free survival. We therefore attempted to address these previous limitations by combining revascularization with JVS-100 and using core laboratory adjudicated wound healing as the primary end point in CLI patients with at least one straight line of flow to the foot post-revascularization, yet evidence of decreased microvascular perfusion as measured by TBI, SPP or TcPO2.
JVS-100 delivers a longer acting form of SDF-1, as demonstrated by local plasmid expression 15–20 days post-administration in preclinical models.16,30 SDF-1 binding to the CXCR4 receptor has been shown to promote multiple mechanisms of ischemic tissue repair inducing angiogenesis, vasculogenesis, recruitment of endogenous stem cells, inhibition of apoptosis and cell death, and reduction in fibrosis to improve tissue perfusion and wound repair.8–11,31 These multiple mechanisms theoretically provide an advantage over other plasmid-based single growth factor therapies, such as vascular endothelial growth factor (VEGF) and non-viral fibroblast growth factor type 1 (nv1-FGF), which were shown to be safe, but ultimately ineffective.32,33
Unfortunately, as a therapy for chronic wounds, the endogenous increase in SDF-1 expression associated with injury is short-lived. 13 Administration of JVS-100, a plasmid-based SDF-1 therapy, provides longer exposure to the benefits of increased SDF-1 expression. SDF-1 gene expression is upregulated in patients with advanced PAD,12,14 but endogenous SDF-1 levels are too low to stimulate adequate microvascular growth to heal wounds. Observations from human skeletal muscle analysis in CLI patients undergoing peri-genicular amputation demonstrate increased expression of CXCR4 in skeletal muscle fibers and microvessels; however, the increased CXCR4 expression lacks sufficient microvascular support to sustain function. 14
Faster wound healing is important because of the growing focus on reducing hospital readmissions, healthcare costs, and improving efficiency, particularly relative to the treatment of patients with CLI. 34 The Affordable Care Act and the US Department of Health and Human Services’ efforts to improve quality over quantity with bundled payments has renewed interest in outcomes beyond MALE and major amputation-free survival in patients with CLI. In a recent analysis, we showed that almost 50% of readmissions for CLI were wound related. 24 Indeed, a significant portion of the cost associated with CLI treatment occurs after discharge.34–36 More importantly, those who heal their wounds within 3 months are less likely to have a major amputation. 24 Therefore, the addition of biologic therapies to revascularization procedures would be appealing if they were able to demonstrate faster wound healing. The STOP-PAD trial was the first trial, to our knowledge, to test this hypothesis of biologic therapy following revascularization procedures, with the goal of improving foot perfusion and wound healing. However, a 3-month follow-up might be a relatively short duration to see the full impact of JVS on wound healing, and therefore we look forward to the 6-month results.
Despite not meeting the primary end point, the STOP-PAD trial highlighted a number of important issues related to CLI and wound healing. Despite advanced revascularization and rigorous follow-up, only 25% of wounds healed at 3 months. In addition, and importantly, almost 20% of wounds increased in size. This was in parallel to the small incremental increase in TBI from pre- to post-revascularization. The small changes in TBI, as shown in Figure 3, are indicative of the current limitations of endovascular and open surgical revascularization to improve microcirculation. This finding confirms the lack of improvement in perfusion post-revascularization that was previously shown by our group.37,38 It is also worth noting that about one-quarter of the patients were not prescribed statins despite the American Heart Association/American College of Cardiology (AHA/ACC) guideline class I recommendations (supplementary Appendix Table 2). 17 In addition, there have been multiple studies discussing the potential benefits of statins in angiogenesis through upregulation of SDF-1 and overexpression of CXCR4.39–43
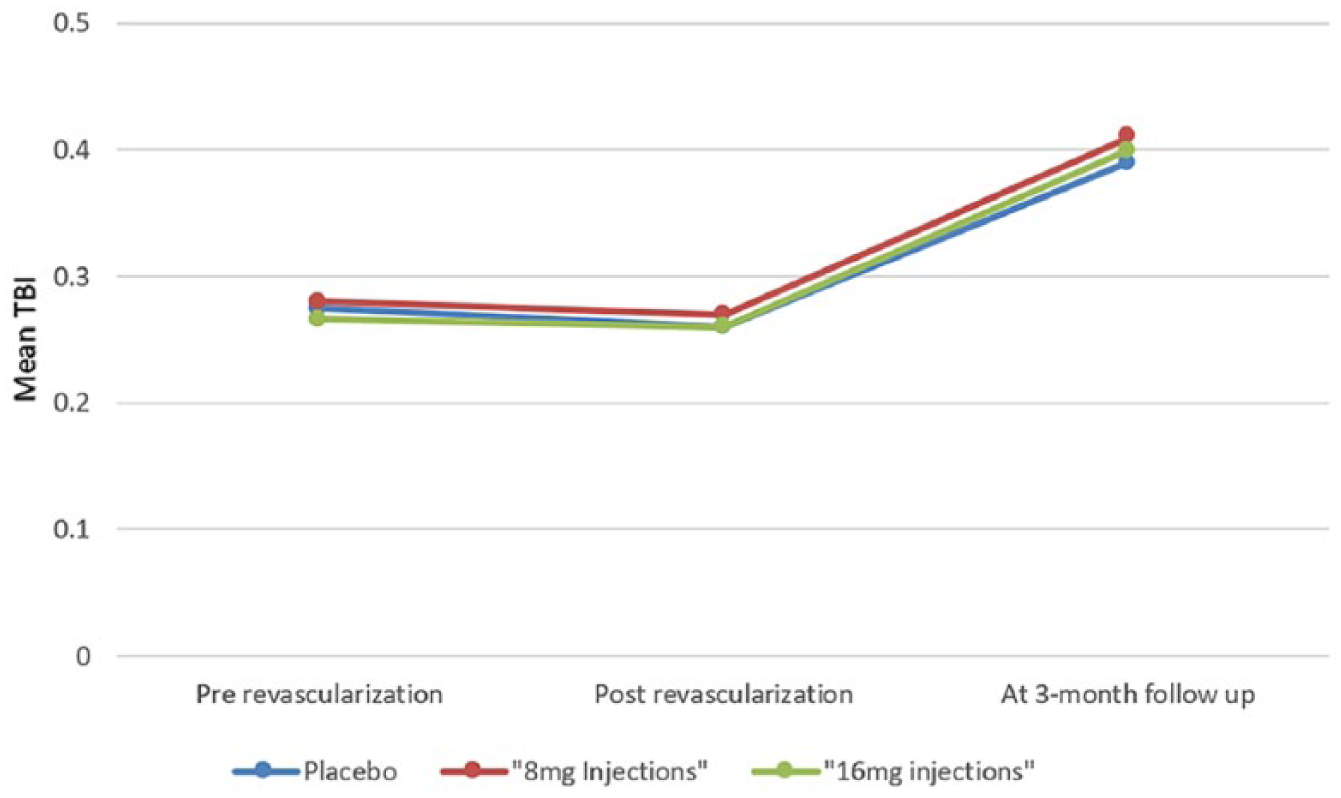
Changes in TBI before revascularization, after revascularization, and at 3 months post-randomization. All three groups had an average TBI of < 0.3 prior to randomization, with only a trivial change post-revascularization, and no meaningful increase at 3 months.
Limitations
There are a number of limitations: first, an angiosome-based revascularization (treating the tibial artery that supplies direct blood flow to the wound) was not mandated but encouraged. This may have resulted in suboptimal macrovascular revascularization and perfusion of the target area; however, the angiosome concept remains controversial and has only a class IIb recommendation by the recent AHA/ACC guidelines. Second, the optimal route and site of intramuscular injection of JVS-100 is unknown. Third, post-revascularization wound care was not standardized; however, this reflects real world practice. Fourth, the 3-month window may be too short to see the full effects of biologic therapies and it is possible that 6 or 12 months may more accurately represent the actual therapeutic effect.
Conclusion
The findings from our study are disappointing but confirm the high morbidity and mortality associated with CLI, even with modern revascularization techniques. Over two-thirds of our patients had diabetes and nearly 10% had ESRD. Rates of death and amputation remain high. While JVS-100 could not impact wound healing at 3 months, our trial design confirmed that wound healing may be useful as a primary end point. Our study suggests that wound healing is a complex process and may require a combination of therapies including both optimized revascularization and a mixture of biologic therapies addressing multiple targets for angiogenesis, vasculogenesis, fibroblast recruitment, and other mechanisms of wound healing.
Supplemental Material
10.1177_1358863X18817610_Supplementary_material – Supplemental material for SDF-1 plasmid treatment for patients with peripheral artery disease (STOP-PAD): Randomized, double-blind, placebo-controlled clinical trial
Supplemental material, 10.1177_1358863X18817610_Supplementary_material for SDF-1 plasmid treatment for patients with peripheral artery disease (STOP-PAD): Randomized, double-blind, placebo-controlled clinical trial by Mehdi H Shishehbor, John Rundback, Matthew Bunte, Tarek A Hammad, Leslie Miller, Parag D Patel, Saihari Sadanandan, Michael Fitzgerald, Joseph Pastore, Vikram Kashyap and Timothy D Henry in Vascular Medicine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Shishehbor served as the National PI for STOP-PAD but did not receive any type of financial reimbursement for this role. Michael Fitzgerald and Joseph Pastore (deceased) are employees of Juventas Therapeutics. The other authors have no relevant disclosures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was funded by Juventas Therapeutics. The authors are responsible for the design and conduct of this study, all study analyses, the drafting and editing of the paper, and final contents.
Supplemental material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
