Abstract
High-intensity statins are recommended for patients with peripheral artery disease (PAD). Critical limb ischemia (CLI) is the most advanced presentation of PAD. The benefit of statins in the CLI population is unclear based on the existent studies. Our objective was to perform a systematic review and meta-analysis regarding the efficacy of statin therapy in patients with CLI. PRISMA guidelines were followed. PubMed, EMBASE, and Cochrane CENTRAL databases were reviewed up to April 30, 2019. The primary outcomes included amputation rates and all-cause mortality. Secondary outcomes included primary patency rates, amputation-free survival and major adverse cardiac or cerebrovascular events (MACCE). Risk of bias was assessed with the Robins-I tool for observational studies. A random-effects model meta-analysis was performed. Heterogeneity was assessed with I2. Funnel plots and Egger’s test were used to assess publication bias. Nineteen studies including 26,985 patients with CLI were included in this systematic review. Among patients with known data on statin status, 12,292 (49.6%) were on statins versus 12,513 (50.4%) not on statins. Patients treated with statins were 25% less likely to undergo amputation (HR 0.75; 95% CI: 0.59–0.95; I2 = 79%) and 38% less likely to have a fatal event (HR 0.62; 95% CI: 0.52–0.75; I2 = 41.2%). Statin therapy was also associated with increased overall patency rates and lower incidence of MACCE. There was substantial heterogeneity in the analysis for amputation and amputation-free survival (I2 > 70%). In conclusion, statins are associated with decreased risk for amputation, mortality, and MACCE, as well as increased overall patency rates among patients with CLI. Future studies should assess whether other lipid-lowering medications in addition to high-intensity statins can further improve outcomes among patients with CLI. (
Introduction
Peripheral artery disease (PAD) affects more than 200 million people worldwide and four to eight million people in the United States.1–3 Among patients older than 60 years of age, PAD prevalence is approximately 12–20% and is even more prevalent in patients with a history of diabetes (DM), smoking, or chronic kidney disease (CKD).4–6 Patients with PAD have a higher mortality risk, driven by higher cardiovascular mortality rates secondary mainly to atherosclerotic coronary artery disease (CAD).3,6–9 Critical limb ischemia (CLI) is the most advanced form of PAD and is associated with up to a 50% 6-month risk for major amputation, a 25% 1-year mortality risk, worsened quality of life measures, and high healthcare costs.10–13
PAD is managed as a CAD equivalent with regards to lipid-lowering therapy and there is a class 1 recommendation in the most recent American College of Cardiology (ACC) and American Heart Association (AHA) guidelines but also for statin therapy in patients with PAD.3,14 Current guidelines suggest that a low-density lipoprotein (LDL) level of less than 100 mg/dL should be the target for high-risk patients with PAD. 12 However, there are no specific randomized clinical trials (RCTs) studying the effect of statins in the CLI population and thus CLI management is based on recommendations derived from evidence in patients with claudication or low ankle–brachial index (ABI) values. Previous observational data have suggested a benefit of statins – and specifically high-intensity statins – in patients with CLI and PAD.15,16 Our objective with this meta-analysis was to systematically review the medical literature examining outcomes of statin use in patients with CLI and to quantitatively synthesize these data in order to reach conclusions about the current role of, and benefits of, statin therapy in CLI.
Methods
This systematic review and meta-analysis were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and was registered in the PROSPERO international prospective register of systematic reviews (PROSPERO registration number: CRD42019134160). 17
Literature search
Three different major databases were reviewed for eligible studies: Cochrane CENTRAL, EMBASE, and Medline (PubMed), up to April 30, 2019. The literature search was conducted by two independent investigators (DGK, AAM), who were blinded to each other. When there was a disagreement, a third investigator was used (SG) to reach consensus. The following keywords in different combinations were used for the literature search: ‘statin’, ‘statins’, ‘atorvastatin’, ‘simvastatin’, ‘rosuvastatin’, ‘fluvastatin’, ‘lovastatin’, ‘pitavastatin’, ‘lipids’, ‘peripheral artery disease’, ‘PAD’, ‘critical limb ischemia’, and ‘CLI’. The following inclusion criteria were applied: (i) studies designed as RCTs or with observational design (prospective or retrospective cohorts or case–control studies) directly comparing statins versus no statins for patients with CLI; (ii) studies that reported quantitative data for at least one of the outcomes of interest; (iii) studies that did not include duplicated populations with other included studies in this meta-analysis in order to avoid counting the same patients twice. The references of the included studies were also manually reviewed to identify further eligible articles. The initial literature search yielded 253 potentially eligible studies. After excluding studies that did not meet the inclusion criteria, a total of 19 studies were deemed eligible for the systematic review. From these, 14 studies were included in meta-analysis. The detailed search algorithm is depicted in the PRISMA flow diagram in Figure 1.
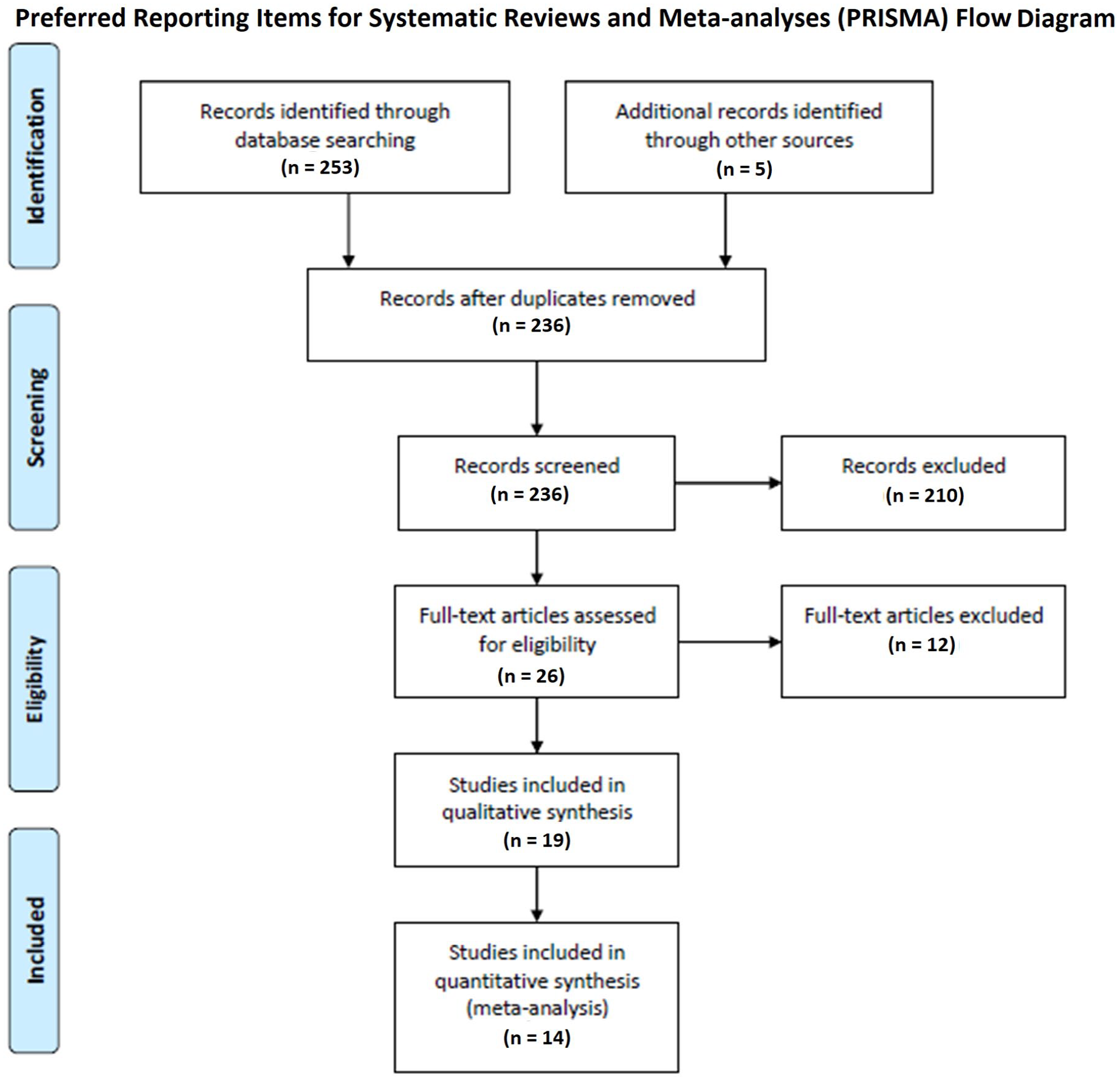
PRISMA flow diagram.
Data extraction and outcomes
Two reviewers blinded to each other (OJ, AKJ) extracted the pertinent data from the eligible studies based on a predefined data extraction form. All disagreements were resolved with discussion and with the involvement of a third investigator (AAM) in order for consensus to be reached. Data extracted included the name of the study and the name of the first author, year of publication, center and country where the study was performed, inclusion and exclusion criteria, patient number in the study and in each of the groups, baseline demographics, pertinent clinical characteristics and laboratory values, and outcomes of interest. Primary outcomes included limb loss and mortality assessment. Secondary outcomes included primary patency, any patency (defined as either primary or primary assisted or secondary), amputation-free survival, and major adverse cardiac or cerebrovascular events (MACCE) rates, as those were defined by the original studies.
Risk of bias assessment
Risk of bias assessment was assessed independently by two investigators (IK, AAM) with the ROBINS-I tool for nonrandomized studies. 18 The nonrandomized studies were evaluated for the following domains: confounding, selection of participants, departure from intended interventions, missing data, measurement of outcomes, and selective reporting.
Statistical synthesis and analysis
Outcomes were reported by the individual studies with one of the following: hazard ratios (HR), odds ratios (OR), relative risk (RR) or event rates. Most of the studies reported HRs and thus it was decided that the pooled outcomes will be presented with this measure of outcomes of interest. OR/RR or events rates were converted to HR with the following approach: HR = OR / (1 – p + (p * OR). Whenever a study reported the outcomes as nonstatin users versus statin users, those were converted to statins users versus nonstatin users. Whenever a study reported the outcomes in an inverse way compared to the rest of the studies (i.e. limb salvage for one study vs limb loss for the rest of the studies), the HRs/ORs were inverted in order to be indicative of the analyzed outcome of interest. Individual study outcomes were pooled based on the original studies definition. When we wanted to analyze an outcome and one of the studies reported this outcome as part of the composite endpoint (i.e. Dosluoglu et al. reported amputation rates only as part of the composite endpoint of death or amputation24–26), a separate (sensitivity) analysis was performed without this study. Given that the follow-up time among studies was variable for outcomes, the latest available follow-up time was utilized in each study when adjudicating dichotomous outcomes. A random-effects model was used to account for heterogeneity among studies. Outcomes were analyzed and presented as HRs and the corresponding 95% CIs. Higgins I2 statistic was used to account for heterogeneity and when it was > 70% we considered the heterogeneity substantial. 19 Forest plots were used to graphically display the effect size in each study and the pooled estimates. Funnel plots and Egger regression tests were used to assess publication bias.20,21 A sensitivity analysis was performed among studies that reported amputation rates (we excluded the study by Dosluoglu et al., which presented a composite endpoint of amputation or death). Statistical analysis was performed using STATA software, version 14.1 (StataCorp, College Station, TX, USA).
Results
Study and patient characteristics
In total, 19 studies and 26,985 individual patients were included.15,16,22–38 Twelve studies were conducted in the US, four in Europe, and three in Japan. No RCTs were identified. Four studies were prospective and 15 were retrospective. Inclusion and exclusion criteria for each individual study are presented in Table 1. Detailed patient characteristics are presented in Table 2. The included number of patients in each study ranged from 39 to 14,825, and the majority of the studies had a predominant male population. The mean age ranged from 67.5 to 77 years among the individual studies. The prevalence of comorbidities such as hypertension (HTN), (Hypelipidemia) HLD, DM, smoking history, CAD, CKD, myocardial infarction (MI) or stroke history, as well as use of aspirin of clopidogrel, can be found in Table 2. Three of the studies were from the same center (Dosluoglu et al. 2010, 2012, 201424–26) and two other studies were also from the same center (Fernandez et al. 2010, Saqib et al. 201330,37). After excluding the duplicated populations and studies that did not report outcomes with HR, 14 studies from 13 different populations (two studies by Dosluoglu were included which had the same population but were not analyzed together for the same outcome) were included in quantitative synthesis out of the 19 initial studies included in this systematic review. The study by Vogel et al. was included in the analysis as two different studies, since they provided two different HRs: one for statin use among patients with CLI Rutherford 4 (rest pain) and one for statin use among patients with CLI Rutherford 5 or 6 without providing an overall pooled HR for all patients with CLI. 36 Among patients included in the meta-analysis, 12,292 patients (49.6%) were known to be on statins versus 12,513 (50.4%) not on statins. All of the studies were assessed as having a low or moderate risk of bias – we did not find any studies with a high risk of bias.
Study characteristics.
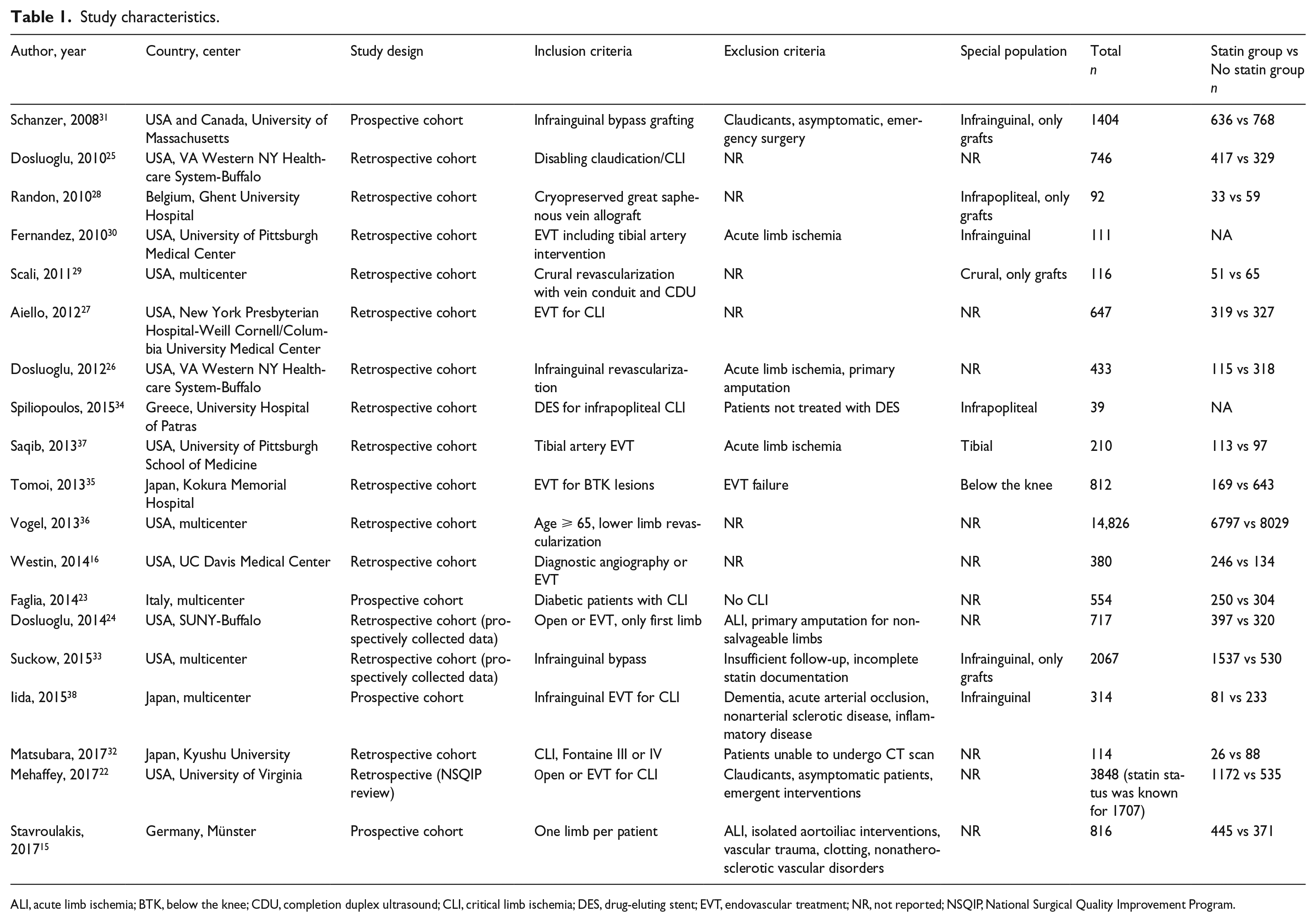
ALI, acute limb ischemia; BTK, below the knee; CDU, completion duplex ultrasound; CLI, critical limb ischemia; DES, drug-eluting stent; EVT, endovascular treatment; NR, not reported; NSQIP, National Surgical Quality Improvement Program.
Patient characteristics of the included studies.
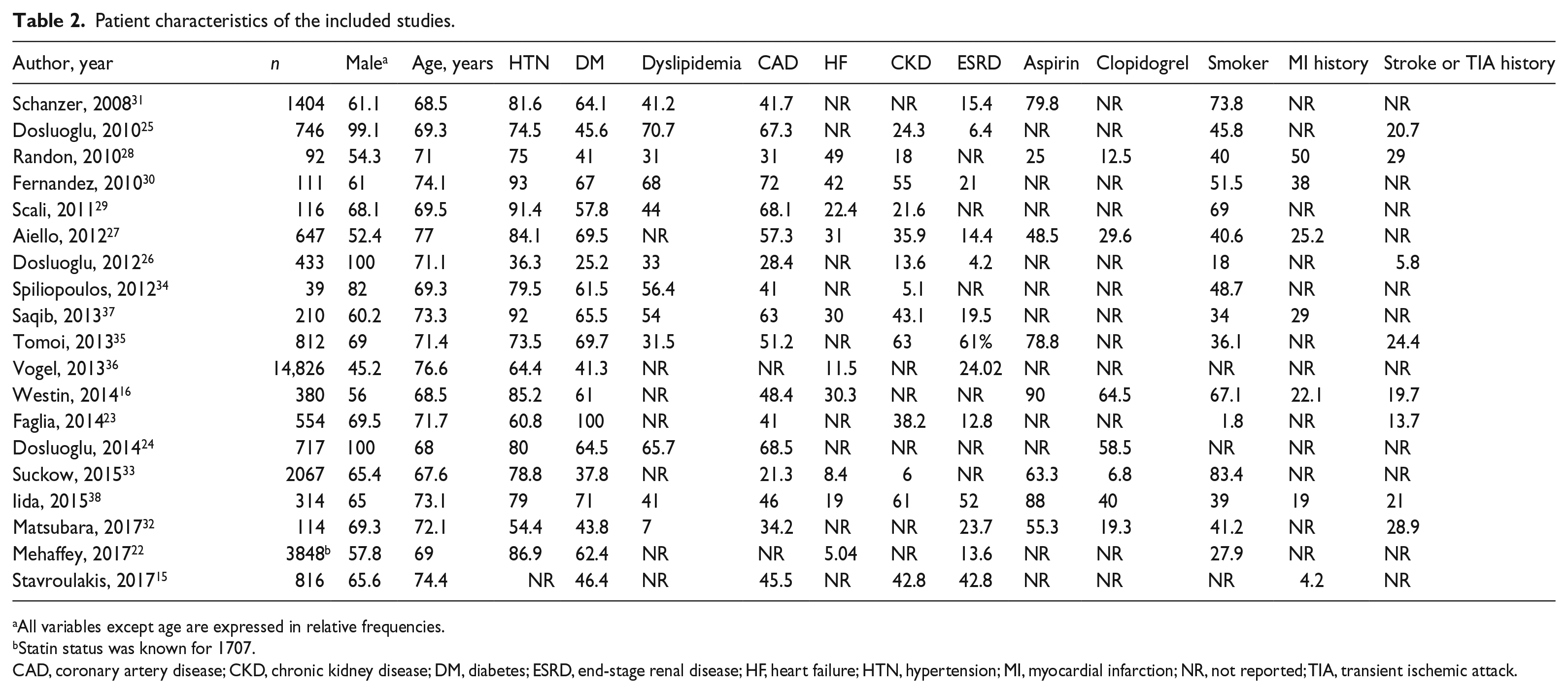
All variables except age are expressed in relative frequencies.
Statin status was known for 1707.
CAD, coronary artery disease; CKD, chronic kidney disease; DM, diabetes; ESRD, end-stage renal disease; HF, heart failure; HTN, hypertension; MI, myocardial infarction; NR, not reported; TIA, transient ischemic attack.
Meta-analysis
Limb loss, mortality, amputation-free survival, primary patency, and MACCE
Seven studies reported limb loss as an outcome. Our pooled analysis showed that statin use was associated with fewer major amputations among patients with CLI (HR 0.75; 95% CI: 0.59–0.95; I2 = 79%) (Figure 2A), although the results had substantial heterogeneity. No publication bias was detected (Egger test: p = 0.6). This association with decreased amputation rates was maintained, even when we performed a sensitivity analysis excluding the study by Dosluoglu et al. (which was the only one that did not use pure amputation rates as outcome but the composite endpoint of mortality or amputation) (HR 0.76; 95% CI: 0.58–1.00; I2 = 79.4%) (Figure 2B). However, the results had substantial heterogeneity once again. No publication bias was detected (Egger test: p = 0.652). Patients treated with statins had lower associated mortality rates compared to patients with CLI who were not treated with statins (HR 0.62; 95% CI: 0.52–0.75; I2 = 41.2%) (Figure 3). No publication bias was detected (Egger test: p = 0.242). Among five studies that provided primary patency as an outcome, there was no significant difference between the two groups (HR 0.86; 95% CI: 0.67–1.10; I2 = 53.2%) (Figure 4). No publication bias was detected (Egger test: p = 0.806). In the pooled analysis among the seven studies that reported any type of patency, patients treated with statins had lower rates of loss of patency (HR 0.80; 95% CI: 0.66–0.96; I2 = 40.9) (Figure 5). No publication bias was detected (Egger test: p = 0.666). Four studies reported amputation-free survival instead of, or in addition to, amputation rates. Among those, statins were associated with increased amputation-free survival (HR 0.71; 95% CI: 0.51–1.00; I2 = 73.7%) (Figure 6), although the heterogeneity was substantial. No publication bias was detected (Egger test: p = 0.509). Finally, patients treated with statins had decreased associated rates of MACCE among the four studies that reported this endpoint (HR 0.50; 95% CI: 0.39–0.66; I2 = 0) (Figure 7). No publication bias was detected (Egger test: p = 0.6). Although the Egger test was not significant for the outcome of mortality, the contour funnel plot shows asymmetry in the plot, indicating that smaller studies were more strongly associated with a protective effect of statins compared to larger studies (Figure 8).
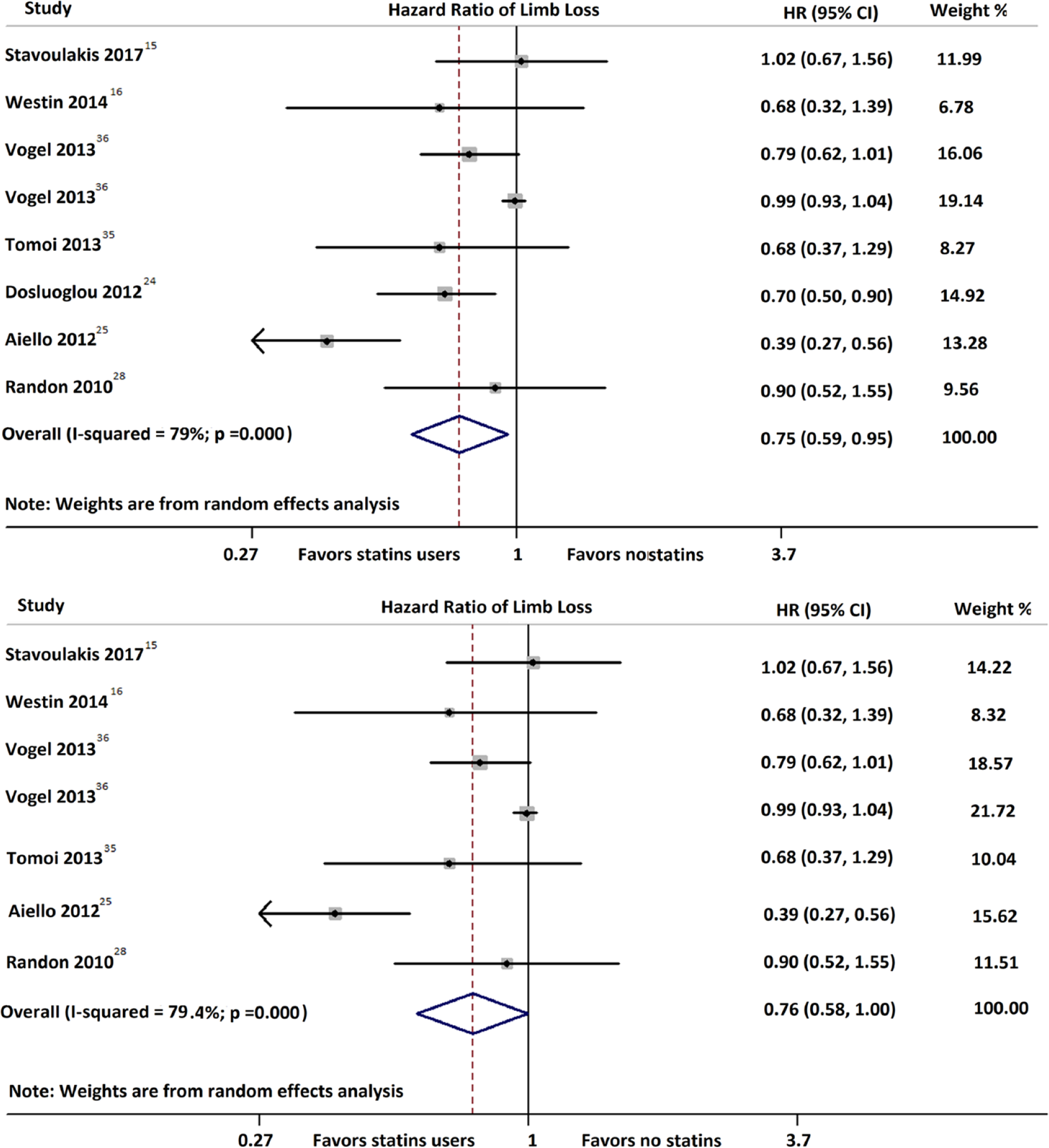
Statins were associated with lower amputation rates among patients with CLI (HR 0.75; 95% CI: 0.59–0.95; I2 = 79%) (A). Similar results were obtained in the sensitivity analysis that excluded the study by Dosluoglu et al., which was the only one that reported a composite endpoint of amputation or death and not pure amputation (HR 0.76; 95% CI: 0.58–1.00; I2 = 79.4%) (B).
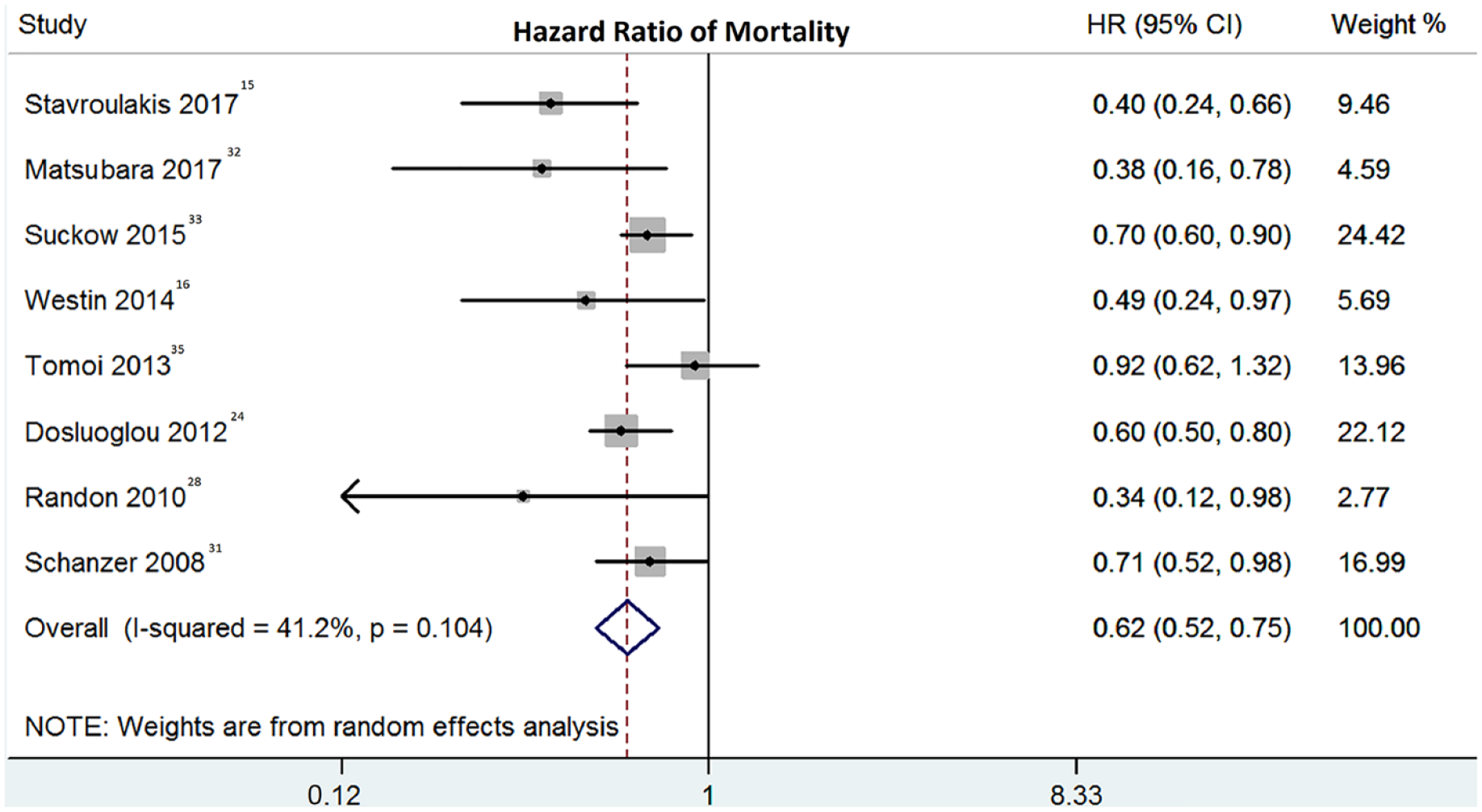
Statins were associated with lower mortality rates among patients with CLI (HR 0.62; 95% CI: 0.52–0.75; I2 = 41.2%).
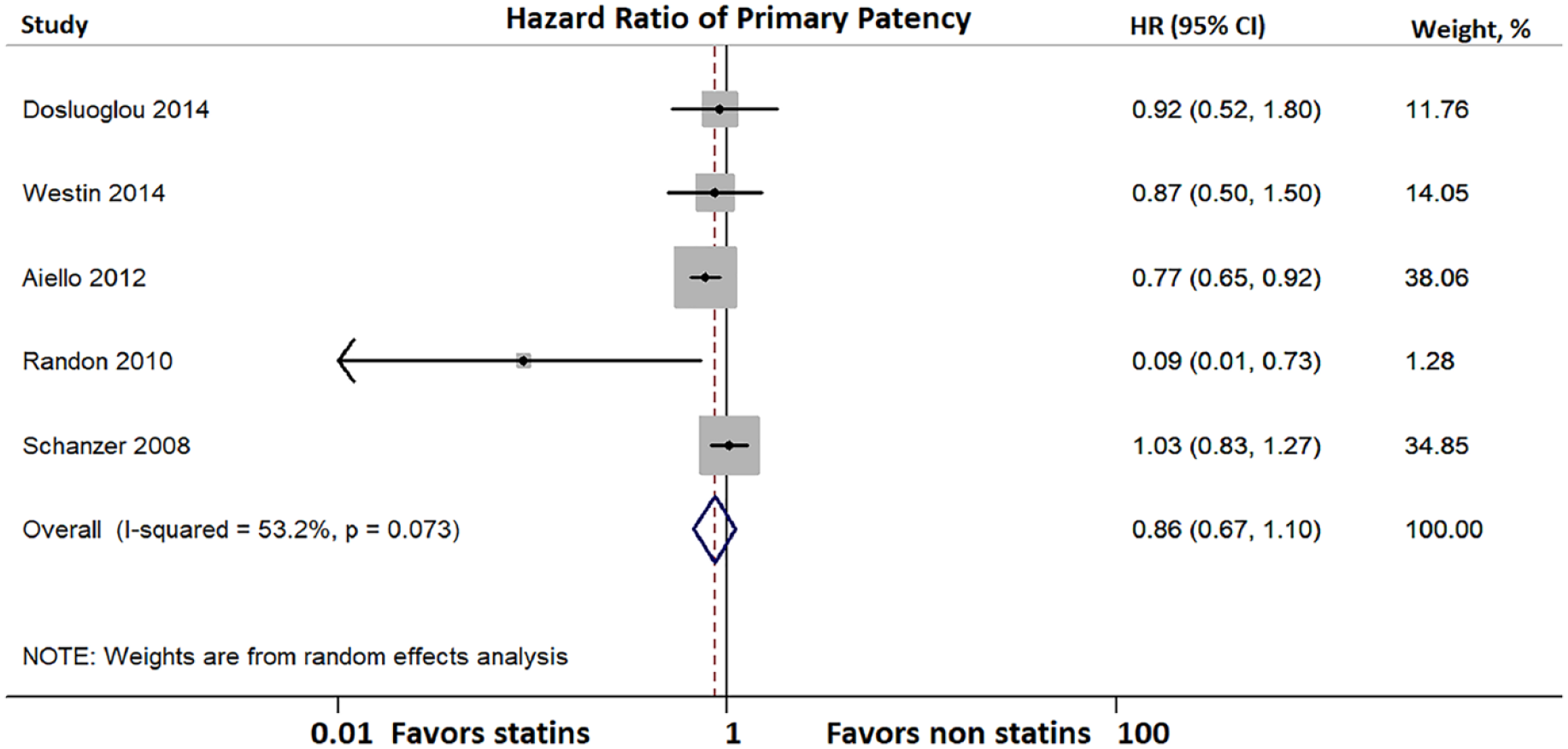
There was no difference between patients treated with or without statins regarding primary patency (HR 0.86; 95% CI: 0.67–1.10; I2 = 53.2%).
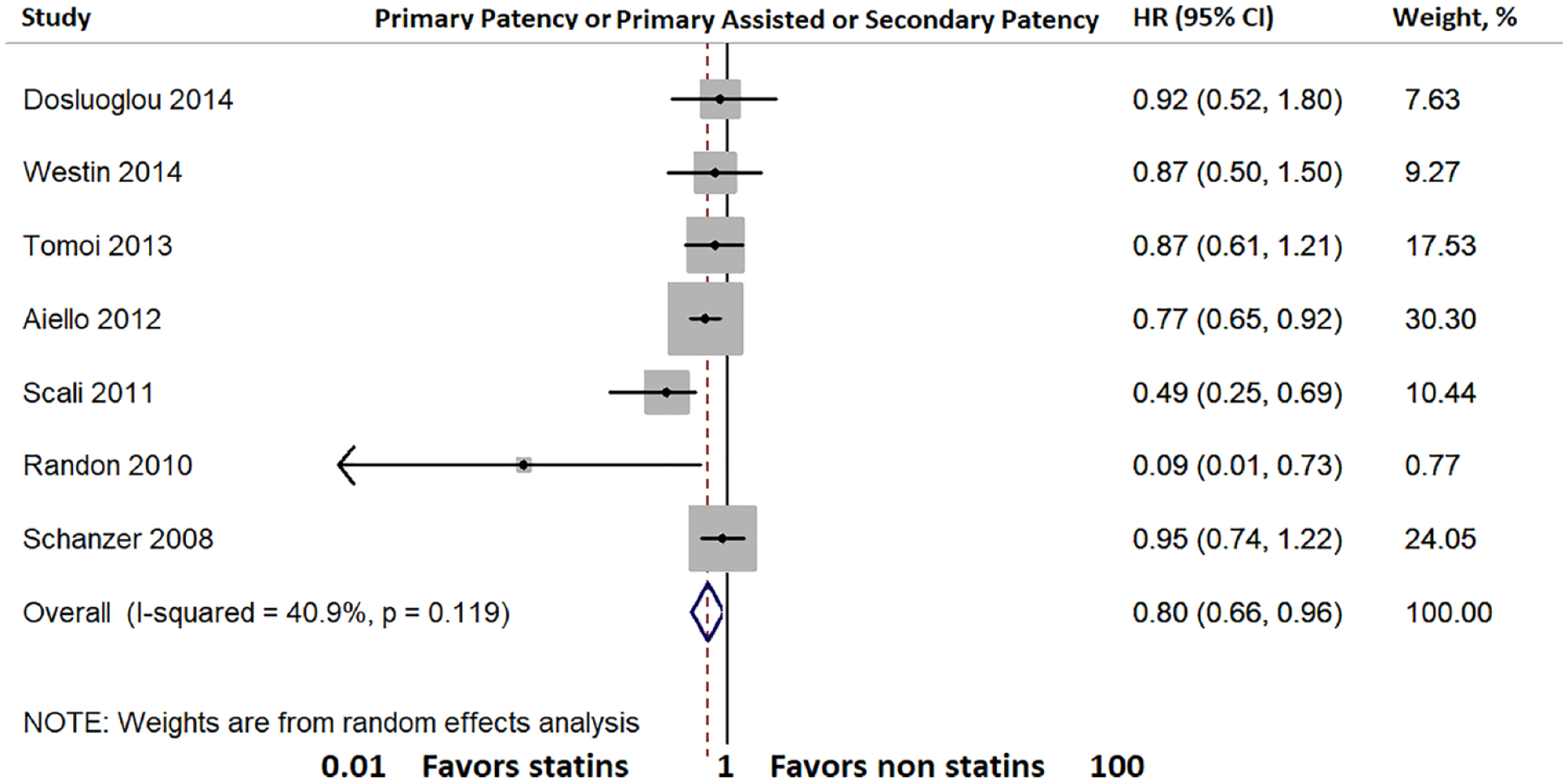
Pooled analysis of studies that report primary patency or primary assisted patency or secondary patency showed that patients treated with statins were more likely to maintain patency compared to their peers not treated with statins (HR 0.80; 95% CI: 0.66–0.96; I2 = 40.9).
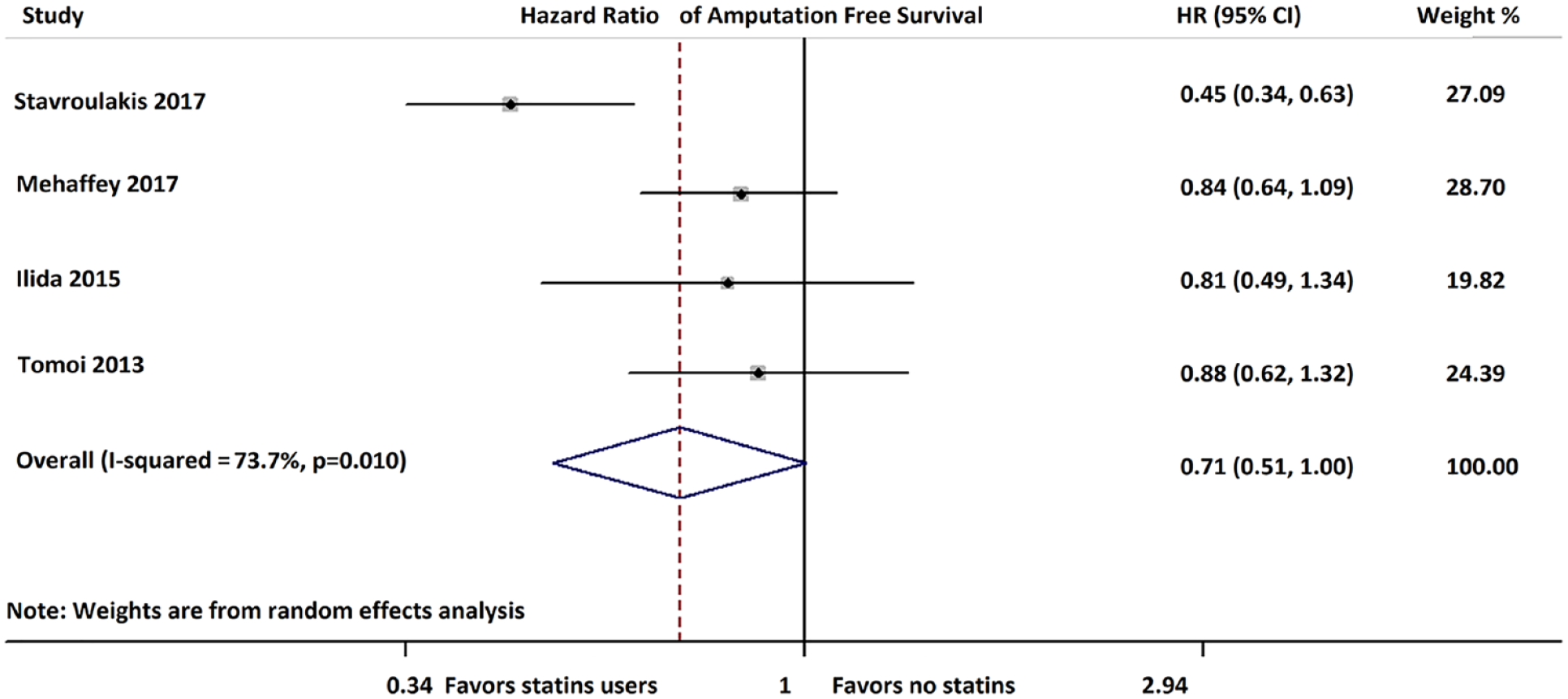
Statins were associated with improved amputation-free survival (HR 0.71; 95% CI: 0.51–1.00; I2 = 73.7%).
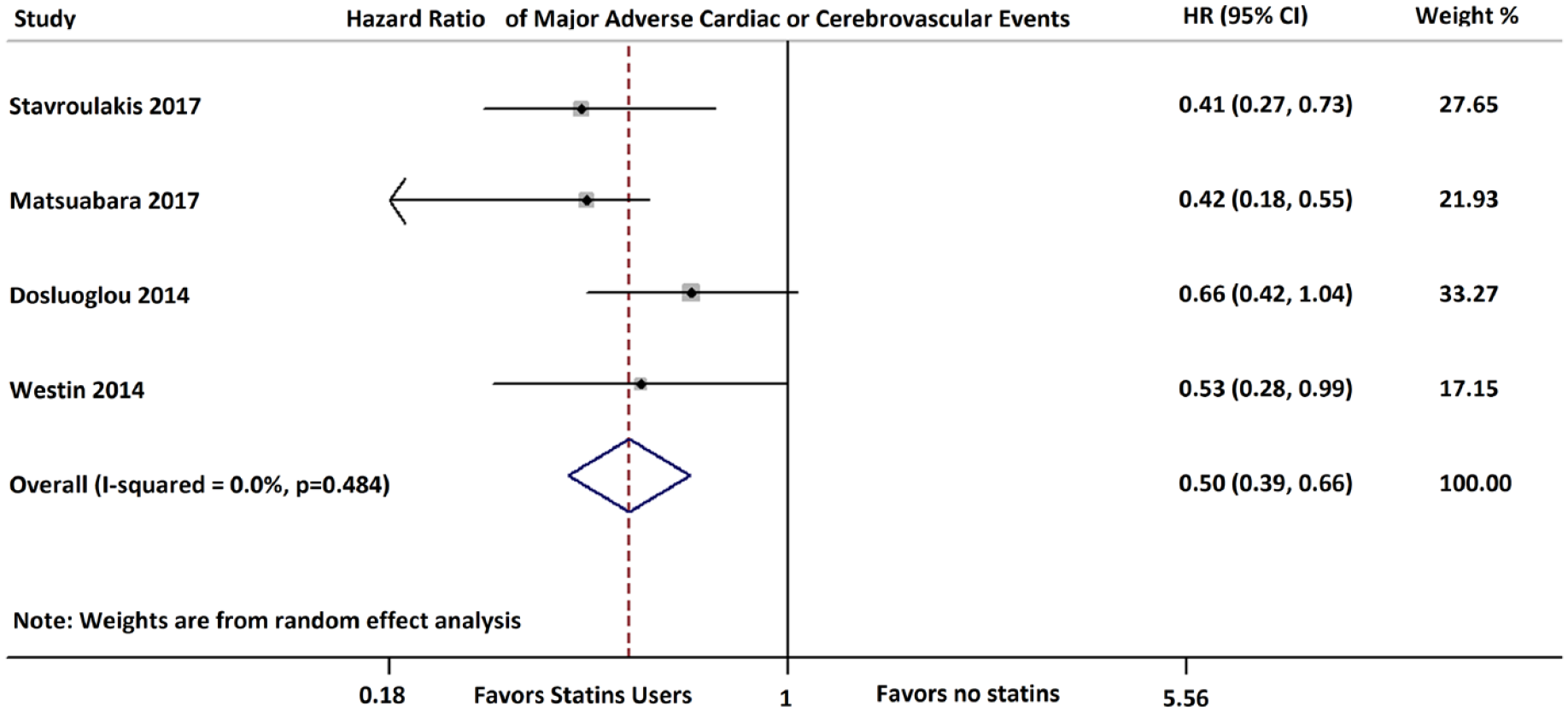
Statins were associated with a decrease in MACCE among four studies that reported this endpoint (HR 0.50; 95% CI: 0.39–0.66; I2 = 0).
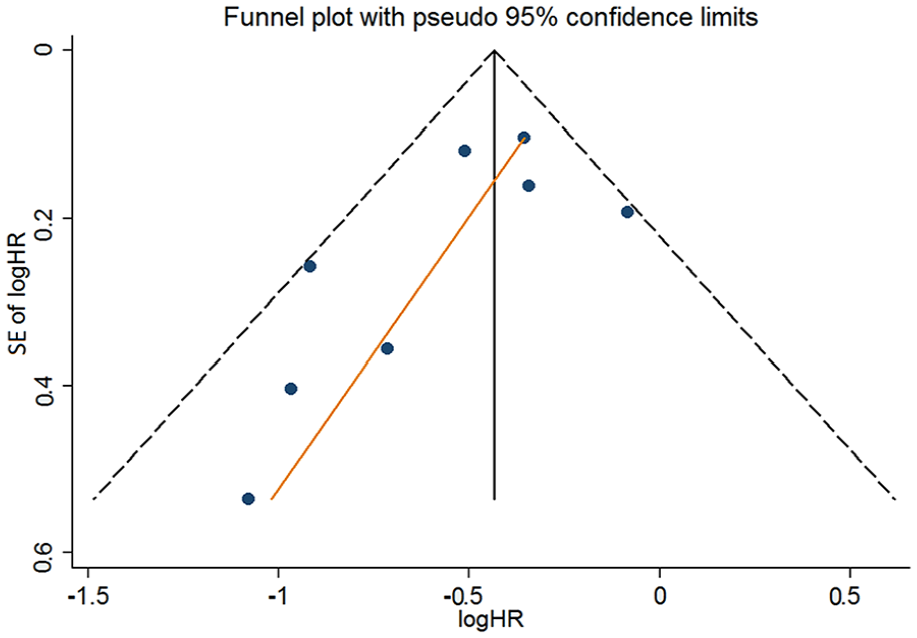
Funnel plot for mortality.
Discussion
This was a systematic review and meta-analysis of the role of statin treatment in patients with CLI. Nineteen studies with a total of 26,985 patients were included. Our results showed that statin use was associated with decreased risk for major amputation, mortality, and MACCE rates, as well as greater amputation-free survival and overall patency rates. No difference was found in primary patency rates. Heterogeneity was substantial for the outcomes of amputation and amputation-free survival.
Although statins have been assumed to have significant benefit among patients with PAD, the Heart Protection Study was the first study to demonstrate significant benefit in PAD. 39 A prespecified subgroup analysis from that study among patients with PAD showed that simvastatin led to an almost 25% reduction of MACCE compared to placebo. Since then, the ACC/AHA guidelines adopted clear recommendations for statin administration in these patients.12,40 However, it is well known that statins are underutilized in PAD compared to CAD. Previous reported rates from a large Danish registry showed that statin utilization was as low as 55% in patients with PAD without concomitant CAD and 65% in patients with PAD and also CAD, while earlier studies in CLI reported rates as low as 50%.27,31,41,42 However, and given the clear recommendations by ACC/AHA for high-intensity statin use in patients with PAD, these rates are slowly increasing, although not yet at the desired levels. 16
Statin medications are 3-hydroxy-3 methylglutaryl-coenzyme A reductase inhibitors and have known benefit in decreasing endothelial inflammation and increasing arterial plaque stabilization. 43 Our study confirmed earlier reports and showed once again that statin use in the CLI population is associated with decreased risk for mortality and MACCE.15,16,26 Although there is enough level of evidence from prior research explaining why and how statins help decrease the risk of MI, stroke, and overall cardiovascular mortality in patients with PAD, the exact mechanism that permits statins to improve PAD and CLI-focused outcomes remains unclear.39,44 Especially for the CLI population, a recent study that examined the pathology of arteries from patients with CLI found that luminal thrombi exist in up to 70% of the CLI population, while almost two-thirds of these thrombi may not be associated with significant atherosclerotic lesions. 45 One could assume that the high burden of atherosclerosis and calcification in these patients could be a limitation for achieving improved outcomes. 15 Earlier studies showed that patients with PAD have improved pain-free walking distance and 6-minute walking test, and an overall physical function when treated with statins.46–51 Subsequent single-center studies confirmed the findings by the Heart Protection Study regarding the benefit of statins in terms of MI or death,15,16 MACCE, while other studies showed a benefit for statins (which was also confirmed by our meta-analysis) in terms of maintaining primary patency or primary assisted patency.27–29 The REACH registry demonstrated that patients with PAD using statins had a lower risk of adverse limb events compared to those not taking statins over 4 years of follow-up. 42 The investigators of the Heart Protection Study failed to find a superiority for the statin group versus no statins in terms of decreasing amputation rates in the PAD population, despite the large sample size.52,53 Earlier reports that focused on CLI populations only either were unable to detect a difference,15,16,28 or showed a superiority for statins only when they examined the composite endpoint of amputation or death and not for amputation only. 16 Recently, in one report from the CRITISCH registry, including patients with CLI treated with either endovascular or open techniques, Stavroulakis et al. also did not find a difference between the two groups in amputation rates, but showed that the amputation-free survival was increased among statins users. 15 A previous study by Arya et al. has shown that high-intensity statin use in patients with PAD has the potential to improve the outcomes, but the CLI rate in this population was only 11%. 44 To the best of our knowledge, our study is the first to suggest a benefit for statins in terms of decreasing amputation rates in the CLI population. From a mechanistic standpoint, we do not have definite evidence of the pathway that improves limb salvage in patients taking statins. However, potential mechanisms include, but are not limited to: plaque stabilization, prevention of further worsening of atherosclerosis, improved wound healing, and a reduction in thrombotic burden.
Statin compliance is only one of the many aspects of optimal medical therapy for patients with CLI. Even if the ACC/AHA guidelines do not mention a specific LDL-cholesterol (LDL-C) target for lipid-lowering therapy, the European Society of Cardiology guidelines provide a Class I (Level of Evidence C) recommendation that all patients with PAD should have their serum LDL-C reduced to ⩽ 1.8 mmol/L (70 mg/dL) or decreased by ⩾ 50% if the initial LDL-C level is between 1.8 and 3.5 mmol/L (70 and 135 mg/dL). 14
Antithrombotic therapy, glycemic control, smoking cessation, close follow-up with focused CLI clinics which should include a multidisciplinary set of CLI specialists (cardiologists, vascular surgeons, podiatrists, nurses, etc.), and advanced limb salvage endovascular techniques are all vital parts of medical care for patients with CLI. Moreover, recent reports have shown that high-intensity statin therapy can further decrease amputation and mortality rates among patients with CLI.54–56 Recently, a post-hoc analysis among patients with PAD from the FOURIER (Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk) trial was published. 57 This study showed that adding PCSK9 (a nonstatin lipid-lowering agent) to moderate or high-intensity statins can be beneficial for patients with PAD, and regardless of MI or stroke history it can decrease major adverse limb event rates. No specific data for the CLI population of FOURIER were reported.
Limitations
This study has a number of limitations. First, this was a meta-analysis of observational studies, and thus should be interpreted in the context of real-world research and its inherent limitations. Given the observational design of the included studies, it is impossible to definitely attribute to statins the lower risk for events in the statin group. It is possible that patients who were taking statins were also more likely to be prescribed and be compliant with antithrombotics, but we did not have access to individual patient level data to adjust for that. Second, the location and the nature of the lesions, as well as the definition of CLI, statins regimen, and duration of treatment among the studies were heterogeneous. Third, the follow-up of the studies was heterogeneous. Fourth, in this meta-analysis, patients from different countries, with different socioeconomic and ethnic backgrounds, are pooled together. Fifth, there was considerable heterogeneity between the studies for the association between statin use and limb loss (I2 = 79%, p < 0.001) and amputation-free survival (I2 = 73.7%, p = 0.010), limiting confidence in the conclusion that statins were associated with lower rates of these endpoints.
Conclusion
Our meta-analysis shows that statin use is associated with decreased amputation and mortality rates, among other improved outcomes in patients with CLI. Given the very high short-term risk that these patients face for amputation and mortality, more research is warranted on this topic. Because of the known benefit of statins in atherosclerotic disease, it is unlikely that an RCT would be conducted, but future studies should try to shed light into the specific statin subtypes and dosage, and the additional medical optimization of care that should be given to CLI patients in order to achieve optimal outcomes.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Armstrong is a consultant to Abbott Vascular, Boston Scientific, Cardiovascular Systems, Janssen, Medtronic, and Philips. Dr Giri has served on advisory boards for AstraZeneca, received research funds to his institution from Abbott Vascular and Recor Medical, and serves on the Board of Directors for the PERT Consortium (501c3 not-for-profit organization). Dr Dharam J Kumbhani has received honoraria from the American College of Cardiology. Dr. Parikh is an advisory board member to Abbott, Boston Scientific, Medtronic, CSI, and Philips. He receives research funding from Shockwave Medical, TriReme Medical, Intact Vascular and Surmodics. He serves as a consultant to Terumo. Dr. Secemsky is a consultant to CSI, Medtronic, and Philips, and has received grants to his institution from AstraZeneca, BD Bard, Cook, CSI, and Medtronic.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
