Abstract
Few prospective studies have examined the factor V paradox: factor V Leiden (FVL) is a stronger risk factor for deep venous thrombosis (DVT) than for pulmonary embolism (PE). The present study, to the best of our knowledge, is the first population-based study aimed to examine the relationship between FVL and incidence of venous thromboembolism (VTE), DVT and PE in a prospective cohort study of middle-aged Swedish individuals. FVL was determined in 4890 subjects (aged 46–68 years, 57% women) from the general population without previous VTE or cancer, who participated in the Malmö Diet and Cancer study between 1991 and 1994. Incident cases of VTE were identified from the Swedish patient register during a mean follow-up of 15.6 years. Of 4890 subjects with determination of FVL (10.2% carriers), 220 had VTE during follow-up (113 DVT, 78 PE, 29 both). Incidence of VTE was significantly higher in subjects with heterozygous and homozygous FVL: adjusted hazard ratios (HR) were 1.8 (95% CI 1.3–2.6, p=0.001) and 6.5 (2.1–21, p=0.001), respectively. The population attributable fraction was 8.7% for FVL. Adjusted HRs for DVT were 2.2 (1.4–3.3, p<0.001) for heterozygotes and 3.3 (0.5–24, p=0.233) for homozygotes. Adjusted HRs for PE were 1.2 (0.65–2.2, p=0.582) for heterozygotes and 8.7 (2.1–36, p=0.003) for homozygotes. The FVL paradox was confirmed for heterozygotes for FVL. However, homozygotes for FVL had a high risk for PE, suggesting that the FVL paradox is related to the carriership of one wild type and one mutated factor V allele.
Keywords
Introduction
Since the discovery of activated protein C (APC) resistance in 1993 by Dahlbäck et al., 1 and later the demonstration of the Arg506Gln mutation in coagulation factor V (factor V Leiden = rs6025) as the causative mutation in the majority of APC-resistant families, a large number of studies have established factor V Leiden (FVL) as the most common thrombophilic disorder.2–6 Heterozygous carriers of FVL have a three to seven times increased risk of venous thromboembolism (VTE), while homozygotes have a 30–100 times increased risk.7–9 A puzzling phenomenon is that FVL in most case–control studies is strongly associated with deep venous thrombosis (DVT) of the legs, and only weakly or not at all associated with pulmonary embolism (PE). 10 Desmarais et al. first reported this observation. 11 This phenomenon is called the factor V paradox because similar risks for DVT and PE are expected, as the majority of cases with PE are believed to be the result of an embolization of a thrombosis from the legs.9–14 Another issue is that most studies have been case–control studies.15–25 In two prospective studies there was no major difference in risk for DVT of the legs or PE among carriers of FVL.26,27 In a prospective study with a nested case–control design by Folsom et al., the age-adjusted odds ratio (OR) for PE was 2.86 and DVT 3.76 among white and African American individuals. 26 A limitation in this study is that FVL is mainly present in Caucasian populations. Only three cases were homozygous for FVL. In another prospective Danish study by Juul et al., the adjusted hazard ratios (HRs) for heterozygous and homozygous FVL carriers were 2.4 (95% confidence interval (CI), 1.3 to 3.8) and 22 (95% CI, 0 to 60) for DVT, and 3.0 (95% CI, 1.7 to 4.9) and 11 (95% CI, 0 to 33) for PE, respectively. 27 Adults in all ages were included in the Danish study. Thus, the two published prospective studies argue against the factor V paradox.
Sweden is probably the country in the world with the highest prevalence of FVL (at 10–15%).28–30 In such a population with a high prevalence of factor V, we wanted to examine whether the factor V paradox is valid or not using a prospective cohort study. The present paper sought to study whether FVL is associated with incident VTE, DVT and PE in the Malmö Diet and Cancer (MDC) prospective cohort study of middle-aged men and women.31–38
Materials and methods
Study population
The MDC study is a prospective cohort study from the city of Malmö in southern Sweden. A total of 28,449 participants (11,246 men, born 1923–1945; 17,203 women, born 1923–1950) participated in the baseline examination between March 1991 and September 1996. Between October 1991 and February 1994, a randomly selected subgroup was invited to an extended study of cardiovascular diseases (CVD).31–38 A total of 6103 subjects accepted and 5540 of them donated fasting blood samples at a separate visit. FVL was available in 5265 subjects. After exclusion of subjects with a history of previous VTE (n=44) and individuals with a diagnosis of cancer before baseline (n=336), the final study population consisted of 4890 subjects (five individuals had both a history of previous VTE and cancer).
All participants provided written informed consent, and the study was approved by the ethics committee at Lund University, Lund, Sweden (LU 51/90).
Measurements and definitions
Information about use of medication, medical history and smoking habits were obtained from a self-administered questionnaire.31–38 Weight and height were measured to the nearest 0.1 kg and 0.5 cm, respectively, with subjects wearing light clothing and no shoes. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared (kg/m2). Blood pressure was measured using a mercury-column sphygmomanometer after 10 minutes of rest in the supine position. Diabetes was defined as a fasting whole blood glucose level greater than 109 mg/dL (i.e. 6.0 mmol/L), self-reported physician’s diagnosis of diabetes or use of anti-diabetic medications. 39 History of atrial fibrillation was defined as a hospital diagnosis of atrial fibrillation and history of coronary heart disease (CHD) was defined as a diagnosis of an acute coronary event. The Swedish Inpatient Register was used for case-retrieval. 40 Subjects were categorized into current smokers (i.e. those who smoked regularly or occasionally) or non-smokers (i.e. former smokers and never smokers). Use of blood pressure (BP) medication, statins, and hormone replacement treatment (HRT) at baseline was also registered.
DNA was extracted from peripheral blood cells. Genotyping was performed at the Broad Institute (Boston, MA, USA) with the Illumina HumanOmniExpressExome BeadChip version 1.0 and the iScan System (Illumina, San Diego, CA, USA), using the AutoCall calling algorithm. The array included >240,000 exome variants and >700,000 markers for coverage of the genome-wide variation. The FVL (rs6025) polymorphism was imputed according to 1000Genomes, build 37, using IMPUTE 2 software [www.1000genomes.org]. 41 Genetic data for imputation of the FVL mutation was available for 5265 individuals.
Ascertainment of endpoint
All subjects were followed from the baseline examination until a first event of VTE (primary or secondary diagnosis), emigration, death or 31 December 2009, whichever came first. VTE was defined as International Classification of Diseases, Eighth Revision (ICD-8, used before 1987) codes 450 (pulmonary embolism, PE) and 451 (deep vein thrombosis (DVT) of the lower limbs); ICD-9 codes (used 1987–1996) 415B (PE) and 451 (DVT of the lower limbs); and ICD-10 codes (used 1997–2009) I26 (PE) and I80 (DVT of the lower limbs). Patients with superficial thrombophlebitis were excluded (ICD-9 code 451A and ICD-10 code I80.0). The Swedish inpatient and outpatients registers and the cause of death register were used for case retrieval.40,42 The inpatient register and cause of death register have been operating during the whole follow-up period, and the outpatient register covers hospital outpatient visits from 2001 and onwards. Validation studies from the patient register have shown high case validity of a diagnosis of VTE (95%), 43 and for other cardiovascular disorders.44,45
Malmö has only one hospital: Malmö University Hospital. All objective assessments for DVT/PE are performed at the Department of Diagnostic Radiology at the Malmö University Hospital. All diagnostic workups were performed by a radiologist and the next day by another radiologist for accuracy. It has been shown that almost all VTE patients in Malmö University Hospital are diagnosed with objective methods such as phlebography, ultrasound or computer tomography (CT). 46 In the study by Isma et al. of VTE patients diagnosed in Malmö between 1998 and 2006, phlebography had been used in 739 (84%) patients, duplex ultrasound in 214 (24%) patients, other diagnostic methods in 13 (1.4%) patients, and 142 (16%) patients had undergone both phlebography and duplex ultrasound. 46 Of 330 PE patients, 313 (95%) had been diagnosed using CT, 19 (6%) with lung scintigraphy, two (1%) with other diagnostic methods, and in six (2%) with both CT and lung scintigraphy. 46 In 58 (7%) DVT cases, the diagnosis was based on clinical symptoms in patients with a concomitant and objectively verified PE. 46 From the study by Isma et al., we know that of 882 DVT patients diagnosed at Malmö University Hospital, 116 (13%) had iliac DVT, 465 (53%) had DVT involving femoropopliteal veins, 246 (28%) had calf DVT, and 55 (6%) had unknown DVT location. 46 Only six patients (0.7%) had bilateral DVT. 46 Among Malmö VTE patients according to Isma et al., the most common acquired risk factors were hormone therapy (24% of female DVT patients and 19% of female PE patients), previous surgery (13% of DVT patients and 19% of PE patients), and malignant disease (12% of DVT patients and 11% of PE patients). 46 Provoked VTE was present in 632 (55%) of the 1140 patients. The remaining 508 (45%) VTE patients had unprovoked VTE. 46
Statistical analysis
The subjects were categorized according to FVL genotype. Cox proportional hazards regression was used to examine the association between FVL genotype and incidence of VTE, DVT, and PE. The time axis was time from the baseline examination until death, emigration, incident VTE, or end of follow-up, whichever occurred first. Age and sex were included as covariates in the sex- and age-adjusted model. Secondly, we also adjusted for potential cardiovascular risk factors due to the reported association between arterial and venous thromboembolic events: 47 history of CHD (acute coronary event), atrial fibrillation, diabetes mellitus, systolic blood pressure (SBP), leucocyte count, BMI, current smoking, use of BP medication, statins, and HRT. Interaction terms were added to the full multivariate model to explore potential interactions between FVL genotype and other variables. The fit of the proportional hazards model was checked visually by plotting the incidence rates over time and by using time-dependent variables in the model. We calculated the population attributable fraction (PAF) as described. 48 PAF describes the fraction of disease occurrence in a population associated with a particular risk factor. It is obtained by dividing the population attributable risk by the total incidence of disease in the population. Population attributable risk is the product of the attributable risk (difference between the incidence in exposed and non-exposed persons) and the prevalence of exposure to the risk factor in the population. 48
Results
Baseline characteristics
A total of 4890 individuals were available for the analysis. The distribution of the factor V allele was in perfect Hardy–Weinberg equilibrium (p-value=0.8995): no FVL allele 4389 (89.75%) observed versus 4388.56 expected; FVL heterozygotes 487 (9.96%) observed versus 487.88 expected; and 14 (0.29%) observed FVL homozygotes versus 13.56 expected. Table 1 presents the baseline characteristics of the study population according to carriership of the FVL allele. Women were more prevalent in the MDC study (Table 1). However, carriership of FVL was similarly common in women and men (10.3% [n=211] vs 10.1% [n=290]).
Baseline characteristics of the included participants (n=4890).
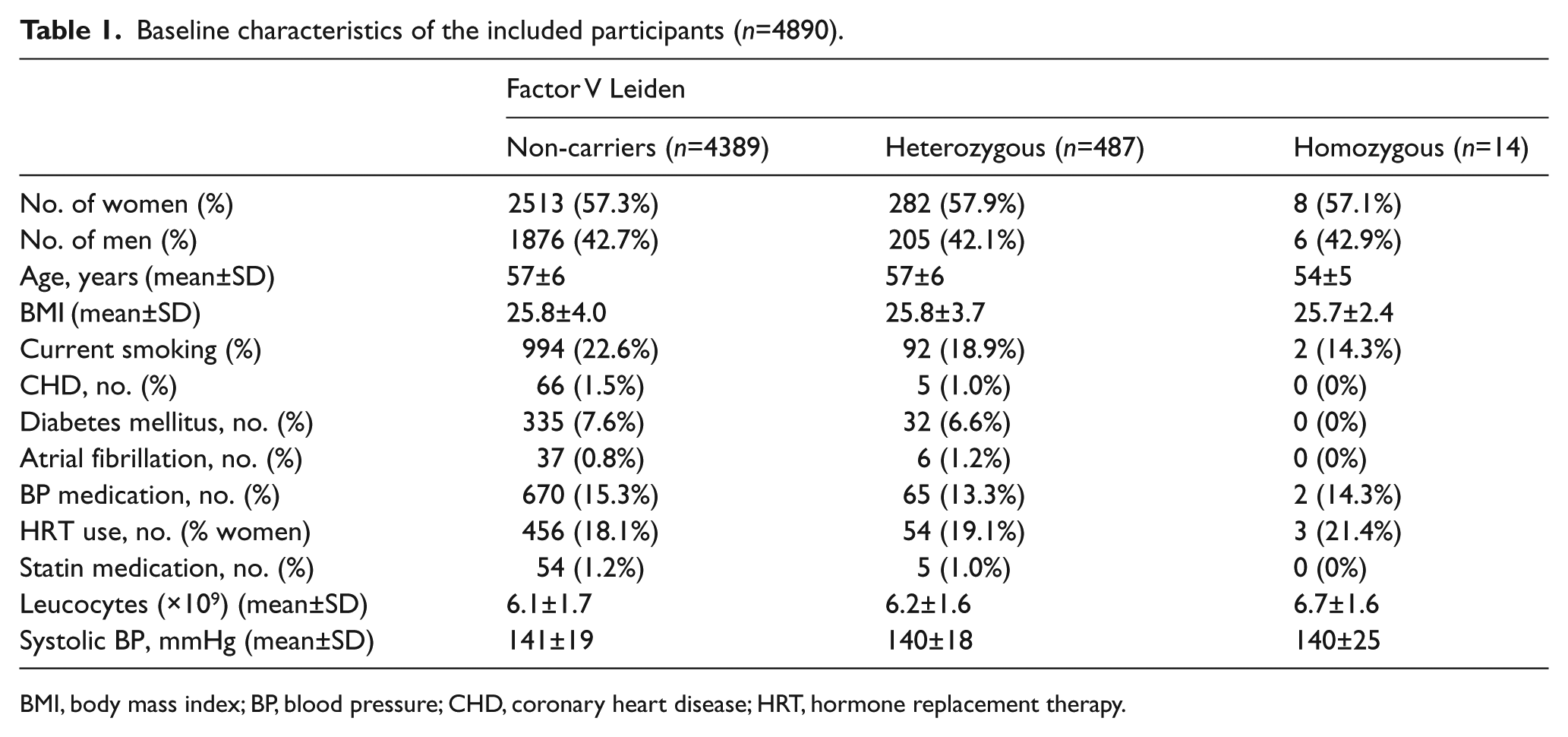
BMI, body mass index; BP, blood pressure; CHD, coronary heart disease; HRT, hormone replacement therapy.
Factor V Leiden and incidence of VTE
During a median follow-up of 16.76 years (interquartile range 16.05–17.40 years), a total of 220 (87 men, 133 women) had VTE (Table 2). The distribution of the different types of VTE manifestations according to number of FVL alleles are shown in Table 2. The sum of the follow-up time was 76,470.22 years, corresponding to a VTE incidence rate of 2.9 (95% CI 2.5–3.3) per 1000 person years. The VTE incidence rates per 1000 person years were for non-carriers, FVL heterozygotes, and FVL homozygotes: 2.6 (95% CI 2.3–3.0), 4.8 (95% CI 3.4–6.5), and 14.0 (95% CI 3.6–38.1), respectively. The PAF was calculated to be 7.6% for FVL heterozygosity and 1.1% for FVL homozygosity. Thus, FVL accounts for 8.7% of all VTE events in the population. The incidence of VTE was significantly higher in heterozygous or homozygous carriers of the FVL alleles compared to those without both in the crude, sex- and age-adjusted, and in the fully adjusted models (Table 3 and Figure 1A). The fully adjusted model was adjusted for the potential cardiovascular risk factors shown in Table 1. The fully adjusted HRs were 1.8 (95% CI 1.3–2.6, p=0.001) for heterozygous carriers and 6.5 (95% CI 2.1–21, p=0.001) for homozygous carriers.
Manifestations of venous thromboembolism (VTE), deep venous thrombosis (DVT), and pulmonary embolism (PE) according to factor V Leiden carriership.

Hazard ratios (HR) according to factor V Leiden (FVL) genotype for venous thromboembolism (VTE), deep venous thrombosis (DVT), and pulmonary embolism (PE).

Adjusted for potential cardiovascular risk factors: age, sex, BMI, leucocyte count, current smoking status, BP medication, statin use, HRT use, CHD, atrial fibrillation, diabetes mellitus, and systolic BP (mmHg).
BMI, body mass index; BP, blood pressure; CHD, coronary heart disease; HRT, hormone replacement therapy.
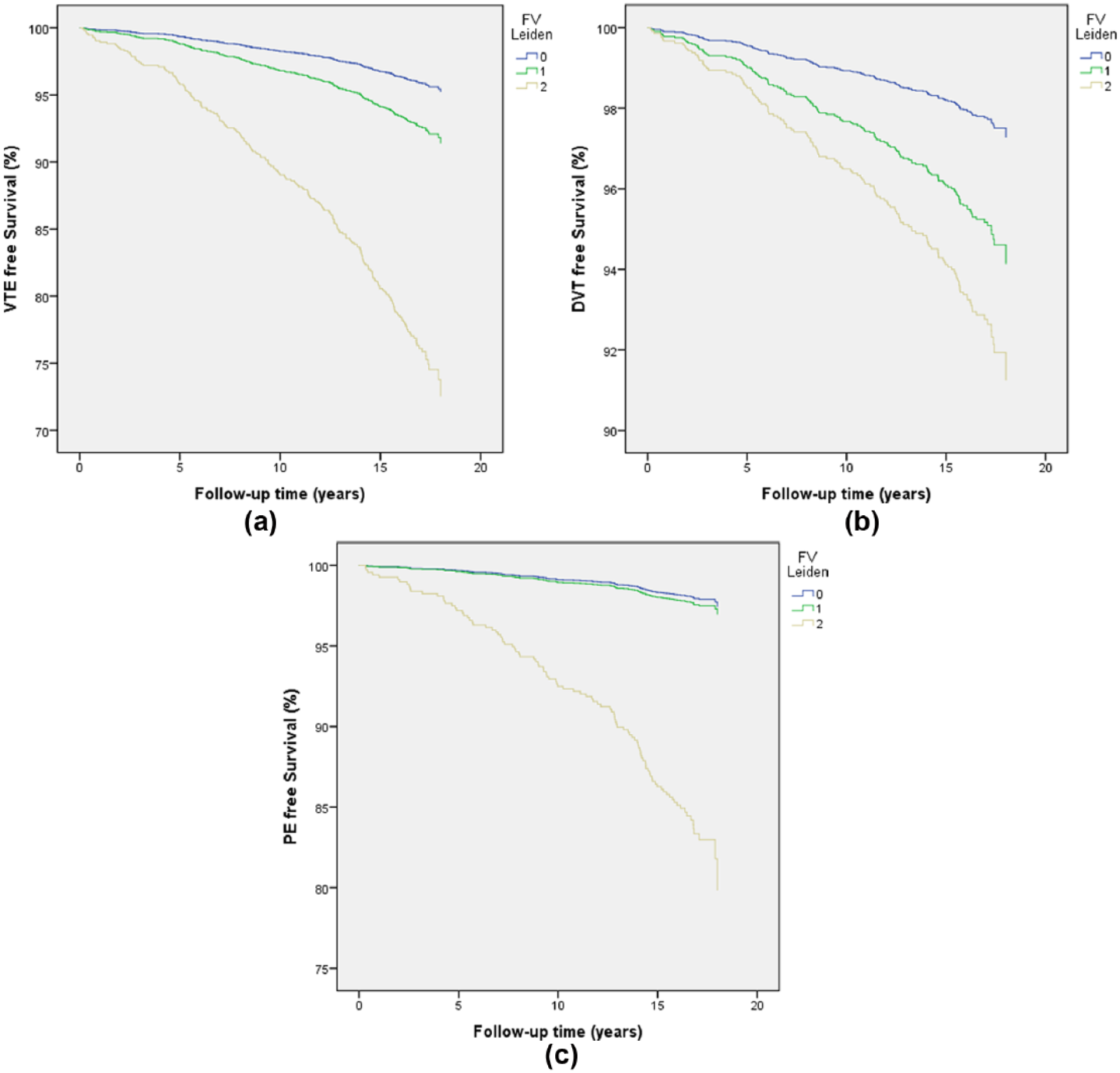
Fully adjusted Cox regression curves showing the first (a) venous thromboembolism (VTE); (b) deep venous thrombosis (DVT); and (c) pulmonary embolism (PE) event-free survival in relation to factor V (FV) Leiden carriership (0, 1, or 2 F V Leiden alleles). Note: Figure is in color online.
Factor V Leiden and incidence of DVT
Incidence of DVT was significantly higher in subjects with heterozygous but not homozygous carriers of the FVL alleles compared to those without both in the crude, sex- and age-adjusted, and the fully adjusted models (Table 3 and Figure 1B). The fully adjusted model was adjusted for all variables shown in Table 1. The fully adjusted HRs were 2.2 (95% CI 1.4–3.3, p<0.001) for heterozygous carriers and 3.3 (95% CI 0.5–24, p=0.233) for homozygous carriers.
Factor V Leiden and incidence of PE
Incidence of PE was significantly higher in subjects with homozygous but not heterozygous carriers of the FVL alleles compared to those without both in the crude, sex- and age-adjusted, and the fully adjusted models (Table 3 and Figure 1C). The fully adjusted model was adjusted for all variables shown in Table 1. The fully adjusted HRs were 1.2 (95% CI 0.65–2.2, p=0.582) for heterozygous carriers and 8.7 (95% CI 2.1–36, p=0.003) for homozygous carriers.
Sensitivity analysis
A sensitivity analysis was done including subjects with a history of previous VTE (n=44) and individuals with a diagnosis of cancer before baseline (n=336), including in total 5265 individuals in the analysis (Table 4). Five patients had both cancer and a history of previous VTE. The results were almost identical to the results in Table 3 with the exclusion of cancer patients and those with previous VTE.
Sensitivity analysis. a

Data given as hazard ratios (HR) according to factor V Leiden (FVL) genotype for venous thromboembolism (VTE), deep venous thrombosis (DVT), and pulmonary embolism (PE).
Patients with previous VTE and cancer before baseline were also included. In total, 5265 individuals were included in the analysis.
Adjusted for potential cardiovascular risk factors: age, sex, BMI, leucocyte count, current smoking status, BP medication, statin use, HRT use, CHD, atrial fibrillation, diabetes mellitus, and systolic BP (mmHg).
BMI, body mass index; BP, blood pressure; CHD, coronary heart disease; HRT, hormone replacement therapy.
Discussion
The present study is the first prospective population-based cohort study of Swedish middle-aged men and women to confirm that the factor V paradox is valid in heterozygotes. The study also confirms that the prevalence of FVL is very high in southern Sweden.28–30 The high prevalence allowed the prospective incidence and risk of VTE to be determined for both FVL heterozygotes and homozygotes. Interestingly, homozygous carriers of FVL had a high risk for PE, though the number of homozygotes was small. Thus, carrying two factor V alleles may increase the risk for PE while one allele was not associated with PE. Thus, the factor V paradox remains a puzzling phenomenon that remains to be solved. However, the present study suggests that the FVL paradox is related to the carriership of one wild type and one mutated factor V allele. Our study is in line with published case–control studies with similar findings (i.e. FVL heterozygosity is not at all or only weakly associated with PE).9–25 Our study contradicts two previously published cohort studies.26,27 Why the results are different is unclear, but might be related to the fact that one of the studies included African Americans, who have a low prevalence of factor V alleles. 26 The Danish study also included younger individuals, and the present study included only Swedish middle-aged individuals without a previous VTE or cancer before baseline. 27 The present study also confirms that Sweden probably has the highest prevalence of FVL in the world.28–30 The prevalence of FVL in the present study is almost identical to previous published allele frequencies in Malmö.28,30 The HR in this population is much lower than the initial estimation of the VTE risk associated with FVL.1–9 This is partly explained by the study design using a randomly selected population-based cohort study. However, the study population is middle aged and it has been shown that familial and genetic factors are much weaker in older age than at a younger age. 42 This could explain why FVL accounted only for 8.7% (PAF) of all VTE events in this middle aged Swedish population. This age dependence of the thrombotic risk attributed from a defined genetic risk factor is an important issue that often is not considered.
The mechanism of the factor V paradox is not known, but different mechanisms have been proposed, as summarized by Corral et al. 13 The factor V paradox might be explained by a reduced embolization risk conferred by the FVL mutation. 13 The reduced embolization risk might be due to an enhanced local thrombin generation, which increases the inflammatory reaction and strengthens the clot structure by activation of thrombin-induced FXIII transglutaminase activity. However, this hypothesis remains to be proven. Another potential mechanism could be related to a potential antifibrinolytic effect of FVL described by Bajzar et al. 49 Parker et al. have, in another study, confirmed this potential antifibrinolytic effect of FVL mutation. 50 However, none of these hypotheses explain why homozygous but not heterozygous carriership of FVL is associated with an increased risk for PE. Factor V is a complex molecule with complex interactions. 51 Factor V regulates both pro- and anticoagulant pathways. It circulates as a single-chain procofactor, which is activated by thrombin or FXa to FVa that serves as cofactor for FXa in prothrombin activation. 51 The cofactor function of FVa is regulated by APC and protein S. Factor V may also act as an anticoagulant APC cofactor in the inhibition of FVIIIa in the membrane-bound tenase complex (FIXa and FVIIIa complex). In later years, it has been shown that Factor V also affects the tissue factor pathway inhibitor (TFPI) anticoagulant pathway. 51 Thus, there are several possible molecular explanations of the Factor V paradox that remain to be elucidated.
Strengths and limitations
A strength is that the endpoints were retrieved from national registers covering hospital outpatient and inpatient care in Sweden. 40 Moreover, the study population were all from Malmö and all patients were diagnosed and treated at a single hospital (Malmö University Hospital). 46 A study of VTE in this register reported a case validity of 95%. 24 Previous studies from Malmö and Sweden reported that almost all VTE patients are diagnosed with objective methods, such as phlebography, ultrasound or CT. 46 It should be noted that a relatively high proportion of DVT patients were diagnosed with phlebography, which is the gold standard method. 46 Nowadays ultrasound is more common. Still, the majority of patients with PE were diagnosed with the more modern CT method. 46 We do not think this has affected the results. However, the endpoint definition obviously does not include cases of VTE that are undetected or do not seek medical care. A further limitation is that we could not distinguish proximal and distal DVT, but this has been extensively described by Isma et al. among Malmö DVT patients. 46 Another question is whether provoked and unprovoked VTE could change the results. Around 55% of Malmö VTE patients have provoked VTE according to Isma et al. 46 At least, the inclusion of cancer-provoked VTE in the sensitivity analysis did not affect the results to any major degree. Moreover, provoked VTE is similarly common in FVL patients and those without FVL. 6 Though the data are slightly old (censored on 31 December 2009), clinical practice concerning the diagnostic procedure for VTE regarding phlebography, ultrasound and CT is still relevant for today’s practice. Though one could argue that the lack of association between heterozygous FVL and PE is due to the limited number of VTE events, this is not a likely explanation due to the fact that the fully adjusted HR was only 1.2 for PE (p=0.582) but 2.2 for DVT (p<0.001). For homozygotes, the fully adjusted HR was 3.3 for DVT (p=0.233) but 8.7 for PE (p=0.003).
The FVL mutation was imputed using a whole genome scan, but the distribution of the FVL allele was in perfect Hardy–Weinberg equilibrium and the prevalence of the factor V allele was identical to previously published smaller studies.28,30 However, our analysis did not include family history or other less common genetic risk factors for VTE. Thus, we had no information about other inherited prothrombotic conditions or antiphospholipid antibodies. Several factors such as BMI and medication were measured only at baseline, which is a limitation. However, FVL is not associated with these factors. All patients with self-reported physician-treated cancer before baseline were excluded. Certain risk factors for VTE were unavailable, which is another limitation of the study. We have no information about major surgery or trauma during the follow-up period. However, the results were almost identical when including patients with previous cancer or history of VTE before baseline in a sensitivity analysis. Adding comorbidities and potential cardiovascular risk factors, including HRT, in the full model did not change the results. Moreover, all baseline characteristics and DNA samples for FVL determination were collected several years before the VTE events; it is unlikely that subsequent trauma or surgery could be a cause of bias in this study. We cannot rule out residual confounding by various comorbidities or concomitant drugs not adjusted for in the analysis. Sweden has had a great influx of immigrants in the last decade, but at the time of study inclusion mainly Caucasians were included. This is an advantage as the prevalence of FVL varies depending on ethnicity,7–9 which might be a problem in studies of mixed ethnicities.
A further limitation is the use of ICD codes that do not allow for distinguishing between proximal and distal DVT. Moreover, we could not separate provoked and unprovoked VTE cases. However, van Stralen et al. have studied these issues extensively without a clear explanation of the FVL paradox. 12 Moreover, this does not change our observation that the FVL paradox is valid for FVL heterozygotes but not for homozygotes in a prospective study of Swedish middle-aged patients.
Clinical implications of the factor V Leiden paradox
What are the clinical implications of the FVL paradox? It is not obvious that the FVL paradox will have any immediate clinical implications because a DVT could also lead to considerable morbidity and even mortality, just as PE. Still, the FVL paradox is important. Elucidation of this paradox could lead to better understanding of why certain DVTs embolize and other do not.
Conclusion
It is concluded that FVL is associated with increased incidence of VTE in this prospective cohort study from a Swedish general middle-aged population. Heterozygous FVL was associated with DVT and not PE, confirming the factor V paradox to be valid in this setting. Homozygous FVL was associated with PE, although few homozygotes were included in the study. The cause of the factor V paradox remains an elusive enigma to be elucidated. However, homozygotes for FVL had a high risk for PE, suggesting that the FVL paradox is related to the carriership of one wild type and one mutated factor V allele.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was supported by the Swedish Heart-Lung Foundation, the Swedish Research Council, and grants from Lund University and Skåne University Hospital (ALF).
