Abstract

Hospitalizations are common in people with lower-extremity peripheral artery disease (PAD), but associations of hospitalizations with response to an exercise intervention in PAD are unknown. 1 Understanding whether and how hospitalization affects response to an exercise intervention, which is first-line therapy for PAD, can potentially assist clinicians in tailoring treatment for patients with PAD who are hospitalized during the time period that they are participating in a walking exercise intervention.
In post hoc analyses, data from two randomized clinical trials of home-based walking exercise for people with PAD, Low Intensity Exercise Intervention in Peripheral Artery Disease (LITE) and Group Oriented Arterial Leg Study (GOALS), were combined to evaluate whether participants hospitalized during the walking exercise intervention improved their 6-minute walk (6MW) distance less than people not hospitalized.
Detailed methods were reported.2,3 The LITE and GOALS clinical trials were selected for analysis because their exercise interventions were highly effective, inclusion criteria were similar, both studies lasted 12 months, and information about hospitalization was available throughout the trials. Participants who completed the 6MW test at both baseline and 12-month follow-up were included. Participants who participated in both studies were included only for the GOALS trial, completed before LITE began. The institutional review boards at participating sites approved the protocols. All participants provided written informed consent.
People with PAD whose walking was primarily limited by PAD, who were not wheelchair bound or otherwise severely disabled, and for whom an exercise program was determined likely to be safe, were included. Participants underwent a 6MW test at baseline and at 12-month follow-up. In the exercise intervention, participants were asked to walk for exercise 5 days per week, working up to 50 minutes per session according to their ability, and had weekly contact with a study coach. Hospitalizations were ascertained via monthly telephone calls and at 6-month and 12-month follow-up visits. Hospitalizations were categorized as: cardiovascular event, coronary revascularization, infection, lower-extremity revascularization, and ‘other,’ which included hospitalizations that did not fall into the former categories such as orthopedic injuries (i.e., fractures) or gastrointestinal events (i.e., bleeding).
Baseline characteristics were compared between those hospitalized versus not hospitalized using a two-sample t-test with unequal variance for continuous variables and chi-squared test or Fisher’s exact test for categorical variables. Among those randomized to exercise or control groups, the mean change in 6MW distance from baseline to 12 months was compared between those hospitalized versus not hospitalized using a two-sample t-test with unequal variance. Among those randomized to exercise or control, analysis of covariance (ANCOVA) examined the between-group differences in 6MW distance at 12 months between those hospitalized versus not hospitalized, adjusting for age, sex, race, baseline 6MW distance, and variables that differed meaningfully between participants hospitalized and those not hospitalized: body mass index (BMI), myocardial infarction, stroke, pulmonary disease, and study (GOALS vs LITE).
Of 432 participants with PAD (mean age: 70 years, 48.8% women, 54.9% Black), 300 (69.4%) were randomized to exercise and 132 (30.6%) were randomized to control. A total of 117 participants (27.1%) were hospitalized at least once, including 87 (29.0%) randomized to exercise and 30 (22.7%) randomized to control (p = 0.18). Of those hospitalized, 38 (43.7%) randomized to exercise and eight (26.7%) randomized to control had two or more hospitalizations (p = 0.10). Online supplemental Figure 1 shows the reasons for hospitalization. At baseline, participants who were hospitalized had a significantly higher BMI, shorter 6MW distance, and higher prevalence of pulmonary disease, myocardial infarction, and stroke compared to those not hospitalized (online supplemental Table 1).
Among participants randomized to exercise, hospitalization during the trial was associated with significantly less 6MW distance improvement compared to no hospitalization (mean change: –0.2 vs +25.5 meters; adjusted between-group difference: –35.1 meters [95% CI: –55.4, –14.8], p = 0.0008) (Table 1). Among participants randomized to exercise, two or more hospitalizations were associated with significantly less improvement in 6MW distance compared to those not hospitalized (–9.0 vs +25.5 meters; adjusted between-group difference: –46.5 meters [95% CI: –76.3, –16.7], p = 0.0023). There was no statistically significant association between hospitalization and change in 6MW distance among participants randomized to control (Table 1).
Association of hospitalization with change in 6-minute walk test at 12-month follow-up among participants with peripheral artery disease randomized to clinical trials of home-based walking exercise and a nonexercise control.
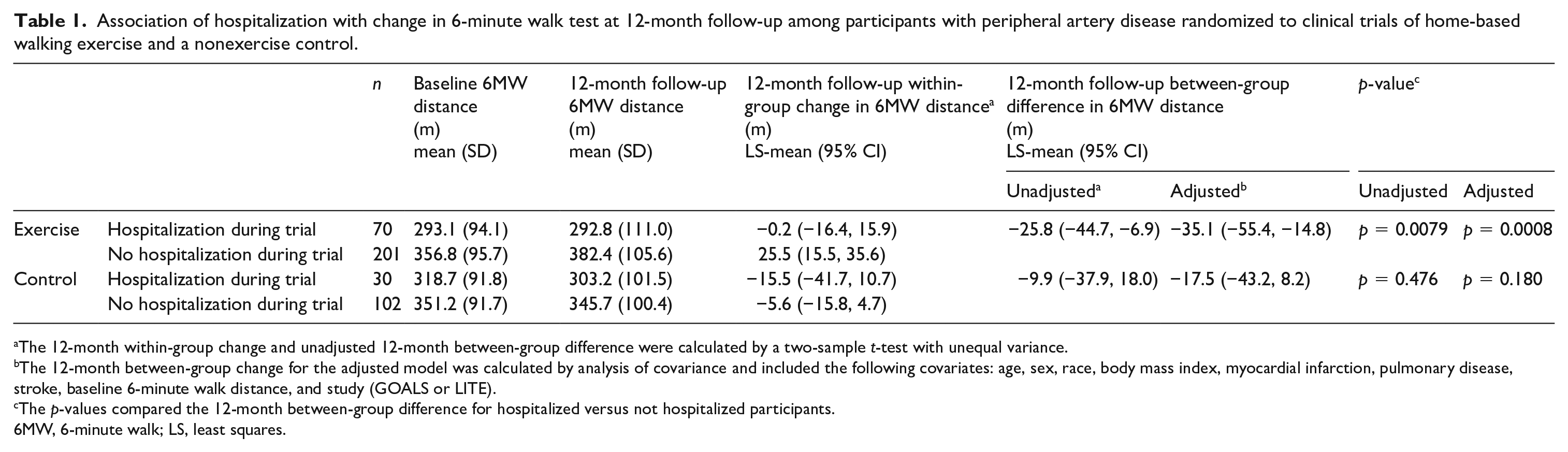
The 12-month within-group change and unadjusted 12-month between-group difference were calculated by a two-sample t-test with unequal variance.
The 12-month between-group change for the adjusted model was calculated by analysis of covariance and included the following covariates: age, sex, race, body mass index, myocardial infarction, pulmonary disease, stroke, baseline 6-minute walk distance, and study (GOALS or LITE).
The p-values compared the 12-month between-group difference for hospitalized versus not hospitalized participants.
6MW, 6-minute walk; LS, least squares.
In this analysis of two randomized clinical trials of highly effective home-based walking exercise interventions for PAD, hospitalization occurred in 29.0% (nearly one-third) of those randomized to exercise, and was associated with significantly poorer improvement in 6MW distance, by 35.1 meters, compared to those not hospitalized. 4 A systematic review of 27 studies (1642 participants) of participants with PAD who were randomized to a home-based exercise intervention for 12–52 weeks reported that home-based exercise was directly related to four adverse events. 5 A separate review of 74 studies (2876 participants with PAD) of duration 2 weeks to 12 months reported that supervised exercise was directly associated with eight adverse events. 6 However, neither systematic review reported whether occurrence of hospitalization during participation in an exercise intervention was associated with poorer response to exercise in people with PAD. Because people with PAD have multiple comorbidities, extreme walking disability, and can respond to relatively small amounts of walking exercise, the effects of hospitalization on response to exercise could differ between people with and without PAD. For example, since relatively small amounts of exercise can improve the 6MW distance in PAD, hospitalization might have a smaller effect on gains from an exercise program. Alternatively, hospitalization-associated deconditioning could be more pronounced in people with PAD, limiting their participation in home-based exercise therapy after hospital discharge, and reducing gains in walking performance. 7
This study has several strengths. First, 55% of participants were Black and approximately 50% were women. Second, the relatively large number of hospitalizations provided statistical power to assess associations of hospitalization with change in 6MW distance. This study also had some limitations. First, analyses were post hoc and require confirmation. Second, the number of participants in the control group was smaller than for the exercise interventions owing to the trial design for LITE. 2 Therefore, there may have been inadequate statistical power to detect statistically significant differences between hospitalized and not hospitalized participants in the control group. Third, details about length of hospital stay or severity of illness during hospitalization were not collected. Fourth, this study focused on associations of hospitalizations with response to highly effective home-based exercise interventions. Results may have differed if ineffective home-based exercise interventions had been included in analyses. 8
Among people with PAD engaged in home-based walking exercise, hospitalizations were common and were associated with less improvement in walking performance compared to those not hospitalized. Further study is needed to identify targeted interventions to help patients with PAD maximize benefits from exercise after a hospitalization.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X231191909 – Supplemental material for Hospitalizations during home-based walking exercise interventions in peripheral artery disease: Results from two randomized clinical trials
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X231191909 for Hospitalizations during home-based walking exercise interventions in peripheral artery disease: Results from two randomized clinical trials by Neela D Thangada, Shujun Xu, Lu Tian, Lihui Zhao, Michael H Criqui, Luigi Ferrucci, W. Jack Rejeski, Christiaan Leeuwenburgh, Todd Manini, Bonnie Spring, Diane Treat-Jacobson, Daniel E Forman, Lydia Bazzano, Jack Guralnik, Robert Sufit, Tamar Polonsky, Melina R Kibbe and Mary M McDermott in Vascular Medicine
Footnotes
Data availability statement
Deidentified data will be shared upon reasonable request to the principal investigator (Dr McDermott) and approval by the institutional review board.
Declaration of conflicting interests
Dr McDermott reported receiving research funding from Regeneron and Helixmith and other research support from Helixmith, ArtAssist, ChromaDex, ReserveAge, Mars Company, and personal fees from Cambrian BioPharma. Dr Spring reported receiving research funding from Actigraph scientific advisory board. The other authors reported no disclosures.
Funding
Research reported in this publication was supported by the National Heart, Lung, and Blood Institute (R01-HL088589) for the GOALS randomized clinical trial and by the National Heart, Lung, and Blood Institute and National Institute on Aging Intramural Division (R01-HL122846) for the LITE randomized clinical trial.
Supplementary material
The supplementary material is available online with the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
